Abstract
Background:
This feasibility study assessed a novel self-adapting closed-loop system which does not require carbohydrate announcement, in adults with type 1 and type 2 diabetes.
Methods:
Single-arm study, comprising a 14-day run-in using participants’ usual insulin therapy with a blinded continuous glucose monitor (CGM), followed by 12 weeks use of the novel closed-loop system. The algorithm adjusted its own parameters after 4, 6, 8, and 10 weeks of use.
Results:
Thirty-two participants with type 1 and 10 participants with type 2 diabetes were enrolled. Mean time in range (TIR; % CGM readings = 70-180 mg/dL) was 37.7% at baseline and 55.9% during the intervention period in type 1 diabetes; 17.6% at baseline and 51.5% during the intervention period in type 2 diabetes. Median time <70 mg/dL during the intervention period was 1.1% in type 1 and 0.0% in type 2 diabetes. Median TIR was 65% following the fourth algorithm adaptation. Median daily insulin delivered by manual bolus was 1.0 units in type 1 and 0.0 units in type 2 diabetes, consistent with no meal announcement. There were four serious adverse events: worsening retinopathy, severe hypoglycemia following a period of paused automation, and two hospitalizations unrelated to the device.
Conclusions:
A closed-loop algorithm that adjusts its own parameters and requires no meal announcement was feasible in a cohort of adults with type 1 and type 2 diabetes. Clinical benefits were most apparent with the fully adapted algorithm.
Keywords
Introduction
Despite significant advances in automated insulin delivery (AID) systems, people living with type 1 diabetes continue to face significant burden associated with diabetes management, including that associated with carbohydrate announcement prior to meals. Multiple AID systems have demonstrated glycemic benefit when compared to sensor-augmented pump therapy in large randomized controlled trials;1-6 however, participants in those trials were instructed to announce carbohydrate intake. These systems have been consequently termed “hybrid closed-loop.”
The utility of carbohydrate announcement in AID systems has a basis in physiology and pharmacology. Continuous glucose monitors (CGMs) measure interstitial glucose, which may lag behind blood glucose by several minutes. 7 Current “rapid-acting” insulin analogues have a relatively slow onset when delivered subcutaneously, with peak activity 30 to 60 minutes after administration, 8 while “ultra-rapid-acting” insulin preparations have demonstrated only modest pharmacokinetic improvement.9,10 Therefore, delivery of a manual insulin bolus prior to planned carbohydrate consumption to pre-emptively prevent postprandial hyperglycemia would be expected to result in tighter glycemic control than an AID system only able to deliver additional insulin once the postprandial hyperglycemia has occurred.
However, a continued requirement for carbohydrate announcement when using AID systems may be perceived as a barrier to uptake by many people with diabetes. 11 Data from the US type 1 diabetes exchange registry shows only a modest increase in insulin pump uptake following the introduction of AID systems. 12 Furthermore, some patient groups (particularly adolescents) are known to use AID and not consistently announce carbohydrates. 13 There is therefore a pressing need for AID systems that are operable as “fully closed-loop,” without carbohydrate announcement.
Although the efficacy of AID in type 1 diabetes is now clearly established, the majority of people with diabetes globally have type 2 diabetes. Despite the introduction of incretin-based therapies and sodium/glucose co-transporter 2 (SGLT-2) inhibitors, a subset of people with type 2 diabetes still require complex insulin regimens with multiple daily injections. Studies have demonstrated feasibility of AID in this cohort.14-17 It is therefore important that trials of new AID systems incorporate people with both type 1 and type 2 diabetes.
This first-in-human feasibility study investigated the safety and efficacy of a novel self-adapting AID system designed to be used without carbohydrate announcement, in adults with type 1 and type 2 diabetes.
Methods
This was a single-arm feasibility study conducted in New Zealand at four clinical sites. The protocol was approved by the Northern A Health and Disability Ethics Committee of New Zealand (2021 FULL 11513). The study was registered with the Australian New Zealand Clinical Trials Registry (ACTRN12622000190707).
Recruitment aimed to include 30 participants with type 1 diabetes and 10 participants with type 2 diabetes requiring insulin. Key inclusion criteria were (1) initial diabetes diagnosis >6 months prior to the screening visit; (2) aged 16 years or older; (3) no prior use of an AID system (prior use of an insulin pump was permitted); (4) mean HbA1c >7.5% (58 mmol/mol) based on available results within 6 months prior to the screening visit; and (5) basal insulin requirement 9.6-96 units per 24 hours (equivalent to basal rates of 0.4 to 4.0 units/h, as permitted by the investigational AID system), determined from basal rate(s) if an existing insulin pump user or basal insulin dose if using multiple daily insulin injections. Key exclusion criteria were (1) pregnancy; (2) alcohol or drug dependence; (3) severe visual impairment; and (4) any other medical or psychological factors that would, on assessment by the investigators, make the person unsuitable for the study.
