Abstract
Background:
We present a digital therapeutic (DTx) using continuous glucose monitoring (CGM) and an advanced artificial intelligence (AI) algorithm to digitally personalize lifestyle interventions for people with type 2 diabetes (T2D).
Method:
A study of 118 participants with non–insulin-treated T2D (HbA1c ≥ 6.5%) who were already receiving standard care and had a mean baseline (BL) HbA1c of 7.46% (0.93) used the DTx for three months to evaluate clinical endpoints, such as HbA1c, body weight, quality of life and app usage, for a pre-post comparison. The study also included an assessment of initial long-term data from a second use of the DTx.
Results:
After three months of using the DTx, there was an improvement of 0.67% HbA1c in the complete cohort and −1.08% HbA1c in patients with poorly controlled diabetes (BL-HbA1c ≥ 7.0%) compared with standard of care (P < .001). The number of patients within the therapeutic target range (< 7.0%) increased from 38% to 60%, and 33% were on the way to remission (< 6.5%). Patients who used the DTx a second time experienced a reduction of −0.76% in their HbA1c levels and a mean weight loss of −6.84 kg after six months (P < .001) compared with BL.
Conclusions:
These results indicate that the DTx has clinically relevant effects on glycemic control and weight reduction for patients with both well and poorly controlled diabetes, whether through single or repeated usage. It is a noteworthy improvement in T2D management, offering a non-pharmacological, fully digital solution that integrates biofeedback through CGM and an advanced AI algorithm.
Keywords
Introduction
Type 2 diabetes (T2D) has reached the scale of a global pandemic. In 2021, the global prevalence of diabetes was 6.1%, of which 96% was T2D. Forecasts for 2050 predict a prevalence of 9.8%, reinforcing concerns about its impact on public health. 1 The rise in these numbers is surpassing the limited capacity of health care professionals to manage this pandemic, clearly highlighting the need for scalable digital therapeutic (DTx) approaches. 2
Non–insulin-treated patients without risk of hypoglycemia typically aim for a glycated hemoglobin (HbA1c) target range of < 7.0%. 3 Lifestyle therapies are the primary treatment in helping to achieve this threshold and are a crucial component in addition to all other treatment options. However, implementing lifestyle changes can be challenging and requires significant effort.4,5 Empowering patients in self-management, offering continuous glucose biofeedback, and daily companionship are essential components of a successful diabetes therapy. Consequently, innovative approaches are necessary to expand therapeutic options.6 -8 The DTx glucura (Perfood GmbH, Lübeck, Germany) combines these aspects and provides a fully digital and therefore scalable therapy that fills gaps in T2D health care.
The DTx has been approved as reimbursable digital health application (“DiGA”) based on data from the pilot study presented here. This application empowers individuals with self-efficacy and focuses on the long-term feasibility of lifestyle therapy implementation. It provides continuous glucose monitoring (CGM) and meal-related glucose feedback. The proprietary advanced artificial intelligence (AI) algorithm continues to make predictions about individual postprandial glucose reactions even after the initial use of a CGM sensor.
The presented pilot study provides evidence for the usage of the DTx as an adjunct to the standard of care for patients with T2D.
Methods
The Prescription Digital Therapeutic
The DTx “glucura” offers a continuous program in a three-month cycle with one CGM sensor phase, followed by an implementation phase that includes weekly goals. The DTx serves as fully digital therapy experience and does not involve complimentary in-person coaching elements. The DTx has been granted provisional approval by the Federal Institute for Drugs and Medical Devices (BfArM) and is therefore completely reimbursed by statutory health insurances. In order to provide additional evidence of improvement in HbA1c, which is the accepted primary endpoint for studies with diabetes patients, 9 the DTx must undergo a randomized controlled trial (RCT). Upon successful demonstration of the positive health care effect on HbA1c, the DiGA is permanently reimbursable. 10
It focuses on various lifestyle adjustments, including adopting a low-glycemic diet, reducing calorie intake, promoting physical activity, enhancing motivation, providing knowledge, addressing sleep hygiene, and engaging in stress management. Compatible with various wearables, smart trackers, and glucose sensors, the DTx can derive personalized nutrition recommendations and track physical activity.
The DTx imports CGM data from validated devices during the up to two-week long sensor phase, generating personalized recommendations for a low-glycemic diet. During this phase, users log their meals in a nutrition diary. Meal responses are analyzed and rated on a meal-score reflecting the maximum change in postprandial glucose levels. Users receive suggestions for targeted meal comparisons to illustrate individual postprandial differences for similar meal types.
The implementation phase setting weekly goals based on CGM data and analyzed information to gradually implement lifestyle modifications. The DTx serves as patient’s daily companion, helping them to implement lifestyle adjustments in small steps so not to overwhelming the patients. Users continue to receive meal feedback with predicted blood glucose curves and meal scores even after the up to two weeks of the CGM sensor phase. This feedback is complemented by knowledge lessons and videos to enhancing patients’ understanding of T2D and their nutrition therapy, all of which promote better and more consistent self-management.
The Advanced Artificial Intelligence Algorithms
The DTx has integrated several advanced AI algorithms into its core functionality. In developing our machine learning model, we adhered to established best practices to ensure the reliability and validity of our results. This includes rigorous data preprocessing, careful selection and tuning of algorithms and hyperparameters, and thorough evaluation and risk assessment.11,12
Traditional methods using the glucose levels at meal initiation are not accurate and, eg, highly sensitive to slight inaccuracies of the meal’s start time or second meal effects.13,15 The first algorithm is designed to establish a baseline (BL) for postprandial glucose levels, a crucial step in accurately assessing the body’s response to meals consumed.
To mitigate these issues, an expert data set was set up. For this, clinical experts independently and manually analyzed postprandial reactions to meals. This accumulated expert knowledge contains manually set BL levels and served as the foundation for training a machine learning algorithm. At its core, this patent-pending process employs a random forest regressor that uses nine statistical features chosen from a vast set of features engineered from several hours of blood glucose data leading up to the meal in question to output a BL, as, eg, several percentiles of the CGM data. 16 This machine learning model achieves an accuracy comparable to the variability observed among different human clinical experts when setting BLs for the same meal. Thus, this model effectively automates human expertise.
Another important algorithm used in the DTx leverages a comprehensive data set to fuel a deep learning-based neural network, specifically designed to learn representations of data using multiple layers, known as deep architectures. 17 Our neural network is engineered to deliver personalized predictions of blood glucose responses to specific meals by utilizing a big data set of meals with detailed compositions and corresponding blood glucose reactions. The input of this machine learning model consists of the meal composition and data regarding the individual, including a description of the CGM. To increase the application’s safety, we implemented two measures. We defined a risk score that penalizes particularly unfavorable errors in the context of diabetes (eg, predicting a poor meal to be a good one) and tuned hyperparameters to minimize this score. In addition, a kernel density estimator is used to exclude meals from the prediction too uncommon relative to the training data. The final accuracy is within the range of the repeatability of blood glucose reactions to the same meal for the same individual. We are constantly monitoring the accuracy of the model in a real-world setting while patients wear CGM sensors comparing our predictions to actual sensor readings.
