Abstract
Background:
To evaluate a new fully closed-loop (FCL) system in people with type 1 diabetes (T1D) with high-carbohydrate and high-fat unannounced meals.
Methods:
After a 1-week Control-IQ run-in period at home with mealtime insulin boluses, ten adults with T1D used the Tandem Freedom FCL System in the hotel setting for 72 hours without meal announcement or mealtime insulin boluses. Participants consumed high-carbohydrate and high-fat meals during their stay. Exercise challenges occurred each day. A Wilcoxon signed-rank test for nonparametric data compared outcomes between periods.
Results:
Mean participant age was 38.6 years, duration of diabetes 15.9 years, total daily insulin 0.71 units/kg/d, and HbA1c 7.3%. There were no diabetic ketoacidosis (DKA) or severe hypoglycemia events. During the hotel study, FCL was active 97.3% of the time, and median meal size was 70.8 g carbohydrate and 53.2 g fat for breakfast, 53.8 g carbohydrate and 40.0 g fat for lunch, and 96.1 g carbohydrate and 53.1 g fat for dinner. Median time in range (TIR) 70 to 180 mg/dL was 61.0% [58.9, 73.0] without any meal announcement or mealtime insulin boluses during the 72-hour FCL period, compared to 56.3% [50.9, 64.0] with their home pump with mealtime insulin boluses during the at-home run-in week (+9.0%, P = .23). Overnight TIR was 95.9% [83.8, 100.0] for FCL versus 69.6% [57.6, 77.8] for the run-in period (+26.1%, P = .01). Time <70 mg/dL was low at 0.4% during FCL.
Conclusions:
FCL insulin delivery with the Tandem Freedom System was safe and effective in adults with T1D with high-carbohydrate, high-fat meals.
Keywords
Introduction
Hybrid closed-loop (HCL) systems have significantly improved glycemic outcomes in people of all ages with type 1 diabetes (T1D). 1 While some studies show that HCL systems can work well without mealtime insulin boluses for smaller meals, most studies on use of these systems without mealtime insulin boluses report either smaller amounts of carbohydrate intake or fail to report the amount of carbohydrate intake altogether.2-4 Results of studies of HCL systems used without mealtime insulin boluses for relatively higher carbohydrate intake, or presumed higher carbohydrate intake, often report significant amounts of hyperglycemia.4,5
A potential solution to use HCL systems without meal boluses for people with T1D is extensive, individualized customization of insulin delivery settings. Some studies have included adjustment of pump settings guided by regular meetings between research staff, up to weekly, to perform frequent personalized settings optimizations to account for missing meal boluses. 6 Others have tried to automatically adapt single components of the system for individual patients, hoping to personalize performance for optimal outcomes. 7 These approaches are not feasible outside of the research setting and lead to persistent diabetes burden by requiring frequent interaction with the pump and sensor. 8
A more robust solution is to employ a fully closed-loop (FCL) system, designed from the outset for use without meal announcement or mealtime insulin boluses. The Tandem Freedom system, an FCL insulin delivery system for people with diabetes, is such a system. It offers a number of features to reduce burden and minimize the need for user interaction, including but not limited to automated meal detection without any form of meal announcement, automated settings initialization and adaptation based on total daily insulin (TDI), 9 and sleep and exercise modes where automated meal detection is disabled.
This feasibility study aimed to evaluate the Tandem Freedom FCL system in adults with T1D with high-carbohydrate and high-fat unannounced meals.
Methods
Study Design
This single-center study was approved by the New Zealand Health and Disability Ethics Committee, and informed consent was obtained from each participant. The study was registered on ClinicalTrials.gov (NCT06834568).