Eligibility criteria did not specify any requirements regarding non-insulin diabetes medications; however, investigators aimed to only enroll participants with type 2 diabetes once non-insulin medications had been optimized. This was defined as regular use (if tolerated) of metformin, an SGLT-2 inhibitor and/or a glucagon-like peptide 1 (GLP-1) receptor agonist, titrated to the maximum tolerated dose, and any other non-insulin medications felt to be clinically indicated. Investigators liaised with the usual health care providers of potential participants to enable medication changes where necessary, prior to study enrollment.
Recruitment of participants with type 2 diabetes prioritized those of Māori or Pacific Island ethnicities, recognizing that these groups suffer significant diabetes-related inequities in health outcomes in New Zealand. 18 Recruitment of participants additionally prioritized people aged 60 years or older with the aim of achieving a wide range of ages in the study cohort.
All participants provided written consent for participation. Once enrolled, participants attended a baseline assessment visit and commenced blinded CGM monitoring using a Dexcom G6 CGM (Dexcom Inc., San Diego, California), used for 14 days while continuing their usual insulin therapy. Participants already using CGM were permitted to continue their personal (unblinded) CGM during this period. CGM data capture >75% was required, and blinded CGM was continued beyond 14 days if needed until sufficient data were obtained.
Participants then proceeded to use of the investigational AID system for 12 weeks. The system comprised a mylife YpsoPump insulin pump with Orbit micro or Orbit soft infusion sets (Ypsomed, Burgdorf, Switzerland), Dexcom G6 CGM, and the investigational algorithm (inControl Auto) located within a modified version of the Dexcom G6 app, installed on an Android smartphone which was paired (via Bluetooth) with both the pump and CGM.
The AID system was initiated with a single basal rate and 2-hour maximum insulin delivery limit. Investigators chose initial settings based on participants’ usual insulin requirements and review of run-in CGM data. The basal rate could be freely changed within the first 14 days of the intervention period; the 2-hour maximum insulin delivery limit could be freely changed at any time.
The AID algorithm was designed to minimize user interaction. The algorithm is modular, with distinct mitigations for hypoglycemia (via attenuation of basal insulin) and for hyperglycemia (via intensification of basal insulin), and uses proprietary signal processing techniques to estimate the user’s metabolic state (glucose and insulin in various compartments), to detect meals, and enable more aggressive treatment of hyperglycemia accordingly. In addition, the algorithm was adaptive, responding (via a static formula) to the user’s experience of out-of-range sensor glucose, incorporating scheduled automated individualization of a parameter with a role analogous to that of the insulin sensitivity factor used to calculate correction boluses in conventional therapy. Adaptation first occurred following 28 days’ use and was repeated every 14 days thereafter. Each adaptation required >75% CGM data capture and no change to the programmed basal rate in the 14 days prior. Participants and investigators could not override the adaptation schedule nor could they manually change the therapy parameter adjusted by the adaptation.
Participants received training regarding the AID system and insulin pump use, including management of hypoglycemia, and recognition and management of AID system and infusion set failure. The insulin pump was programmed with a backup basal insulin delivery profile, which was delivered if the pump did not otherwise receive insulin dosing instructions from the study smartphone. Participants were able to deliver manual insulin boluses by interacting directly with the pump, which could be used if there was an AID system failure; if in automation, they were instructed to deliver a manual insulin bolus if CGM glucose was >400 mg/dL (>22.2 mmol/L), but to avoid delivering manual insulin boluses otherwise.
The study smartphone was configured to upload CGM recordings to Dexcom Clarity, allowing investigators to remotely review recordings in real time. Participants received training regarding CGM use, including the setup of CGM alarms, which were individualized for each participant. During the first seven days of the intervention phase, investigators reviewed CGM recordings daily and altered the basal rate if time >270 mg/dL was >30% or time <70 mg/dL was >4%, or as per investigator discretion otherwise. Investigators then reviewed CGM recordings at least twice in the second week of the intervention phase and at least once per week thereafter. Investigators did not receive CGM alerts outside of these scheduled reviews; however, participants were able to contact a member of the study team at any time if they had concerns.