Study Design
This was a six-month study with a three-month intraindividual retrospective phase (three months to BL) mirroring standard of care and a three-month prospective data collection from the use of the DTx (BL to +3 months). All participants had already received standard treatment (eg, medication) for at least six months prior to the start of the program and continued to receive it unchanged during the treatment.
The primary hypothesis is that the arithmetic mean of the intraindividual HbA1c is significantly reduced after completion of the digital program (+3 months, after the intervention) compared to BL (before the start). Further endpoints were assessed at BL and after three months. The time window was defined as 90 days (ie, “three months”) plus a maximum of 28 days for the final HbA1c assessment. After using the DTx once, the patients were offered to use it a second time. The endpoint was assessed upon completion of the digital program for the second time (+6 months, after the second intervention; Figure 1).
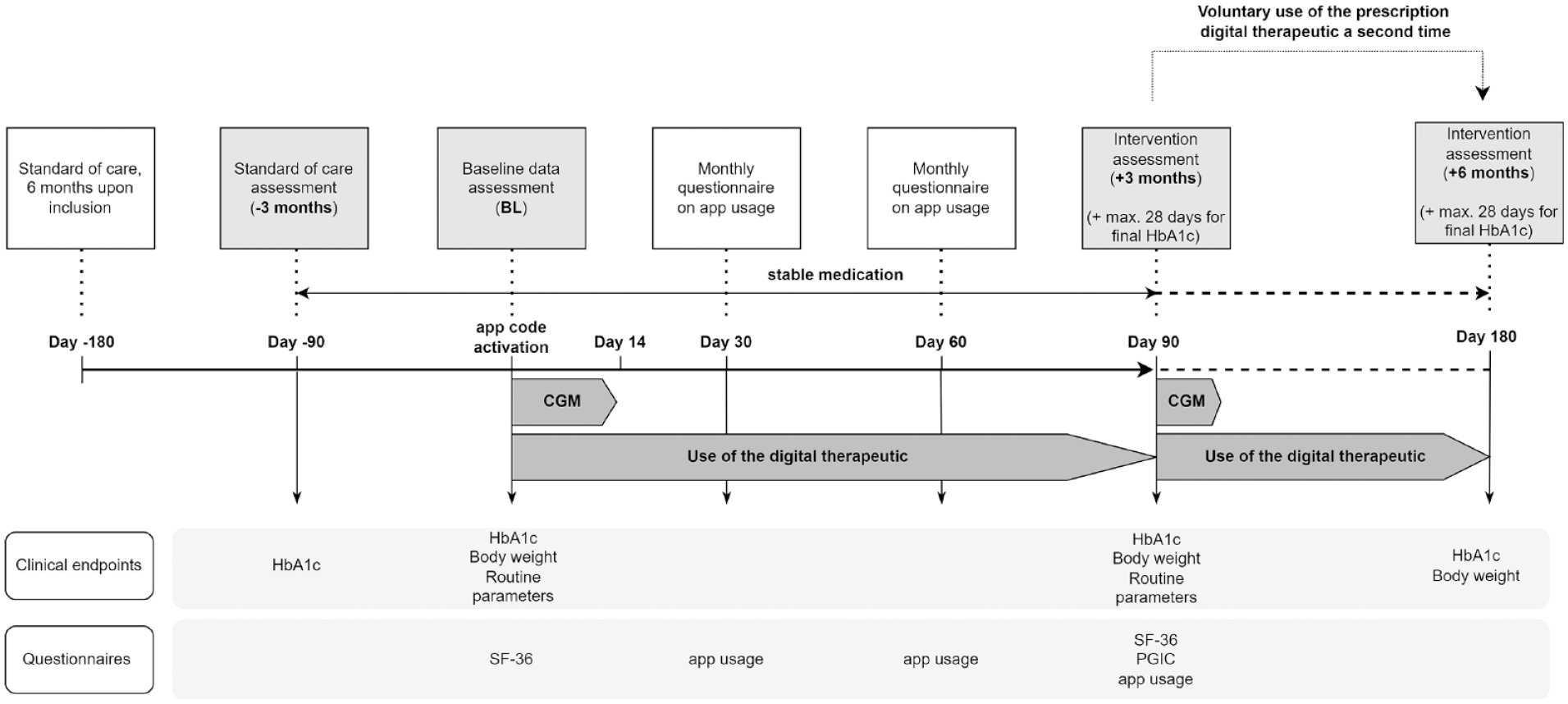
Study design including time points for endpoint assessment. Patients used the continuous glucose monitoring (CGM) system for up to two weeks, the prescription digital therapeutic (DTx) over the course of three months and voluntarily continued using the DTx for additional three months. Clinical endpoints were assessed at −3 months, baseline, after +3 months, and after +6 months. Questionnaires were assessed within the three months of DTx use.
Study Population
Patients with non–insulin-treated T2D aged 18 years and older according to the International Statistical Classification of Diseases and Related Health Problems–Tenth Edition (ICD-10) diagnosis code E11. were eligible for inclusion when the following criteria were met: HbA1c ≥ 6.5% or higher, a body mass index (BMI) ≥ 25 kg/m2, stable weight (±5% in the last three months), enrollment in the Disease Management Program (DMP) reflecting standard of care, and no current pregnancy or lactation. In addition, participants needed to maintain their diabetes-specific medication during the study.
We present the data of n = 118 patients who reported complete HbA1c data at BL and after three months, 103 patients reported retrospective HbA1c data and 67 patients after six months. Patients were recruited direct-to-patient or via the treating physicans. From the aspect of patient recruitment, the study cohort can be considered to be representative for the anticipated DiGA target population.
Clinical Endpoints and Questionnaires
HbA1c is used as a primary endpoint. Patient-reported outcomes were assessed using validated questionnaires. For this, quality of life was measured using the Short-Form 36 questionnaire (SF-36). 18 Patients’ global impression of change (PGIC) was assessed (scale from “much better” (+3) to “much worse” [−3]). In addition, patients were asked about their satisfaction and use of the DTx.
Statistical Analysis
For the complete data set the missing retrospective values of n = 15 patients (from −3 months) were conservatively replaced using the individual BL data assuming no change within the control phase under standard care. All other data were analyzed if information was available at both measurement times (complete case analysis). A subgroup analysis was carried out for patients out of the HbA1c treatment target range at BL (HbA1c ≥ 7.0%, “poorly controlled”) 3 and for those who used the DTx a second time (+6 months).
