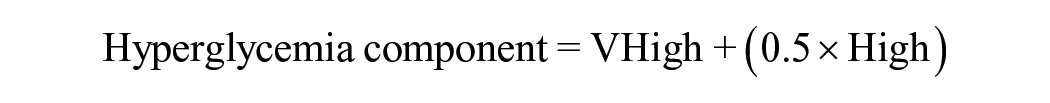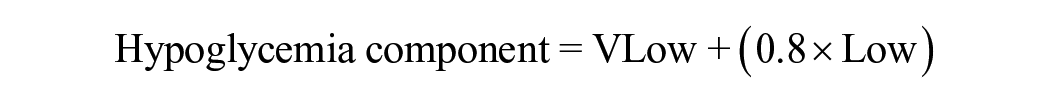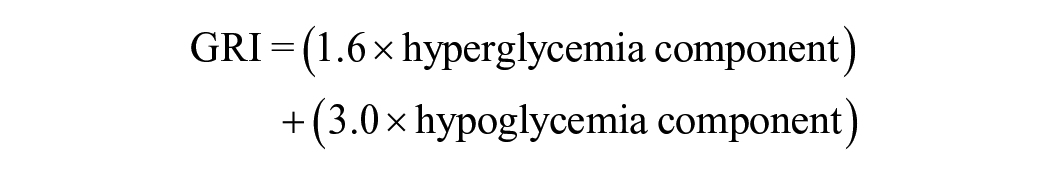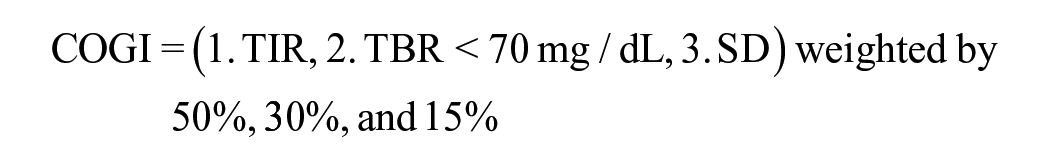Abstract
Background:
The glycemia risk index (GRI) is a novel composite continuous glucose monitoring (CGM) metric composed of hypoglycemia and hyperglycemia components and is weighted toward extremes. This study aimed to investigate the association between GRI and the risk of albuminuria in type 1 diabetes.
Methods:
The 90-day CGM tracings of 330 individuals with type 1 diabetes were included in the analysis. Glycemia risk index was divided into five risk zones (A-E), and hypoglycemia and hyperglycemia components were divided into quintiles. Albuminuria was defined as a spot urine albumin-to-creatinine ratio ≥30 mg/g. Associations of albuminuria with GRI and its hypoglycemia and hyperglycemia components were estimated.
Results:
Mean GRI and glycated hemoglobin (HbA1c) were 40.9 ± 21.3 and 7.3 ± 1.0%, respectively, and the overall prevalence of albuminuria was 17.6%. Prevalence of albuminuria differed significantly by GRI zone (P = .023). In logistic regression analysis, the adjusted odds ratio (OR) of albuminuria per increase in the GRI zone was 1.70 (95% confidence interval [CI]: 1.19-2.41) after adjusting for various factors affecting albuminuria. The association remained significant after adjusting for achievement of the recommended target of time in range (70-180 mg/dL; >70%) or HbA1c (<7%). The hyperglycemia component of GRI was also associated with albuminuria, and the association remained significant even after adjusting for HbA1c level itself (adjusted OR 1.44, 95% CI: 1.05-1.98).
Conclusions:
Glycemia risk index is significantly associated with albuminuria in individuals with type 1 diabetes.
Keywords
Introduction
Continuous glucose monitoring (CGM) is standard clinical practice for insulin users with diabetes.1,2 Several randomized clinical trials have shown that CGM can help glycemic control, including reducing hypoglycemia episodes and glycated hemoglobin (HbA1c) levels.3-17 Continuous glucose monitoring enables clinicians to comprehensively assess glycemic quality by providing information about glycemic variability and proportions of time in hyperglycemia, euglycemia, and hypoglycemia.18,19
Glycemia risk index (GRI) is a novel composite CGM metric, calculated as 3.0 × very low-glucose hypoglycemia (VLow; <54 mg/dL; level 2 hypoglycemia) + 2.4 × low-glucose hypoglycemia (Low; 54-<70 mg/dL; level 1 hypoglycemia) + 1.6 × very high-glucose hyperglycemia (VHigh; >250 mg/dL; level 2 hyperglycemia) + 0.8 × high-glucose hyperglycemia (High; >180-250 mg/dL; level 1 hyperglycemia). 20 Glycemia risk index is different from time in range (TIR), the representative CGM metric, in three ways. First, GRI more heavily weights level 2 hypoglycemia (VLow) and hyperglycemia (VHigh) than it does level 1 hypoglycemia (Low) and hyperglycemia (High). Second, it more heavily weights hypoglycemia than it does hyperglycemia. Third, it can be visualized on a GRI grid, a two-dimensional plot with the hypoglycemia component of GRI as the horizontal axis and the hyperglycemia component of GRI as the vertical axis. Glycemia risk index has been demonstrated to have a stronger correlation with clinician assessments of the glycemic quality of CGM tracings than TIR. 20 However, the associations between GRI and diabetic complications have not been extensively studied.
Exposure to hyperglycemia increases the risk of microvascular complications of diabetes.21-24 Diabetic kidney disease is one of the major microvascular complications of diabetes, and albuminuria is a representative marker of kidney disease.25,26 The relationship between GRI and albuminuria in type 1 diabetes has not been reported.
Therefore, we aimed to investigate the association between GRI and albuminuria in individuals with type 1 diabetes.
Methods
Data Source and Study Population
Ninety-day CGM tracings of individuals with type 1 diabetes were obtained from three tertiary hospitals in the Republic of Korea (Samsung Medical Center, Yonsei University Wonju College of Medicine, and Korea University Anam Hospital), from January 2019 to December 2023. Type 1 diabetes was diagnosed according to the current reimbursement criteria of the Korean National Health Insurance Service when individuals used insulin and met at least one of the following criteria: fasting C-peptide ≤0.2 nmol/L; glucagon- or meal-stimulated C-peptide ≤0.6 nmol/L; positive for glutamic-acid-decarboxylase or other autoantibodies; 24-hour urine C-peptide <30 µg/day; or a history of diabetic ketoacidosis at the time of diabetes diagnosis. 27 Blood samples for fasting C-peptide were collected after fasting for over eight hours. Meal-stimulated levels were defined as those taken within five hours of a meal. Clinical data at the initial CGM data collection, including age, sex, duration of diabetes, HbA1c, body mass index (BMI), diagnosis of hypertension, systolic blood pressure, diastolic blood pressure, estimated glomerular filtration rate (eGFR), and use of renin-angiotensin system (RAS) inhibitors, were investigated. Albuminuria was defined as a spot urine albumin-to-creatinine ratio (uACR) of ≥30 mg/g within six months. If multiple uACR measurements were available, the value closest to the CGM date was used. Written informed consent was waived by the institutional review board of each center, and this study protocol (No. 2018-07-132-009) was approved in accordance with the ethical guidelines of the Declaration of Helsinki and Korea Good Clinical Practice.
