Abstract
Background:
Reliable blood glucose (BG) measurements are important for people with diabetes to manage their therapy as well as in point-of-care testing (POCT) performed by health care professionals to monitor BG of patients or even to diagnose diabetes. Among other factors, endogenous and exogenous substances present in blood samples can impact the measurement results. To ensure and prove that blood glucose monitoring systems (BGMSs) are robust in terms of potential interferents, manufacturers have to perform extensive evaluations.
Method:
An interference screening test was performed for three reagent system lots of a POCT system and of a BGMS for self-monitoring of BG. A paired-difference approach based on ISO 15197:2013 and CLSI guideline EP07 was used with venous whole blood samples at two different glucose concentrations. Seventy potential interferents expected to be common in people with diabetes were evaluated.
Results:
The interference effects were determined as normalized biases between test samples and corresponding control samples. For 69 of the 70 investigated potential interferents, both systems met the predefined acceptance criteria, with the normalized biases falling within ±10 mg/dL or ±10% at glucose concentrations ≤100 mg/dL or >100 mg/dL, respectively, for each of the three evaluated reagent system lots.
Conclusions:
The BGMS investigated in this study were found to be robust with respect to the 70 evaluated potential interferents. Interference effects were observed only for N-Acetyl-L-cysteine. Extensive evaluations of potential interfering factors can make an important contribution to ensure reliability of BGMS.
Introduction
Reliable blood glucose (BG) measurements are an important prerequisite for people with diabetes to manage their therapy as well as in point-of-care testing (POCT) performed by health care professionals to monitor BG of patients or even to diagnose diabetes. Besides, for example, deficiencies in production processes of BG meters and reagent systems or handling issues during the BG measurements, there are also influence quantities present in the blood sample, which may impact the measurement result. These influence quantities include the hematocrit, although nowadays many blood glucose monitoring systems (BGMSs) are able to compensate the effect of hematocrit. Other interfering factors are substances of either endogenous or exogenous origin. Endogenous substances like uric acid may, for example, cause interference when present in elevated levels due to disease. 1 Other potential interferents are drugs, metabolites, food components, but also substances added during sample preparation like anticoagulants. 2
To ensure and prove that BGMSs brought to the market are robust in terms of potential interferents, manufacturers have to perform extensive evaluations. According to the In-Vitro Diagnostic Medical Device Regulation (IVDR), manufacturers have to identify and control relevant interfering factors in the context of their implemented risk management systems. 3 In addition, data collected at the time of market introduction should also be reviewed and updated regularly after a BGMS has been placed on the market, for example, to account for newly available drugs.
Numerous studies dealing with the accuracy of BGMSs were published in the last decade. In contrast, there are few publications on comprehensive evaluations of interference effects with most of them addressing only a small number of substances.4-10
The ISO standard ISO 15197:2013 specifies requirements for interference testing in BGMSs for self-testing. 11 This standard was harmonized with the regulations of the European Union and thus can be applied in conformity assessments of BGMSs for self-testing, however, respective regulations for POCT systems do not exist. In contrast, the U.S. Food and Drug Administration (FDA) provides two Guidances for Industry and FDA staff, one of them refers to BGMSs for self-testing, the other to POCT systems.12,13 Both guidances address interference evaluations applying identical requirements for BGMSs for self-testing and POCT systems. A general guideline for interference testing in clinical chemistry (CLSI EP07) and supplemental tables, including recommended test concentrations for numerous potential interferents published in an additional document (CLSI EP37), are provided by the Clinical and Laboratory Standards Institute (CLSI).2,14 The interference screening procedure described by the CLSI guideline EP07 is also referenced by ISO 15197:2013; however, ISO 15197 provides more detailed requirements regarding testing procedures and acceptance criteria which CLSI EP07 leaves to the discretion of the user of the guideline. In case interference is observed for an evaluated factor, ISO 15197:2013 recommends to perform a dose-response evaluation to determine the lowest concentration at which interference occurs and to describe the interference effect in the instructions for use.
In this study, interference screening testing was performed for a POCT system and a BGMS for self-monitoring of BG based on the requirements of ISO 15197:2013 and procedures described by CLSI EP07. The substances required to be evaluated according to ISO 15197:2013 Annex A had been investigated previously (data for 24 substances on file at manufacturer), however, this study focused on a subset of potential interferents expected to be common in people with diabetes.