Specifically, we compared the mean of the intraindividual differences using a two-sided t-difference test. When the sample size was < 25, the sign test was performed instead.19,20 The 95% confidence interval (CI) for the mean of the change is calculated when using the t-difference test. The significance level was set at P < .05 for both tests.
Results
Baseline Demographics and Disease Severity
We present data of n = 118 patients (Table 1). Patients were between 31 and 78 years old with a mean age of 55.5 years (SD = 8.7). Participants were in 58% female (n = 69), and the mean duration of diabetes diagnosis was 7.2 years (SD = 5.2). The mean HbA1c was 7.46% (SD = 0.93) with a minimum of 6.50% and a maximum of 11.00%. With a mean body weight of 100.1 kg (SD = 18.2), the mean BMI was 33.7 kg/m2 (SD = 5.7).
Baseline Demographics and Disease Severity of the Complete Cohort.
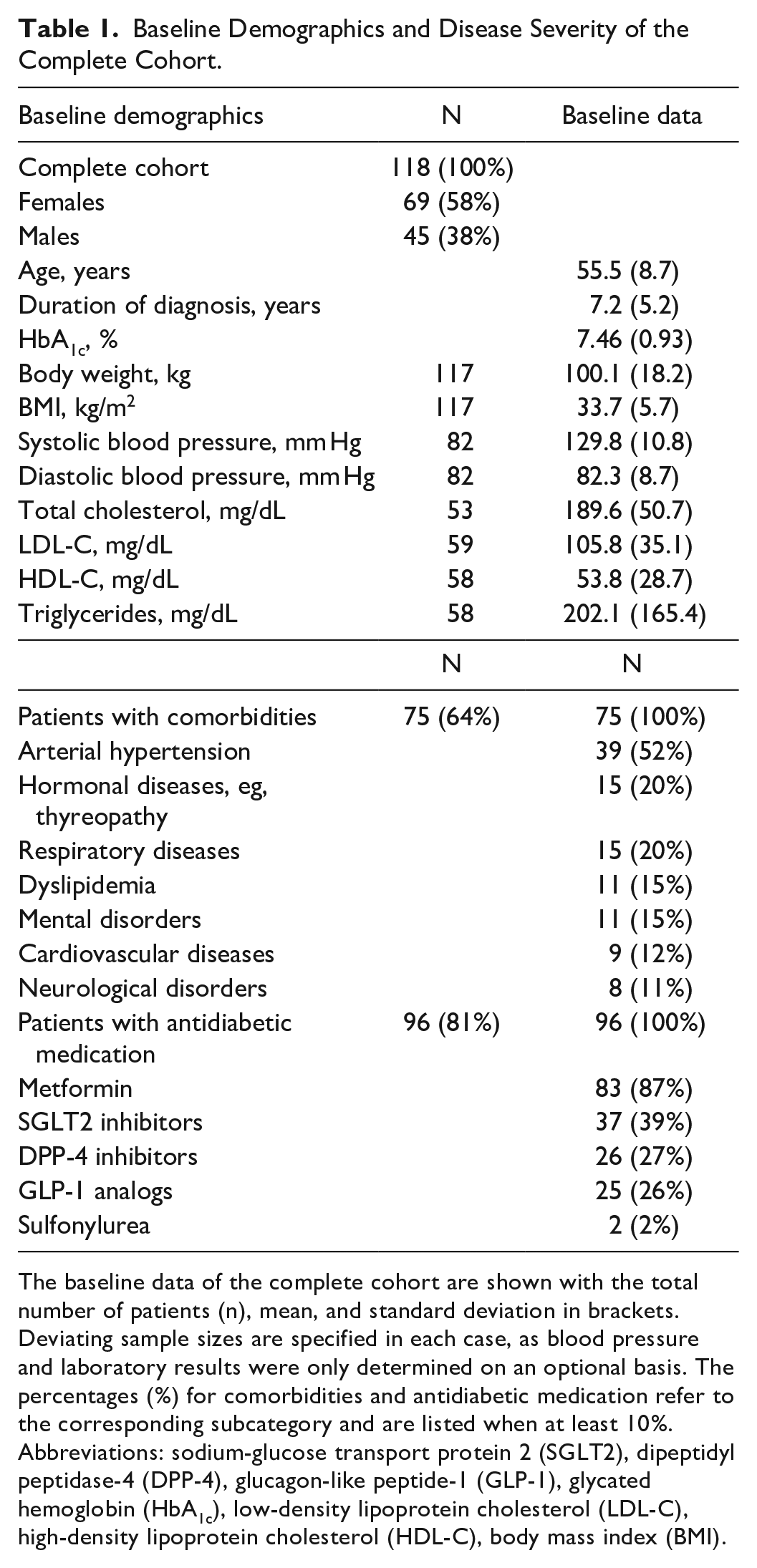
The baseline data of the complete cohort are shown with the total number of patients (n), mean, and standard deviation in brackets. Deviating sample sizes are specified in each case, as blood pressure and laboratory results were only determined on an optional basis. The percentages (%) for comorbidities and antidiabetic medication refer to the corresponding subcategory and are listed when at least 10%.
Abbreviations: sodium-glucose transport protein 2 (SGLT2), dipeptidyl peptidase-4 (DPP-4), glucagon-like peptide-1 (GLP-1), glycated hemoglobin (HbA1c), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), body mass index (BMI).
Additional data from routine clinical diagnostics were available for blood pressure and serum lipids. The mean diastolic blood pressure was 82.3 mm Hg (SD = 8.7), the mean total cholesterol was 189.6 mg/dL (SD = 50.7), and the mean serum triglycerides was 202.1 mg/dL (SD = 165.4).
About 75 patients (64%) reported comorbidities, with arterial hypertension being the most commonly mentioned. About 96 patients (81%) were taking non-insulinotropic antidiabetic medications, with 87% using metformin. Combination medications were present with the combination of metformin and SGLT-2 inhibitors and metformin and DPP-4 inhibitors.
Therapeutic Effect on Long-term Blood Glucose Level
The three-month period before initiating the use of the digital program reflects standard of care. In the complete cohort, the HbA1c remained unchanged in that period with 7.43% (SD = 0.94) at −3 months and 7.46% (SD = 0.93) at BL (mean change +0.03%-points [−0.09, 0.14], n = 118, P = .65). However, after starting the DTx, the HbA1c significantly improved by a mean of 0.65%-points ([−0.79, −0.50], n = 118, P < .001) down to 6.81% (SD = 0.65). In comparison to the individual standard of care period, the mean treatment effect in the complete cohort was an improvement of HbA1c of −0.67% points ([−0.89, −0.46], n = 118, P < .001; Table 2 and Figure 2).