Initially, 470 individuals with type 1 diabetes using the Dexcom G5 or G6 (Dexcom, San Diego, California), the Medtronic Guardian Sensor 3 (Minneapolis, Minnesota), or the Freestyle Libre 1 (Abbott Diabetes Care, Witney, Oxon, UK) were included in the analysis (Supplementary Figure 1). Among them, 140 individuals who did not meet the criterion of sensor wear time ≥70%; who had missing uACR, eGFR, or HbA1c data; or who had an eGFR <30 mL/min/1.73 m2 or an HbA1c level ≥10% were excluded. Finally, 330 individuals with type 1 diabetes were included in the analyses.
Composite CGM Metrics
Glycemia risk index and its hyperglycemia and hypoglycemia components were calculated as follows 20 :
Glycemia risk index ranges from 0 to 100 and is divided into five risk zones (A: 0-20; B: 21-40; C: 41-60; D: 61-80; and E: 81-100). Zone A indicates the highest quality of glycemia, while zone E indicates the lowest.
We also calculated the composite continuous glucose monitoring index (COGI), 28 which consists of three components: TIR, time below range (TBR) <70 mg/dL, and standard deviation (SD), weighted by 50%, 35%, and 15%, respectively. Continuous glucose monitoring index ranges from 0 (worst) to 100 (best). The COGI equation is as follows:
Linear scoring between 0% TIR (assigned 0 points) and 100% TIR (assigned 100 points)
Linear scoring between 0% TBR (assigned 100 points) and ≥15% TBR (assigned 0 points)
Linear scoring between an SD of ≤18 mg/dL (assigned 100 points) and an SD of ≥108 mg/dL (assigned 0 points)
Statistical Analyses
Continuous variables are presented as mean ± SD or as median (interquartile range [IQR]), while categorical variables are presented as number and percentage (%). Baseline characteristics of participants were compared according to the GRI zone by analysis of variance, Kruskal-Wallis test, Pearson’s χ2 test, or Fisher’s exact test.
The proportions of individuals with albuminuria according to GRI zone and quintile of hyperglycemia (Q1: ≤8.3, Q2: >8.3-15.0, Q3: >15.0-22.3, Q4: >22.3-32.0, and Q5: >32.0) and hypoglycemia components (Q1: ≤0.6, Q2: >0.6-1.2, Q3: >1.2-2.4, Q4: >2.4-4.8, and Q5: >4.8) were compared by Pearson’s χ2 test. In addition, GRI and its components were compared between individuals with and without albuminuria by Student’s t-test or the Wilcoxon rank sum test.
Logistic regression analyses were conducted to estimate the odds ratio (OR) for albuminuria per increase in the GRI zone and quintiles of GRI components. Unadjusted and adjusted ORs (models 1-6) are presented. Model 1 was adjusted for age and sex; model 2 was further adjusted for duration of diabetes, hypertension, eGFR, and use of RAS inhibitors; model 3 was further adjusted for BMI; model 4 was adjusted for variables in model 2 plus achievement of the recommended TIR target >70%; model 5 was adjusted for variables in model 2 plus achievement of the recommended HbA1c target <7%; and model 6 was adjusted for variables in model 2 plus HbA1c. Subgroup analyses were conducted based on age (<40 vs ≥40 years), sex, diabetes duration (<10 vs ≥10 years), hypertension, eGFR (≥90 mL/min/1.73 m2 vs <90 mL/min/1.73 m2), use of RAS inhibitors, TIR (>70% vs ≤70%), and HbA1c (<7% vs ≥7%). Subgroup analyses were conducted in model 2, due to the missing rate for BMI. In addition, ORs for albuminuria per 1 SD change in GRI, TIR, and COGI were calculated. Sensitivity analyses were conducted among individuals who measured the spot uACR at least twice, defining albuminuria as a spot uACR ≥30 mg/g on at least two occasions.
Pearson’s correlation analyses between log-transformed uACR and GRI or its components were also conducted. Moreover, univariable and multivariable linear regression analyses of log-transformed uACR were performed. Age, sex, hypertension, duration of diabetes, eGFR, and GRI or its hyperglycemia component were included in the multivariable linear regression analyses.
All statistical analyses were conducted using R version 4.2.2 (R Foundation for Statistical Computing, www.R-project.org), SPSS version 23 (IBM SPSS Inc. New York, New York), or GraphPad Prism version 9 (GraphPad Software, San Diego, CA, USA). Statistical significance was set at a two-sided P value < .05.
Results
Baseline Characteristics of Participants
The baseline characteristics of participants according to the GRI zone are presented in Table 1. Mean age was 45.1 ± 15.7 years, and 46.7% (154/330) were male. There were no significant differences in age or sex according to the GRI zone. The median diabetes duration of the total population was 10 years (IQR, 4-19 years), but zone A participants had a shorter diabetes duration than zone B to E participants. Blood pressure, eGFR, and use of RAS inhibitors did not significantly differ by GRI zone. Among the total population, 30.0% (99/330) had hypertension, and the mean eGFR was 100.7 ± 21.5 mL/min/1.73 m2. Mean HbA1c, TIR, and GRI were 7.3 ± 1.0%, 65.9 ± 16.8%, and 40.9 ± 21.3, respectively. As the GRI zone increased, HbA1c increased and TIR decreased. HbA1c values of zone A and zone E were 6.5 ± 0.8% and 8.4 ± 0.7%, respectively, and the TIR values were 88.8 ± 5.3% and 34.0 ± 9.5%. Among the 330 participants, 92 (27.9%) used the Dexcom G5, 117 (35.5%) used the Dexcom G6, 8 (2.4%) used the Medtronic Guardian Sensor 3, and 113 (34.2%) used the Freestyle Libre 1. No individuals were using a hybrid-closed loop system.
Baseline Characteristics of Participants According to GRI Zone (N = 330).
