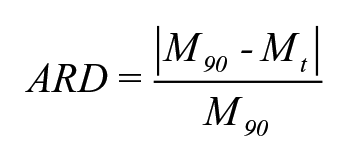Abstract
Background and Objective:
It is not clear how the short-term continuous glucose monitoring (CGM) sampling time could influence the bias in estimating long-term glycemic control. A large bias could, in the worst case, lead to incorrect classification of patients achieving glycemic targets, nonoptimal treatment, and false conclusions about the effect of new treatments. This study sought to investigate the relation between sampling time and bias in the estimates.
Methods:
We included a total of 329 type 1 patients (age 14-86 years) with long-term CGM (90 days) data from three studies. The analysis calculated the bias from estimating long-term glycemic control based on short-term sampling. Time in range (TIR), time above range (TAR), time below range (TBR), correlation, and glycemic target classification accuracy were assessed.
Results:
A sampling time of ten days is associated with a high bias of 10% to 47%, which can be reduced to 4.9% to 26.4% if a sampling time of 30 days is used (P < .001). Correct classification of patients archiving glycemic targets can also be improved from 81.5% to 91.9 to 90% to 95.2%.
Conclusions:
Our results suggest that the proposed 10-14 day CGM sampling time may be associated with a high correlation with three-month CGM. However, these estimates are subject to large intersubject bias, which is clinically relevant. Clinicians and researchers should consider using assessments of longer durations of CGM data if possible, especially when assessing time in hypoglycemia or while testing a new treatment.
Keywords
Introduction
The use of continuous glucose monitoring (CGM) sensor technology has provided clinicians and scientific investigators with an accurate tool to evaluate diabetes control beyond the use of HbA1c and spot measurement of glucose.1-3 This is particularly useful in assessing the effects of new treatments on the percentage of glucose values within target ranges (TIR) and time spent in hyperglycemia (TAR) and hypoglycemia (TBR).4-6
However, the effective use of CGM technology in routine clinical practice remains relatively low. This may be due to the need for a more standardized use of CGM; therefore, recently, an international consensus on the use of CGM was published. 7 The general recommendation is to sample CGM for 10 to 14 days, and targets for glycemic control should be TIR >70%, TAR <25%, and TBR <4%. The recommendation on sample time is based on several studies pointing toward the fact that the correlation between long-term CGM sampling (three months) and short-term CGM sampling does not improve substantially beyond 14 days of sampling.8,9 However, the correlation has several limitations and does not provide a practical interpretation of how large a bias certain sampling times could yield.10,11 Moreover, recent studies have shown that the proposed sampling time is not optimal for all types of patients.12,13 In particular, assessing the target for hypoglycemia could be heavily biased when using a sample time of 14 days. 12
It is still not clear how a selected CGM sampling time could influence the bias in estimating long-term glycemic control. A large bias could, in the worst case, lead to incorrect classification of patients achieving targets, nonoptimal treatment, and false conclusions about the effect of new treatments. Therefore, we sought to investigate the impact of CGM sample time in relation to the expected bias in glycemic CGM target measurements and classification.
Materials and Methods
To clarify the limitations of different CGM sampling times, this study pooled patients from three previous studies with long-term use of CGM in type 1 diabetes patients. The methods and results of the three studies for which data were utilized in the analyses have been published.14-16 The first study (CITY) 15 aimed to determine the effect of CGM on glycemic control in adolescents and young adults with type 1 diabetes. The second study (REPLACE-BG) 14 aimed to determine whether the use of CGM without confirmatory blood glucose monitoring measurements is safe and effective in adults with well-controlled type 1 diabetes. The third study (WISDM) 16 aimed to determine whether CGM is effective in reducing hypoglycemia compared with standard blood glucose monitoring in older adults with type 1 diabetes.
