Abstract
The clinical benefits of continuous glucose monitoring (CGM) in adults with type 1 diabetes is well known. 1 However, the benefits of CGM initiation early after type 1 diabetes onset are not known.
In this retrospective, proof of concept, pilot study, we analyzed the electronic health records of adults (age ≥18 years) diagnosed with type 1A diabetes (T1D) between June 2013 and June 2015 who had at least 1 year of follow up at the Barbara Davis Center for Diabetes Adult Clinic. T1D is defined by at least two positive islet autoantibodies. Adults with other types of diabetes, fewer than three glycosylated hemoglobin (A1C) values during study, antidiabetic medications beside insulin, CGM initiation after 6 months or discontinuation within 3 months, previous participation in immune intervention studies, and pregnant women were excluded. Patients who initiated CGM within 6 months of their diagnosis of T1D were included as the “CGM group” and those who did not use CGM from T1D diagnosis to the study end served as “controls.” Baseline was defined as CGM initiation for CGM group and 3-4 months after diagnosis for controls. The differences in the A1C between CGM and control groups were assessed at diagnosis, baseline, 3, 6, and 12 months after baseline using Student’s
A total of 19 adults (6 CGM group and 13 controls) with T1D were included in the present analysis. CGM was initiated within a mean of 3.8 months of diagnosis in the CGM group.
There were no differences in age (24.8 ± 4.9 vs 21.3 ± 3.9,