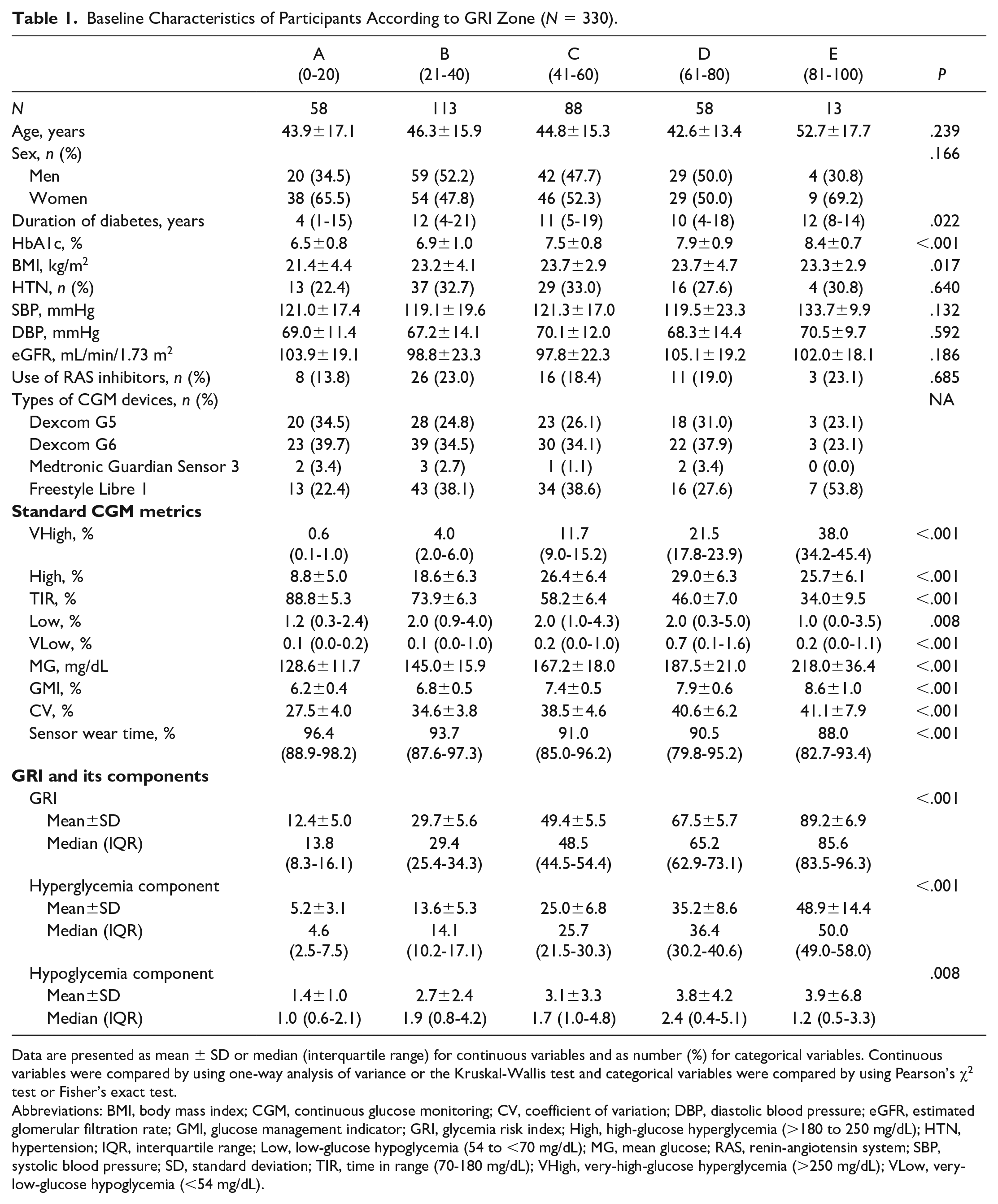
Data are presented as mean ± SD or median (interquartile range) for continuous variables and as number (%) for categorical variables. Continuous variables were compared by using one-way analysis of variance or the Kruskal-Wallis test and categorical variables were compared by using Pearson’s χ2 test or Fisher’s exact test.
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; CV, coefficient of variation; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; GMI, glucose management indicator; GRI, glycemia risk index; High, high-glucose hyperglycemia (>180 to 250 mg/dL); HTN, hypertension; IQR, interquartile range; Low, low-glucose hypoglycemia (54 to <70 mg/dL); MG, mean glucose; RAS, renin-angiotensin system; SBP, systolic blood pressure; SD, standard deviation; TIR, time in range (70-180 mg/dL); VHigh, very-high-glucose hyperglycemia (>250 mg/dL); VLow, very-low-glucose hypoglycemia (<54 mg/dL).
Prevalence of Albuminuria According to GRI Zone
The prevalence of albuminuria was 17.6% and differed significantly by GRI zone (Figure 1a). The prevalence of albuminuria was 5.2% in zone A, 15.0% in zone B, 22.7% in zone C, 25.9% in zone D, and 23.1% in zone E (P = .023 for between-group differences).
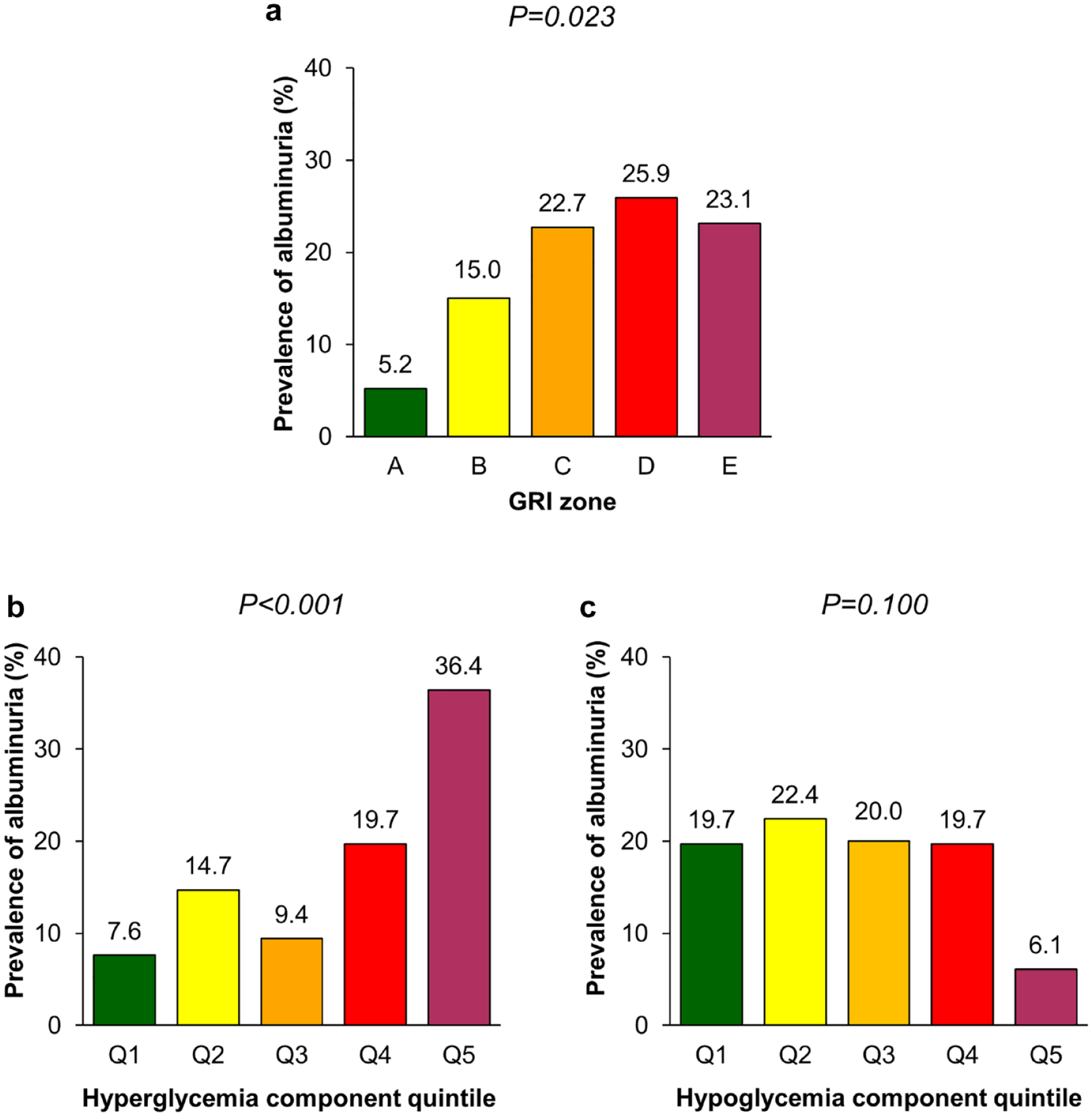
Prevalence of albuminuria according to GRI zone (a), quintile of the hyperglycemia component (b), and quintile of the hypoglycemia component (c). Quintiles of hyperglycemia component: Q1 (≤8.3), Q2 (>8.3-15.0), Q3 (>15.0-22.3), Q4 (>22.3-32.0), and Q5 (>32.0). Quintiles of hypoglycemia component: Q1 (≤0.6), Q2 (>0.6-1.2), Q3 (>1.2-2.4), Q4 (>2.4-4.8), and Q5 (>4.8).
