Abstract
Aims:
According to the 2023 International Consensus, glucose metrics derived from two-week-long continuous glucose monitoring (CGM) can be extrapolated up to 90 days before. However, no studies have focused on adults with type 1 diabetes (T1D) on multiple daily injections (MDIs) and with second-generation intermittently scanned CGM (isCGM) sensors in a real-world setting.
Methods:
This real-world, retrospective study included 539 90-day isCGM data from 367 adults with T1D on MDI therapy. For each sensor metric, the coefficients of determination (R2) were computed for sampling periods from 2 to 12 weeks versus the whole 90-day interval. Correlations were considered strong for R2 ≥0.88.
Results:
The two-week sampling period displayed strong correlations for time in range (TIR, 70-180 mg/dl; R2 = 0.89) and above range (TAR, >180 mg/dl; R2 = 0.88). The four-week sampling period showed additional strong correlations for time in tight range (TITR, 70-140 mg/dl; R2 = 0.92), for the coefficient of variation (CV; R2 = 0.88), and for the glycemia risk index (GRI; R2 = 0.92). The six-week sampling period displayed an additional strong correlation for time below range (TBR, <70 mg/dl; R2 = 0.90). After stratification by clinical variables, lower R2 values were found for older age quartiles (>40 years), higher CV (>36%), lower sensor use (≤94%), and higher HbA1c (>7.5%).
Conclusion:
In patients with T1D on MDI, two- to six-week intervals of isCGM use can provide clinically useful estimates of TIR, TAR, TITR, TBR, CV, and GRI, which can be extrapolated to longer (up to 90 days) time intervals. Longer intervals might be needed in case of older age, higher glucose variability, lower sensor use, and higher HbA1c.
Introduction
For people with diabetes mellitus, continuous glucose monitoring (CGM) data can provide more information than Hb1Ac alone to support therapeutic decisions, particularly related to hypoglycemia, postprandial hyperglycemia, and glucose variability. 1 Indeed, the use of CGM devices should be considered since the onset of insulin-dependent diabetes,2,3 and it is recommended as a feasible first-line method of glucose monitoring for all patients affected by insulin-treated diabetes. 4
The use of CGM devices among patients with type 1 diabetes (T1D) in the United States increased from 7% in 2010 to 2012 to 30% in 2016 to 2018, according to the data of the T1D Exchange Registry. 5 Moreover, the SAGE Study, 6 a multinational, multicentric study in subjects with T1D, reported that the proportion of subjects using CGM devices decreased with age and that the multiple daily injections (MDI) therapy is still the most used therapy worldwide.
Continuous glucose monitoring profiles can provide information about within-day, between-day, and between-week variability. Based on current evidence and in accordance with the most recent international consensus statement, 1 14 consecutive days of CGM data with at least 70% of sensor use might predict the glycemic profile of up to a three-month period. However, as already pointed out in the former 2019 consensus on time in range (TIR), 7 in people with T1D, even if the most recent 14 days have strong correlations with three months for mean glucose (MG), TIR, and hyperglycemia metrics, the time-to-time correlations of time below range (TBR) and of the coefficient of variation (CV) are weaker than those of other metrics, and longer sampling periods might be needed as glucose variability worsens. 1 ,7-11 Moreover, a novel composite sensor metric, the glycemia risk index (GRI), was introduced in the latest years to describe the quality of the glucose control according to the composite risk of higher and lower glucose values 12 and, according to a recent study, 13 even for this metric 14 days might be an optimal sampling interval to reflect 90 days of CGM data as well.
Previous studies investigated the correlations between CGM metrics and the duration of sampling periods, but they often involved different CGM devices and different insulin treatment strategies. 1 Only two studies have been carried out in a real-world setting: one in childhood/adolescence 14 and one in adulthood. 15
Therefore, aims of this real-life study performed in a large cohort of adults with T1D with the same isCGM sensors and the same treatment strategy (MDI) were: (1) to investigate the correlations between several different time windows of sensor-derived data and three months of isCGM metrics, (2) to gauge which variables might affect the strength of such correlations, and (3) to find out any significant differences in the classification of glucose profiles from such time intervals versus three months according to the recommended metrics targets. 7
Methodology
In this multicentric, real-world, retrospective observational study, we collected glucose sensor data from the routine visits of adult subjects with T1D who attended the Outpatient Diabetes Clinics from January 1, 2023 to December 31, 2023 at the University Hospital of Verona and from January 1, 2023 to April 30, 2023 at the Papa Giovanni XXIII Hospital of Bergamo. Inclusion criteria were: age ≥18 years, diagnosis of T1D, treatment with MDI, use of a second-generation isCGM sensor (AbbottTM Freestyle Libre 2). Data of subjects with sensor use <70% during the 90 days before clinical visits were excluded, and visits done less than 90 days after previously included visits of the same subject were excluded as well, in order to avoid the repeated use of the same time series of isCGM readings in the same subject.
The primary goal of this study was to find the shortest sampling interval with a clinically acceptable strong correlation with 90 days of isCGM data. The primary outcome variable was the R2 of the correlations between the metrics of shorter time intervals and the metrics of the full 90-day interval (see Statistical Analysis for further details).
Secondary goals of this study were: (1) to unveil which clinical variables affect the degrees of correlation between the metrics during different sampling durations and the reference metrics of the full three-month time period; (2) to compare the proportions of glucose trends classified as on-target or off-target in shorter time intervals versus the full three-month period.
For each included visit, we collected isCGM-derived data regarding the 90 days prior the visit and calculated the following metrics: TIR 70 to 180 mg/dl, time in tight range (TITR) 70 to 140 mg/dl, time above range >180 mg/dl (TAR), TBR <70 mg/dl, CV, MG, and the GRI, calculated according to the previously published formula. 12 Then, we created composite time intervals with consecutive, additive two-week intervals in order to assess the relationship of the 90-day sensor metrics with the metrics of six progressively longer time periods preceding each visit, that is 1 to 14, 1 to 28, 1 to 42, 1 to 56, 1 to 70, and 1 to 84 days; metrics values for such composite sampling intervals were the weighted means of the metrics values for the percentage of sensor use of each two-week interval.
The study was conducted in compliance with the World Medical Association’s Declaration of Helsinki and it was approved by the ethics committees of both hospitals.
Statistical Analysis
Continuous variables are presented as mean and standard deviation or as median and interquartile range, according to their distribution shapes. Categorical variables are presented as absolute and relative (percentage) frequencies. Shapiro-Wilk tests were performed to assess normality for continuous variables.
In order to account for the presence of multiple visits (and thus isCGM readings) of the same subjects in the database, all data of each subject were analyzed as a data panel, and subjects and visits were included as factors in the statistical model used for the analysis.