After completing an initial feasibility trial in ten adults with T1D with 24 hours of FCL use (NCT06428591), this trial expanded FCL use to 72 hours and had no limits on meal sizes. Ten adults with T1D were recruited for the trial. Key inclusion criteria were: (1) age ≥18 years with diagnosis of T1D for at least one year, (2) current t:slim X2TM insulin pump with Control-IQTM technology (Control-IQ) user, having been prescribed Control-IQ for at least three months, and (3) HbA1c ≤10%, recorded in the last three months. Key exclusion criteria were (1) more than one episode of diabetic ketoacidosis (DKA) or severe hypoglycemia (needing assistance) in the past six months, (2) pregnant or planning pregnancy, (3) significant chronic kidney disease or hemodialysis, (4) history of adrenal insufficiency, or (5) a condition, which in the opinion of the investigator or designee, would put the participant or study at risk. Use of any insulin other than U-100 lispro or aspart was not allowed. Use of any noninsulin glucose-lowering agents other than metformin was also not allowed.
Participants first completed a 1-week run-in period with their personal Control-IQ pump and study-provided Dexcom G6 sensor at home. Participants changed out their G6 sensor 48 ± 12 hours before the hotel stay. Upon arriving at the hotel on day 1, participants bolused for lunch with their home pump, then switched to the Tandem Freedom system in the afternoon. The Freedom system was initialized with a single TDI value, determined by investigator review of the participant’s home pump download.
The 72-hour study analysis period for FCL use began at 6 a.m. day 2 and ended at 6 a.m. day 5. Participants were provided three meals per day. They were also allowed to have desserts with meals and extra snacks between meals. Macronutrient content of meals was calculated by a registered dietitian, but the calculated carbohydrate content was not used in any way by the Tandem Freedom FCL system. No insulin boluses were given for any meals or snacks. There was no meal announcement of any kind by the user. There was also no restriction on meal sizes.
Participants were also encouraged to participate in a daily exercise activity of at least 45 minutes in the late morning, followed by a supervised outing to different activities in the afternoon each day. These afternoon activities included bowling, mini-golf, and a zip line course in the forest.
Details of the Tandem Freedom System
For the study, the Tandem Freedom FCL algorithm v3.3.2 ran on a Tandem t:slim X2 insulin pump. Initialization of closed loop required only entering a value for estimated TDI, at which point the system automatically derived settings for basal rate, carbohydrate ratio, and correction factor. These closed-loop settings were stored internally in the pump and were not visible to the user. After a full 24-hour period of system use, at midnight each night these closed-loop profile settings were automatically adjusted by the system, as previously described by Shah et al. 9
Tandem Freedom is intended to be used as an FCL system, meaning without any form of meal announcement or user-initiated mealtime insulin boluses. While users could enter a meal bolus or a correction dose using a bolus calculator screen, the goal of this study was to use the system without any user interaction for meals. The system offered numerous features to differentiate it from HCL systems that are sometimes used without mealtime insulin boluses. First, the system had automatic meal detection, aiming to give a larger percentage of mealtime insulin dosing as close to the meal as possible. To reduce burden, no form of meal announcement was used in the study. Second, it derived closed-loop profile settings from TDI, which only needed to be entered once at system initiation, increasing ease of use. The derived settings were adapted every night at midnight automatically by the system and were not visible to the user, offering a simpler user experience. Tandem Freedom also included user-initiated sleep and exercise modes. Both modes disable automated meal detection while in use. In addition, exercise mode reduced the nominal insulin dose to prevent exercise-induced hypoglycemia. Sleep mode was scheduled to be active from 11 p.m. to 6 a.m. each night during the hotel stay. For participants that exercised, exercise mode was started 30 to 90 minutes before exercise as per investigator instructions to the participant for the late morning exercise period.
Statistical Analysis
The sample size of ten adult participants represented a convenience sample for the feasibility study and was not powered. The primary outcome of the study was safety, assessing for severe hypoglycemia, defined as hypoglycemia requiring assistance because of an altered cognitive state, and DKA defined by the criteria established in the Diabetes Control and Complications Trial. 10 Secondary outcomes for routine continuous glucose monitoring (CGM) metrics included time in range (TIR) of 70 to 180 mg/dL, time in tight range of 70 to 140 mg/dL, time above range of >180 mg/dL and >250 mg/dL, time below range of <70 mg/dL and <54 mg/dL, mean sensor glucose, sensor glucose standard deviation (SD), and sensor glucose coefficient of variation (CV), compared between the Control-IQ run-in week and the 72 hours of FCL use with Tandem Freedom in the hotel setting. Insulin use was also analyzed. A Wilcoxon signed-rank test for nonparametric data compared outcomes between periods.