The prevalence of albuminuria differed significantly by quintile of the GRI hyperglycemia component (Figure 1b; P < .001). While 7.6% of the participants in the first quintile of the hyperglycemia component had albuminuria, 36.4% of those in the fifth quintile had albuminuria. In contrast, there were no significant differences according to the quintile of the hypoglycemia component of GRI (Figure 1c; P = .100). Figure 2 depicts the GRI grid. Within the same GRI zone, participants with a higher value of hyperglycemia component had albuminuria.
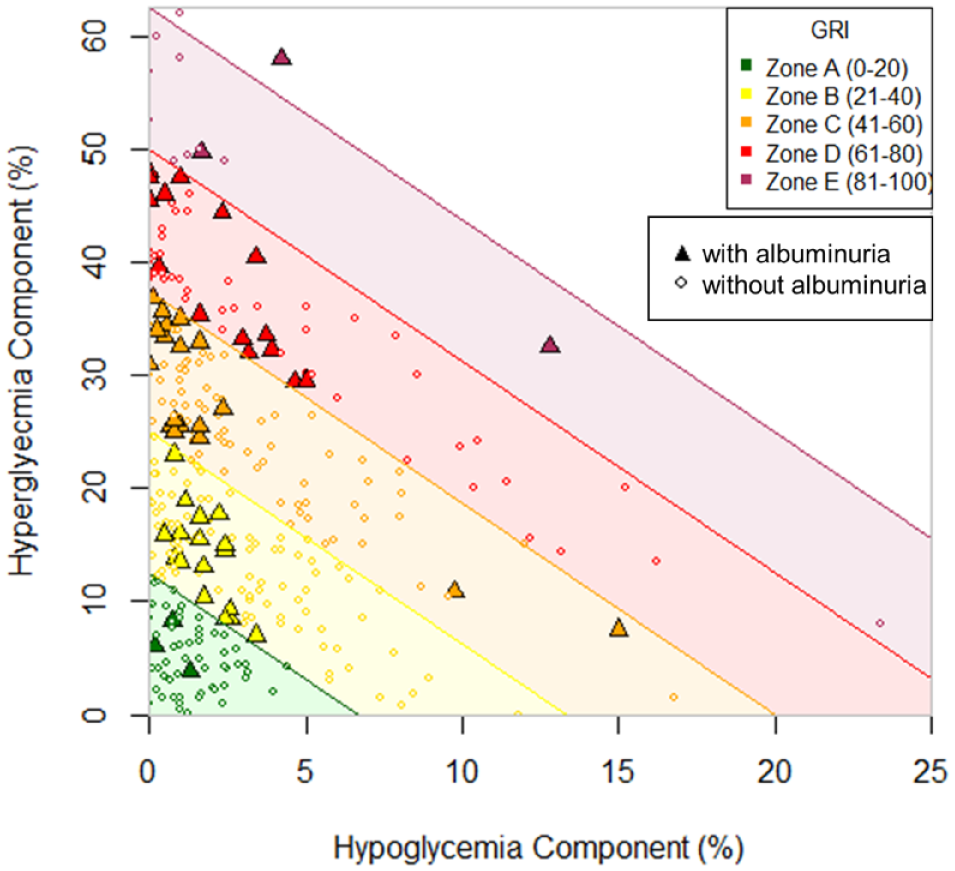
GRI grid.
We also compared the GRI score and its components between participants with and without albuminuria (Figure 3). Participants who had albuminuria had a higher GRI than those without albuminuria (49 vs 39, P = .002). The hyperglycemia component was also higher in those with albuminuria (26 vs 19, P < .001), while the GRI hypoglycemia component was similar between groups (2 vs 3, P = .063).
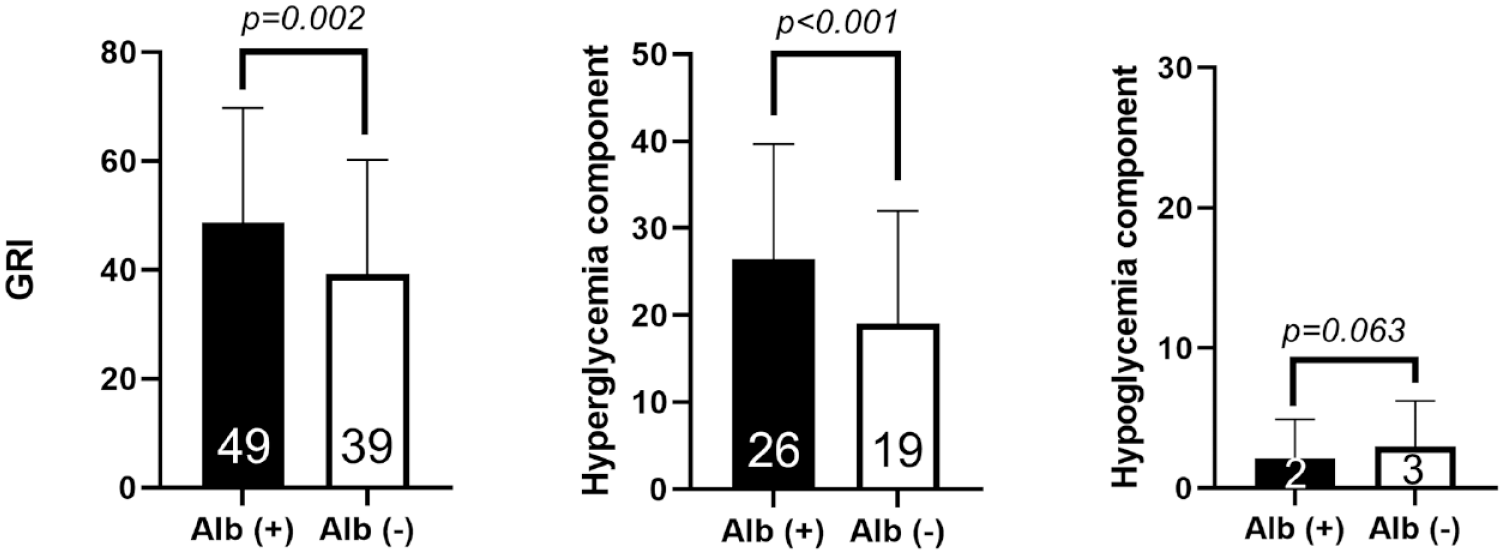
Comparison of GRI and its components according to the presence of albuminuria. Means with standard deviation are presented.