As this was a feasibility study and was not powered to reach conclusions regarding glycemic efficacy, there was no pre-defined primary outcome or pre-defined comparative statistical analysis. Outcomes included HbA1c at baseline and study end; CGM metrics (% time in range [TIR] 70-180 mg/dL, % time in tight range 70-140 mg/dL, % time <70 mg/dL, % time <54 mg/dL, % time >180 mg/dL, % time >250 mg/dL, mean sensor glucose, glucose variability measured by coefficient of variation [CV], glycemia risk index [GRI]); 19 insulin delivery metrics (total daily dose, percentage of daily insulin delivered by manual bolus); body mass index (BMI) at baseline and study end; and any adverse events. Overall outcomes were reported separately for type 1 and type 2 diabetes.
In a secondary analysis to investigate the effect of algorithm adaptations, data from type 1 and type 2 diabetes were combined and stratified by the number of sequential adaptations that had occurred. This secondary analysis only incorporated data from a “per protocol” population with sufficient CGM capture, defined as participants where (1) the participant had ≥10 days with ≥70% CGM data capture following the relevant adaptation; (2) daily mean time spent in automation was >90% across the period of interest; and (3) daily mean of the proportion of insulin delivered as manual boluses was <10% across the period of interest.
Adverse events were only reportable if related to the use of the investigational medical device (adverse device effect) or if serious (resulting in death, life-threatening illness or injury, permanent impairment of a body structure or body function, or required medical or surgical intervention to prevent any of the above, or otherwise resulted in hospitalization). Hypoglycemia was only reportable as an adverse event if severe, defined as requiring the assistance of another person due to altered consciousness to actively administer carbohydrate, glucagon, or other resuscitative actions; this definition did not include any specified blood glucose value. Hyperglycemia was only reportable as an adverse event if severe, defined as blood glucose >300 mg/dL (>16.7 mmol/L) with blood ketones >1.5 mmol/L.
Results
Thirty-two participants with type 1 diabetes and 10 participants with type 2 diabetes enrolled in the study between March 2022 and October 2023. Baseline data are presented in Table 1. All participants with type 2 diabetes were prescribed an SGLT-2 inhibitor and/or a GLP-1 receptor agonist at enrollment.
Baseline Characteristics.
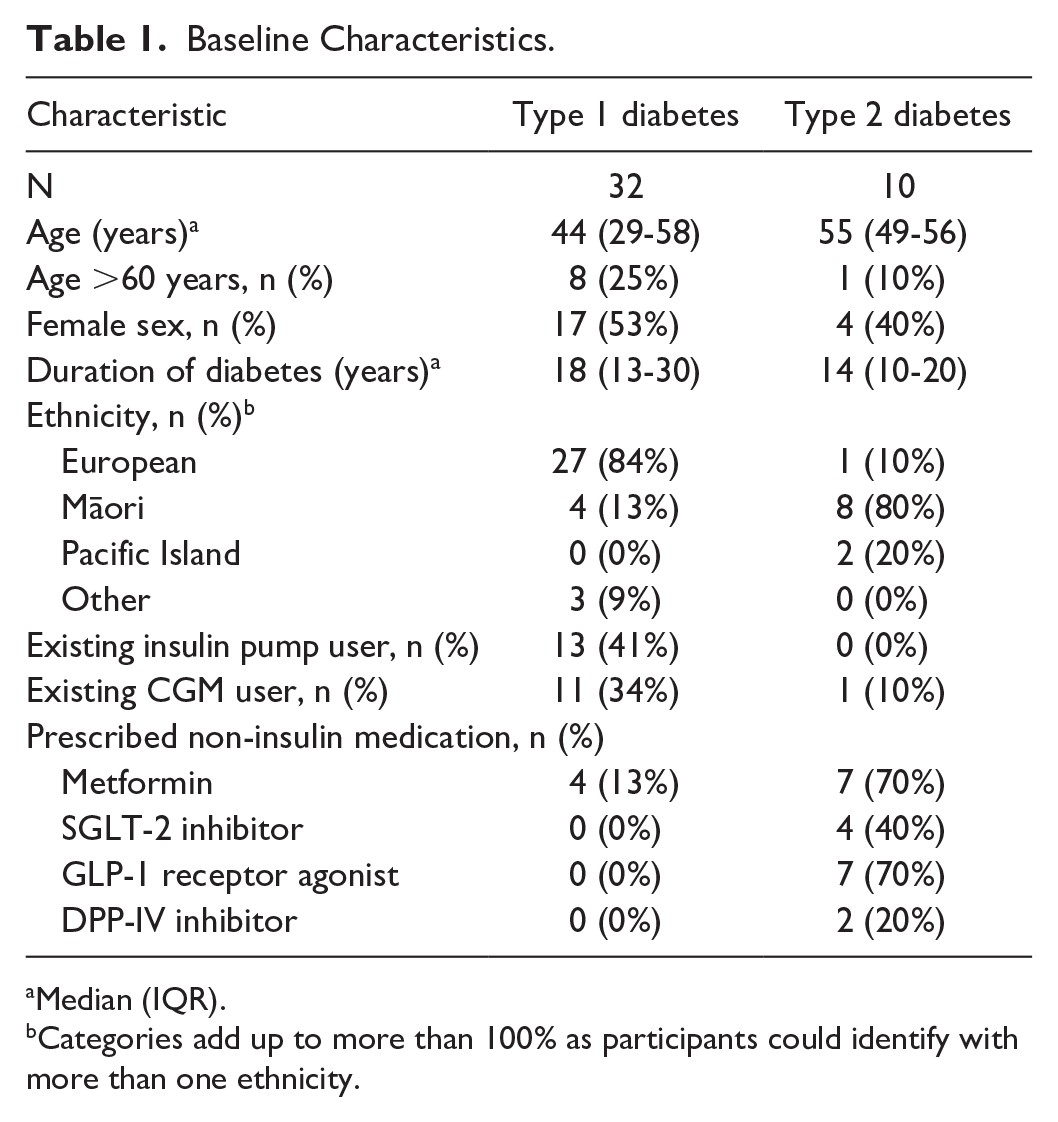
Median (IQR).