Methods
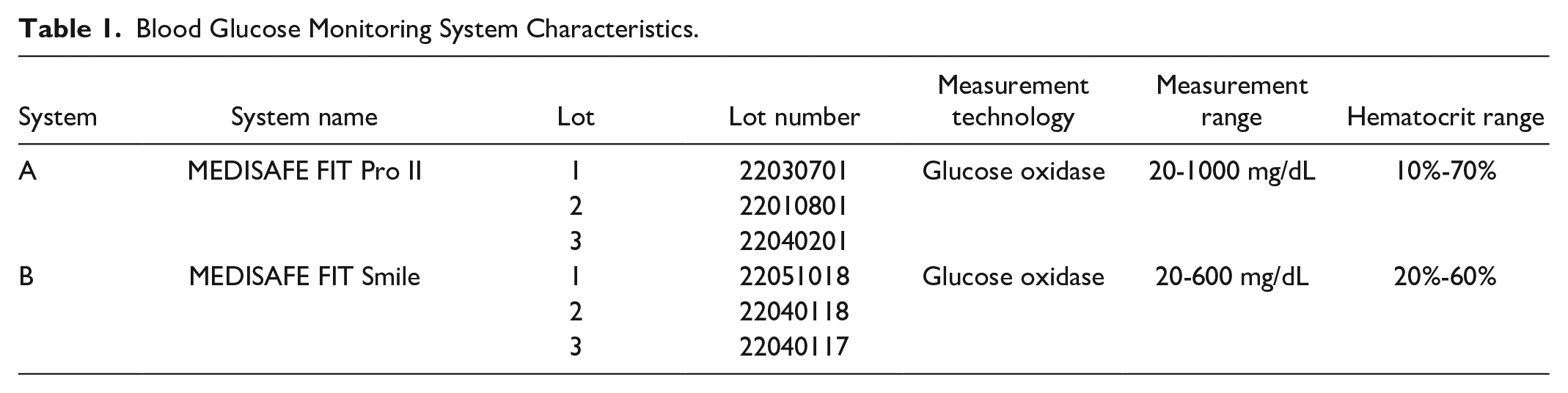
In this study, interference testing was performed for three reagent system lots each of a POCT system (MEDISAFE FIT Pro II, Terumo Corporation, Tokyo, Japan [System A]) and a BGMS for self-monitoring of BG (MEDISAFE FIT Smile, Terumo Corporation [System B]). Both systems use test tips with a glucose oxidase enzyme (Table 1). The evaluation was performed at the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm (IfDT) in Germany. The Institute is accredited as a testing laboratory according to ISO/IEC 17025:2017 by the Deutsche Akkreditierungsstelle GmbH (DAkkS) in terms of performance evaluations according to ISO 15197:2013, including interference testing. Study procedures were based on the requirements of ISO 15197:2013 and, in addition, taking specifications and procedures of CLSI EP07 into consideration. Both systems were maintained and operated in accordance with their respective instructions for use.
Blood Glucose Monitoring System Characteristics.
Seventy potential interferents expected to be common in people with diabetes were tested with one test concentration in a paired-difference testing approach (Table 2, Figure 1). Test concentrations were taken from CLSI EP37, if available. 14 For potential interferents not listed in EP37, test concentrations were either calculated as stipulated by CLSI EP07, taking at least three times the concentration expected to be present in blood following a therapeutic drug dose, or—in case of two substances—test concentrations were chosen based on requests obtained from clinical technicians. A dose-response-evaluation as recommended by ISO 15197:2013 was not included in this study.
Potential Interferents and Test Concentrations Evaluated in This Study.
