Abstract
Objectives:
Time in range (TIR, 70-180 mg/dL) is an established marker of glycemic control. More recently, time in tight range (TTR, 70-140 mg/dL) has been proposed as well. The aim of this study was to examine the relationship between TIR, TTR, and HbA1c in youth and young adults with type 1 diabetes (T1D) in the German/Austrian/Luxembourgian/Swiss Diabetes Prospective Follow-up (DPV) registry.
Methods:
Data of youth and young adults aged ≤25 years with T1D for >3 months, documented in the DPV registry between 2019 and 2022 were analyzed. The most recent available HbA1c and corresponding continuous glucose monitoring (CGM) profiles in the 12 preceding weeks with at least 80% completeness were included. Associations were investigated using correlation and adjusted regression models.
Results:
1901 individuals (median age 14.0 years [IQR 10.4-16.9]) were included in the analysis. TIR and TTR correlated strongly, r = 0.965 (95% CI [0.962, 0.968]), P < .001. TTR estimates predicted from TIR were significantly higher in the group with high coefficient of variation (CV group ≥ 36%), P < .001. Correlations between TIR or TTR and HbA1c were both strong, r = −0.764 (95% CI [−0.782, −0.745]) and r = −0.777 (95% CI [−0.795, −0.759]), both P < .001, with no significant difference (P = .312) However, adjusted regression models indicated a slightly better fit for the prediction of HbA1c from TIR compared with TTR.
Conclusions:
Based on large, real-world data from a multinational registry, TIR and TTR correlated strongly, and both showed a good prediction of HbA1c. TTR estimates predicted from TIR were significantly higher in people with high glucose variability (CV).
Keywords
Introduction
Over the past decade, the use of continuous glucose monitoring (CGM) systems has increased exponentially in everyday management of type 1 diabetes (T1D) across all age groups.1-5 The CGM-derived marker time in range (TIR, percentage of glucose values between 70 and 180 mg/dL) has thus become an important component in the assessment of metabolic control.6,7 It has been shown that lower TIR is related to the progression of microvascular complications.8,9 Current consensus guidelines recommend an HbA1c <7% and a TIR of >70% as optimal metabolic control.6,10 However, even in people with T1D meeting these targets, the cardiovascular mortality is twofold higher than in individuals without T1D. 11
Achieving the recommended treatment goals is still a major challenge for children and adolescents living with T1D. 12 Recent advances in diabetes technology, such as automated insulin delivery (AID) systems, allow to attain a TIR of 70% or higher. 13 Children and adolescents without diabetes spend 96% of the time in the glucose range between 70 and 140 mg/dL. 14 Therefore, another CGM-derived marker, Time in Tight Range (TTR, percentage of glucose values spent between 70 and 140 mg/dL) has been proposed as a new marker to further assess and improve glycemic control and reduce the risk of micro- and macrovascular complications.
Time in range and HbA1c show a negative correlation. 15 An analysis of TIR and HbA1c from multiple randomized controlled trials in adults with T1D revealed a wide spread of HbA1c levels associated with a specific level of TIR. 16 This relationship has only been studied in few reports on T1D youth.17,18 For the proposed new marker TTR, the aforementioned Swedish study that examined CGM profiles of just over 100 children and adolescents described a nonlinear relationship between TTR and HbA1c. 17 A recent investigation assessing the relationship between TIR and TTR using sensor data from different studies in T1D or T2D in adults and children found a nonlinear relationship between TIR and TTR. 19 Furthermore, the relationship varied according to the coefficient of variation (CV) and time below range (TBR). 19 While there are studies that TTR may serve as an important additional marker of glycemic control in children and adolescents with T1D,20-22 these associations (TIR vs TTR and HbA1c vs TIR/TTR) have not been studied in larger numbers of children, adolescents, and young adults with T1D.
Hence, the aim of this study was to examine the relationship between TTR, TIR, and HbA1c and to examine to what extent HbA1c can be predicted from TIR and TTR in youth and young adults with T1D.