Therapeutic Effect of the DTx on HbA1c of the Complete Cohort and of Patients With Poorly Controlled Diabetes (BL-HbA1c ≥ 7.0%).
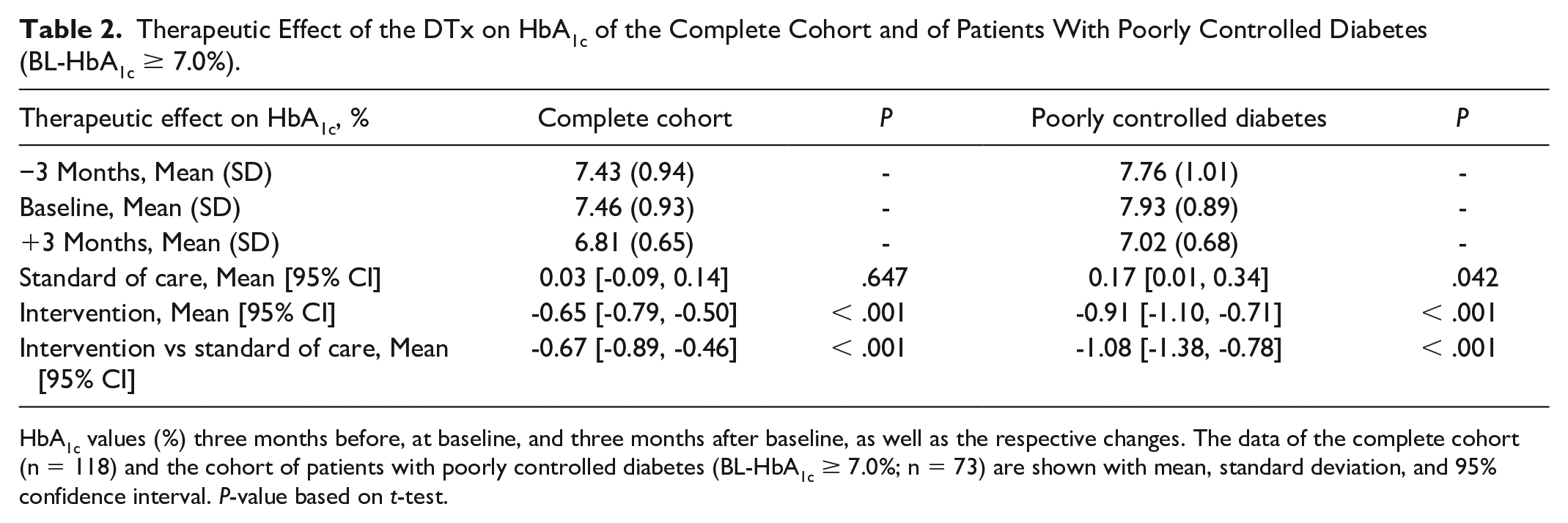
HbA1c values (%) three months before, at baseline, and three months after baseline, as well as the respective changes. The data of the complete cohort (n = 118) and the cohort of patients with poorly controlled diabetes (BL-HbA1c ≥ 7.0%; n = 73) are shown with mean, standard deviation, and 95% confidence interval. P-value based on t-test.
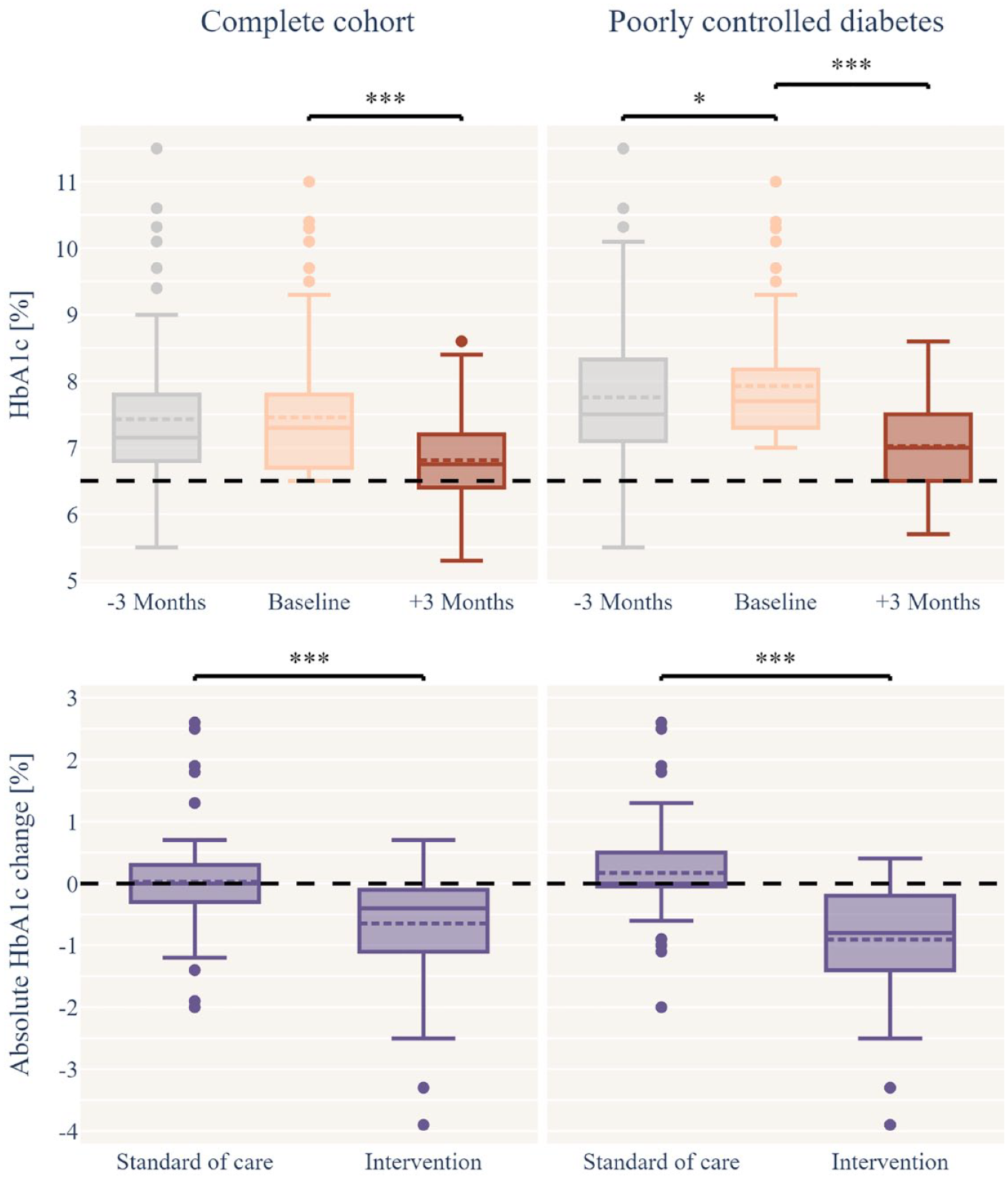
Therapeutic effect of the DTx on HbA1c of the complete cohort (n = 118) and of patients with poorly controlled diabetes (BL-HbA1c ≥ 7%; n = 73). HbA1c values (%) three months before, at baseline, and three months after baseline are present above. The respective absolute HbA1c changes of standard of care (−3 months vs baseline) and intervention (baseline vs +3 months) is present below. Statistical significances based on t-test are represented with asterisks: * for P-values between < .05 and .01; *** for P < .001.