For this study, the inclusion criteria were a useful CGM wear period for three months (defined as 90 days) with a successful number of days CGM worn ≥80%. We included a total of 329 patients from the three studies (CITY n = 106; REPLACE-BG n = 113; WISDM n = 110). The idea of using these three study populations was to pool a large heterogeneous population that could reflect the differences in the general population of type 1 diabetes patients from young to old. Although some patients had multiple nonoverlapping three-month periods with suitable data, the analysis only included one period from each patient to avoid biasing the results.
We calculated the following metrics from CGM sampling intervals 1 to 90 days: time in range (TIR): % of readings and time 70 to 180 mg/dL (3.9-10.0 mmol/L), time above range (TAR): % of readings and time >180 mg/dL (>10.1 mmol/L), and time below range (TBR): % of readings and time <69 mg/dL (> 3.8 mmol/L). The absolute relative difference % (ARD) was calculated from each sampling period (Mt) compared with the full sampling period of three months (M90). The concept of calculating ARD was to give a realistic estimation of how much bias could be present when interpreting a metric from a specific sampling interval. The results are presented as the median and 25th/75th percentile due to the nonparametric distributions:
The correlation coefficient for each sampling period compared with the full sampling period was calculated. The analysis also included a classification assessment, analyzing whether each sampling interval would result in a correct classification of the patients achieving the proposed clinical targets for each metric compared with a full sampling period of three months. 7 The targets were defined as >70% TIR, <25% TAR, and <4% TBR.
Results
The included patients had a median age of 40 [20; 65] years, range of 14 to 86 years. A total of 49.9% of the patients were male, and the median duration of diabetes was 18 years [8; 33], range of 0 to 64 years. Table 1 shows the characteristics of the three study cohorts included in the analysis.
Characteristics of the Three Study Populations Included in the Analysis.
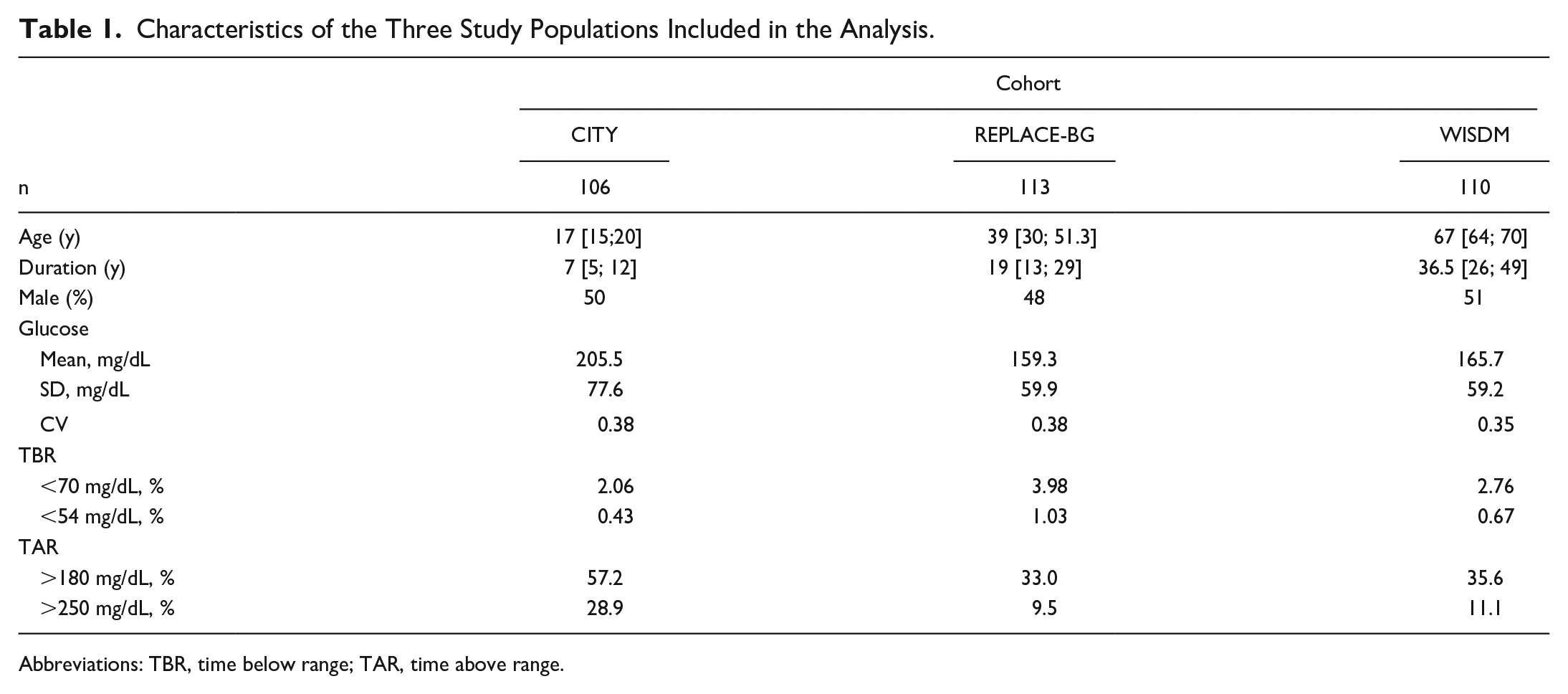
Abbreviations: TBR, time below range; TAR, time above range.