To evaluate the relationship for each metric in each composite sampling period with the respective 90-day reference value, random-effects linear regression models (fitted to data panel) were computed, and the strength of such relationship was assessed through the resulting overall squared correlation coefficient (R2), also known as coefficient of determination, as it represents the proportion of the variability of a given dependent variable that is explained by the independent variable 16 : A R2 value of 0.88 was chosen as a threshold to define the strength of the correlation, according to the most recent studies. 17
For each sensor metric and for each composite time interval, median absolute difference (MAD) from the correspondent value at 90 days of glucose monitoring was also calculated, in order to provide a statistical estimate of the deviation of each metric from the 90-day reference period.
Further subgroup analyses were performed with the calculation of R2 for each metric after stratifying by sex, age quartiles, HbA1c (under or above median value), total daily insulin dose (under or above median value), CV (under or above 36%), and sensor use (under or above median value). Multiple regression analyses were also computed to assess the potential influence of relevant clinical variables on the relationship between strongly correlated short sampling intervals and the 90-day reference period; in these analyses, given the presence of multiple regressors, R2 was the squared coefficient of multiple correlation for each adjusted model.
For each composite sampling interval and for the full 90-day period, the proportions of isCGM-derived data which reached the recommended targets for TIR, TAR, TBR, and CV 7 were calculated, and McNemar’s χ2 tests were performed in order to detect any significant differences between the proportion of on-target isCGM readings during different time intervals versus the 90-day reference time window. Sensitivity and specificity of isCGM reading classification were calculated for each metric and each sampling interval versus the 90-day reference time period. Finally, Spearman’s nonparametric correlation coefficients were computed for each of these metrics in order to assess the relationship of the classification of readings according to targets during different time intervals versus the 90-day reference time window.
Kruskal-Wallis tests were also performed within each metric between each sampling period to find whether any sampling period was associated to significantly different metrics.
Since this was an exploratory study, no corrections for multiple comparisons were made and significance testing was not performed.
Statistical analyses were performed using STATA software, Version 14.2 (STATA, College Station, Texas, USA).
Results
The analysis included 539 90-day intervals of isCGM data, collected in 367 Italian subjects of Caucasian ethnicity (one to four visits per subject, average 1.4 visits/subject) with T1D (M/F = 203/164), ranging in age from 19 to 84 years. The main clinical variables are shown in Table 1. No significant differences of the main glucose metrics between the single two-week periods were found (Supplementary Figure 1).
Clinical Variables and isCGM-Derived Metrics of the Full 90-Day Period in the Whole Sample of 539 90-Day Intervals Collected in 367 Subjects.
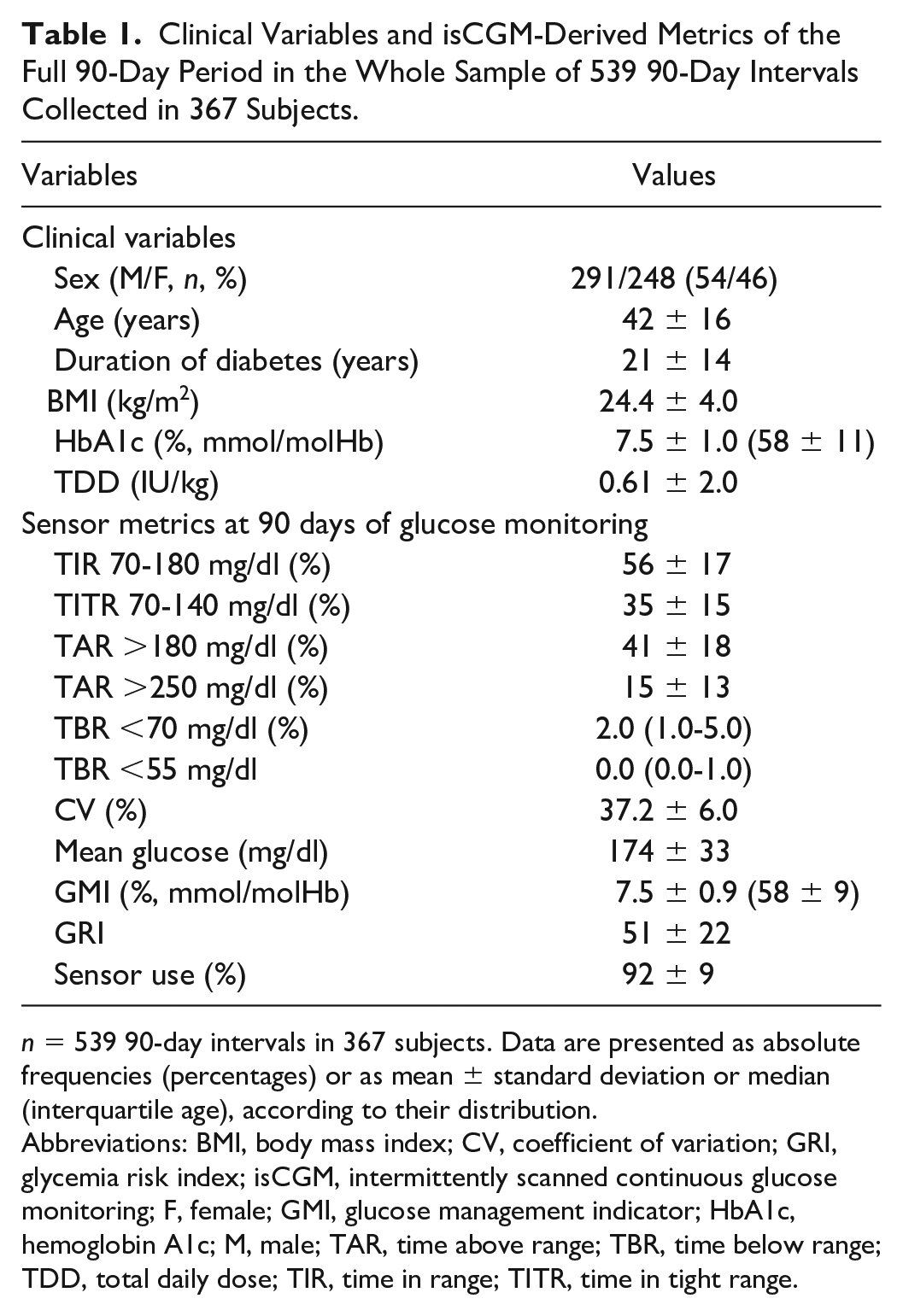
n = 539 90-day intervals in 367 subjects. Data are presented as absolute frequencies (percentages) or as mean ± standard deviation or median (interquartile age), according to their distribution.
Abbreviations: BMI, body mass index; CV, coefficient of variation; GRI, glycemia risk index; isCGM, intermittently scanned continuous glucose monitoring; F, female; GMI, glucose management indicator; HbA1c, hemoglobin A1c; M, male; TAR, time above range; TBR, time below range; TDD, total daily dose; TIR, time in range; TITR, time in tight range.
Assessing the Strength of the Correlation of Sampling Intervals Versus 90 Days
Scatter plots were done for each metric to provide a graphical representation of the relationship between each sampling interval and the full 90-day period (Supplementary Figures 2–8).