At BL, only 38% (n = 45) were within the therapeutic treatment range (HbA1c < 7.0%). After using the DTx, this increased to 60% (n = 71). In addition, after using the DTx, n = 39 (33%) of the patients had an HbA1c below 6.5% and were potentially on the path to remission. Patients with poorly controlled diabetes (BL-HbA1c ≥ 7.0%) and thereby outside the treatment target range showed an even higher responsiveness. Their improvement in HbA1c levels was 0.91% ([−1.10, −0.71], n = 73, P < .001) down to 7.02% (SD = 0.68). In relation to the individual standard of care period, the mean treatment effect in the patients with poorly controlled diabetes was an improvement of HbA1c of −1.08% points [1.38, 0.78], n = 73, P < .001, Table 2 and Figure 2).
Effect on Body Weight and Additional Clinical Parameters
The overall body weight of the complete cohort demonstrated a significant improvement, decreasing by −3.5 kg ([−4.27, −2.71], n = 116, P < .001) from the initial body weight of 100.1 kg (SD = 18.2) to 96.4 kg (SD = 18.2). Consequently, there was a notable improvement in BMI, decreasing by −1.2 kg/m2 ([−1.45, −0.93], n = 116, P < .001, Table 3).
Change in Body Weight, BMI and Routine Parameters.
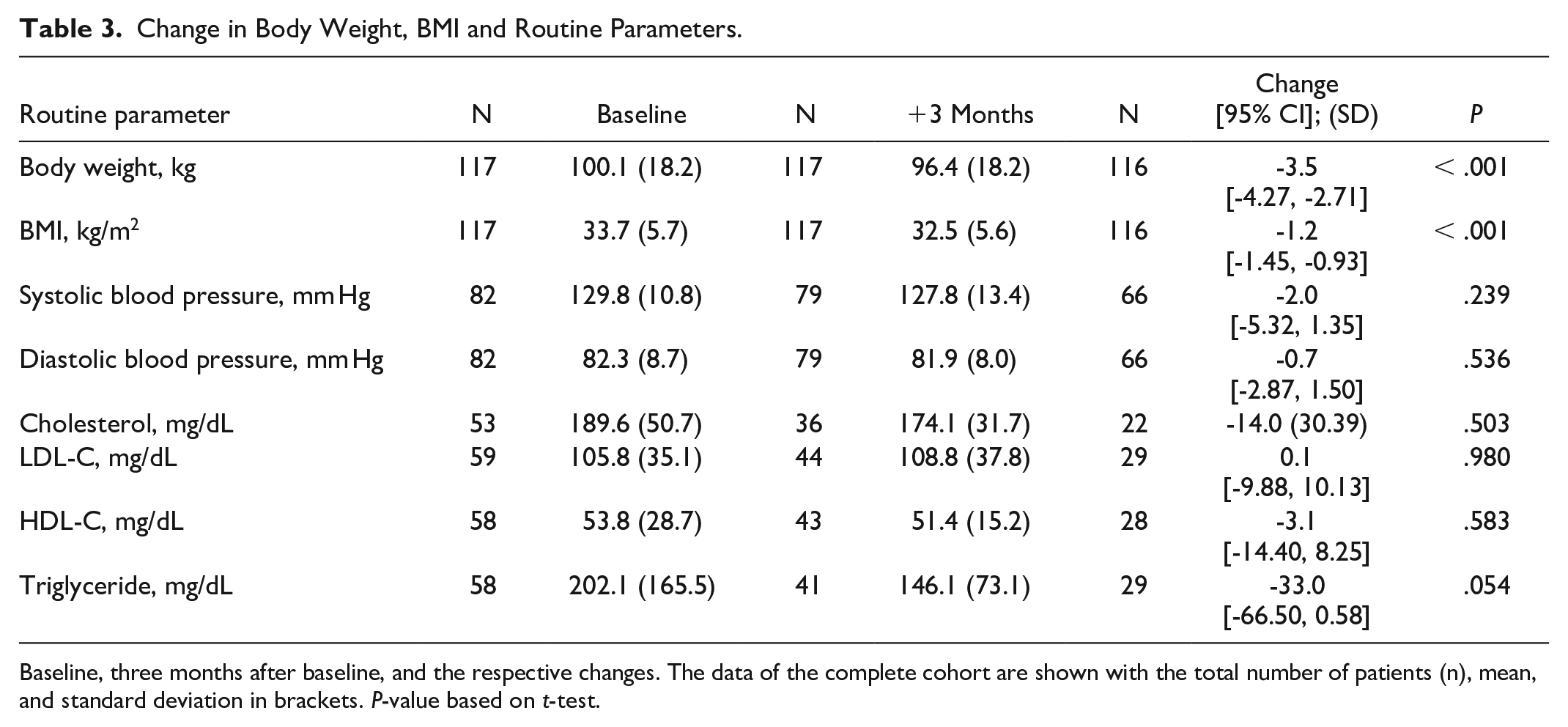
Baseline, three months after baseline, and the respective changes. The data of the complete cohort are shown with the total number of patients (n), mean, and standard deviation in brackets. P-value based on t-test.
Individuals with poorly controlled diabetes at BL experienced a significant improvement in body weight by −4.0 kg ([5.09, −2.99], n = 72, P < .001) from the initial body weight of 100.3 kg (SD = 17.85) to 95.77 (SD = 18.14). This led to a significant improvement in BMI by −1.4 kg/m2 ([1.72, 1.02], n = 72, P < .001).
Routine data on blood pressure and serum lipids at BL indicated mean values within the treatment target range. Notably, after three months of using the DTx, there was no significant impact on blood pressure and serum lipids (Table 3).