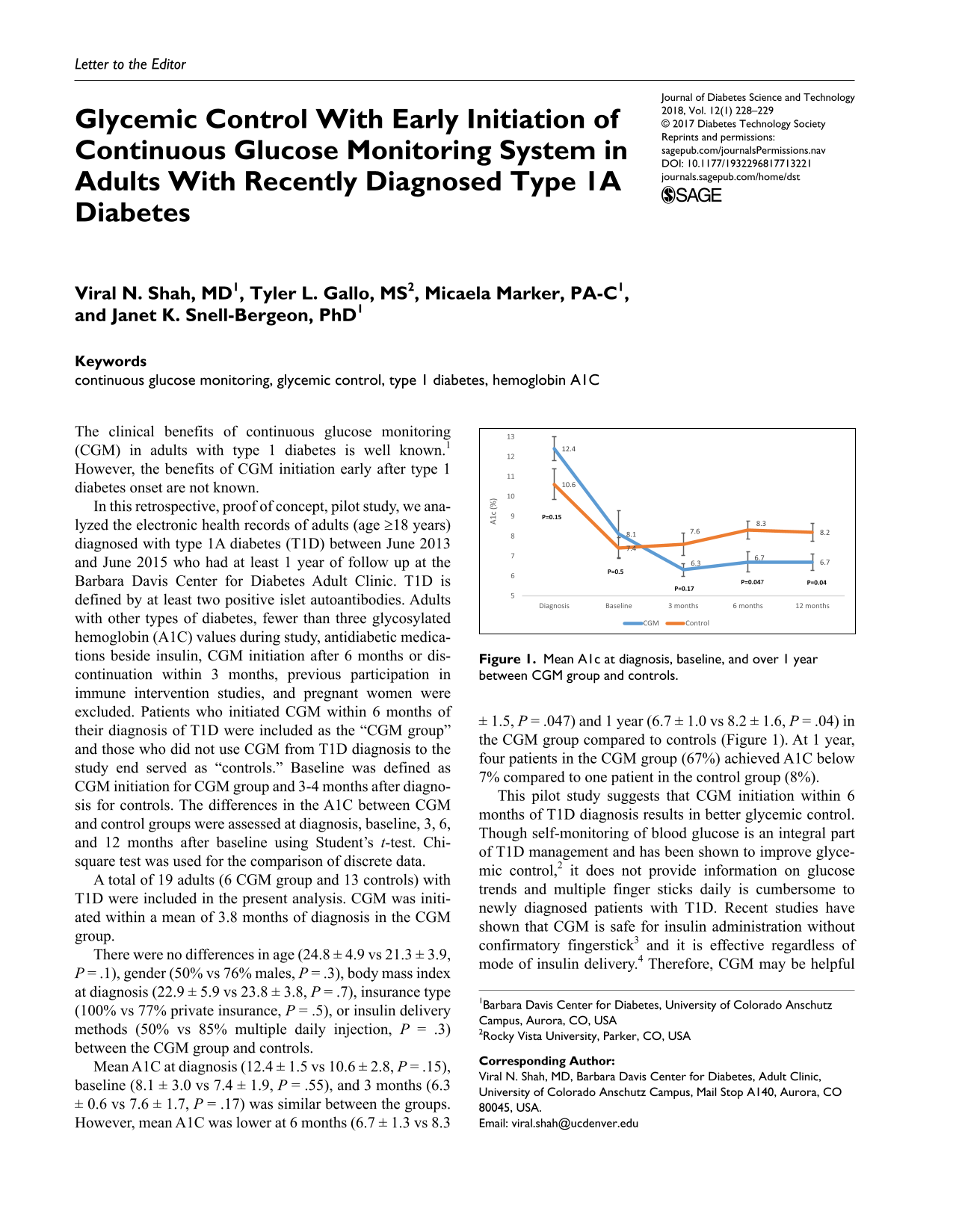
Mean A1C at diagnosis (12.4 ± 1.5 vs 10.6 ± 2.8,
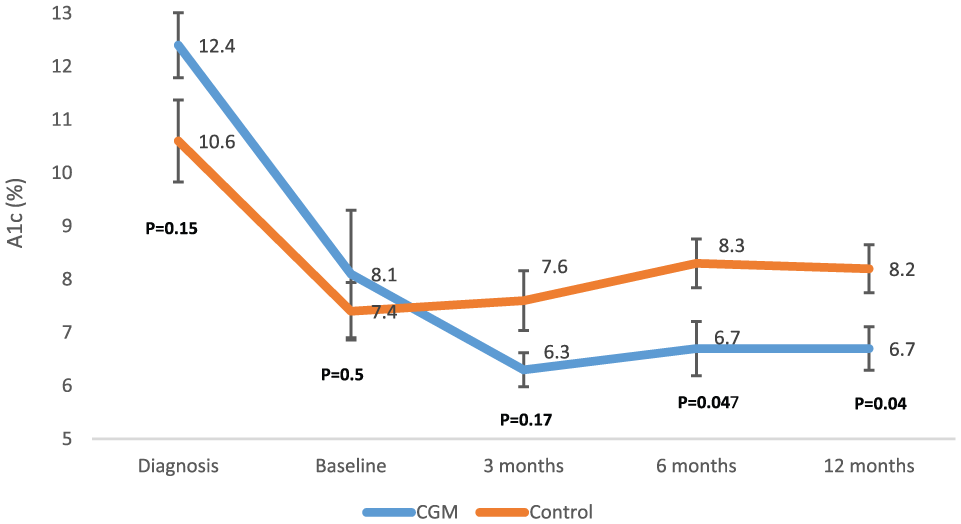
Mean A1c at diagnosis, baseline, and over 1 year between CGM group and controls.
This pilot study suggests that CGM initiation within 6 months of T1D diagnosis results in better glycemic control. Though self-monitoring of blood glucose is an integral part of T1D management and has been shown to improve glycemic control, 2 it does not provide information on glucose trends and multiple finger sticks daily is cumbersome to newly diagnosed patients with T1D. Recent studies have shown that CGM is safe for insulin administration without confirmatory fingerstick 3 and it is effective regardless of mode of insulin delivery. 4 Therefore, CGM may be helpful for newly diagnosed patients and provide greater understanding of the effects of insulin, food, exercise, and stress on glucose fluctuations. Its use may ease management of diabetes. An adequately powered randomized control trial is needed to confirm the findings of our study.
Footnotes
Abbreviations
AIC, glycosylated hemoglobin; CGM, continuous glucose monitoring; TID, type 1A diabetes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VNS’s employer has received research funding from T1D Exchange and Sanofi. VNS received speaking fees from the Dexcom Inc. TLG, MM, and JKS report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Dexcom Inc. However, the sponsor had no role in devising the study concept, data collection, analysis, and interpretation.