The results are presented in Table 2 and are graphically represented in Figures 1 and 2. As expected, the more days used for sampling, the less bias and the higher the correlation the estimate introduces. Notably, the median ARD % was significantly reduced when comparing samples from 10 to 20 or 30 days. In addition, as illustrated in Figure 1 from the boxplots, there is a large variance in the bias between patients. Looking at the TIR bias from 10 to 30 days of sampling, the ARD is reduced from 10.2 [4.5; 17.6] to 4.9 [2.5; 10.7]. To exemplify this, when achieving an estimated 70% TIR from 10 days of sampling, the long-term TIR would, on average, be 70% ± 7.1. However, it is also important to emphasize the large gap between the 25th and 75th percentiles of 4.5 to 17.6.
Pooled Results From the Combined Studies.
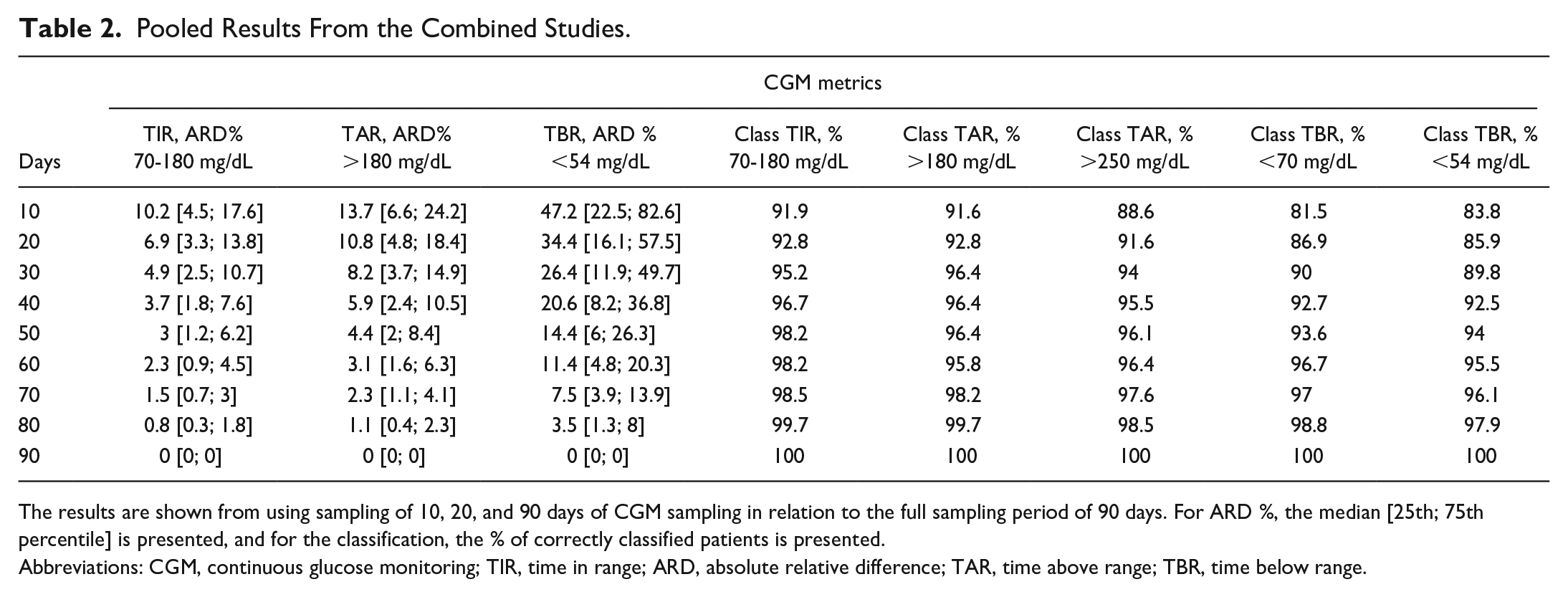
The results are shown from using sampling of 10, 20, and 90 days of CGM sampling in relation to the full sampling period of 90 days. For ARD %, the median [25th; 75th percentile] is presented, and for the classification, the % of correctly classified patients is presented.