Methods
Study Design
This observational cross-sectional study analyzed data from the population-based Diabetes Prospective Follow-Up (DPV) registry, a collaborative database that collects information from people with diabetes including CGM profiles 23 from diabetes centers in Germany, Austria, Luxembourg, and Switzerland for quality improvement, and health care research. Data are transferred pseudonymized to the Institute of Epidemiology and Medical Biometry at Ulm University in Germany twice every year. Implausible data are reported back to each participating center for validation and/or correction. Subsequently, prior to analysis, data are completely anonymized. For this analysis, the March 2023 data set was used.
Study Population
Data of children, adolescents and young adults aged 0.5 to 25 years with T1D for more than three months and documented insulin therapy between 2019 and 2022 were included. Further inclusion criteria comprised availability of CGM profiles with a CGM data collection rate of at least 80% over 12 weeks preceding an HbA1c measurement. The most recent available HbA1c and corresponding CGM profiles were identified and analyzed.
Variables
Demographic data analyzed included age, duration of diabetes, gender, migration status (defined as birthplace outside Austria, Germany, Luxembourg, or Switzerland for the individual with T1D or for one or both parents), BMI (kg/m2) z-score using reference data from the German obesity working group in childhood and adolescence (AGA), 24 insulin therapy (multiple daily injections [MDI], continuous subcutaneous insulin infusion [CSII] and AID, also called hybrid closed-loop [HCL] systems). In the present analysis, CSII and AID use has been defined as any use in the 12-week period selected. People using CSII with the predictive low glucose suspend (PLGS) or low glucose suspend (LGS) algorithm were categorized to the CSII group. AID users included commercially available HCL systems and open artificial pancreas systems (OAPS). Glycemic variables included HbA1c (% or mmol/ mol) and CGM metrics calculated from available CGM profiles: TIR, TTR, time below range (TBR, percentage of glucose values below 70 mg/dL), time above range (TAR, percentage of glucose values time above > 180 mg/dL), mean sensor glucose (mg/dL), and CV (standard deviation of sensor glucose divided by mean sensor glucose).
Statistical Analysis
Descriptive statistics are given as median (lower and upper quartile) for continuous variables and percentage for binary variables. Differences between subgroups were compared using Wilcoxon rank sum test for continuous outcomes or χ2 test for binary outcomes. P-values were adjusted for multiple testing using the Bonferroni-Holm method.
Spearman correlation coefficients for TIR, TTR, and HbA1c were calculated for the overall cohort and for subgroups including gender, age (<6 years, 6-<12 years, 12-<18 years, and 18-25 years), insulin therapy (MDI, CSII, AID), HbA1c groups (<7%, 7%-<8.5%, ≥8.5%), and diabetes duration (0.25-2 years and > 2 years).
The relationship between TTR and TIR was further explored fitting locally estimated/weighted Scatterplot Smoothing (LOESS) lines, overall and for people in different groups as specified above. Further analysis explored the relationship stratified by CV ≥/<36%. In addition, we performed beta-regression models to analyze the association between TTR und TIR, adjusting for gender, age, diabetes duration, insulin therapy and CV groups. We also investigated the effect of CV on the TTR-TIR association by including a CV by TIR interaction term in the model. Using these regression analyses, we calculated the median adjusted estimated TTR for TIR subgroups.
To estimate HbA1c depending on TIR or TTR, quadratic polynomial regression models were applied adjusting for gender, age, diabetes duration, and insulin therapy groups. Regression models were compared using the Akaike information criterion (AIC) and the Bayesian information criterion (BIC). Using these regression analyses, we calculated the median adjusted estimated HbA1c for TIR and TIR subgroups.
A two-sided P-value <.05 was considered statistically significant. All analyses were performed with SAS version 9.4 (build TS1M8; SAS Institute, Inc., Cary, NC) on a windows server mainframe.