Categories add up to more than 100% as participants could identify with more than one ethnicity.
Overall outcome data are presented in Table 2. The CGM metrics incorporate all available CGM recordings, including periods not in automation. Overall median time in automation was 98% (interquartile range [IQR] = 95%-99%). Mean % CGM TIR for the intervention period was 55.9% (standard deviation 8.0%) in type 1 diabetes and 51.5% (standard deviation 25.2%) in type 2 diabetes. Median HbA1c at study completion was 7.5% (59 mmol/mol) (IQR = 7.3%-8.0%, 56-64 mmol/mol) in type 1 diabetes and 7.6% (60 mmol/mol) (IQR = 7.2%-8.7%, 55-72 mmol/mol) in type 2 diabetes. Figure 1 presents baseline and intervention TIR and HbA1c for each individual participant, demonstrating improved metrics for many participants with suboptimal baseline glycemia.
Overall Outcome Data.
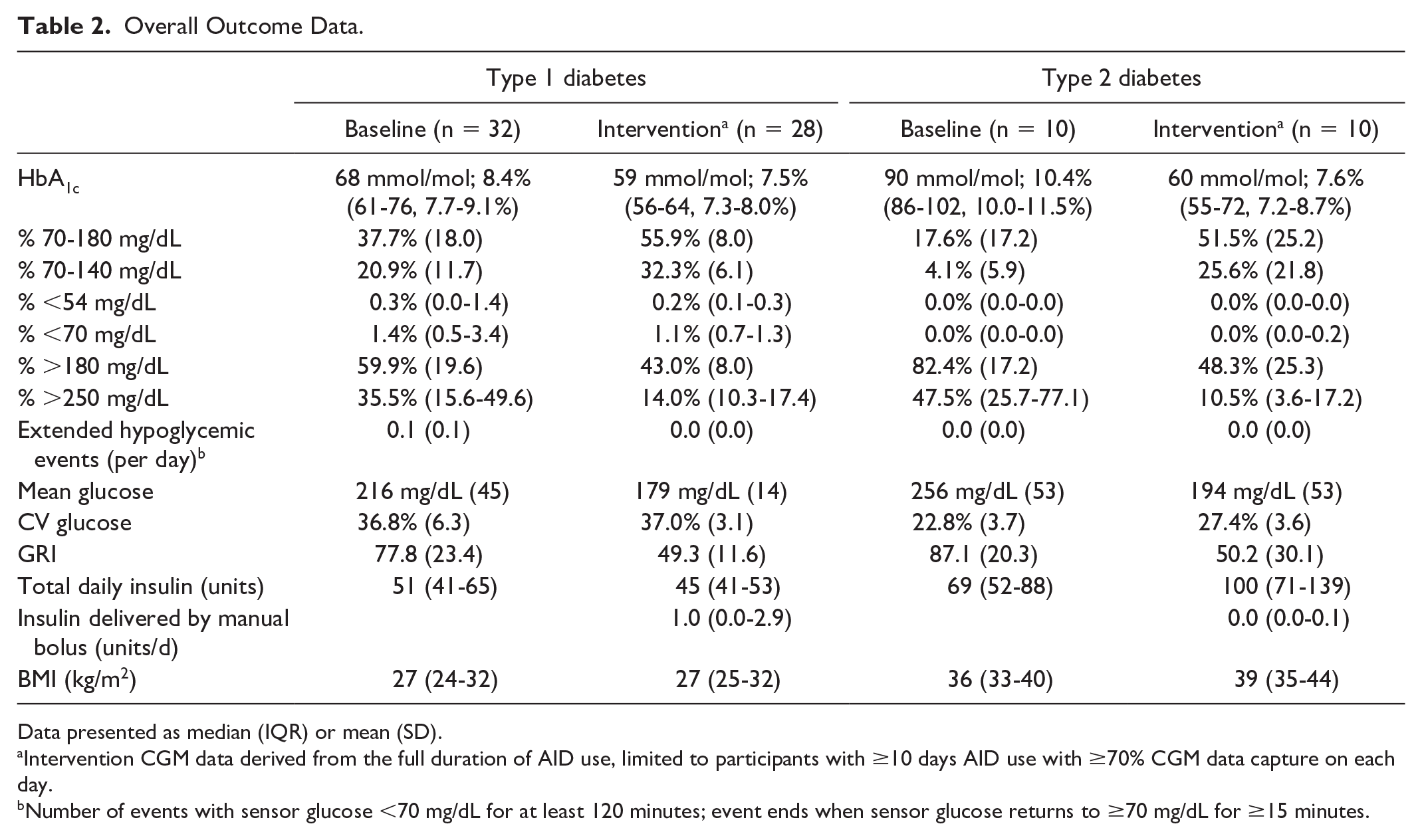
Data presented as median (IQR) or mean (SD).