The values of sensor metrics, the coefficient of determination R2, and the MADs of the metrics of each composite time interval versus the 90-day period are shown in Table 2. The strength of the correlations with the metrics of the entire 90-day time window, as assessed by R2, increased with the duration of sampling intervals (Figure 1). Time in range, TAR, and MG showed R2 values equal to or higher than 0.88 at two weeks (0.89, 0.89, and 0.90, respectively); TITR, CV, and GRI crossed the cut-off value only as of the four-week interval (0.92, 0.88, and 0.92, respectively). TBR needed at least six weeks of data collection to reach an R2 of 0.90. The MAD decreased gradually from shorter to longer sampling periods for each sensor metric (Figure 2) as well as for GRI (Supplementary Figure 9), coherently with the increase of R2.
Sensor Metrics Through Different Sampling Intervals, Coefficients of Determination of the Correlations Between Increasing Sampling Intervals and 90-Day Intervals of isCGM Data and Median Absolute Differences of Metrics Values at Sampling Intervals Versus 90 Days.
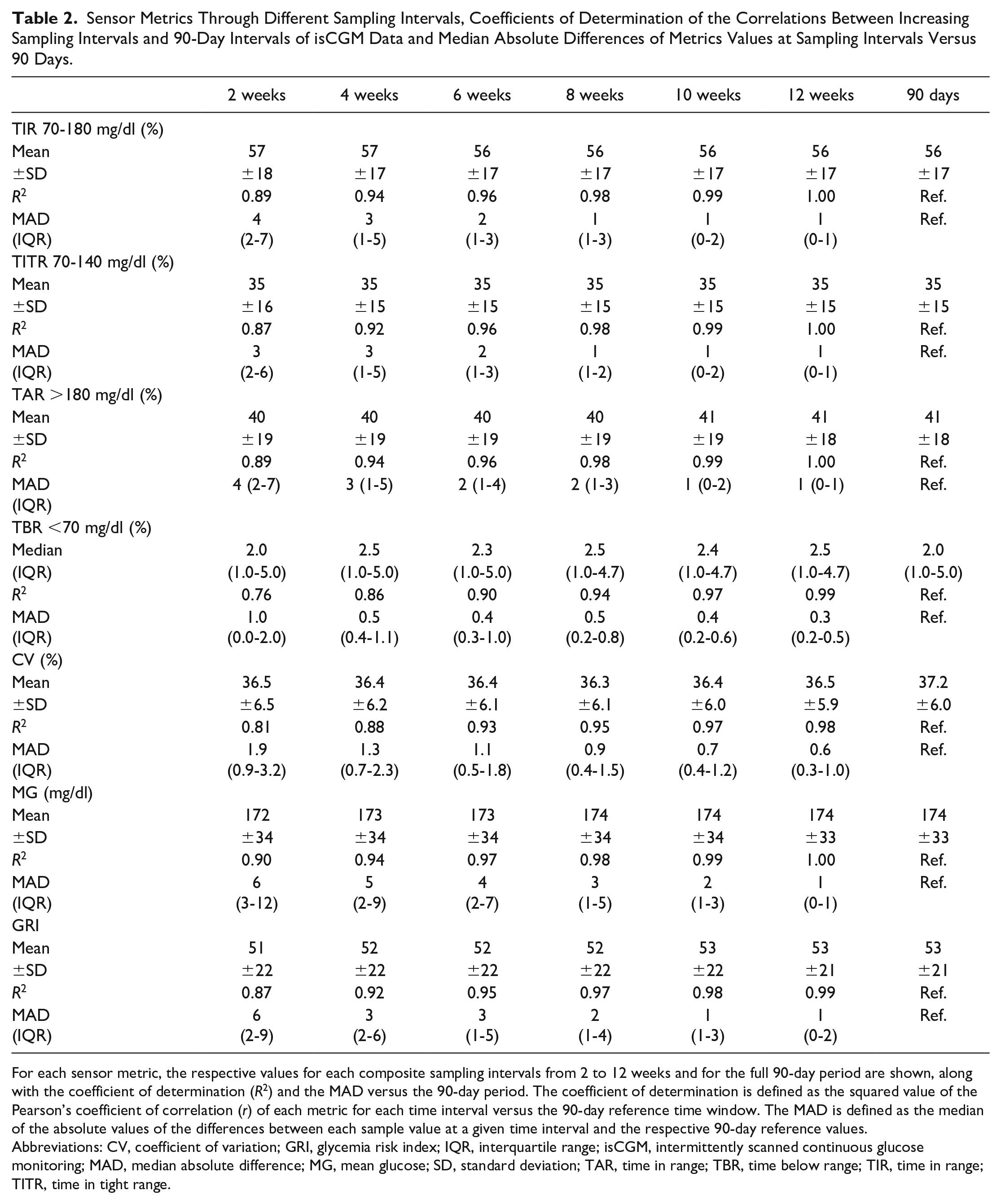
For each sensor metric, the respective values for each composite sampling intervals from 2 to 12 weeks and for the full 90-day period are shown, along with the coefficient of determination (R2) and the MAD versus the 90-day period. The coefficient of determination is defined as the squared value of the Pearson’s coefficient of correlation (r) of each metric for each time interval versus the 90-day reference time window. The MAD is defined as the median of the absolute values of the differences between each sample value at a given time interval and the respective 90-day reference values.
Abbreviations: CV, coefficient of variation; GRI, glycemia risk index; IQR, interquartile range; isCGM, intermittently scanned continuous glucose monitoring; MAD, median absolute difference; MG, mean glucose; SD, standard deviation; TAR, time in range; TBR, time below range; TIR, time in range; TITR, time in tight range.
