Abstract

Keywords
The main therapeutic objective for people with type 1 diabetes is to improve glycemic control in the aim of preventing and delaying the onset of complications. Recommendations indicate that the use of continuous glucose monitoring (CGM) improves glycemic control. 1 Furthermore, the use of decision support systems (DSS) and applications can help patients to make appropriate decisions. In this context, we developed the first version of our application, Insulin Pump-RT Advisor (IPRA) which showed that patients’ spontaneous behavior was not always optimal and that the use of applications could correct inappropriate and/or maladaptive behavior. 2 Better-adapted behaviors can potentially lead to better glycemic control. Then, a new version of the application, Insulin Real-Time Advisor (IRTA), was developed, with new functionalities (like bolus wizard calculator).
We developed an interventional, prospective, monocentric, single-group, before-after study which included a first phase of 3 months without IRTA (between visit 1 [V1] and visit 2 [V2]), and a second one of 3 months with IRTA (between visit 2 and visit 3 [V3]), which delivered personalized advice.
A total of 64 patients, with treatment by Multiple Daily Injections of insulin (MDI) or insulin pump, monitored at Rennes University Hospital and trained to use FreeStyle Libre (FSL, Abbott Diabetes Care, Alameda, CA) have been analyzed to try to show an improvement of 5% of time spent in the glycemic target 70-180mg/dL (Time In Range, TIR). The average age of patients was 48.3. Most were diagnosed with diabetes before 2000 (36 patients, 56.25%), with mean and median disease duration of, respectively, 23.1 and 21 years. Forty patients were on an insulin pump (62.5%), which they had had on average for 10.6 years, and 24 were on MDI (37.5%). The initial mean and median HbA1c were 7.8% (minimum of 6% and maximum of 10.5%).
Our clinical trial failed to show any improvement in glycemic control with IRTA, in terms of time on glycemic target over the month preceding the V2 and V3 consultations, HbA1c and other indirect parameters (Table 1). 3
Glycemic and Glucose Variability Measures Outcomes.
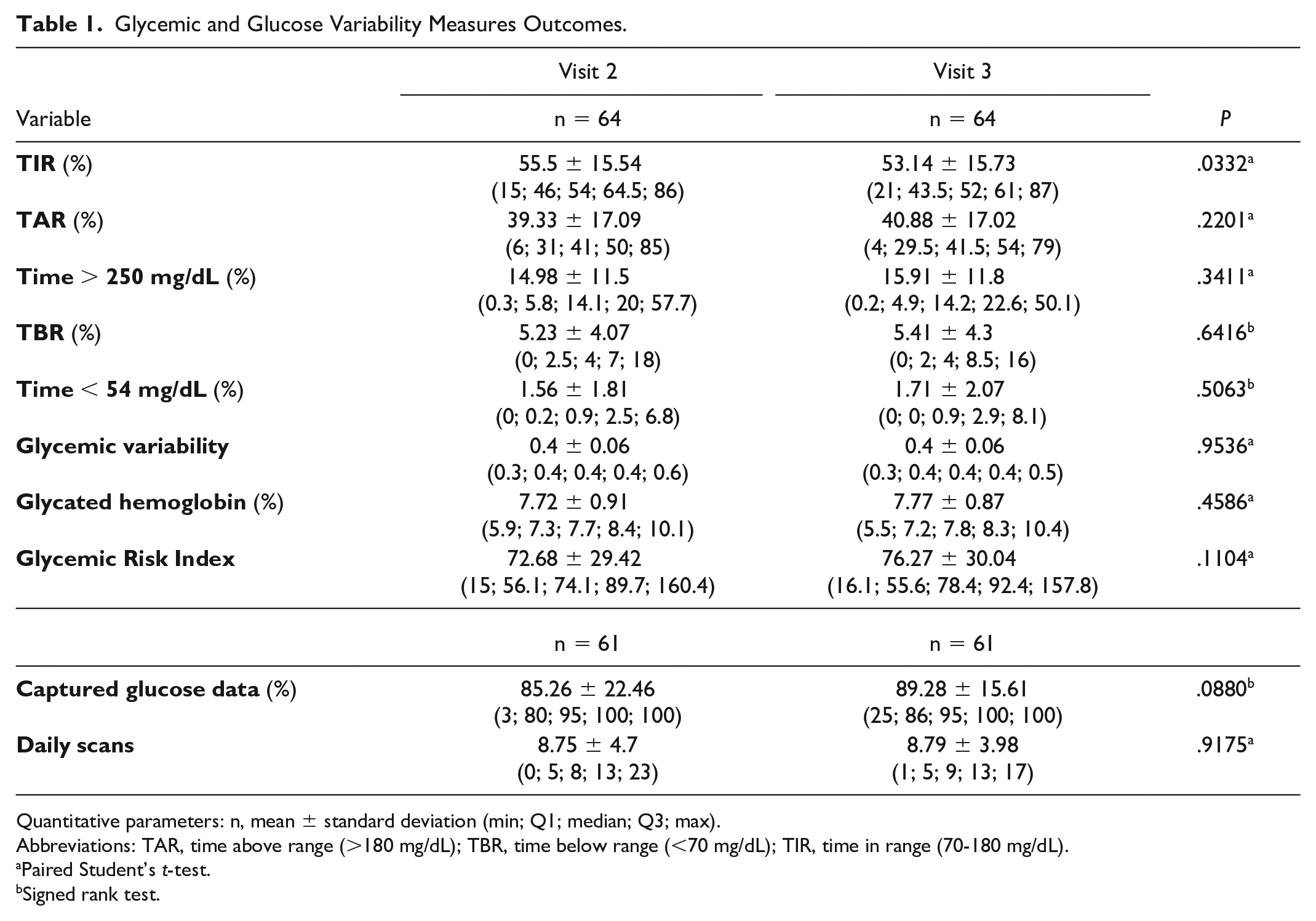
Quantitative parameters: n, mean ± standard deviation (min; Q1; median; Q3; max).