Results
Demographics
Ten adults with T1D (30% female) enrolled and completed the study. Mean (SD) participant age was 38.6 (19.6) years, duration of diabetes was 15.9 (3.9) years, TDI was 0.71 (0.18) units/kg/d, body mass index (BMI) was 29.4 (5.2) kg/m2, and HbA1c was 7.3 (0.8) %. No participants were taking metformin or any other noninsulin glucose-lowering medication. Full demographics are listed in Table 1.
Participant Demographics (N = 10).

Safety
There were no DKA or severe hypoglycemia events.
Meal Sizes
During the 72-hour FCL period in the hotel study, median meal size for breakfast was 70.8 g carbohydrate, 36.7 g protein, and 53.2 g fat; for lunch 53.8 g carbohydrate, 31.4 g protein, and 40.0 g fat; and for dinner 96.1 g carbohydrate, 67.6 g protein, and 53.1 g fat. Median macronutrient intake per day was 230.5 g carbohydrate/d, 144.6 g protein/d, and 158.8 g fat/d, with participants consuming as much as 339 g carbohydrate/d, 189 g protein/d, and 275 g fat/d.
Glycemic and Insulin Outcomes
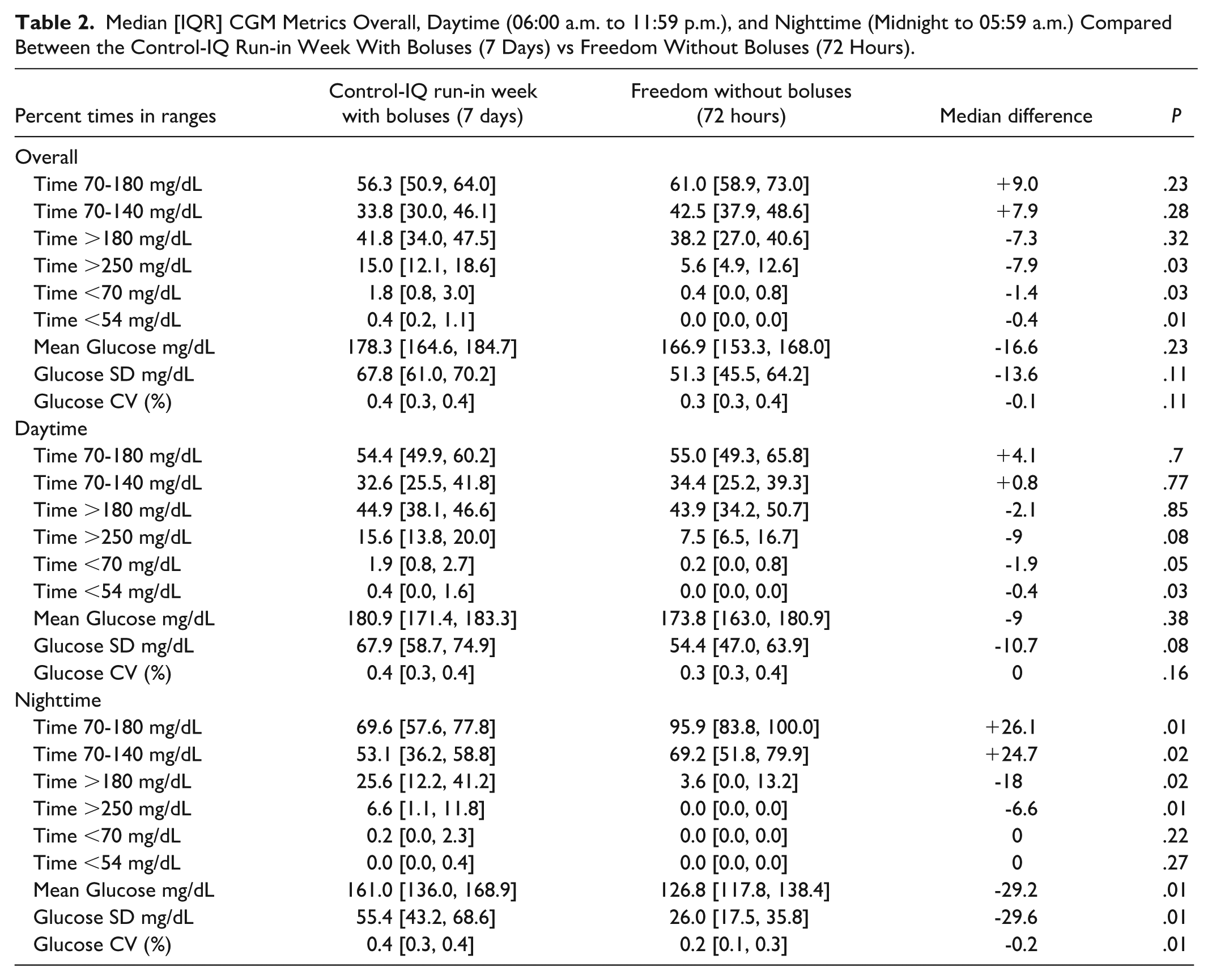