Patient-Reported Outcomes: Quality of Life and Use of the App
The patients reported a significant improvement in health-related quality of life (SF-36) as measured by the physical summation scale of +1.92 ([0.67, 3.18], n = 110, P = .003) and the psychological summation scale of +3.28 ([1.11, 5.45], n = 110, P = .003). In the subgroup analysis of patients with poorly controlled diabetes at BL, an even higher improvement was observed, in the physical summation scale of +3.06 ([1.50, 4.62], n = 69, P < .001) and in the psychological summation scale of +4.64 ([1.90, 7.39], n = 69, P = .001).
Of total, 73% (n = 83 of a total of n = 114) of patients reported that their general condition (PGIC) had improved after using the DTx (score 1-3). Of total, 76% (n = 55 of a total of n = 72) in the subgroup analysis with a BL-HbA1c ≥ 7.0%.
The patients used the DTx on an average of 4.7 to 6.5 days per week (n = 103-114). Satisfaction, comprehensibility, willingness to continue therapy and involvement in disease management was rated high (n = 101-113). These data show that the DTx is highly usable, and that the patient could interact well with the application (Table 4).
Data on the Use and Usability of the DTx.
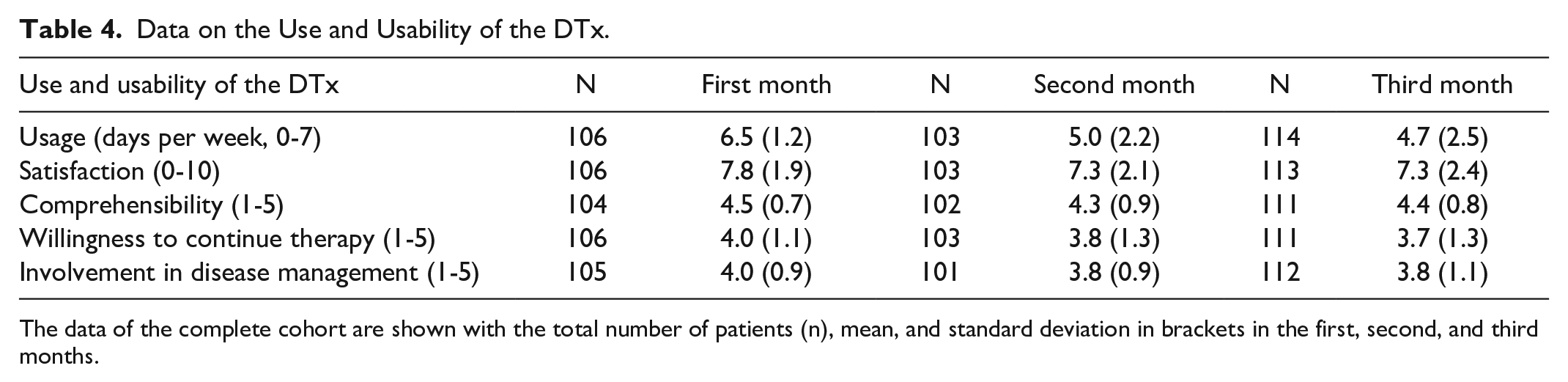
The data of the complete cohort are shown with the total number of patients (n), mean, and standard deviation in brackets in the first, second, and third months.
Long-term Therapeutic Effect
The study focused on an intervention over three months. However, we could also collect data from n = 67 subjects after six months, of whom n = 51 used the DTx a second time and n = 16 did not.
Of note, patients who used the DTx a second time (second intervention) showed an improvement of their HbA1c by an additional −0.12%-points ([−0.26, 0.03], n = 51, n.s.) from 6.78% (SD = 0.70) down to 6.67% (SD = 0.70). In total, patients reduced their HbA1c by −0.76% ([−1.02, −0.50], n = 51, P < .001) in six months. However, patients who did not use the DTx a second time (follow-up) showed no additional improvement in HbA1c (change: 0.01%, SD = 0.31, n = 16, n.s.). In total, these patients reduced their HbA1c by 0.63% (SD = 0.74, P = .035) in six months.
Patients who used the DTx a second time reported an additional improvement of their body weight by a mean of −2.85 kg ([−4.76, −0.94], n = 50, P = .004) from 96.41 kg (SD = 17.92) down to 93.53 kg (SD = 15.99). In total, patients reduced their body weight by a mean of −6.84 kg ([−9.45, −4.22], n = 51, P < .001) in six months. Consequently, there was a notable improvement in BMI over six months by −2.34 kg/m2. However, patients who did not use the DTx a second time reported no further improvement but an increase of +1.16 kg (SD = 3.26, n = 16, n.s.). In total, patients reduced their body weight by −1.71 kg (SD = 3.08, P = .022) in six months. Consequently, there was an improvement in BMI over the six months, decreasing by 0.63 kg/m2 (Table 5).
Long-term Therapeutic Effect of the DTx on HbA1c and Body Weight.
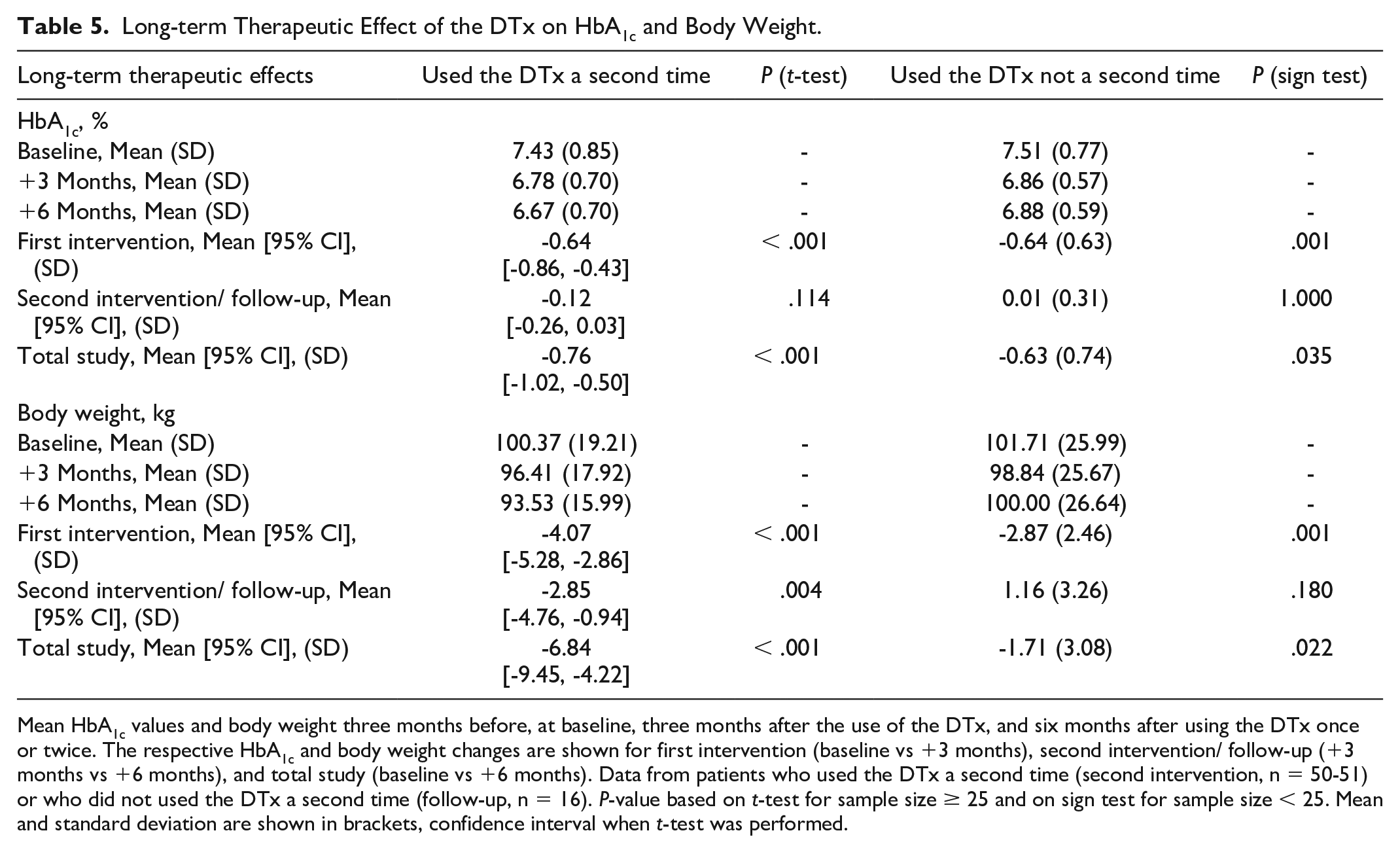
Mean HbA1c values and body weight three months before, at baseline, three months after the use of the DTx, and six months after using the DTx once or twice. The respective HbA1c and body weight changes are shown for first intervention (baseline vs +3 months), second intervention/ follow-up (+3 months vs +6 months), and total study (baseline vs +6 months). Data from patients who used the DTx a second time (second intervention, n = 50-51) or who did not used the DTx a second time (follow-up, n = 16). P-value based on t-test for sample size ≥ 25 and on sign test for sample size < 25. Mean and standard deviation are shown in brackets, confidence interval when t-test was performed.
Discussion
All patients had previously received standard of care and continued to receive it. Throughout the observation period, it was ensured that no alterations were made to the antidiabetic and other medication. Parameters such as age, BMI, gender distribution, duration of diabetes diagnosis, and the use of diabetes medication were within the expected range for patients with T2D.5,21,22 Despite these arguments, it can be assumed that the cohort is a rather technically skilled and motivated group in relation to the complete group of patients with non–insulin-treated T2D. However, this comes with the requirements to be willing to engage with an app and implement lifestyle changes. Although this is not representative to all patients, it represents the target group for the DTx and is a suitable cohort to evaluate the clinical effectiveness of the DTx glucura.