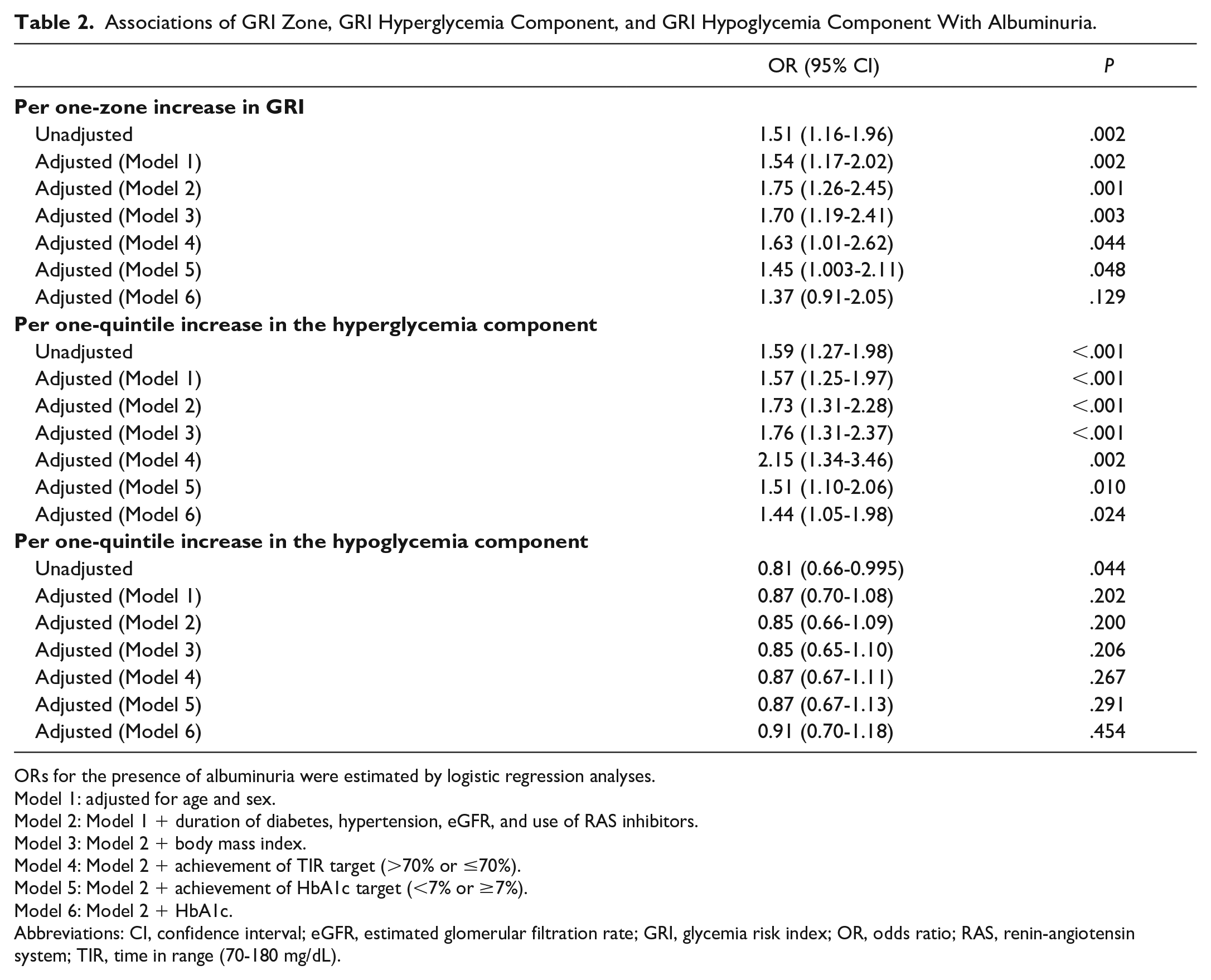
Associations of GRI Zone, GRI Hypoglycemia Component, and GRI Hyperglycemia Component With Risk of Albuminuria
Table 2 presents the associations of GRI and its components with albuminuria, estimated by logistic regression analyses. Unadjusted OR of albuminuria per increase in the GRI zone was 1.51 (95% confidence interval [CI] 1.16-1.96). After adjustment for confounding factors of age, sex, duration of diabetes, hypertension, eGFR, use of RAS inhibitors, and BMI (model 3), the OR of albuminuria per one-zone increase in the GRI was 1.70 (95% CI: 1.19-2.41). The association was also significant after adjustment for variables in model 2 and achievement of the recommended TIR target >70% (model 4, adjusted OR 1.63, 95% CI: 1.01-2.62) or recommended HbA1c target <7% (model 5, adjusted OR 1.45, 95% CI: 1.003-2.11). However, the association lost significance after adjustment for the HbA1c level itself (model 6, adjusted OR 1.37, 95% CI: 0.91-2.05).
Associations of GRI Zone, GRI Hyperglycemia Component, and GRI Hypoglycemia Component With Albuminuria.
ORs for the presence of albuminuria were estimated by logistic regression analyses.
Model 1: adjusted for age and sex.
Model 2: Model 1 + duration of diabetes, hypertension, eGFR, and use of RAS inhibitors.
Model 3: Model 2 + body mass index.
Model 4: Model 2 + achievement of TIR target (>70% or ≤70%).
Model 5: Model 2 + achievement of HbA1c target (<7% or ≥7%).
Model 6: Model 2 + HbA1c.
Abbreviations: CI, confidence interval; eGFR, estimated glomerular filtration rate; GRI, glycemia risk index; OR, odds ratio; RAS, renin-angiotensin system; TIR, time in range (70-180 mg/dL).
The GRI hyperglycemia component was also significantly associated with albuminuria and remained significant after adjusting for various factors including achievement of the recommended targets for TIR (model 4, adjusted OR 2.15, 95% CI: 1.34-3.46) and HbA1c (model 5, adjusted OR 1.51, 95% CI: 1.01-2.06). Moreover, the association remained significant even after adjusting for the HbA1c level itself (model 6, adjusted OR 1.44, 95% CI: 1.05-1.98). In contrast, the hypoglycemia component was not significantly associated with albuminuria after adjustment for confounding factors.
Table 3 presents the results of the subgroup analyses. Increases in the GRI zone and quintile of the GRI hyperglycemia component were generally associated with an increased risk of albuminuria, without significant interactions between subgroups. Both GRI and its hyperglycemia component were significantly associated with albuminuria among individuals at increased risk for albuminuria, such as older individuals, those with a longer diabetes duration, hypertension, lower eGFR, or higher HbA1c. Among those with HbA1c ≥7%, the adjusted ORs per increase in the GRI zone and quintile of its hyperglycemia component were 1.57 (95% CI: 1.02-2.42) and 1.76 (95% CI: 1.19-2.59), respectively. The hyperglycemia component was also significantly associated with albuminuria among those with TIR ≤70% (adjusted OR 2.76, 95% CI: 1.50-5.09).
Subgroup Analysis.