During the hotel study, FCL was active 97.3% [96.1, 97.7] of the time. Median TIR of 70-180 mg/dL was 61.0% [58.9, 73.0] without any mealtime insulin boluses during the 72-hour FCL period, compared to 56.3% [50.9, 64.0] with their home pump and a median of 3.0 [2.25, 4.5] user-initiated insulin boluses per day with carbohydrate entry during the at-home run-in week (+9.0%, P = .23). Overnight TIR was 95.9% [83.8, 100.0] for FCL vs 69.6% [57.6, 77.8] for the run-in period (+26.1%, P = .01). Hypoglycemia was significantly reduced with FCL use, with time <70 mg/dL 0.4% [0.0, 0.8] during FCL vs 1.8% [0.8, 3.0] during the run-in week (−1.4%, P = .03). Full CGM metrics for overall, daytime, and nighttime comparing the run-in week to the 72 hours of FCL use are listed in Table 2. Change in TIR per participant is shown in Figure 1.
Median [IQR] CGM Metrics Overall, Daytime (06:00 a.m. to 11:59 p.m.), and Nighttime (Midnight to 05:59 a.m.) Compared Between the Control-IQ Run-in Week With Boluses (7 Days) vs Freedom Without Boluses (72 Hours).
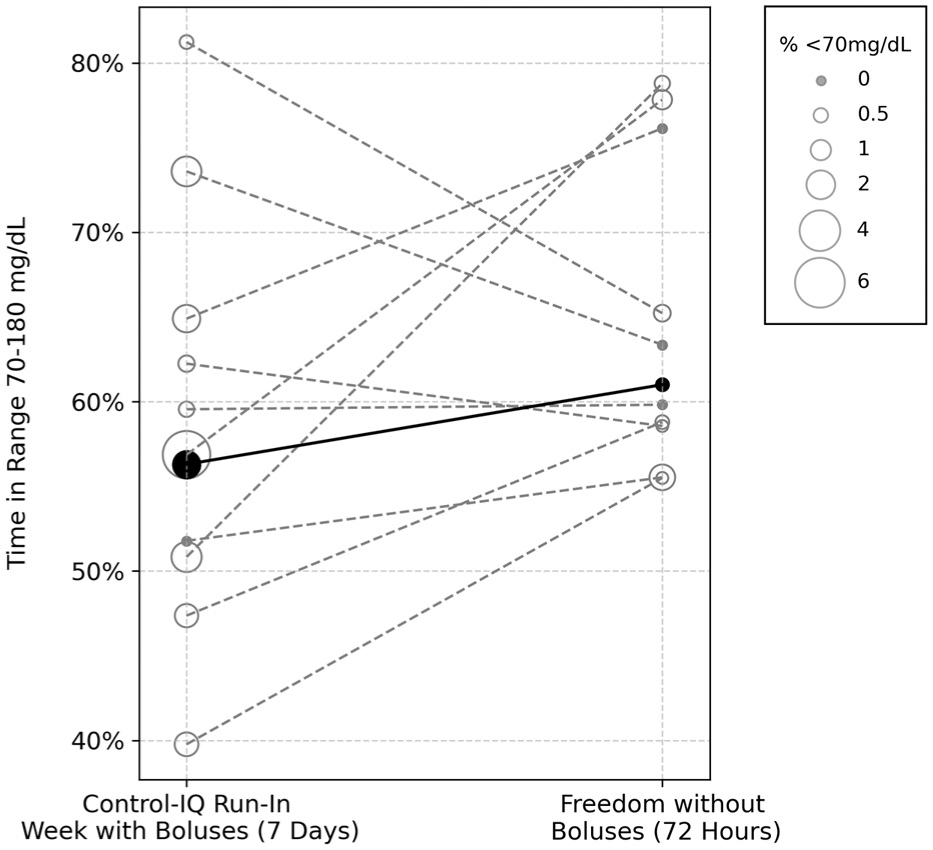