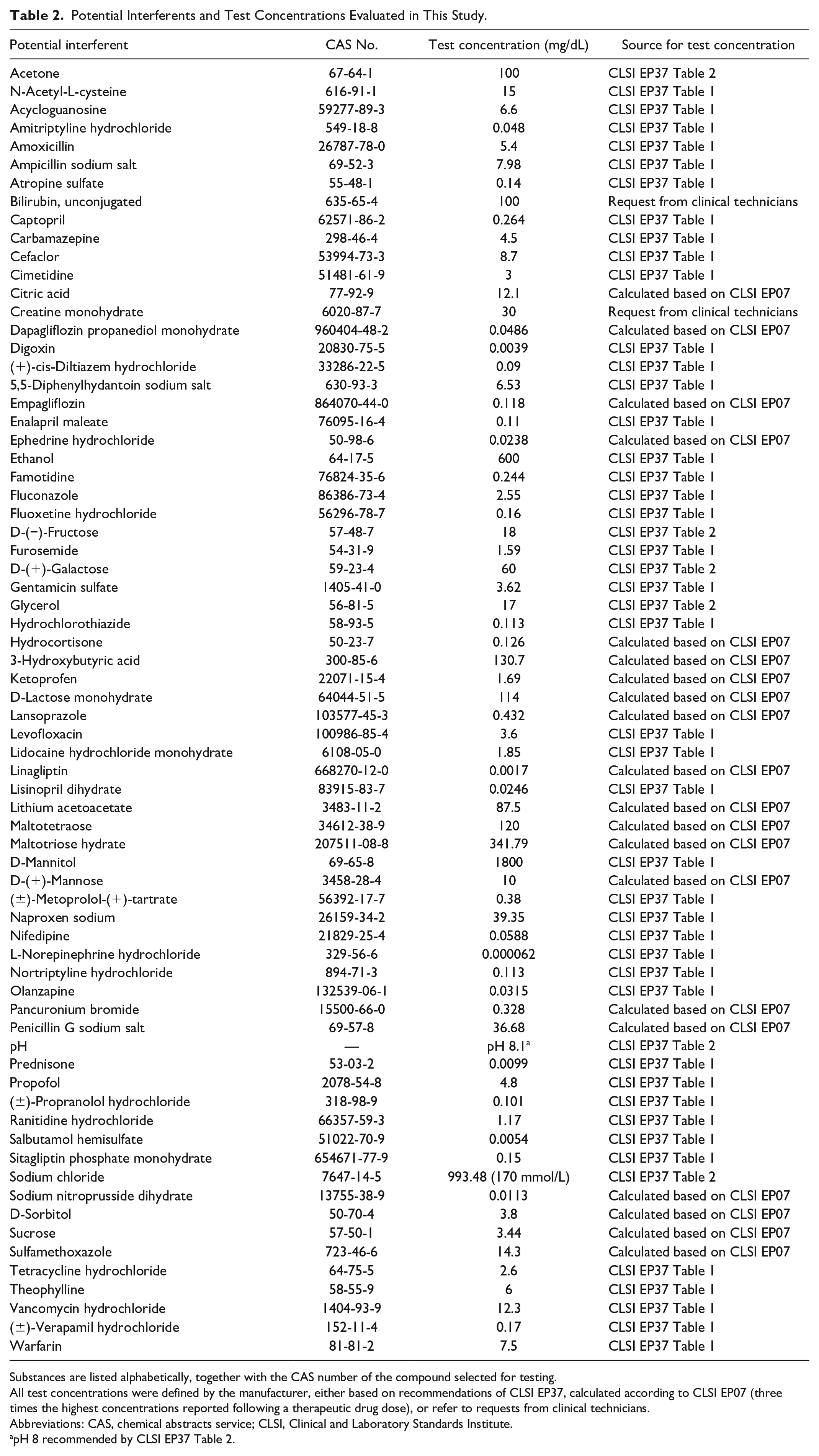
Substances are listed alphabetically, together with the CAS number of the compound selected for testing.
All test concentrations were defined by the manufacturer, either based on recommendations of CLSI EP37, calculated according to CLSI EP07 (three times the highest concentrations reported following a therapeutic drug dose), or refer to requests from clinical technicians.
Abbreviations: CAS, chemical abstracts service; CLSI, Clinical and Laboratory Standards Institute.
pH 8 recommended by CLSI EP37 Table 2.