Results
Study Population
Of 45 426 people diagnosed with T1D for more than three months and aged 25 years or younger in DPV, CGM data were available in 7321 up to 12 weeks before a HbA1c measurement. 1901 individuals had a HbA1c value and CGM profiles with ≥80% completeness over the preceding 12 weeks and were included, see Supplemental Figure 1. More than 70% of the CGM profiles date from the calendar year 2022. Median (lower and upper quartile) age of the cohort was 14.0 years (10.4-16.9) and median diabetes duration 4.4 years (2.1-8.0), 1141 (60%) were insulin pump users. Median TIR, TTR, and HbA1c were 57% (45–67), 35% (27-44), and 7.4% (6.8-8.0), respectively. Table 1 depicts demographic and glycemic parameters of the study population.
Demographics and Glycemic Parameters.

Values are presented as median (lower; upper quartile) or n (%).
Time in range and TTR were highest in the 598 young people achieving current international targets with a HbA1c < 7%: TIR was 69% (62-77) and TTR 46% (30-54) respectively. Furthermore, AID users and children/adolescents with shorter diabetes duration (0.5-2 years) had highest TIR and TTR values, Supplemental Table 1. In 122 young people with excellent glycemic control with a TIR of >80% (median TIR 85% [82-89] and a median HbA1c of 6.2% [5.7-6.5]), TTR was 63% (59-71) and 98% achieved TTR >50% (results not shown). Within this specific group, median CV was 26.8% (24.5-29.3), TBR 2% (1-4), and average glucose was 131 mg/dL (124-138).
Association Between TIR and TTR
Time in range and TTR correlated strongly, r = 0.965 (95% CI [0.962, 0.968]), P < .001. As indicated by the LOESS curve on Figure 1, the association of TTR depending on TIR was not strictly linear and the slope was higher for people already achieving high levels of time in range, in TIR levels above 80% with corresponding TTR levels above 60%.
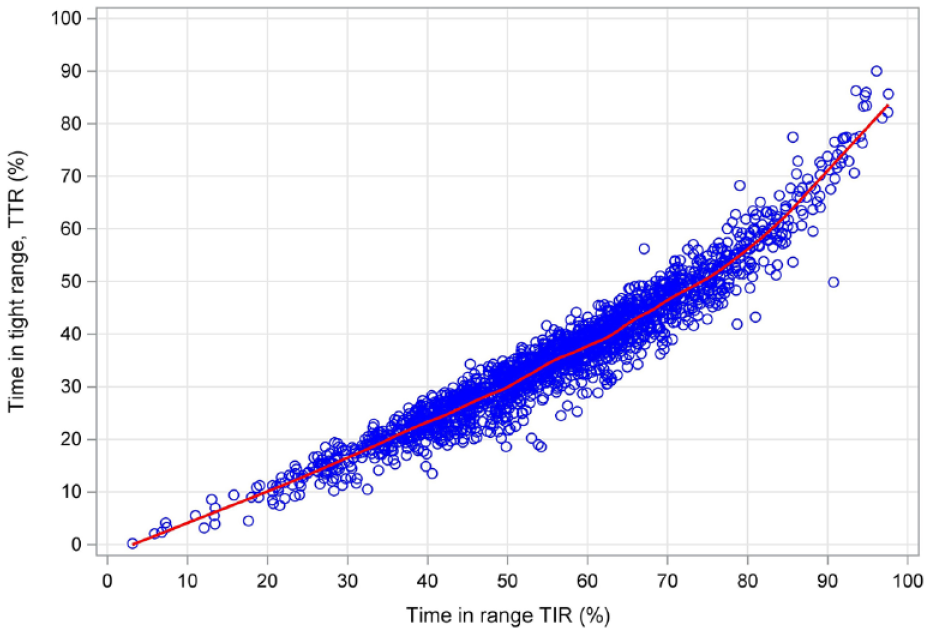
Locally estimated regression curve (LOESS) for the association between TIR (70-180 mg/dL) and TTR (70-140 mg/dL).
Time in range and TTR also correlated strongly in all subgroups examined, Supplemental Table 1. The LOESS curves (not shown) assessing the relationship of TTR with TIR were similar after stratification for gender, different age groups, treatment groups, and HbA1c groups.
Table 2 shows predicted TTR for given TIR ranges derived from beta-regression adjusted for gender, age, diabetes duration, insulin therapy and CV groups. For a TIR between 70% and 75% predicted TTR was 47.9% (47.5-48.4).