Intervention CGM data derived from the full duration of AID use, limited to participants with ≥10 days AID use with ≥70% CGM data capture on each day.
Number of events with sensor glucose <70 mg/dL for at least 120 minutes; event ends when sensor glucose returns to ≥70 mg/dL for ≥15 minutes.

Baseline and study intervention TIR and HbA1c.
Total daily insulin dose and BMI were similar between baseline and intervention periods in participants with type 1 diabetes. In participants with type 2 diabetes, median total daily insulin dose was 69 units (IQR = 52-88 units) at baseline and 100 units (IQR = 71-139 units) during the intervention period; median BMI was 36 kg/m2 (IQR = 33-40) at baseline and 39 kg/m2 (IQR = 35-44) at study end. Consistent with the use of the AID system as “fully closed-loop,” median insulin delivered by manual bolus was 1.0 units (IQR = 0.0-2.9) in type 1 and 0.0 units (IQR = 0.0-0.1) in type 2 diabetes.
Participants could undergo up to four algorithm adaptations during AID use. Table 3 and Figure 2 present CGM metrics stratified by number of preceding adaptations. Median TIR was 56% prior to any adaptation, 70% following the third adaptation, and 65% following the fourth adaptation.
CGM Data Stratified by Number of Preceding Algorithm Adaptations.
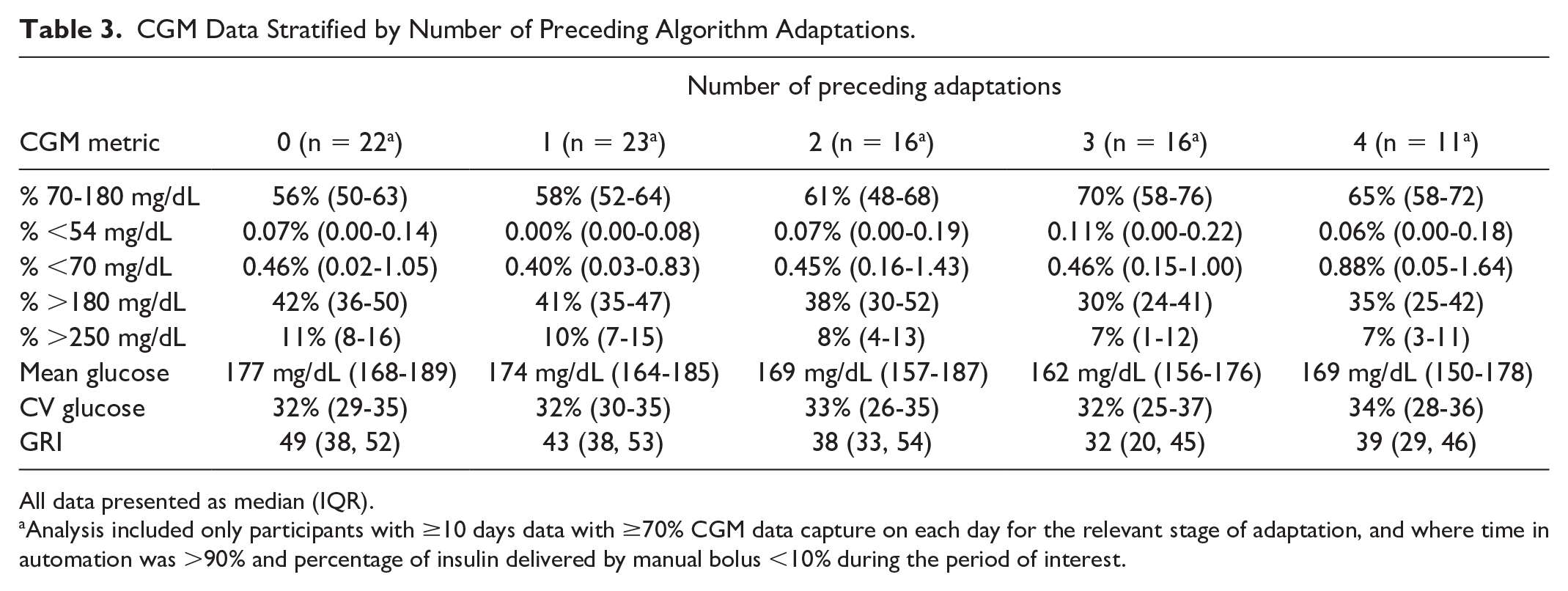
All data presented as median (IQR).