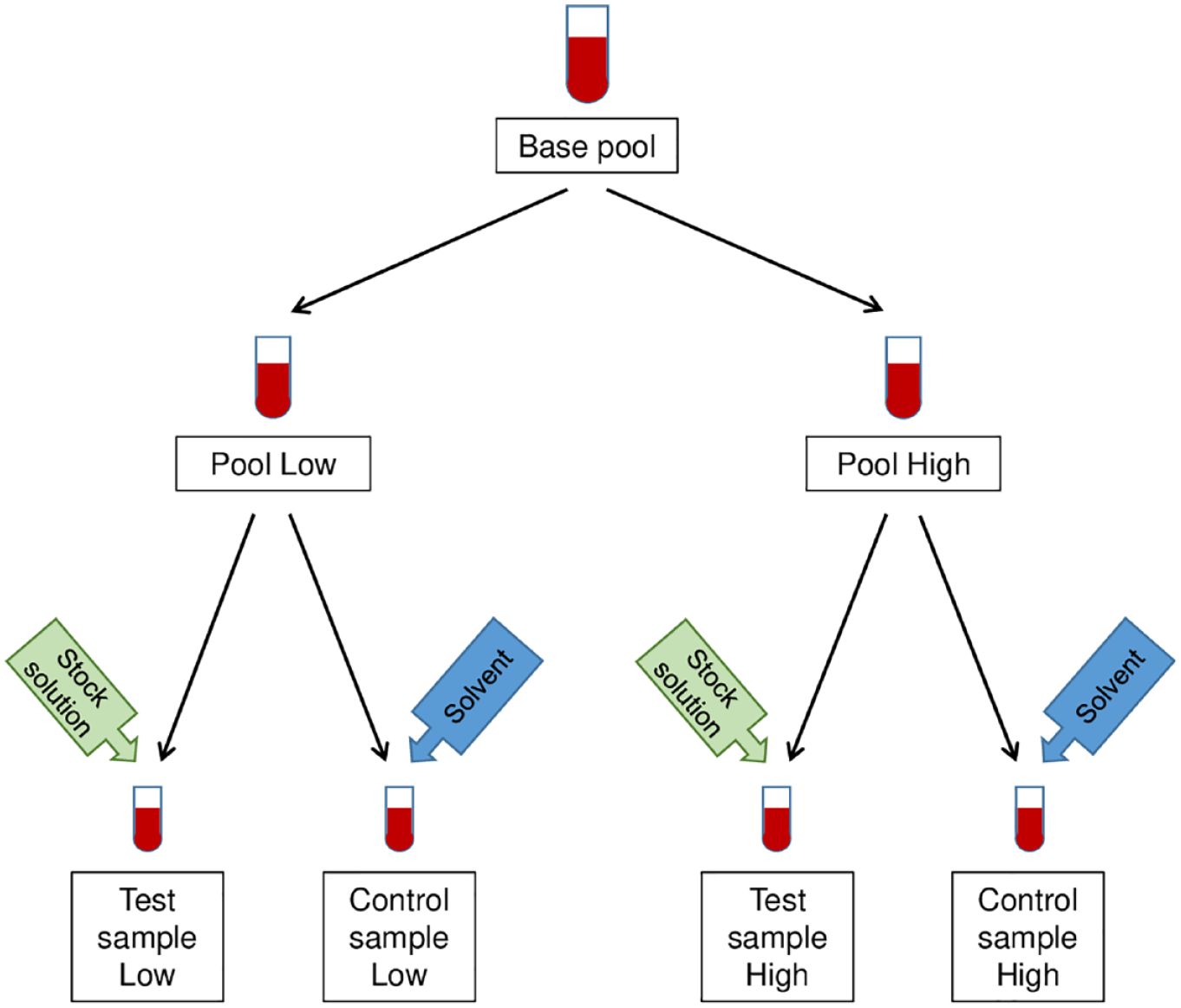
Preparation of test and control samples for one potential interferent. A base pool, prepared with blood collected from one donor, was split into two subpools, one for testing at a low glucose concentration (50-100 mg/dL), the other for testing at a high glucose level (250-350 mg/dL). From each of these subpools (low and high glucose) two aliquots were taken to prepare test samples and control samples, respectively. The test samples were spiked with a stock solution of the potential interferent, whereas the identical volume of the respective solvent was added to the designated control sample.
Testing was performed using venous whole blood samples obtained in lithium-heparin tubes from 38 volunteers (six of them participating twice), not taking vitamin C or any medications within the last 48 hours besides insulin, thyroxin, or contraceptives. In addition, the hematocrit values of the samples were determined and verified to fall within the BGMSs’ labeled ranges (10%-70 % [system A], 20%-60% [system B]), but preferably between 35% and 50%.