Median Predicted TTR for Given TIR Groups.

Predicted TTR is derived from beta-regression model adjusted for gender, age, diabetes duration, therapy, and CV groups.
Association Between TIR and TTR Depending on CV
The LOESS curve for the association between TIR and TTR stratified by CV groups indicated that CV >= 36 % resulted in a higher TTR for a given TIR, Figure 2a. Beta regression confirmed that TTR estimates depending on TIR adjusted for CV were significantly higher in the CV group ≥ 36%, P < .001, Figure 2b. However, the increase in TTR for 10% increase in TIR was almost identical in both CV groups: Relative Risk (RR) for CV <36: 1.278 (95% CI [1.275, 1.282]) and RR for CV >=36: 1.276 (95% CI [1.269, 1.284]). The ratio of both RRs was 0.999 (95% CI [0.992, 1.005]), P = .661. Accordingly, the ratio of TTR in CV < vs ≥ 36% was similar at very low TIR (at minimum TIR of 18.7%: 1.160; 95% CI [1.144, 1.176]) and very high TIR (at maximum TIR 77.5%: 1.150; 95% CI [1.143, 1.157]). The effect of the CV group on the TTR—TIR relationship remained similar in each gender and age, diabetes duration, and insulin therapy group.
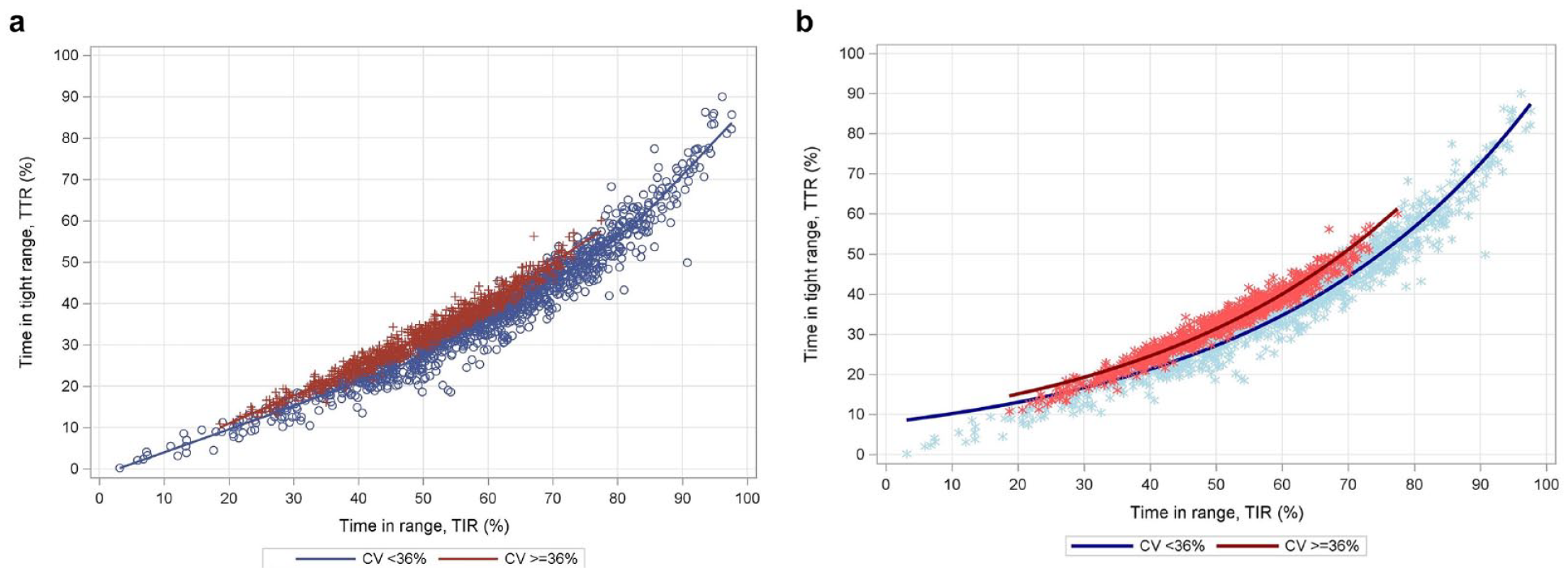
(a) Locally estimated regression curve (LOESS) for the association between TIR (70-180 mg/dL) versus TTR (70-140 mg/dL) stratified by Coefficient of Variation (CV) ≥ 36%/< 36% and (b) Beta-regression curve for the association between TIR (70-180 mg/dL) versus TTR (70-140 mg/dL) by interaction with CV ≥ 36%/< 36%.