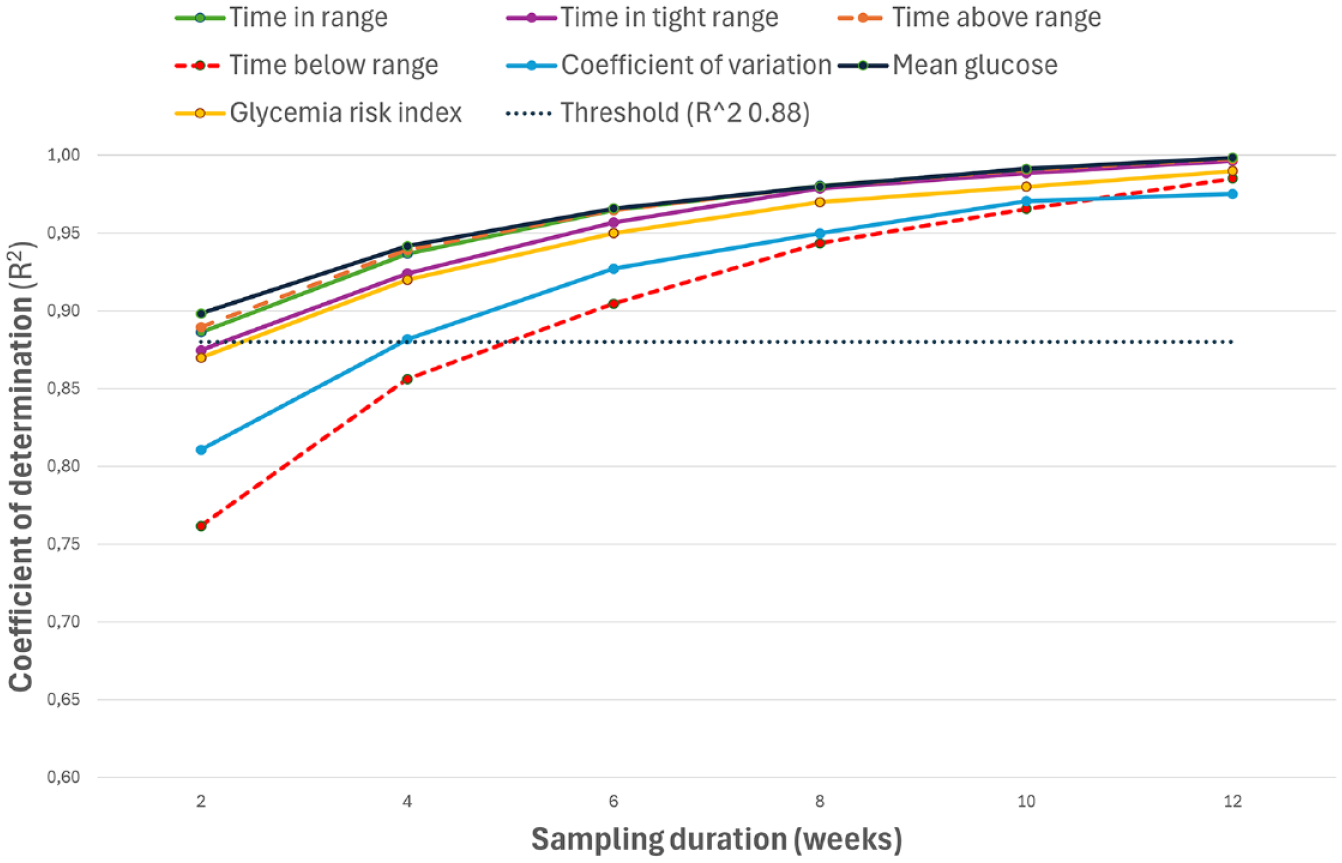
R2 values for sampling intervals from 2 to 12 weeks compared with the 90-day interval. The coefficient of determination (R2) is defined as the squared value of the Pearson’s coefficient of correlation (r) of each metric for each time interval versus the 90-day reference. For each sensor metric, the R2 values between isCGM data over each composite sampling interval and isCGM data over the full 90-day period were plotted. R2 values were calculated from 539 isCGM intervals from 367 subjects. For all correlation analyses, P < .001.
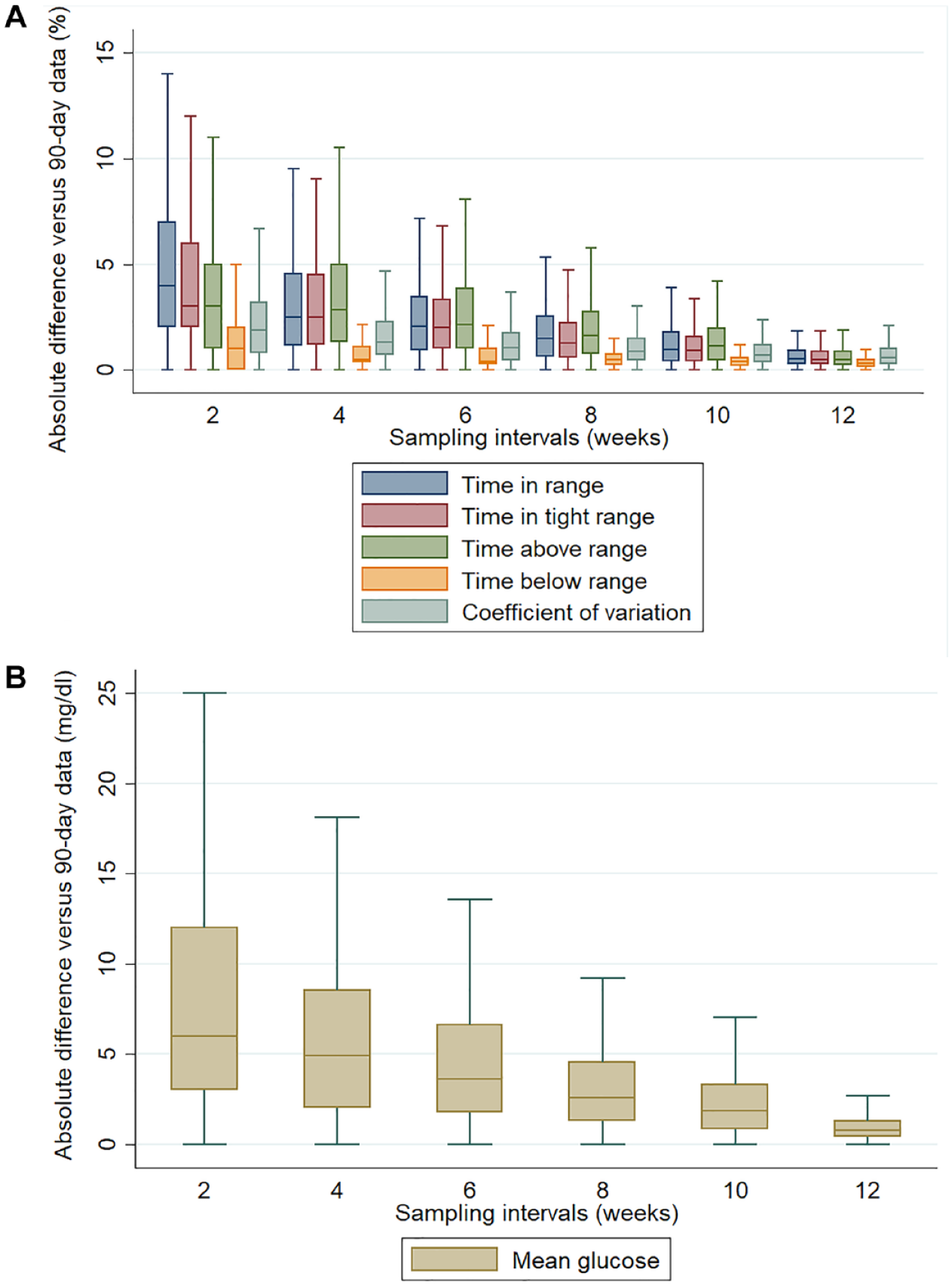
Absolute difference (AD) at each isCGM sampling interval from the 90-day values for TIR, TITR, TAR, TBR, CV (a) and for mean glucose (b). For each sensor metric, the distribution of the AD from each sampling interval versus the full 90-day time period was shown through box-plots with median and IQR (box) and minimum and maximum values (whiskers); outliers (data higher than the third quartile or lower than the first quartile respectively for more than 1.5 times the IQR) were not shown. AD values were calculated from 539 isCGM intervals from 367 subjects.