Analysis included only participants with ≥10 days data with ≥70% CGM data capture on each day for the relevant stage of adaptation, and where time in automation was >90% and percentage of insulin delivered by manual bolus <10% during the period of interest.
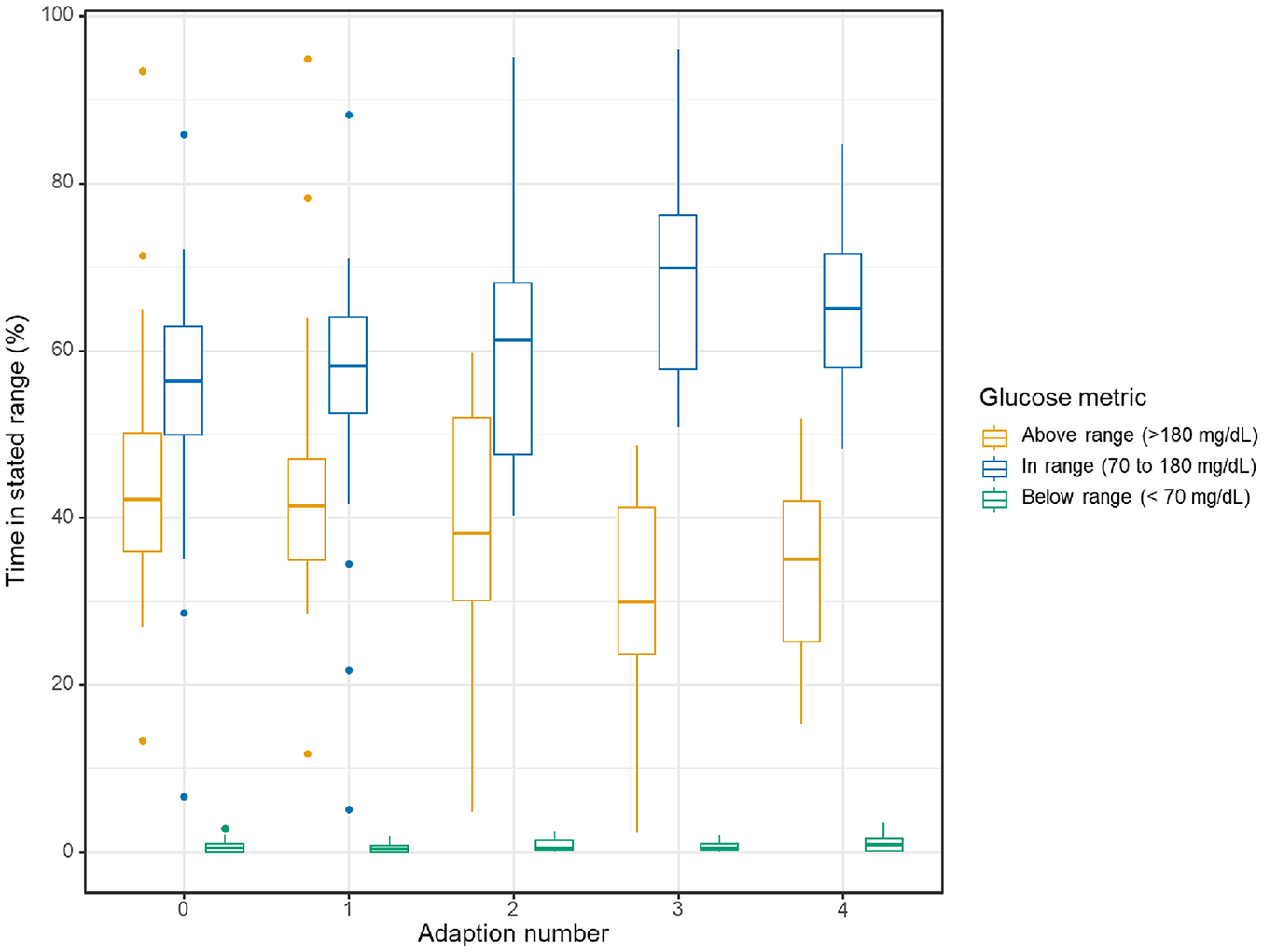
Key CGM metrics by number of preceding algorithm adaptations. Plots present median (IQR). Analysis included only participants with ≥10 days data of with ≥70% data capture on each day for the relevant stage of adaptation and where time in automation was >90% and percentage of insulin delivered by manual bolus <10% during the period of interest.