Associations of TIR and TTR With HbA1c
Time in range and TTR were negatively correlated with HbA1c, r = −0.764 (95% CI [−0.782, −0.745]), and r = −0.778 (95% CI [−0.795, −0.759]), both P < .001, respectively, with no significant difference (P = .312), Figure 3. Quadratic polynomial regression adjusted for gender, age, insulin therapy, and diabetes duration both predicted HbA1c well, according to AIC and BIC the model with HbA1c prediction from TIR indicates a slightly better fit (TIR vs TTR: AIC 3696.86 vs 3749.80, BIC 3747.85 vs 3810.80, respectively).
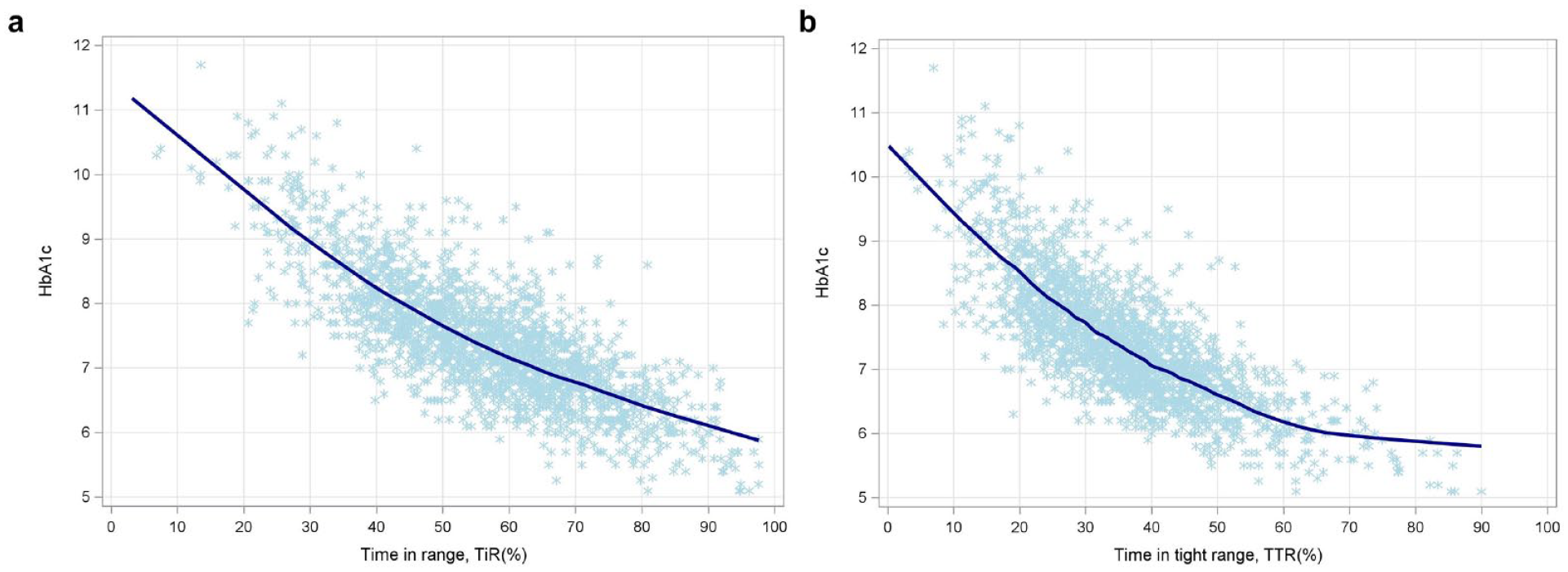
Relationship between HbA1c and TIR and TTR, respectively. Estimation of HbA1c from quadratic regression depending on (a) TIR and (b) TTR.