Clinical Variables Affecting the Correlation
As shown in Table 3, higher R2 values for TIR, TAR, and MG for two-week periods and for TITR for four-week periods were found for CV lower than 36% (R2 for CV ≤36% vs >36%: 0.94, 0.94, 0.95, 0.96 vs 0.76, 0.79, 0.81, 0.84, respectively), for lower HbA1c values (R2 for HbA1c ≤7.5% vs >7.5%: 0.85, 0.85, 0.84, 0.90 vs 0.75, 0.75, 0.82, 0.81, respectively), for lower age quartiles (R2 for first-second vs third-fourth age quartiles: 0.92-0.90, 0.92-0.91, 0.92-0.92, 0.94-0.94 vs 0.86-0.85, 0.86-0.86, 0.86-0.87, 0.91-0.88, respectively), and for higher percentages of sensor use (R2 for sensor use >94% vs ≤94: 0.91, 0.91, 0.92, 0.94 vs 0.87, 0.87, 0.87, 0.91, respectively). TBR, instead, showed more pronounced increments of R2 for lower HbA1c (R2 for HbA1c ≤7.5% vs >7.5%: 0.75 vs 0.68 at two weeks, 0.86 vs 0.79 at four weeks, 0.91 vs 0.85 at six weeks). As for GRI, higher R2 values at four weeks were found especially for lower CV (R2 for CV ≤36% vs >36%: 0.96 vs 0.83), with slighter improvements for lower HbA1c (R2 for HbA1c ≤7.5% vs >7.5%: 0.90 vs 0.86) and younger age quartiles (R2 for first-second vs third-fourth age quartiles: 0.94-0.94 vs 0.91-0.88).
Coefficients of Determination of the Correlations Between Increasing Sampling Intervals and 90-Day Interval of isCGM Data After Stratifying by Clinical Variables.
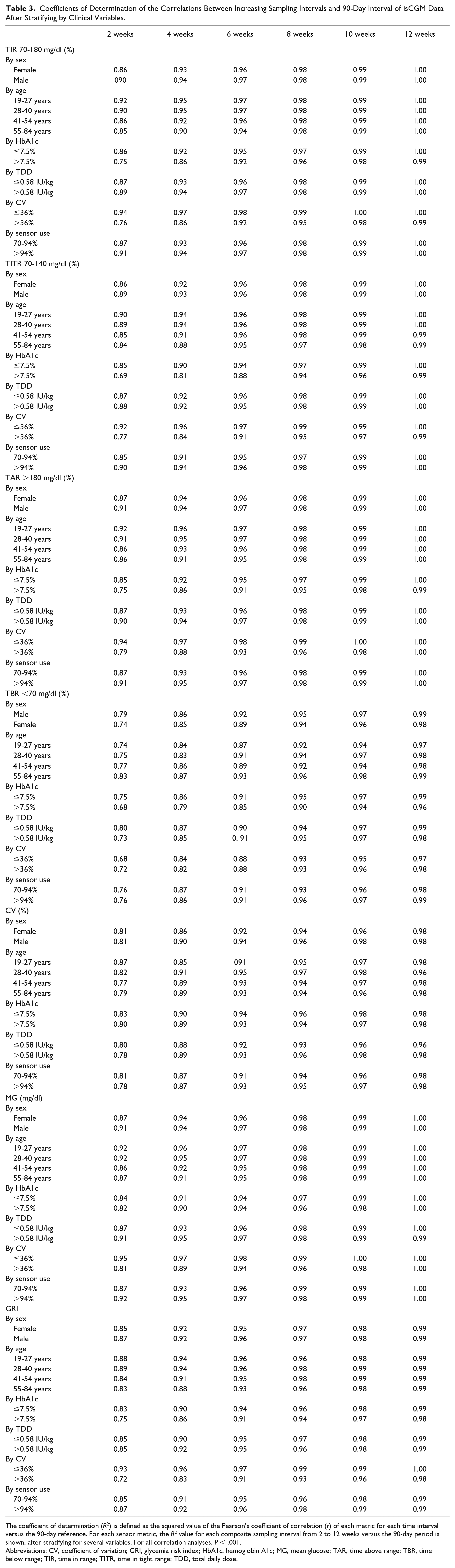
The coefficient of determination (R2) is defined as the squared value of the Pearson’s coefficient of correlation (r) of each metric for each time interval versus the 90-day reference. For each sensor metric, the R2 value for each composite sampling interval from 2 to 12 weeks versus the 90-day period is shown, after stratifying for several variables. For all correlation analyses, P < .001.
Abbreviations: CV, coefficient of variation; GRI, glycemia risk index; HbA1c, hemoglobin A1c; MG, mean glucose; TAR, time above range; TBR, time below range; TIR, time in range; TITR, time in tight range; TDD, total daily dose.
Further, adjusted multiple regression models between the metrics at 90 days and the metrics at the previously detected shortest optimal sampling interval confirmed a statistically strong, albeit quantitatively minor, contribution of HbA1c to the overall R2, whereas other covariates like sensor use, age, and CV played a lesser role (Supplementary Tables 1–6). For GRI, instead, the main contribution to the overall R2 in the adjusted regression models came from CV, while other covariates showed a lesser influence (Supplementary Table 7).
Evaluating the Accuracy to Detect On-Target Readings
As reported in Table 4, we calculated the percentages of 90-day isCGM readings which reached TIR>70%, TAR<25%, TBR<4%, and CV≤36% at 90 days and at the several sampling intervals: When considering the shortest time intervals for R2 value to be equal to or higher than 0.88 (two weeks for TIR and TAR, four weeks for CV, and six weeks for TBR), higher proportions of isCGM readings achieved the targets, when compared to the 90-day reference, for TIR (23.7% at two weeks vs 20.0% at 90 days, P = .008), TAR (22.5% at two weeks vs 18.6% at 90 days, P = .004), TBR (69.3% at six weeks vs 63.8% at 90 days, P < .001), and CV (47.8% at four weeks vs 42.9% at 90 days, P = .001). Moreover, both sensitivity (for the detection of actual on-target readings for the 90-day period) and specificity (for the detection of actual off-target readings for the 90-day period) increased with longer sampling intervals, still maintaining values around 90% at the aforementioned selected times (except for TBR, which failed to achieve such levels of specificity even at their shortest time of strong correlation with 90 days): respectively, 88.6% and 92.9% for TIR at two weeks, 90.9% and 93.8% for TAR at two weeks, 97.0% and 84.1% for TBR at six weeks, and 94.2% and 87.8% for CV at four weeks.