Abbreviations: TAR, time above range (>180 mg/dL); TBR, time below range (<70 mg/dL); TIR, time in range (70-180 mg/dL).
Paired Student’s t-test.
Signed rank test.
We consider the result on TIR as clinically insignificant. Our disappointing results can be explained. It is more difficult to achieve an improvement in glycemic control in well-controlled patients with extensive diabetes experience than in poorly controlled patients, as described in studies of Advanced Hybrid Closed Loop (AHCL) systems. 4 It is difficult to conclude on the robustness of the results of our poorly handled application (only 9 patients requested more than 100 recommendations each). Finally, inclusion in the study began just before the first lock-out in 2020, leading to delays in inclusion and longer call-out times. It may have led to disinterest, non-compliance, loss of enthusiasm. Our results are in line with those available in literature, particularly those obtained from the PEPPER predictive DSS or the DailyDose system.5,6 AHCL were not available in France when we conducted the study.
Further investigations need to be undertaken on other populations with diabetes to define subpopulations in which IRTA would be the most relevant.
Footnotes
Acknowledgements
The authors thank the participants for their time and involvement in the realization of this study. They would also like to thank the students and researchers of CentraleSupelec Rennes Engineering School in Rennes for their help in creating the application. The University Hospital of Rennes provided financial and human support for this study. The Institut d’Electronique et des Technologies du NuméRique took part in the project. E-MEDYS contributed to the development of the application. The work described and the opinions expressed in this manuscript are the fruit of a collaborative effort carried out within the Diabetology Department of the Rennes University Hospital.
Abbreviations
AHCL, Advanced Hybrid Closed Loop; CGM, Continuous Glucose Monitoring; DSS, Decision Support Systems; FSL, FreeStyle Libre; IPRA, Insulin Pump-RT Advisor; IRTA, Insulin Real-Time Advisor; MDI, Multiple Daily Injections; TAR, Time Above Range; TBR, Time Below Range; TIR, Time In Range.
Clinical Trial Registration
ClinicalTrials.gov Identifier NCT04211220.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