The study was approved by the Ethics Committee (Ethikkommission bei der Landesärztekammer Baden-Württemberg, Application number MP-2018-010), exempted from approval by the Competent Authority (Bundesinstitut für Arzneimittel und Medizinprodukte), and conducted in compliance with the Guideline for Good Clinical Practice and under consideration of the Declaration of Helsinki (revised edition, Fortaleza 2013). All participants gave their informed consent before study-related procedures were started.
Preparation of Blood Samples
Each potential interferent was tested using blood from one donor (base pool). The base pool was split into two subpools, one for testing at a low glucose concentration (50-100 mg/dL), the other for testing at a high glucose level (250-350 mg/dL), as required by ISO 15917:2013. The respective glucose levels were adjusted by adding a concentrated glucose solution, if required. Glucose concentrations were assigned using a laboratory analyzer (Cobas Integra 400 plus, Roche Diagnostics GmbH, Mannheim, Germany). From each of both subpools (low and high glucose), two aliquots were taken to prepare test samples and control samples, respectively. The test samples were spiked with a stock solution of the respective potential interferent (see below), whereas the identical volume of the solvent was added to the designated control sample, to account for effects of solvent and dilution (Figure 1).
Preparation of Stock Solutions of Potential Interferents
Powdered or liquid substances were dissolved in suitable solvents before being added to the blood samples, if required. Adequate concentrations of these stock solutions were chosen considering the respective substance’s targeted test concentration and solubility. However, excess dilution of the test samples leading to changes in the sample matrix had to be avoided. As recommended by CLSI EP07, stock solutions with at least 20 times the designated test concentration were used, except for D-Mannitol, Maltotetraose and Bilirubin where only 10-fold stock solutions could be prepared due to limited availability of substance or lack of solubility.
A similar approach as for substances was also used to adjust the pH value. A diluted sodium hydroxide solution was added to the designated test sample until a pH value of 8.1 was reached whereas an identical volume of 0.9% NaCl was added to the control sample.
Measurement Procedure
Measurements were performed according to ISO 15197:2013 under standardized temperature conditions, that is, room temperature was kept in the range of 23°C ± 5°C, and samples were set to a starting temperature of 23°C ± 5°C and kept within ±3°C during measurements. Humidity was verified to be within the range given in the instructions for use (30%-85%). Ten consecutive measurements were performed on test samples and control samples with each of three reagent system lots, using 10 meters for each system. Before and after these 30 measurements with each system, sample aliquots were removed for plasma separation, followed by duplicate glucose measurements with a laboratory analyzer. These measurements were intended to verify sample stability and to calculate normalized biases (see below).
Measurements of test samples and control samples were completed within 1 hour of spiking with stock solution or solvent, respectively.
Trueness and precision of the laboratory analyzer were verified during the experimental phase based on requirements of the Guideline of the German Medical Association on Quality Assurance in Medical Laboratory Examinations (Rili-BÄK). 15
Data Analysis
Sample stability over the course of the measurement series was verified by comparing the mean glucose concentrations measured with the laboratory analyzer before and after the measurements with the BGMSs. Samples were rated stable, if the calculated deviations were ≤4 mg/dL or ≤4% for glucose concentrations <100 mg/dL or ≥100 mg/dL, respectively.
In deviation from ISO 15197 and CLSI EP07, interference effects were determined by calculating the normalized bias between test sample and corresponding control sample. For this purpose, the difference between the mean BGMS results obtained from test sample and control sample was normalized by subtracting the difference calculated between mean results from test sample and the control sample measured with the laboratory analyzer. This normalization is suitable to eliminate other confounding factors, for example, glycolysis effects varying between test sample and control sample. As this approach is based on the assumption that the laboratory analyzer does not show relevant interference caused by the evaluated substances, influence quantities labeled for the analyzer have to be considered. In this study, a test concentration of 100 mg/dL was used for Bilirubin, exceeding the concentrations declared to not show interference with the laboratory analyzer and reagent system used. 16 After critically reviewing the data, the results obtained for Bilirubin were considered valid.