Proportions of On-Target isCGM Readings Classified According to Recommended Glucose Sensor Targets Through Increasing Sampling Intervals and Comparison With Proportion of On-Target Readings at 90 days of isCGM Data.
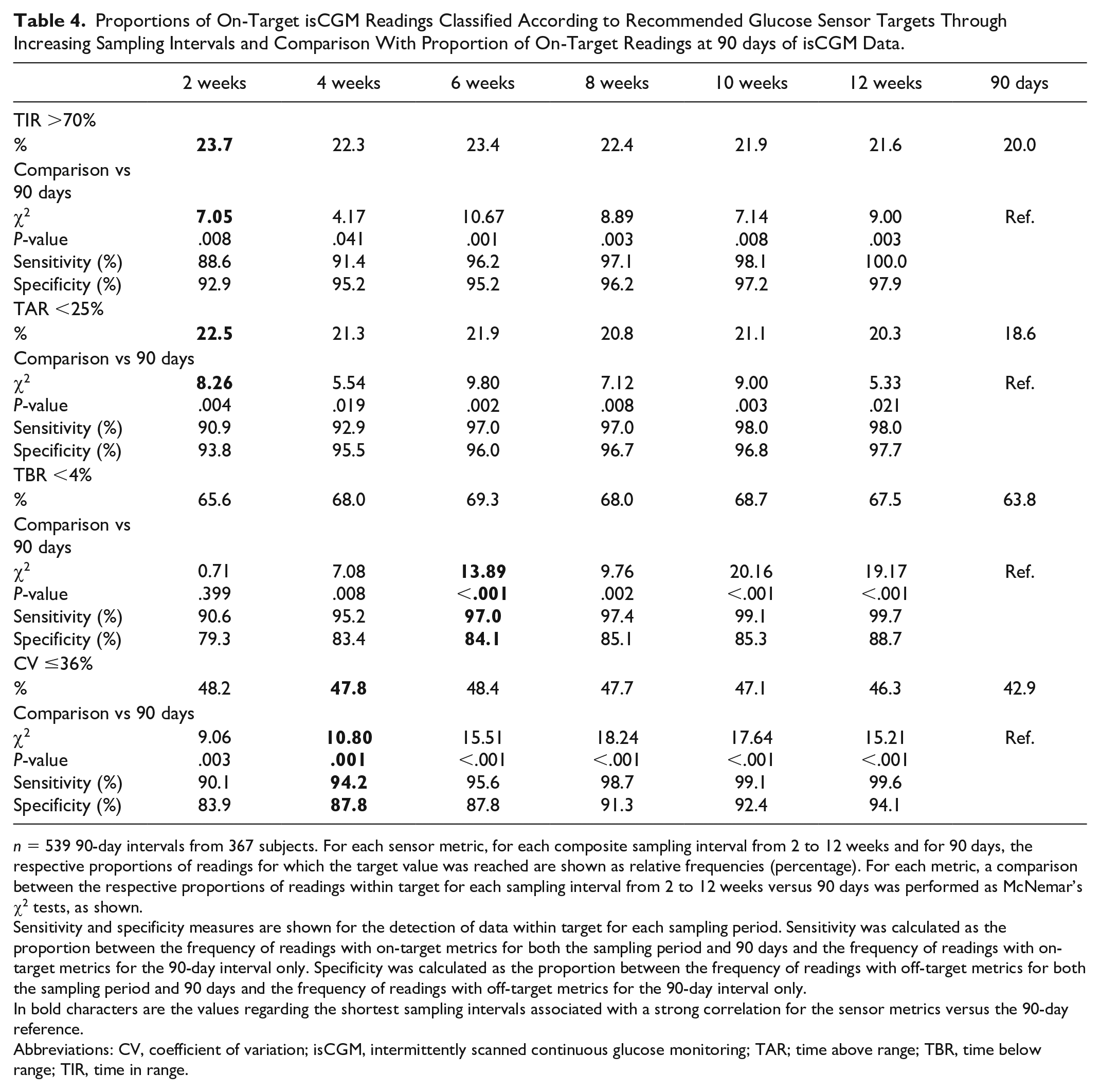
n = 539 90-day intervals from 367 subjects. For each sensor metric, for each composite sampling interval from 2 to 12 weeks and for 90 days, the respective proportions of readings for which the target value was reached are shown as relative frequencies (percentage). For each metric, a comparison between the respective proportions of readings within target for each sampling interval from 2 to 12 weeks versus 90 days was performed as McNemar’s χ2 tests, as shown.
Sensitivity and specificity measures are shown for the detection of data within target for each sampling period. Sensitivity was calculated as the proportion between the frequency of readings with on-target metrics for both the sampling period and 90 days and the frequency of readings with on-target metrics for the 90-day interval only. Specificity was calculated as the proportion between the frequency of readings with off-target metrics for both the sampling period and 90 days and the frequency of readings with off-target metrics for the 90-day interval only.
In bold characters are the values regarding the shortest sampling intervals associated with a strong correlation for the sensor metrics versus the 90-day reference.
Abbreviations: CV, coefficient of variation; isCGM, intermittently scanned continuous glucose monitoring; TAR; time above range; TBR, time below range; TIR, time in range.
Further Spearman’s correlations (Figure 3) between the proportions of isCGM readings for the four main metrics (TIR, TAR, TBR, and CV) within target at each sampling time versus 90 days confirmed strong correlations for each metric when considering the same time intervals for which R2 values ≥0.88 were previously achieved for the actual metrics and the strength of the correlations increased with the duration of sampling times.