Change in time in the range of 70 to 180 mg/dL per participant, comparing the control-IQ run-in week with mealtime insulin boluses (7 days) vs freedom without mealtime insulin boluses (72 hours). The black line represents the median change in time in range, which improved from 56.3% with control-IQ to 61.0% with Tandem freedom. The size of each circle represents the time in hypoglycemia, which was significantly reduced with Tandem freedom.
Total daily insulin use was significantly reduced in the FCL period, from 0.9 [0.7, 1.1] units/kg/d during the run-in period to 0.6 [0.4, 0.7] units/kg/d with FCL use (−0.4 units/kg/day, P = .02).
Median cumulative postprandial insulin delivery by hour is shown in Table 3. The system delivered up to 80% of the 4-hour total postprandial insulin in the first 2 hours after breakfast.
Median Cumulative Postprandial Insulin Delivery After Unbolused Meals With the Tandem Freedom System.
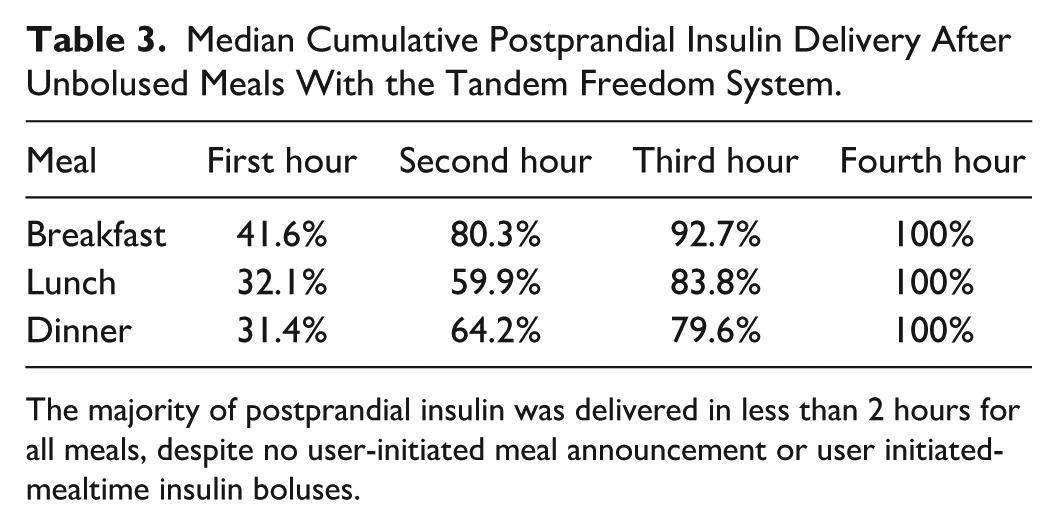
The majority of postprandial insulin was delivered in less than 2 hours for all meals, despite no user-initiated meal announcement or user initiated-mealtime insulin boluses.