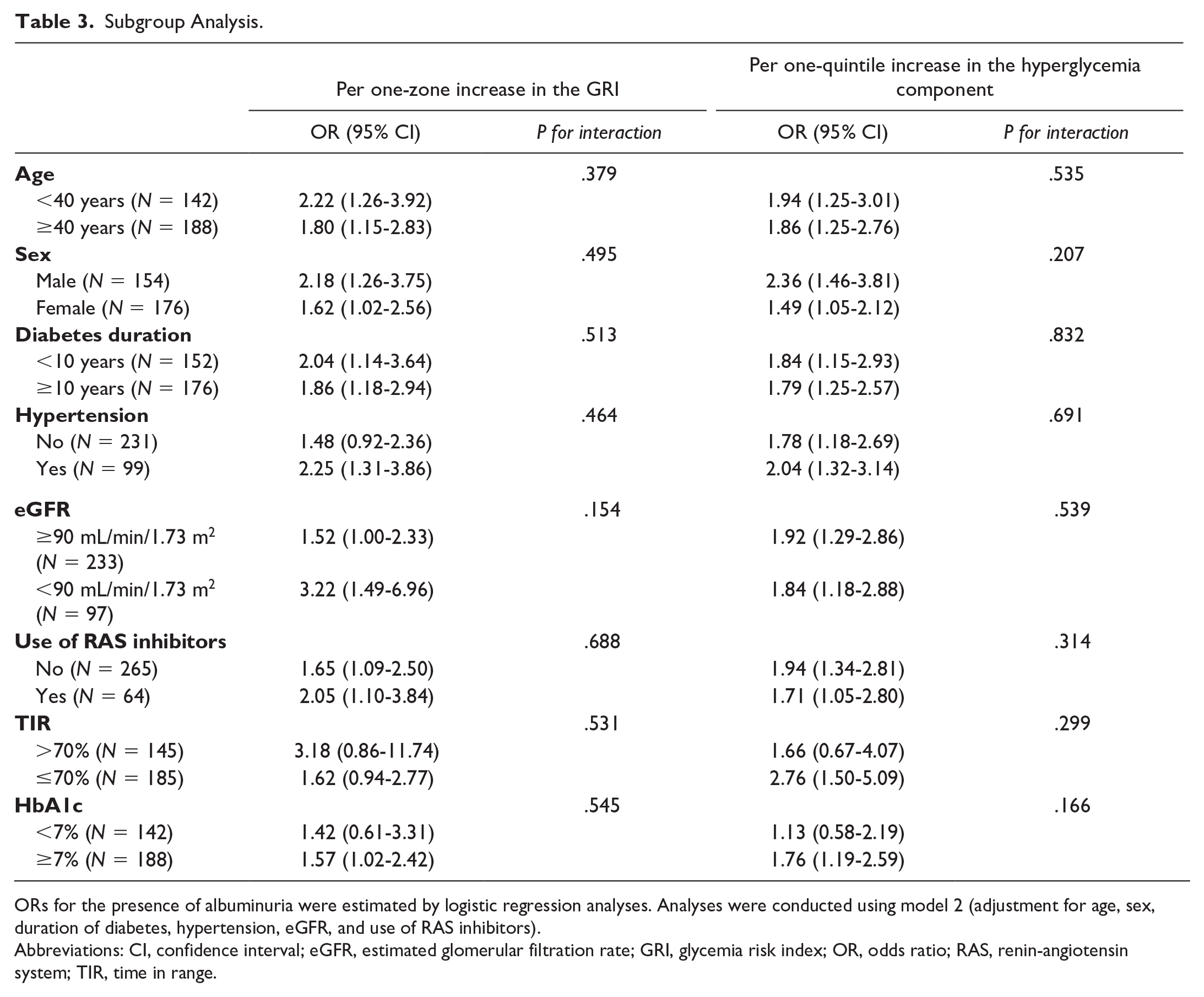
ORs for the presence of albuminuria were estimated by logistic regression analyses. Analyses were conducted using model 2 (adjustment for age, sex, duration of diabetes, hypertension, eGFR, and use of RAS inhibitors).
Abbreviations: CI, confidence interval; eGFR, estimated glomerular filtration rate; GRI, glycemia risk index; OR, odds ratio; RAS, renin-angiotensin system; TIR, time in range.
Association of Other CGM Metrics, Including TIR and COGI, With Prevalence of Albuminuria
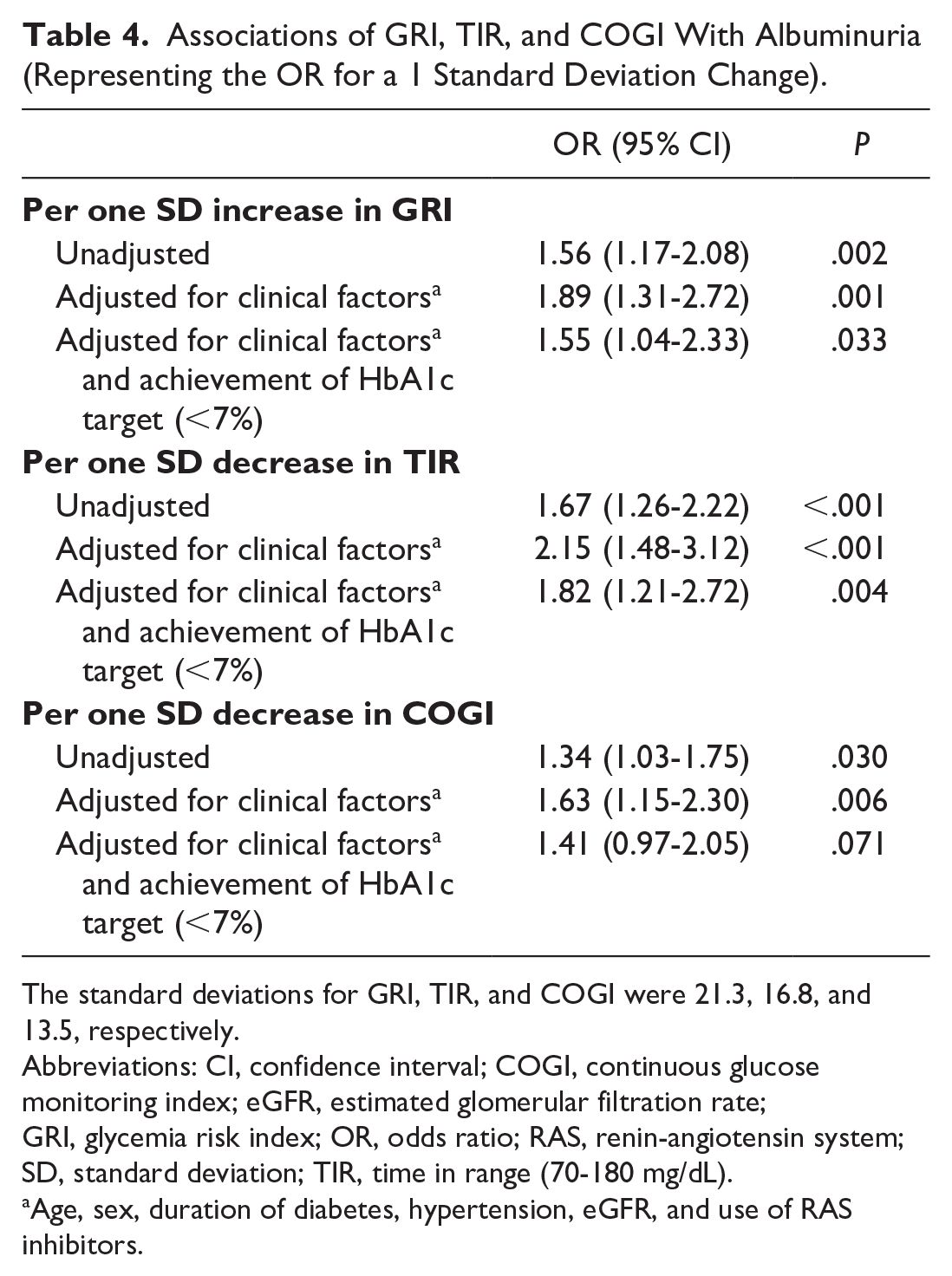
We investigated whether other CGM metrics, including TIR and COGI, are as significantly associated with albuminuria as GRI (Table 4). Adjusted ORs for albuminuria per 1 SD change in GRI, TIR, and COGI were calculated. Both TIR and COGI were also significantly associated with the risk of albuminuria. Adjusted OR per one SD change was the highest for TIR (2.15 [95% CI: 1.48-3.12]), followed by GRI (1.89 [95% CI: 1.31-2.72]) and COGI (1.63 [95% CI: 1.15-2.30]), after adjusting for age, sex, duration of diabetes, hypertension, eGFR, and use of RAS inhibitors.