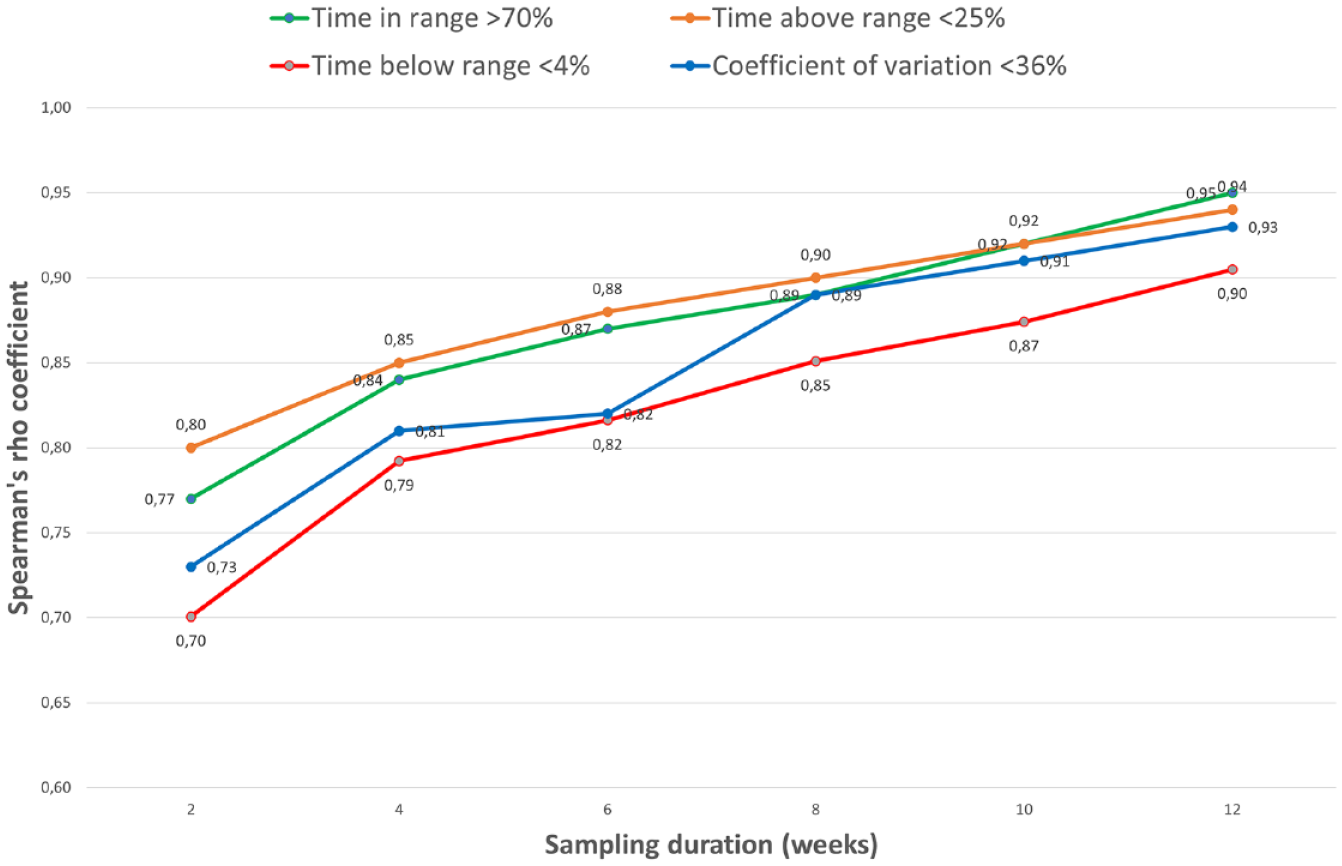
Spearman’s correlations for the proportion of intervals within target, according to the recommendations for each metric, for sampling intervals from 2 to 12 weeks compared with the 90-day interval. For each sensor metric, the Spearman’s coefficients of correlation (ρ) between the proportion of each composite sampling interval and isCGM data over the full 90-day period were plotted. Coefficient values were calculated from 539 isCGM intervals from 367 subjects. For all correlation analyses, P < .001.
Discussion
In this real-world retrospective study of second-generation isCGM devices, conducted in adult patients with T1D on MDI therapy, we confirmed that longer time samplings were associated to stronger correlations (R2) with the full 90-day interval8,10,15,18,19 and that, for most glucose metrics (TIR, TAR, MG), the shortest sampling interval to obtain a strong correlation with 90 days was two weeks.8,15,17,18 However, as suggested by previous works,7,17 our results showed the need for longer sampling intervals for CV (four weeks) and especially for TBR (six weeks). As a novelty of our work, we also found that four weeks were needed for a proper assessment of TITR. A further addition of our work was the examination of the increasing values of R2 through longer sampling intervals not only for the main sensor metrics, but also for the novel composite metric of GRI, with R2 trends which were similar to those previously reported by Shah. 13 Moreover, after stratifying by several clinical variables, TIR, TITR, and TAR, in each sampling period, showed weaker correlations to the full 90-day period in patients with older age, higher HbA1c, higher glucose variability, and lower sensor use. As for TBR, lower R2 values were found for higher HbA1c. Regarding GRI, lower R2 values were detected especially for lower CV.
Our results, however, were based on an R2 threshold ≥0.88, as suggested in the most recent review. 17 This threshold still is under debate 17 : Indeed, Riddlesworth proposed an R2 threshold ≥0.84, 8 and even lower values were used in a former study by Xing. 18 More recent studies opted for R2 around 0.88 to 0.90.15,19 As for GRI, in their previous work, Shah et al. considered as optimal a sampling duration, for which a R2 value of 0.88 was reached as well. 13 Furthermore, even if our results seem to be in accordance with previous studies regarding the acceptable time interval of two weeks to achieve an adequate threshold value of R2 for TIR,8,15,17 our study presented some relevant differences, as it only included patients on MDI treatment with only one type of isCGM, whereas most previous studies often involved several combinations of isCGM and real-time CGM (rtCGM) devices and MDI and Continuous Subcutaneous Insulin Infusion (CSII) therapies. 8 ,13-15, 18 Indeed, the use of isCGM, when compared to rtCGM, is associated to slightly lower TIR, to higher time spent in hyper and hypoglycemia, and, consequently, to higher glucose variability. 20 The latter factor may influence the time needed to obtain a close correlation with the fully complete set of data. 9 Also, our study purposely did not include patients on insulin pumps, which are associated to better and more stable glycemic control. 20 Furthermore, our data were collected in a real-world setting, thereby excluding a potential Hawthorne effect bias related to the awareness of being part of clinical trials. Along these same lines, in a recent work regarding pediatric patients with T1D, the optimal sampling interval to achieve metrics strongly related to 12-week metrics was four weeks, and, after stratifying by CGM devices (isCGM vs rtCGM), the cohort with isCGM showed a less relevant improvement of R2 at two and four weeks of data. 14 Moreover, in the recent work by Shah regarding the optimal sampling duration for GRI, 13 a two-week-long period was seen as optimal for the assessment of GRI: Indeed, according to our data, although four weeks resulted as an optimal choice, 14 days might still be a good sampling interval, since the R2 at two weeks was only slightly lower than the chosen cut-off value; however, it should be noted that the study population considered by Shah included people with a different rtCGM, which might partly explain the slight differences we found. 20 Thus, the fact that, despite of such differences, our results are still in accordance with the ones presented in such previous works might also be influenced by the differences in R2 thresholds,8,15,18 in the types and combinations of devices,8,9,13,14,18,19 in the age of sample population, 14 and by lower sample sizes.8,9,15,18,19 Moreover, the R2 calculated by Xing et al., 18 as well as the one calculated by subsequent authors,8,14,15 is not mathematically superimposable to the one computed in the present report. For such reasons, the aforementioned previous studies could be only partially compared with ours.