A TIR between 70% and 75%, corresponds to a median HbA1c 6.7% (95% CI [6.7-6.8]), whereas a TTR between 50% and 55% corresponds to a median predicted HbA1c of 6.5% (95% CI [6.4, 6.6]). Table 3a shows median predicted HbA1c for given TIR groups and Table 3b for given TTR groups. Supplemental Table 2 shows corresponding median TIR and TTR values for predicted HbA1c values.
Median Predicted HbA1c for Given TIR Groups Derived From Quadratic Polynomial Regression Model Adjusted for Gender, Age, Diabetes Duration, and Therapy Groups.

Median [95% CI] Predicted HbA1c Levels (%) for Given TTR Groups Derived From Quadratic Polynomial Regression Model Adjusted for Gender, Age, Diabetes Duration, and Therapy Groups.

Discussion
This study showed a strong correlation between TIR and TTR in a large real-world sample in youth and young adults with T1D. The relationship is not strictly linear, with an increase of the slope (higher TTR depending on TIR) in those achieving already high TIR and TTR also reflected by HbA1c <7%. The relationship was similar regardless of gender, age group, and insulin therapy. Regression analysis revealed that TTR estimates derived from TIR were significantly higher in the group with high glycemic variability (CV ≥ 36%). Of note, people with very high TIR (>80%) or TTR (>60%) rarely have high variability (Figure 2a and b). These results are in line with a study of Beck et al 19 that investigated the relationship of TIR and TTR in data collected in different clinical studies of participants with type 1 diabetes and type 2 diabetes. A recent study analyzing a large set of real-world data from CGM users in people with T1D and T2D also confirmed that the TIR-TTR relationship is varying depending on CV. 25
Secondly, this study showed a strong negative nonlinear correlation between TTR/TIR and HbA1c, both TIR and TTR predicted HbA1c well, with a minimally better fit of the model of HbA1c prediction from TIR. This may be explained by the fact that TIR is derived from a larger range of glucose values and likewise, HbA1c is derived from the overall spread of glucose values such as TIR, TAR and TBR. A recent study evaluating the relationship between TIR and HbA1c showed a similar negative correlation. 18 A Swedish study evaluated TTR and the relationship to HbA1c in children and adolescents and found a curvilinear relationship. 17 Similar to our study, they showed that a TTR of 50% corresponds to a HbA1c-value of 6.5%. 17
Median TTR in our study population was around 35%, comparable with TTR reported in other clinical studies. 20 Similar to our study, highest TTR was found in children and adolescents using an AID systems.20,21 A recent real-world analysis in users of an advanced hybrid closed-loop system reports TTR values of almost 49% on average. 26
Similar to the Swedish study, a treatment goal of TTR > 50% is suggested.17,26 Currently, most children and adolescents with T1D do not reach this proposed target. With the increasing use of AID systems, TTR may serve as an additional marker of glycemic control that may be advantageous for education and motivation to achieve physiological glucose levels. However, negative consequences of focusing on this narrow target such as increased diabetes distress or diabetes burden are also possible, therefore studies that investigate the psychosocial impact of using TTR as an additional and more stringent glycemic outcome are warranted.
In addition to the particular interest in TTR in studies investigating the use and efficacy of AID systems, TTR can also serve as an important CGM-derived marker in people with stage 1 and 2 T1D, for example, to predict progression to clinical diagnosis of T1D. 27 Furthermore, during pregnancy, international consensus guidelines regarding glycemic goals already recommend a target range of 63 to 140 mg/dL corresponding to near-normal, physiological target range. 6 It may be advantageous to define a universally valid near-normal, physiological target range, regardless of the stage or type of diabetes.