Four serious adverse events occurred. One participant with type 1 diabetes and baseline HbA1c 10.1% (87 mmol/mol) was found to have worsening macular edema following commencement of AID. Macular edema resolved with ophthalmology treatment and cessation of AID. One participant had an episode of severe hypoglycemia, which occurred after switching automation off for several hours overnight, resulting in hyperglycemia for which a large automated bolus was delivered when automation was reactivated, on waking in the morning. On analysis of algorithm data, it was thought unlikely that the severe hypoglycemia would have occurred if the participant had remained in automation throughout the night prior, as the hyperglycemia prompting the automated bolus would have been unlikely to develop. Two participants had unplanned hospital admissions; however, both occurred during the blinded CGM run-in phase and were therefore unrelated to the study AID system.
Three participants were withdrawn by investigators due to the above serious adverse events. A further four participants chose to withdraw due to device intolerance, with stated reasons for withdrawal including dislike of wearing an insulin pump, dislike of AID system alerts, and difficulty trusting the AID system. All four of these participants had not previously used an insulin pump. No participants with type 2 diabetes withdrew from the study. Data from participants who withdrew were included in analyses.
Duration of AID use was 0 to 56 days in participants who withdrew and 75 to 112 days in participants who completed the study intervention. The range in AID duration reflected a software error affecting the first 13 participants to enroll, which resulted in periods of time not using AID and in a need for multiple resets of the adaptation schedule. Investigators extended the duration of intervention for these participants on an individual basis, to compensate for periods of time off AID and to facilitate further adaptations. The software error was resolved mid-study and did not affect the remaining participants; no adverse events were attributed to the software error.
Discussion
In this first-in-human feasibility study, use of a novel fully automated AID system by adults with type 1 and type 2 diabetes resulted in improved CGM TIR and HbA1c for a number of individual participants, particularly those with suboptimal glycemia at baseline and those able to proceed through automated adaptations incorporated into the system.
Two serious adverse events related to system use occurred: one (worsening diabetic maculopathy) is a known risk of rapidly improved glycemia following commencement of AID, 20 while the second (severe hypoglycemia) was attributable to the system being out of automation for several hours prior.
Four participants chose to withdraw from the study, citing intolerance of the devices used. All were pump-naïve. Elevated HbA1c has been shown to be a predictor of pump hesitancy and AID discontinuation;21,22 therefore, it is not necessarily surprising that some participants in an AID-naïve cohort with elevated HbA1c encountered device intolerance. All participants with type 2 diabetes completed the full study, emphasizing that clinicians should not regard this group as being unsuited to new technologies.
The AID system used in this trial (inControl Auto) was specifically designed to require minimal user input, requiring a single basal rate as the only adjustable parameter and containing no option within the app interface to announce carbohydrates. The constraints on user input may be perceived as a system limitation; however, they may also increase the appeal of the system for some users. Previous studies have demonstrated that Control-IQ, MiniMed 780G, CamAPS, and AndroidAPS, despite being developed as hybrid closed-loop, may have utility as fully closed-loop systems.16,23-25 However, the relative simplicity of the inControl Auto interface may be perceived favorably by users who have not previously started an AID system when compared to systems initially designed to be used with carbohydrate announcement.
Although AID systems are an established component of care for people with type 1 diabetes, use of AID in people with type 2 diabetes is an emerging area. In this study, substantial improvements in glycemia were observed in several participants with type 2 diabetes. This occurred in association with an increase in total daily insulin dose and an increase in BMI.