Regarding our subgroup analyses, when we focused only on data with CV over the recommended target of 36% 1 or with a low percentage of sensor use, or in the two oldest age quartiles, the threshold for a strong correlation was achieved at longer sampling intervals for TIR, TITR, and TAR, as well as for GRI (especially for lower CV), coherently with the known reduced need for data in case of lower glucose variability1,9 and in case of higher CGM use. 15 As to age, previous studies found similar R2 values among different age groups,14,18 and the differences we reported in this study are small, thus their clinical significance might be questioned. However, the fact that the oldest quartile showed the weakest correlations might support the need of greater attention when proposing such devices to older people. 21
In our study, the group with HbA1c ≤7.5%, when compared to the group with higher HbA1c, showed higher R2 values for TIR, TITR, TAR, TBR, MG, and GRI, especially for the shortest time intervals, so that for most metrics (except for TIR and GRI) two-week longer sampling times were needed to achieve strong correlations with 90 days when selecting data with higher HbA1c values. These results confirm and extend the report by Piona et al. in children/adolescents with T1D, 14 but they apparently are at variance with the paper by Xing et al. 18 However, the latter one included patients from 8 to 75 years old, of whom only 12% were on MDI, and it referred to CGM devices used in the first decade of this century, the accuracy and reproducibility of which was significantly worse than the isCGM of the present study.
Thus, our subgroup analyses support the view that longer (greater than two weeks) sampling intervals might be needed to assess glucose metrics related to the three-month time period in selected subgroups of patients based on their glucose variability, their sensor use, their age, and their HbA1c.
A further novelty of our study was to examine the implications of the selection of specific sampling intervals on the accuracy of classifying patients according to the suggested targets in the main metrics of TIR, TAR, TBR, and CV.1,7 Indeed, the strength of the correlation for the proportion of on-target readings at the various sampling intervals versus 90 days increased with longer time samplings, coherently with the actual metrics, but for each aforementioned metric, when selecting the shortest time intervals for which a strong correlation with 90 days was previously found, there still was a significant overestimate of the proportion of on-target isCGM readings, when compared to 90 days. It might be noted that, even when considering the lowest difference in the proportion of on-target readings versus 90 days, as for TBR at two weeks versus 90 days, it had the lowest specificity as well, resulting in an actual mismatch between on-target visits at the observed time interval versus 90 days. Thus, our results seem to suggest that, even when selecting a sampling interval which strongly correlates with the full three-month time period, there is still a clinically significant risk to overestimate the proportion of subjects with metrics within targets when comparing with 90 days. These observations do not diminish the clinical utility of assessing shorter time periods, which might still provide important clinical information for an effective evaluation of the patients according to their own situational needs and conditions. The trade-off may be a suboptimal representation of longer time periods when considering these sampling intervals.
Strengths
To the best of our knowledge, this is the largest single study about optimal CGM sampling intervals involving adult subjects with T1D on MDI using the same second-generation isCGM device. In our country, this is the most common condition by far and reflects a specific large part of the Italian population affected by T1D. A further strength of our study consisted in its retrospective real-world setting: This makes it a study of effectiveness, 22 thereby providing the type of data needed to assess the cost-effectiveness of this specific, quite common setting of insulin treatment and CGM in adult patients with T1D. An additional strength of our work is that it reports also the TITR, a metric which has not been taken into consideration in other previous works. Finally, our work evaluated how the choice of assumingly adequate time intervals to assess the main metrics at 90 days might still involve a persistent inaccuracy in the detection of readings within or out of the recommended targets, with possible consequences in the clinical practice.
Limitations
This study has some limitations, since only two-week intervals, and composite intervals made from their combinations, were studied, thereby neglecting shorter time periods. Moreover, all the available clinical visits of each patient, according to the inclusion criteria, were included, thereby endowing the subjects with more visits with more statistical weight. We tried to mitigate such imbalance by including only visits with sensor use >70%, similarly to previous studies, 18 but also by accounting for the multiple observations per subject according to data panel analyses, as previously detailed. Finally, using R2 values to assess the shortest optimal sampling intervals has its own limitations, not only because the threshold value of R2 still is to some degree an arbitrary selection, 17 but also because this “correlation approach” is strongly dependent on the total duration of the data set, thereby limiting the generalization of the results. However, although alternative methods have been explored to overcome such limitations,10,23 we adopted this approach for the sake of consistency and comparability with most previous studies on this same research topic. 8 ,13-15,17-19
Conclusions
In adult Caucasian patients with T1D using a second-generation isCGM device and a MDI insulin scheme, followed by diabetes specialists in Italy, a European country with a National Health Service, two weeks of isCGM data correlate well with three-month data for TIR, TAR, and MG, whereas four weeks are needed to reflect three months of data for TITR, CV, and GRI and six weeks are needed for TBR. However, in patients with higher glucose variability, higher HbA1c, older age, and lower sensor use, longer intervals of data collection might be needed to correlate effectively with the last three months. Furthermore, even when selecting an adequate time interval to assess glucose sensor metrics, there is still an overestimation of the proportion of on-target readings, when compared to the full 90-day interval. Thus, even if shorter sampling intervals might maintain their clinical value according to the specific patients’ needs and circumstances, clinicians who might may be willing to assess a whole three-month interval may rely on shorter than three-month time intervals to assess the relevant metrics of glucose control and to inform their clinical decision-making in this specific class of patients, while being cautious when they face patients with clinical characteristics which might suggest the choice of longer intervals and being aware of the possibility to overestimate the percentage of on-target metrics, even when selecting appropriate intervals.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241308564 – Supplemental material for Different Times for Different Metrics: Predicting 90 Days of Intermittently Scanned Continuous Glucose Monitoring Data in Subjects With Type 1 Diabetes on Multiple Daily Injection Therapy. Findings From a Multicentric Real-World Study
Supplemental material, sj-docx-1-dst-10.1177_19322968241308564 for Different Times for Different Metrics: Predicting 90 Days of Intermittently Scanned Continuous Glucose Monitoring Data in Subjects With Type 1 Diabetes on Multiple Daily Injection Therapy. Findings From a Multicentric Real-World Study by Alessandro Csermely, Nicolò D. Borella, Anna Turazzini, Martina Pilati, Sara S. Sheiban, Riccardo C. Bonadonna, Roberto Trevisan, Maddalena Trombetta and Giuseppe Lepore in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AD, absolute difference; ADA, American Diabetes Association; CGM, continuous glucose monitoring; CV, coefficient of variation; HbA1c, hemoglobin A1c; IQR, interquartile range; isCGM, intermittently scanned CGM; MAD, median absolute difference; MDI, multiple daily injection; MG, mean glucose; rtCGM, real-time CGM; SD, standard deviation; T1D, type 1 diabetes; TAR, time above range; TBR, time below range; TIR, time in range; TITR, time in tight range.
Author Contributions
AC and NDB designed the study. AC, NDB, AT, MP, and SSS collected the clinical data. AC, NDB, MT, GL, and RT contributed to data interpretation. AC performed the statistical analyses. AC and NDB wrote the first draft of the article. MT, GL, RT, and RCB contributed to further revisions of the article. All the authors contributed to the final version of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by research grants from the University of Verona (to MT and AC), from the project for “AdvaNced Technologies for Human-centred Medicine” (ANTHEM; to RT), and by “FIL” funds for research from the University of Parma (to RCB).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