To our knowledge, this is the first study examining the relationship of TIR or TTR and HbA1c in a large multicenter, multinational real-world sample in youth and young adults with T1D. A strength of this study is the large real-world dataset of children, adolescents and young adults with T1D and evaluation of CGM profiles with at least 80% of data completeness over a period of 12 weeks. Compared with other data sets,19,25 the study population is more homogenous, analyzing data from children, adolescents and young adults with T1D only. A limitation of this study may be that the relationship between TIR and TTR potentially also depends on other factors not analyzed such as C-peptide levels, type of AID systems or AID settings, nutrition, physical activity or psychosocial factors. Furthermore, the association of TIR and TTR and the impact of TTR on micro- and macrovascular complications needs to be assessed. Data from the Swedish registry did not show a difference in the risks of retinopathy and microalbuminuria in people with HbA1c levels below 6.5% and between 6.5% and 6.9%. 28 Furthermore, in the DCCT data, the risk of microvascular complications is not substantially increased up to a HbA1c level above 8%. 29
A recent study examining the association of TTR and TIR with the presence of microvascular complications and cerebrovascular insults in adults with type 1 diabetes found an inverse association with the presence of microvascular complications and cerebrovascular accidents. 30 Future research will show, if TTR is a better predictor than TIR for micro- and macrovascular complications.
Conclusions
In conclusion, based on real-world data from a multicenter, multinational registry, TIR and TTR correlated strongly and the relationship to HbA1c showed a good prediction of HbA1c from TIR and TTR in youth with T1D. TTR estimates predicted from TIR were significantly higher in people with high CV. This emphasizes the complex relationship of TIR and TTR with other CGM-derived parameters. While there seems no additional benefit of using TTR instead of TIR to assess glycemic control, TTR might be used as additional, physiological target to provide more information specifically for people with very high TIR and near-normal average glucose levels such as individuals with stage 1 and 2 diabetes or AID users; groups expected to grow over the next years. Furthermore, TTR may have advantages for education purposes and motivation.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241288870 – Supplemental material for What is the Relationship Between Time in Range, Time in Tight Range, and HbA1c in Youth and Young Adults With Type 1 Diabetes? Results From the German/Austrian/Luxembourgian/Swiss Diabetes Prospective Follow-Up Registry
Supplemental material, sj-docx-1-dst-10.1177_19322968241288870 for What is the Relationship Between Time in Range, Time in Tight Range, and HbA1c in Youth and Young Adults With Type 1 Diabetes? Results From the German/Austrian/Luxembourgian/Swiss Diabetes Prospective Follow-Up Registry by Marie-Anne Burckhardt, Marie Auzanneau, Joachim Rosenbauer, Elisabeth Binder, Jantje Weiskorn, Melanie Hess, Christof Klinkert, Joaquina Mirza, Lara-Sophie Zehnder, Sandra Wenzel, Kerstin Placzek and Reinhard W. Holl in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank A. Hungele and R. Ranz for DPV documentation software support. The authors acknowledge all participating centers in Germany, Austria, Luxembourg, and Switzerland that contributed data to this analysis, a list with all participating centers can be found in the supplement.
Abbreviations
AID, automated insulin delivery; CGM, continuous glucose monitoring; CV, coefficient of variation; GMI, glucose management indicator; HbA1c, glycosylated Hemoglobin; T1D, type 1 diabetes; TAR, time above range; TBR, time below range; TIR, time in range; TTR, time in tight range.
Author Contributions
MAB: Contribution to design, interpretation of the results, manuscript writing (first draft and review and editing).
MA: Contribution to design, development of the statistical analysis plan, data analysis, interpretation of the results, and critical review of the manuscript. JR: statistical analysis plan, interpretation of the results, and critical review of the manuscript. EB, JW, MH, CK, JM, LSZ, SW and KP: critical review and editing of the manuscript.
RWH: Contribution to design, development of the statistical analysis plan, interpretation of results, critical review of manuscript. Coordination of DPV registry.
All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MAB received honoraria for lectures from Medtronic not related to this publication. JW received honoraria for lectures from Amryt Pharma not related to this publication. MA, JR, EB, MH, CK, JM, LSZ, SW, KP, and RWH have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DPV was supported through the German Federal Ministry for Education and Research within the German Center for Diabetes Research (DZD, grant number 82DZD14E03), the German Robert Koch Institute (RKI), and the German Diabetes Association (DDG).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