Abbreviations: CGM, continuous glucose monitoring; TIR, time in range; ARD, absolute relative difference; TAR, time above range; TBR, time below range.
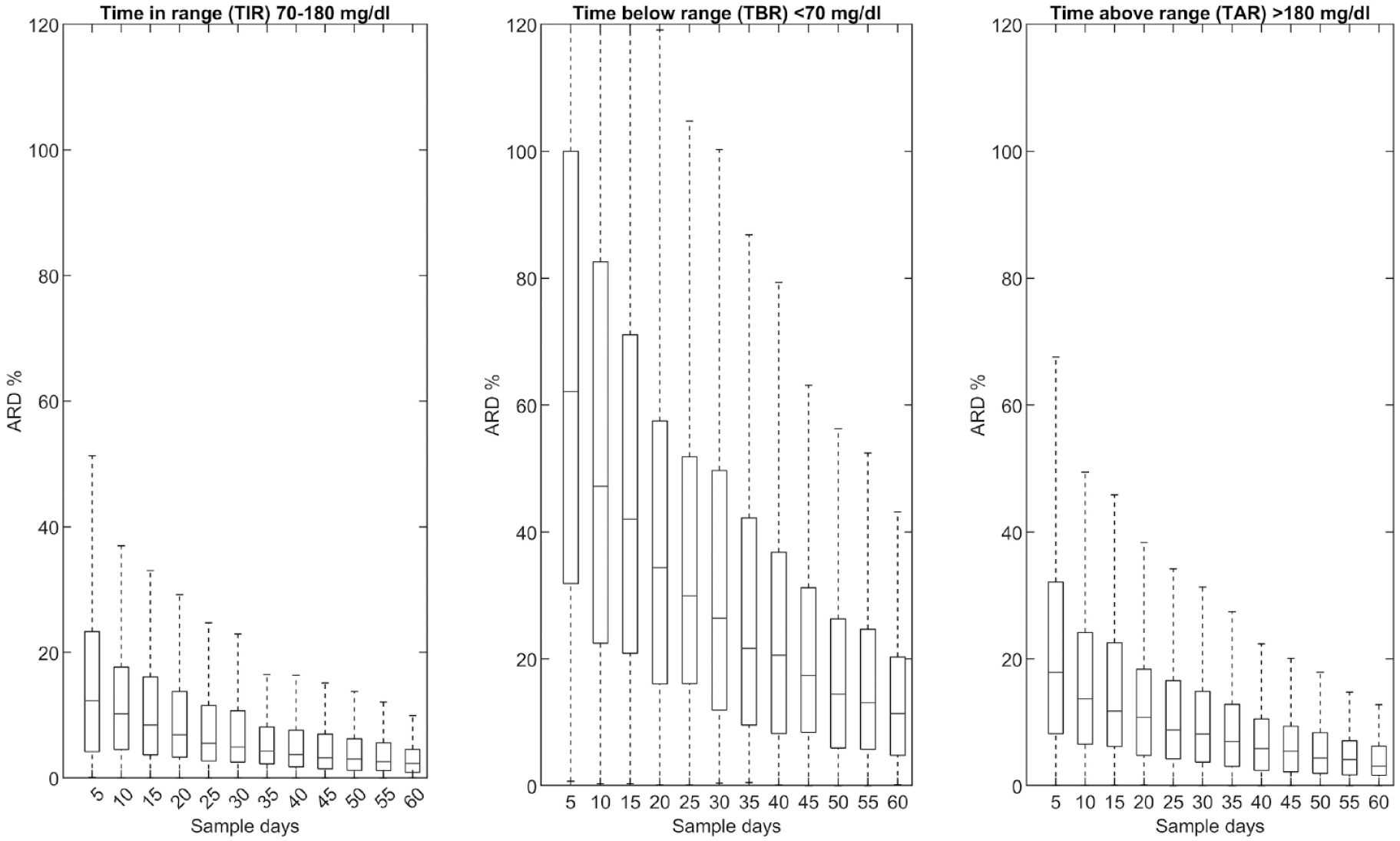
Bias as relative absolute difference (ARD) % associated with sample time compared with long-term sampling (90 days). The bias is presented as a boxplot where the horizontal line is the median and the box is the 25th to 75th percentile.
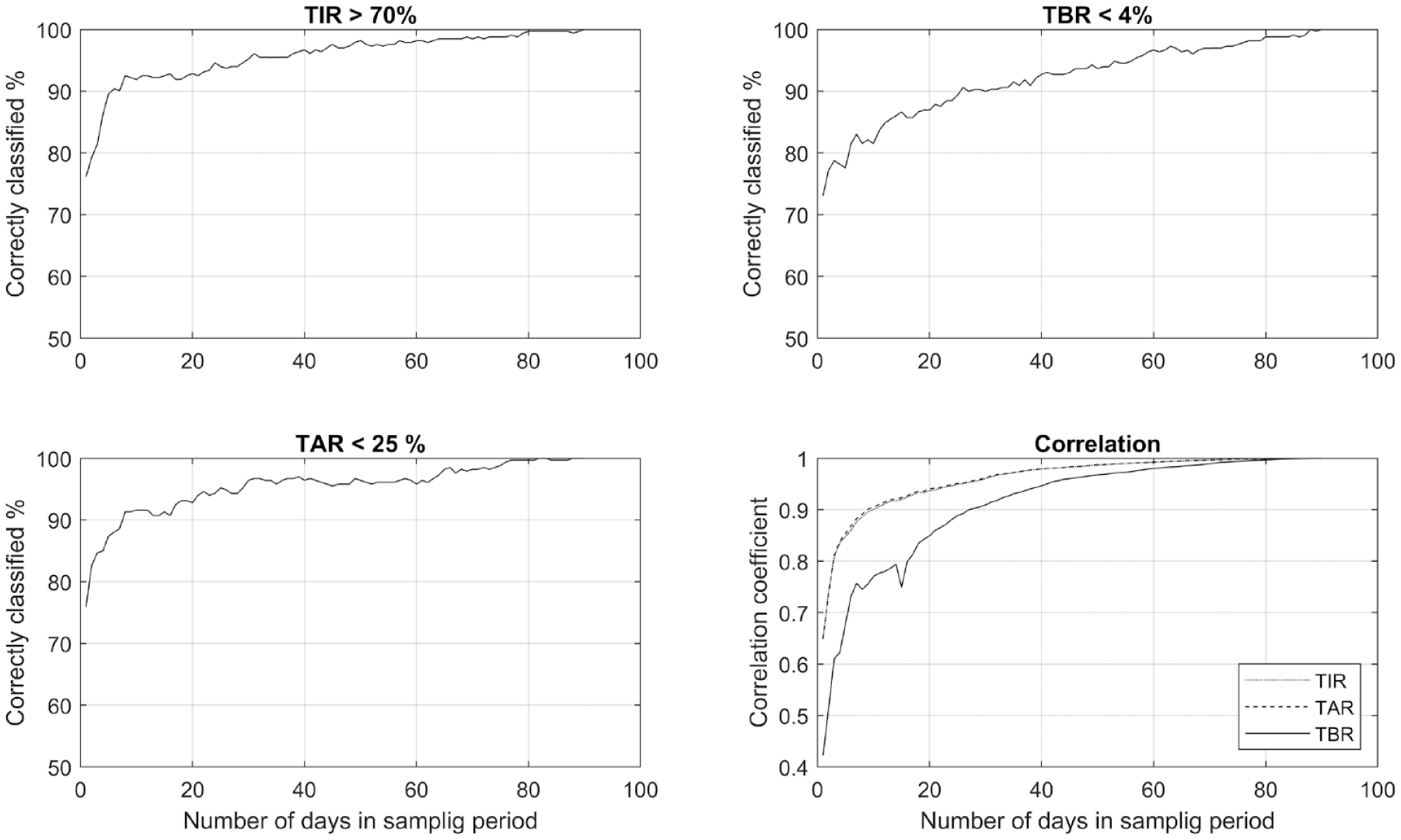
Percentage of patients classified correctly as archiving glycemic target based on the number of days in the sampling period (top & bottom left). The correlation (bottom right) shows the correlation between TIR, TAR, TBR from sampling days compared with a full sampling period of 90 days. Abbreviations: TIR, time in range; TAR, time above range; TBR, time below range.
Looking at the TBR bias from 10 to 30 days of sampling, the ARD is reduced from 47.2 [22.5; 82.6] to 26.4 [11.9; 49.7]. Again, to exemplify this, if achieving an estimate of 4% TBR from 10 days of sampling, the long-term TBR would on average be 4% ± 1.9.
To take another point of view and assess what the consequences could be in terms of misclassifying patients into achieving/not achieving the glycemic targets. Comparing 10 to 30 days of sampling, there is a small gain regarding TIR and TAR, from 91.9%/91.6% to 95.2%/96.4% correctly classified. Regarding TBR, the results show that the correct classification could be substantially improved from 81.5% to 90% using 30 instead of 10 sampling days. However, to achieve the same levels as TIR/TAR, a sampling period of up to 60 days could be necessary.
Discussion
The current study presents the bias and classification error from estimating long-term glycemic control from short-term CGM usage. These are important results, because clinicians and clinical researchers need to know and understand the limitations of assessing short-term CGM as an estimation of long-term glycemic control in patients with diabetes. The findings suggest that 10 to 14 days of sampling may provide a high correlation coefficient with long-term monitoring, but it is associated with a large bias. In particular, noteworthy is that the variance in bias is high when sampling short-term, making the assessment of individual patients difficult.