Associations of GRI, TIR, and COGI With Albuminuria (Representing the OR for a 1 Standard Deviation Change).
The standard deviations for GRI, TIR, and COGI were 21.3, 16.8, and 13.5, respectively.
Abbreviations: CI, confidence interval; COGI, continuous glucose monitoring index; eGFR, estimated glomerular filtration rate; GRI, glycemia risk index; OR, odds ratio; RAS, renin-angiotensin system; SD, standard deviation; TIR, time in range (70-180 mg/dL).
Age, sex, duration of diabetes, hypertension, eGFR, and use of RAS inhibitors.
In sensitivity analyses conducted among individuals who had measured spot uACR at least twice (N = 225, Table 5), with albuminuria being defined as spot uACR ≥30 mg/g on at least two occasions, only GRI remained significantly associated with albuminuria after further adjusting for achievement of the HbA1c target (<7%).
Sensitivity Analyses Among Individuals Who Measured Spot Urine Albumin-to-Creatinine Ratio at Least Twice (N = 225).
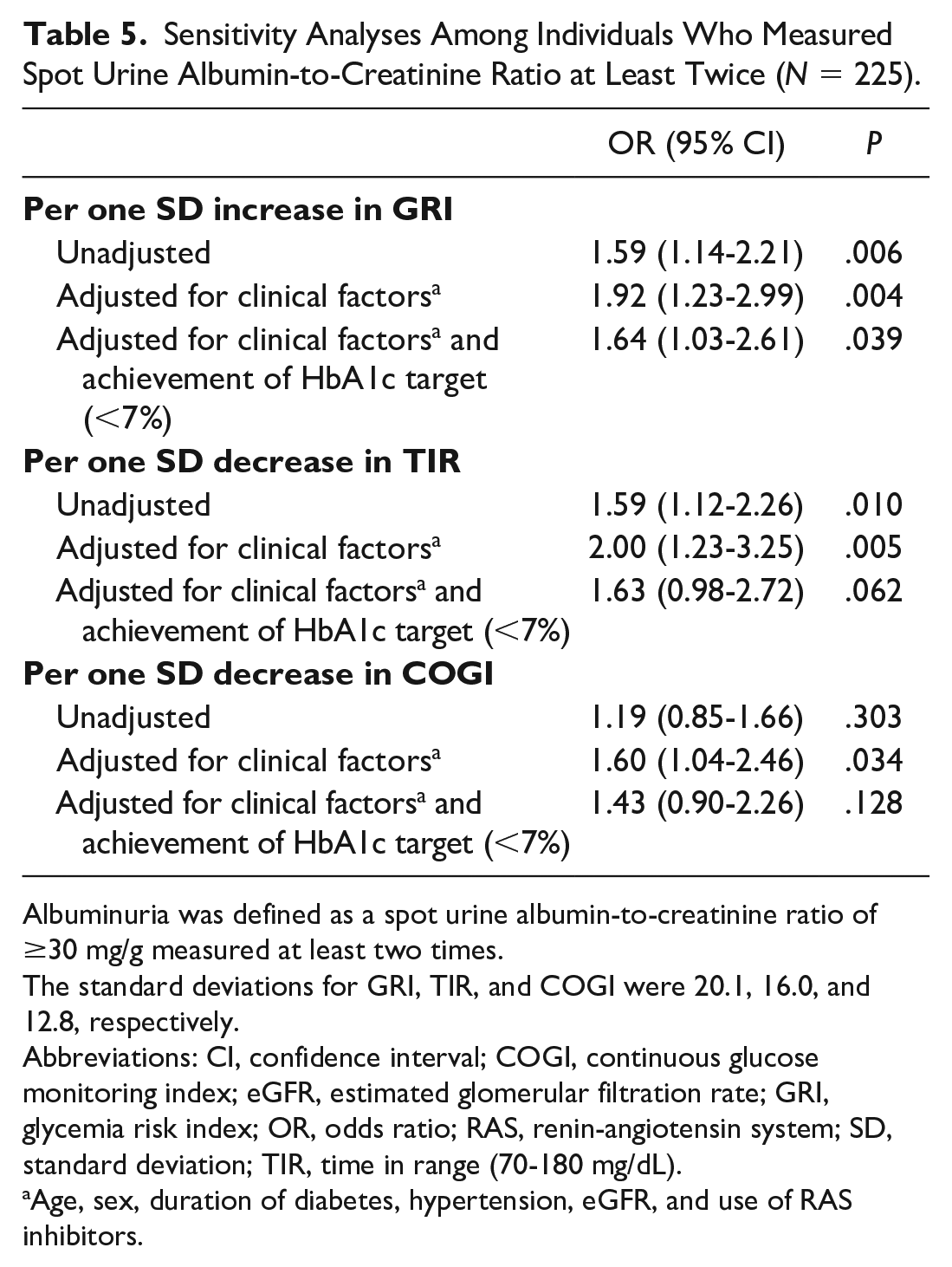
Albuminuria was defined as a spot urine albumin-to-creatinine ratio of ≥30 mg/g measured at least two times.
The standard deviations for GRI, TIR, and COGI were 20.1, 16.0, and 12.8, respectively.
Abbreviations: CI, confidence interval; COGI, continuous glucose monitoring index; eGFR, estimated glomerular filtration rate; GRI, glycemia risk index; OR, odds ratio; RAS, renin-angiotensin system; SD, standard deviation; TIR, time in range (70-180 mg/dL).
Age, sex, duration of diabetes, hypertension, eGFR, and use of RAS inhibitors.
Association Between uACR and GRI
We estimated the correlation between log-transformed uACR and GRI (Supplementary Figure 2). Glycemia risk index (r = .16, P = .004) and its hyperglycemia component (r = .21, P < .001) had a significantly positive association with log-transformed uACR. In contrast, the GRI hypoglycemia component did not have a significant association with log-transformed uACR (r = −.11, P = .057).
In multivariable regression analyses, both GRI and its hyperglycemia component were significantly associated with log-transformed uACR after adjustment for age, sex, hypertension, duration of diabetes, and eGFR (Supplementary Table 1). Among these variables, the most significant factor affecting albuminuria was eGFR (standardized β = −0.32, P < .001), followed by hypertension (standardized β = .27, P < .001), and GRI (standardized β = 0.16, P = .001) or GRI hyperglycemia component (standardized β = 0.19, P < .001).
Discussion
In this study, GRI was significantly associated with the risk of albuminuria in individuals with type 1 diabetes. Each one-zone increase in GRI was associated with a 70% higher risk of albuminuria after adjustment for age, sex, diabetes duration, eGFR, hypertension, BMI, and use of RAS inhibitors, mainly due to the hyperglycemia component of GRI. The association was also significant after adjustment for achievement of the recommended TIR target (>70%) or HbA1c target (<7%).