Conclusion
While HCL systems have achieved excellent results in people with all forms of diabetes,11,12 they retain the burden of requiring some form of user-initiated meal announcement to achieve optimal results. Early studies evaluated closed-loop performance in people with T1D with a single missed meal boluses in a controlled setting. 13 Subsequently, most studies of individuals with T1D using HCL systems in a manner consistent with FCL therapy, namely instructing patients not to bolus for meals, generally do not achieve ideal results, especially when consuming larger meals.2,5
For an FCL system to be effective, it must deliver insulin as close to the meal as possible. In addition, these systems need to be easy to setup and use. Finally, it is important they limit hypoglycemia as well, as giving too much insulin can lead to postprandial hypoglycemia, or hypoglycemia with exercise.
Our results show that use of the Tandem Freedom FCL system, without any form of user-initiated meal announcement or user-initiated mealtime insulin boluses, achieved over 60% TIR in adults with T1D despite some participants consuming over 300 g carbohydrate and over 250 g fat per day, with very little hypoglycemia. Participants also received less insulin than they normally did at home. Prior studies have shown that missed and mistimed insulin doses contribute to higher HbA1c and lower TIR. 14 It may be these mistimed and missed doses also contribute to excessive insulin delivery at the wrong times, contributing to hypoglycemia, especially if user-initiated boluses are given after meals. The advantage of an FCL system is the user does not have to intervene, and the system is designed to gradually bring down glucose levels without causing postprandial hypoglycemia.
We found TIR was slightly higher than that achieved in another recent study of FCL used without meal boluses in people with T1D. 15 In that study, no meal size information was reported. In our study, even with very large meals, the system was able to give the majority of insulin dosing close to the mealtime, with up to 80% of the postprandial insulin delivered within the first 2 hours after breakfast, which is often the most challenging meal of the day. It will be very important to understand how FCL systems work with large meals, as these systems need to work in real-life situations that are not constrained by research protocols.
Strengths of the study include the large meal sizes and having daily activities and exercise to challenge the system. Limitations include it was a small feasibility study in a hotel setting, with participants who were well experienced in use of diabetes technology. Baseline A1c was 7.3%, suggesting this population had above average glycemic control, so the results may not generalize to all populations. Because of the shorter duration of the study, it was not possible to determine the full impact of automated settings adaptation that was built into the system. That will require a longer follow-up period. We also did not collect a food or exercise log during the run-in period, so we could not definitively compare meal sizes or activity levels between the two time periods. Finally, the study only had participants use insulin lispro or insulin aspart, whereas other recent studies have examined the use of ultra-rapid insulin analogs with FCL. 16
In conclusion, use of the Tandem Freedom FCL system in adults with T1D, without user-initiated meal announcement of any kind or mealtime insulin boluses, showed very promising results in this supervised hotel study. The system was safe to use, even with high-carbohydrate, high-fat meals. Future studies will include longer periods of system use in the outpatient setting to allow for the daily adaptation to take effect over weeks to months, larger sample size, patient-reported outcome surveys, and different populations to include children with T1D and adults with type 2 diabetes, as they all will likely benefit from FCL insulin delivery.
Footnotes
Acknowledgements
We would like to acknowledge Alex Wheatcroft, Nicole Piquette, Karen Carmelo, Kirstin White, Peter Zhao, Larry Fu, Jared Villanueva, Chantal Condon, and Mike Michaud for their assistance with this project.
Abbreviations
CGM, continuous glucose monitoring; Control-IQ, t:slim X2 insulin pump with Control-IQ technology; DKA, diabetic ketoacidosis; FCL, fully closed loop; HCL, hybrid closed loop; TIR, time in range; TDI, total daily insulin; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TMW, MIdB, RM, and SH received research funding to their institution from Tandem Diabetes Care, Inc. RS-K, AT, JRR, NS, MS, BM, DG, SR, EWD, PB, JPC, TRU, and JEP are employees of Tandem Diabetes Care, Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Tandem Diabetes Care, Inc.
Prior publication
Data from this study was presented in abstract form at the Advanced Technologies & Treatment for Diabetes—Asia (ATTD Asia) meeting in December 2025.