The DTx comprises four core active elements, low-glycemic diet, modest calorie restriction, exercise, and education. These elements are collectively responsible for the clinical efficacy observed. The medical benefit is primarily demonstrated by a reduction in HbA1c. 9 How much the single elements attribute to the clinical effects on HbA1c remains elusive because the DTx as a whole is investigated. However, the focus is on personalized nutrition recommendations for a low-glycemic diet. This enables small changes in the nutrition without strict calorie restriction or a low-carb diet to improve HbA1c.23 -25 A simultaneous reduction in body weight and improvement in BMI are typically observed with lifestyle interventions. Weight management can positively influence the course of the disease 26,27 and has the potential to achieve remission of the disease. 5 A comparison of HbA1c and body weight at three and six months reveals that the primary HbA1c effect is achieved within three months, with the weight improvement potentially continuing in a linear manner. It can be postulated that the improvement in HbA1c is not merely a consequence of weight improvement, but it is primarily due to the low-glycemic diet. Furthermore, the weight improvement itself is also due to the low-glycemic diet.
In patients with poorly controlled diabetes (BL-HbA1c ≥ 7.0%) who needed treatment optimization, the DTx improved the HbA1c by 1.08%-points compared with standard of care without an additional pharmaceutical escalation. A simultaneous reduction in body weight is associated with a reduction of cardiovascular risk and improvements in quality of life. The patients with poorly controlled diabetes experienced a reduction in body weight of 4.0 kg and a decrease in BMI of −1.4 kg/m2. In addition, their quality of life improved, and patients reported high satisfaction with both the use and usability of the DTx.
The consensus for a clinically relevant improvement in HbA1c is ≥ −0.50%.28,29 In comparison, the DTx resulted in a clinically relevant improvement of 0.67% HbA1c in the complete cohort and of 1.08% HbA1c in patients in need of treatment optimization, without an additional pharmaceutical escalation. This provides a significant added value for these patients, as the results from the United Kingdom Prospective Diabetes Study (UKPDS) demonstrate that a 0.9% reduction in HbA1c is associated with a 25% reduction in microvascular complications, a 10% decrease in diabetes-related mortality, and a 6% decrease in overall mortality. 30 At BL, only 38% were within the therapeutic treatment range of < 7.0% HbA1c. After using the DTx for three months, this increased to 60%. Moreover, after the use of the DTx, 33% even had an HbA1c below 6.5% and were potentially on the way to remission. 31
After using the DTx once, the patients were offered to use it a second time to provide first indications of the long-term effects of the DTx. 9 Many patients took up the offer, which is in line with the mean score (3.7 out of a scale from 1 to 5) for the willingness to continue the therapy after the initial three-month period. Furthermore, a total of 33% of patients had already achieved an HbA1c below 6.5% after the first round, which is a plausible reason why some patients were satisfied with the results and did not require a second course of the DTx. We showed that using the DTx twice led to a total HbA1c reduction of 0.76% and improvement in body weight by 6.84 kg. The use of the DTx once resulted in a total reduction of HbA1c by 0.63% and an improvement in body weight by 1.71 kg in patients observed for six months. The study found a difference in body weight improvement between patients who used the DTx once and those who used it twice after six months. A weight loss of 5% or more in cases of obesity32,33 and specifically in patients with T2D26,27 is considered to lead to a measurable clinical benefit. It is linked to a significantly reduced risk of cardiovascular disease. 27 The results of our analysis indicate that using the DTx for six months resulted in a clinically relevant weight reduction of 6.3%, providing significant benefits by potentially reducing cardiovascular risks.