Weight gain is a well-described complication of intensification of insulin therapy in type 2 diabetes: in the pivotal UKPDS trial, participants randomized to intensive control with insulin and sulfonylureas had a significantly reduced risk of microvascular endpoints but also a significant increase in weight. 26 The specific impact of AID on weight in type 2 diabetes is less well described. A trial of Control-IQ in type 2 diabetes reported an adjusted group difference (compared to CGM alone) of 0.6 kg/m2 BMI after 13 weeks of use, in a cohort with higher baseline TIR (mean 48 ± 24%) than seen in this trial, and no increase in total daily insulin attributable to AID. 27 A trial of Omnipod 5 showed no change in weight associated with the commencement of AID in type 2 diabetes; however, total daily insulin also did not increase. 14 Two trials have investigated CamAPS in type 2 diabetes: one included only participants with end-stage renal failure requiring dialysis (complicating any assessment of weight change), 15 while the second did not report weight as an outcome. 16
The association between insulin therapy and weight gain may favor non-insulin agents in type 2 diabetes; however, in this study, participants all had an elevated baseline HbA1c despite optimization of non-insulin therapies including GLP-1 receptor agonists and SGLT-2 inhibitors. For many, there was an urgent need to intensify glycemic control even if this would result in weight gain. Although incretin-based therapies are demonstrating increasing benefit in type 2 diabetes, 28 it is likely that there will remain a subset of people with type 2 diabetes who require complex insulin regimens to achieve glycemic targets, for whom fully closed-loop systems with simple user interfaces may be of benefit.
This study had important limitations. By design it was a single-arm first-in-human feasibility study not powered for comparative statistical analyses, and therefore cannot be used to reach conclusions regarding overall glycemic efficacy. Significant interruptions to AID use occurred due to a software issue, which, although resolved, resulted in heterogeneity in the duration and nature of AID use by affected participants. However, the study results are sufficiently promising to justify a prospective randomized controlled study to assess the efficacy and safety of the AID system.
An important strength of this study was recruitment of a “high-risk” cohort. Eligibility was limited to participants with HbA1c >7.5% (>58 mmol/mol), resulting in a high baseline median HbA1c. Participants were AID naïve, and only a minority were existing users of insulin pumps or CGM. Recruitment of participants with type 2 diabetes focused on ethnicities (Māori and Pacific Island) which face substantial inequities in diabetes outcomes. 18 The results need to be interpreted in this context. Although many individual participants did not reach the consensus TIR target of >70%, 29 this may reflect the study cohort rather than the investigational AID system. A recently published trial, also conducted in New Zealand in a “high-risk” cohort, used the MiniMed 780G advanced hybrid closed-loop system. 30 In that study, intervention mean TIR was 57%, representing a significant glycemic benefit compared to a control group but also not meeting the consensus target, likely due to inaccurate carbohydrate counting and late or missed meal boluses (a well-described behavior in “high-risk” populations using insulin pump therapy). 31 In contrast, MiniMed 780G reached mean TIR of >70% in other trials.1,32 The failure of multiple AID systems to achieve TIR of >70% in people with suboptimal glycemia emphasizes the importance of ongoing development of AID systems suited to this cohort. It is promising that median TIR was 70% (IQR = 58-76) after the third algorithm adaptation. Although median TIR was 65% (IQR = 58-72) after the fourth algorithm adaptation and this feasibility study was not powered for comparative statistical analysis, it is possible that an updated version of the study AID system, able to progress through more adaptations, may assist in achieving glycemic targets.
Conclusions
In this first-in-human feasibility study, a novel closed-loop algorithm that adjusts its own parameters and requires no meal announcement demonstrated feasibility in a cohort of adults with type 1 and type 2 diabetes. Clinical benefits were most apparent with the fully adapted algorithm, and larger studies are justified to assess glycemic efficacy in adults able to progress through algorithm adaptations. Ongoing development of fully closed-loop systems is likely to be of particular benefit in populations with historically low uptake of diabetes technologies or who are known not to announce carbohydrates with conventional hybrid closed-loop systems.
Footnotes
Acknowledgements
The authors would like to acknowledge the support of Ypsomed, who provided insulin pumps and consumables for the study and review of the manuscript, and Dexcom Inc, who provided phones and CGM for the study, technical support, and review of the manuscript.
Abbreviations
AID, automated insulin delivery; BMI, body mass index; CGM, continuous glucose monitor; GLP-1, glucagon-like peptide 1; SGLT-2, sodium/glucose co-transporter 2; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Martin de Bock, Ryan Paul, and Benjamin Wheeler have received honoraria from Dexcom Inc. Martin de Bock and Ryan Paul are current members of the New Zealand Advisory Board for Dexcom Inc. Enrique Campos-Náñez and Steve Patek are employees of Dexcom Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study funding was provided by Dexcom Inc. Dexcom Inc and Ypsomed provided the devices used in the study. Neither company was involved in the data collection or data analysis.