Prior studies8,9 have reported that a plateau of the correlation coefficient is reached at approximately 10 to 14 days of CGM sampling, with less improvement in correlation compared with using a longer sampling time. These findings are in line with the results from this study; when comparing the correlation coefficient with these studies, we found a similar pattern. However, translating these findings from a correlation into recommendations for sampling time should be done with caution when expected bias is not considered. The results from our study demonstrated that short-term sampling is associated with a high bias and could be substantially reduced if a longer sampling time is used. In particular, if hypoglycemia is an object for assessment, the estimates from 10 days of sampling are associated with a median bias of 47.2 [22.5; 82.6] %. The bias from short-term sampling time could, in the worst case, lead to incorrect conclusions about new treatments or nonoptimal treatment of the individual patient.
A recently published study by Herrero et al 17 concluded, in line with our findings, that a longer sampling period is necessary to achieve a robust estimate of TIR, TBR, TAR, standard deviation, and coefficient of variation. The study by Herrero et al was based primarily on a homogeneous sample cohort and performed an extensive investigation of several additional metrics for glycemic variability as well as TIR, TAR and TBR. Differences were observed between the Herrero et al study, 17 and the results from this study in relation to CGM sampling days needed to achieve a certain tolerance of median ARD. Differences worth mentioning are, for instance, if a tolerance of a median 10% ARD is chosen, the results from our study indicate that sampling days would be approximately 10, 20, and 60 days for TIR, TAR, and TBR, respectively. The corresponding days from the Herrero study are 5, 34-54, and 90+.
There are some practical considerations when considering sampling time; longer sampling time would also mean a higher cost and the fact that most commercially available CGM devices have a sensor duration of 10 to 14 days before they need to be changed. 18
There are several strengths and limitations to our study. First, the analysis only included patients with type 1 diabetes, and the findings cannot be transferred to people with type 2 diabetes, as the glycemic dynamics are fundamentally different. Second, patients from the three studies in the analysis all used Dexcom G4/G5 sensors. Other sensors have different accuracies at different levels of blood glucose. Third, this study analyzed a heterogeneous cohort of type 1 diabetes patients from young to old. This makes it possible to conclude that, not so surprisingly, type 1 patients are not a uniform group and the optimal sampling time should consider this observation as one sampling time is not sufficient for all patients. Moreover, the chosen definition of long-term glycemic control is based on some general consensus, which is based on pragmatism and previous results. However, the “best” definition for long-term glycemic control related to outcomes for patients is still under debate.
Future studies should investigate this matter further; it would be useful if factors for patients with large bias from short-term CGM could be identified. This could lead to an individualized determination of sampling time for each patient to obtain a precise estimate of long-term glycemic control. Previous studies have explored the potential of using machine learning to predict patients at risk in regard to other aspects of the management of diabetes, and a similar approach might provide a valuable clinical tool.19,20
In conclusion, our results suggest that the proposed 10 to 14 day CGM sampling time may be associated with a high correlation with the three-month CGM. However, these estimates are subject to large bias, which is clinically relevant. Clinicians and researchers should consider using assessments of longer durations of CGM data if possible. A longer time should especially be considered when assessing the time in hypoglycemia or when testing a new treatment where it is conceivable that there is a run-in period before blood glucose dynamics stabilize.
Footnotes
Abbreviations
The absolute relative difference (ARD); continuous glucose monitoring (CGM); CGM Intervention in Teens and Young Adults with T1D (CITY); time in range (TIR); time above range (TAR); time below range (TBR); Wireless Innovation for Seniors with Diabetes Mellitus (WISDM); A Trial Comparing Continuous Glucose Monitoring With and Without Routine Blood Glucose Monitoring in Adults With Type 1 Diabetes (REPLACE-BG)
Author Contributions
SLC had access to all of the data analyzed in this study. OH, MHJ, and SLC take responsibility for the integrity and accuracy of the study data analysis and results. SLC, MHJ, and OH were involved in the study design, concept, analysis, and interpretation of the data. SLC drafted the manuscript and performed the statistical analysis. OH and MHJ were involved in critical revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclaimer
The source of the data is the T1D Exchange, but the analyses, content, and conclusions presented herein are solely the responsibility of the authors and have not been reviewed or approved by the T1D Exchange.