Hyperglycemia increases the risk of albuminuria by increasing levels of reactive oxygen species, inflammatory cytokines, and growth factors, which causes endothelial dysfunction.29-33 As expected, the hyperglycemia component of GRI was significantly associated with albuminuria. Notably, the association remained significant after adjustment for various factors known to be associated with renal function, such as age, diabetes duration, hypertension, and BMI. 34
In contrast, the hypoglycemia component of GRI was not associated with albuminuria in our study. Considering that some studies have reported a relationship between hypoglycemia and albuminuria,35,36 the characteristics of our study population, particularly the low proportion of participants with hypoglycemia, might have influenced the results. Indeed, albuminuria was more prevalent in GRI zone D than in E in Figure 1, likely due to the higher hypoglycemia component in zone D (2.4% vs 1.2%). While hyperglycemia was the primary driver of albuminuria in our study, previous research by Varghese et al 35 highlighted the impact of hypoglycemia using intermittently scanned (is) CGM. Their study reported a higher TBR (6.7%) than our population’s lower TBR (1%-2%), which may explain the differences. In addition, the accuracy of isCGM regarding low glucose levels is known to be less satisfactory than that of real-time (rt) CGM, 37 which might also have affected their results. Furthermore, as Figure 1 shows unadjusted prevalence, other factors may have influenced the results. Even considering these points, albuminuria appears to be more closely related to hyperglycemia than to hypoglycemia. 29
Several studies have reported an association between the risk of albuminuria and HbA1c.38-40 Therefore, it is notable that the association between GRI and albuminuria remained significant after adjustment for achievement of the recommended HbA1c target. Glycemia risk index might reflect the risk of albuminuria well because it gives more weight to extreme hyperglycemia (VHigh) than to less extreme hyperglycemia (High). However, the association between GRI and albuminuria lost its significance after adjustment for HbA1c level itself. This may be because hypoglycemia is weighted more heavily than hyperglycemia in the GRI calculation. In contrast, the hyperglycemia component of GRI, which only considers VHigh and High, was significantly associated with albuminuria even after adjustment for HbA1c level.
In subgroup analyses, GRI and its hyperglycemia component were significantly associated among participants with risk factors for albuminuria, such as older age, longer diabetes duration, hypertension, lower eGFR, and higher HbA1c. Therefore, GRI and its hyperglycemia component might further differentiate the risk of albuminuria among individuals who are at increased risk of albuminuria.
Standard CGM metrics including TIR, VLow, Low, VHigh, High, coefficient of variation, mean glucose, and glucose management indicators should be considered simultaneously to assess glycemic quality. 41 However, it is challenging to assess all of these components quickly.42,43 TIR has been proposed as a single representative metric useful for assessing glycemia. 44 In our study, TIR was also significantly associated with the presence of albuminuria, consistent with findings from previous studies.29,45 The adjusted OR for albuminuria per 1 SD change in TIR (2.15) was even higher than that per 1 SD change in GRI (1.89). This may be related to the hypoglycemia component of GRI not being significantly associated with the risk of albuminuria in our study population (probably due to the low proportion of participants with hypoglycemia) and GRI placing more weight on the hypoglycemia component than on the hyperglycemia component. However, in the sensitivity analysis, TIR lost its significant association with albuminuria after adjusting for the achievement of the HbA1c target. Moreover, GRI has the advantage that it can reflect hypoglycemia better than TIR. 46 When experienced clinicians ranked the glycemic quality of CGM tracings, the GRI showed a stronger correlation with their rankings compared with TIR. 20
Continuous glucose monitoring index, another composite CGM metric that considers TIR, hypoglycemia, and glycemic variability, was also significantly associated with albuminuria. However, the adjusted OR for albuminuria per 1 SD change in COGI (1.63) was lower than that per 1 SD change in GRI (1.89), and COGI lost its significant association with albuminuria after adjusting for the achievement of the HbA1c target. Sensitivity analyses among individuals who had measured spot uACR at least twice showed similar results. These findings indicate that GRI is a useful metric for clinicians.
To the best of our knowledge, this is the first study to report an association between GRI and albuminuria in individuals with type 1 diabetes. Strengths of our study include the use of 90-day CGM tracings from multi-centers and adjustment for various factors affecting albuminuria, as well as the achievement of recommended TIR and HbA1c targets.
We previously reported a significant association between GRI and albuminuria in individuals with type 2 diabetes. 47 Therefore, GRI seems to be associated with albuminuria universally, in both type 1 diabetes and type 2 diabetes. Wang et. al reported an association between GRI and diabetic retinopathy among individuals with type 2 diabetes. 48 The association between GRI and other diabetic complications needs to be elucidated.
This study had several limitations. First, due to its cross-sectional design, it was not possible to determine a causal relationship between GRI and albuminuria. Further studies with longitudinal design are needed. Second, as mentioned earlier, the proportion of participants with hypoglycemia was low. Therefore, our findings regarding the association between the hypoglycemia component of GRI and albuminuria should be interpreted with caution. Third, our study is limited by the combined use of isCGM and rtCGM. Generally, isCGM is less accurate than rtCGM, especially during hypoglycemia, where it tends to overestimate low glucose levels compared with rtCGM. This may have influenced our hypoglycemia measurements and underestimated the impact of hypoglycemia on albuminuria. However, similar associations were observed when we included only Dexcom G6 users (Supplementary Table 2). Fourth, although albuminuria has been reported to be associated with smoking,49,50 this analysis did not account for smoking status due to the lack of comprehensive information in the dataset. We acknowledge this limitation and will consider it in future studies to enhance the scope of our analysis. Lastly, our study lacks diversity in the population. The participants were predominantly homogenous in terms of ethnicity, as they were all of sub-Asian (Korean) descent, and were concentrated in specific geographic areas. In addition, all clinics specialized in type 1 diabetes and reported relatively low rates of hypoglycemia. This lack of diversity may limit the generalizability of our findings to broader, more varied populations. Future research should strive to include a more diverse sample and range of clinical settings to enhance the applicability of the results across different demographic and diabetic groups.
Conclusions
Glycemia risk index and its hyperglycemia component were significantly associated with the risk of albuminuria in individuals with type 1 diabetes. Glycemia risk index appears to be a useful index for assessing glycemia and predicting the risk of albuminuria.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241310850 – Supplemental material for Glycemia Risk Index is Associated With Risk of Albuminuria Among Individuals With Type 1 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968241310850 for Glycemia Risk Index is Associated With Risk of Albuminuria Among Individuals With Type 1 Diabetes by Ji Yoon Kim, Jee Hee Yoo, Nam Hoon Kim and Jae Hyeon Kim in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
BMI, body mass index; CGM, continuous glucose monitoring; CI, confidence interval; COGI, continuous glucose monitoring index; eGFR, estimated glomerular filtration rate; GRI, glycemia risk index; HbA1c, glycated hemoglobin; High, high-glucose hyperglycemia; IQR, interquartile range; isCGM, intermittently scanned CGM; Low, low-glucose hypoglycemia; OR, odds ratio; RAS, renin-angiotensin system; rtCGM, real-time CGM; SD, standard deviation; TBR, time below range; TIR, time in range; uACR, urine albumin-to-creatinine ratio; VHigh, very high-glucose hyperglycemia; VLow, very low-glucose hypoglycemia.
Author Contributions
JYK, JHY, and JHK contributed to the design of the study. JYK, JHY, NHK, and JHK collected data. JYK conducted the analyses. JYK, JHY, NHK, and JHK interpreted the results. JYK wrote the initial draft of the manuscript, with revisions by all authors. The final manuscript was approved by all authors. JHK is the guarantor of this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of Data and Materials
Data are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.