The quality of life was assessed using the SF-36 tool, with generally accepted minimal clinically important differences (MCIDs) ranging from 2 to 4 points across various scales and scores.34,35 For individuals with diabetes, a decrease of 1 point in specific SF-36 scales is linked to a noteworthy 9% higher risk of mortality and a 12% increased risk of inability to work, considered clinically significant. 36 For participants with a BL-HbA1c level of ≥ 7.0%, there was a substantial improvement, with a 3.06-point increase in the physical summation scale and a 4.64-point increase in the psychological summation scale. This improvement implies a relevant enhancement in the quality of life, potentially translating to improved daily functioning and productivity.
The DTx is designed to improve diabetes management by implementing personalized lifestyle changes. It is primarily intended to guide patients toward self-efficacy through low-threshold support without feeling overwhelmed. This can be achieved through regular, but not necessarily daily use of the app. This fits with the observation that the participants used the app on average 4.7 to 6.5 days per week.
The ongoing shift toward digital technologies in health care has the power to drive positive change.2,37 The DTxs are evidence-based interventions powered by high-quality software programs designed to prevent, manage, or treat medical conditions. Overall, DTx provide intelligent and accessible tools that empower patients and clinicians and address various conditions through safe and effective data-driven interventions. 38 For instance, an umbrella review from 2020 classified the effects of generic digital approaches for managing T2D between −0.30% and −0.80% HbA1c. 22 The recent Digital Diabetes Management Assessment Report (2024) concluded that six DTx facilitating behavior and lifestyle modification achieved an incremental clinical benefit, defined as an HbA1c benefit of between 0.23% and 0.60% compared with standard care. 28 The evidence base is heterogeneous due to differences in approach, regulatory oversight, and integrated in-person coaching elements. Therefore, it is necessary to differentiate between the DTx. In Germany, DTxs are regulated as DiGA and are approved by the BfArM for reimbursement following a rigorous regulatory, technical, security as well as evidence assessment. 10 Consequently, the DTx that was examined here is evidence-based and clinically relevant.
Often, DTx have in-person coaching elements embedded alongside the digital component, making them only partially digital and more difficult to scale. However, the scalability of DTx approaches is important to assure cost effectiveness, as the prevalence of T2D will rise faster than the limited resource available to health care professionals to cope with this public health emergency.2,39 The DTx used in this study can overcome this hurdle as it is a fully digital intervention and does not rely on complimentary in-person coaching elements.
This study has limitations as patients self-reported their outcomes, which increases the risk of data quality being influenced by recall or social desirability bias. To minimize this risk, measures such as validated questionnaires, clear instructions, and efforts to create a comfortable and trusting environment for participants were taken.
The study cohort represents a suitable cohort for the target population of the DTx; however, it can be assumed that the cohort is a rather technically skilled and motivated group in relation to the complete group of patients with non–insulin-treated T2D.
The pilot study, using a pre-post comparison design, offers initial insights into the efficacy of the DTx by measuring outcomes before and after the intervention within the same group. However, to definitively prove the effectiveness of the DTx, an RCT with a parallel control group is essential. This minimizes biases and external influences, providing more robust and conclusive evidence of the DTx’s effectiveness. The RCT study that rectifies this limitation is registered with the German Registry of Clinical Studies as DRKS00032537 and is currently in progress.
Conclusions
In conclusion, the DTx represents a significant advancement in T2D management, providing a non-pharmacological, fully digital, and scalable solution that integrates continuous glucose biofeedback and daily companionship which are essential components of successful diabetes therapy that are now available in combination of standard therapeutic components of lifestyle therapy. This is made possible by the DTx’s proprietary advanced AI algorithm, which predicts individual postprandial glucose responses.
The study demonstrates the efficacy and holistic benefits of the DTx through observed reductions in HbA1c values, improvements in body weight, and enhancements in quality of life metrics. It also highlights the broader paradigm shift toward digital technologies in health care and emphasizes the importance of evidence-based digital interventions.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241266821 – Supplemental material for Unlocking Potential: Personalized Lifestyle Therapy for Type 2 Diabetes Through a Predictive Algorithm-Driven Digital Therapeutic
Supplemental material, sj-docx-1-dst-10.1177_19322968241266821 for Unlocking Potential: Personalized Lifestyle Therapy for Type 2 Diabetes Through a Predictive Algorithm-Driven Digital Therapeutic by Swantje Kannenberg, Jenny Voggel, Nils Thieme, Oliver Witt, Kim Lina Pethahn, Morten Schütt, Christian Sina, Guido Freckmann and Torsten Schröder in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank all patients who voluntarily gave consent to use their data for this data analysis.
Abbreviations
BL, baseline; BMI, body mass index; CGM, continuous glucose monitoring; DiGA, digital health application (German: Digitale Gesundheitsanwendung); DTx, digital therapeutic; DPP-4, dipeptidyl peptidase-4 inhibitors; T2D, type 2 diabetes; BfArM, Federal Institute for Drugs and Medical Devices (German: Bundesinstitut für Arzneimittel und Medizinprodukte); GLP-1, glucagon-like peptide-1; HbA1c, glycated hemoglobin; ICD, International Statistical Classification of Diseases and Related Health Problems; PGIC, Patients’ Global Impression of Change Scale; SF-36, Short-Form 36 Questionnaire; SGLT2, sodium-glucose transport protein 2 inhibitors.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.V., N.T., K.L.P., O.W., and T.S. are employed at Perfood GmbH. T.S. and C.S. are co-founders of Perfood GmbH and minority shareholders. G.F. is general manager and medical director of the Institute for Diabetes Technology (Institut für Diabetes-Technologie (IfDT) Forschungs-und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies, eg, with medical devices for diabetes therapy on its own initiative and on behalf of various companies. G.F. and IfDT have received speakers’ honoraria or consulting fees in the last three years from the following companies: Abbott, Berlin Chemie, Boydsense, Dexcom, Lilly Deutschland, Novo Nordisk, Perfood, Pharmasens, Roche, Sinocare, Terumo, and Ypsomed. M.S. runs an Institute for Diabetology and has received speakers’ honoraria or consulting fees in the last three years from Novo Nordisk, Lilly Deutschland, Astra Zeneca, Sanofi, Boehringer Ingelheim, Berlin Chemie, Novartis, and Perfood.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Perfood GmbH, the manufacturer of the prescription digital therapeutic “glucura”.
Ethics Approval
This is a clinical trial according to Article 74 (1) MDR of a device already CE-marked according to Article 20 (1) MDR within the scope of its intended purpose, which serves the purpose of further evaluation. All data sets were anonymized before analysis. This procedure was approved beforehand by the ethics committee of the University of Lübeck (AZ 2023-804).
Informed Consent Statement
All patients gave their consent to the use of their anonymized data for these scientific purposes.
Data Availability Statement
The data sets used and/or analyzed during this study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