The normalized bias at both glucose concentration ranges is calculated for each reagent system lot separately, as well as for the three lots taken together.
Based on the criteria stipulated by ISO 15197 for interferents which have to be described in the instruction for use, the following acceptance criteria were defined: For glucose concentrations ≤100 mg/dL or >100 mg/dL, the normalized bias shall be within ±10 mg/dL or ±10%, respectively.
Results
Interference screening was performed using venous whole blood samples with glucose concentrations distributed in two different concentration ranges of 50 to 100 mg/dL and 250 to 350 mg/dL, respectively. The interference effects were determined as normalized biases between test samples and corresponding control samples (Table 3, Figure 2). For 69 of the 70 investigated potential interferents, both systems met the predefined acceptance criteria, with the normalized biases falling within ±10 mg/dL at glucose concentrations ≤100 mg/dL and ±10% at glucose concentrations >100 mg/dL for each of the three evaluated reagent system lots. For one substance, N-Acetyl-L-cysteine, the acceptance criteria were met only in the high glucose concentration range. At low glucose concentrations, one of three reagent system lots of systems A and B fell outside the acceptance criteria with a normalized bias of −10.6 and −14.3 mg/dL, respectively. For systems A and B, normalized biases of 66 and 67 potential interferents, respectively, fell within ±5 mg/dL at glucose concentrations ≤100 mg/dL and ±5% at glucose concentrations >100 mg/dL with all three evaluated reagent system lots.
Results.
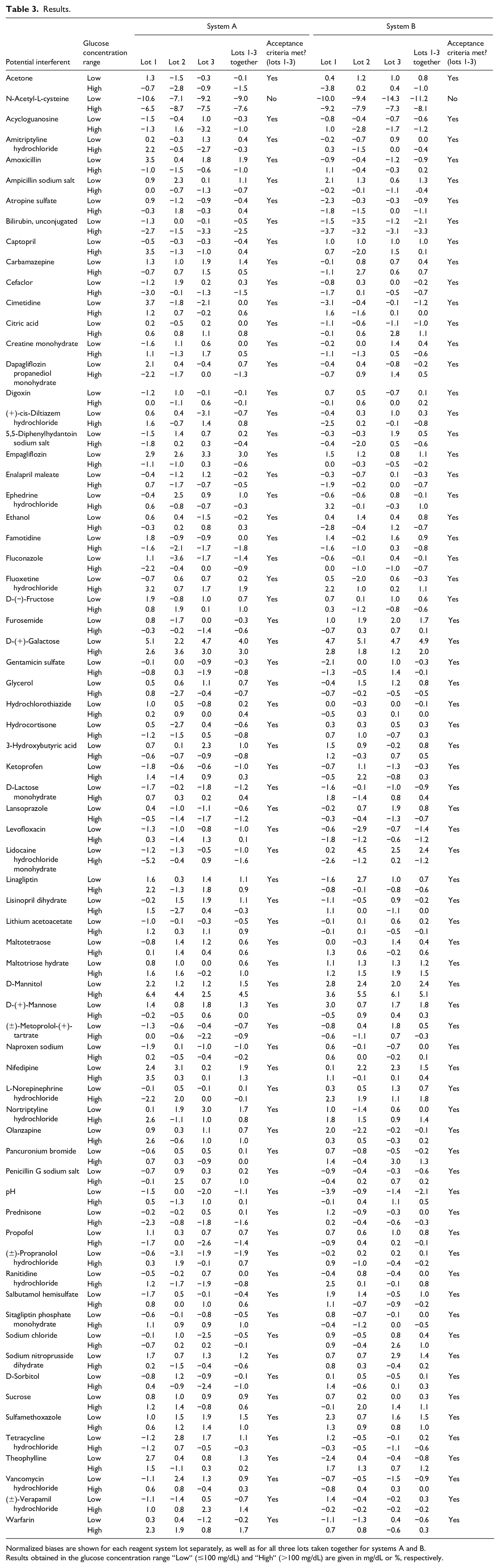
Normalized biases are shown for each reagent system lot separately, as well as for all three lots taken together for systems A and B. Results obtained in the glucose concentration range “Low“ (≤100 mg/dL) and “High“ (>100 mg/dL) are given in mg/dL or %, respectively.
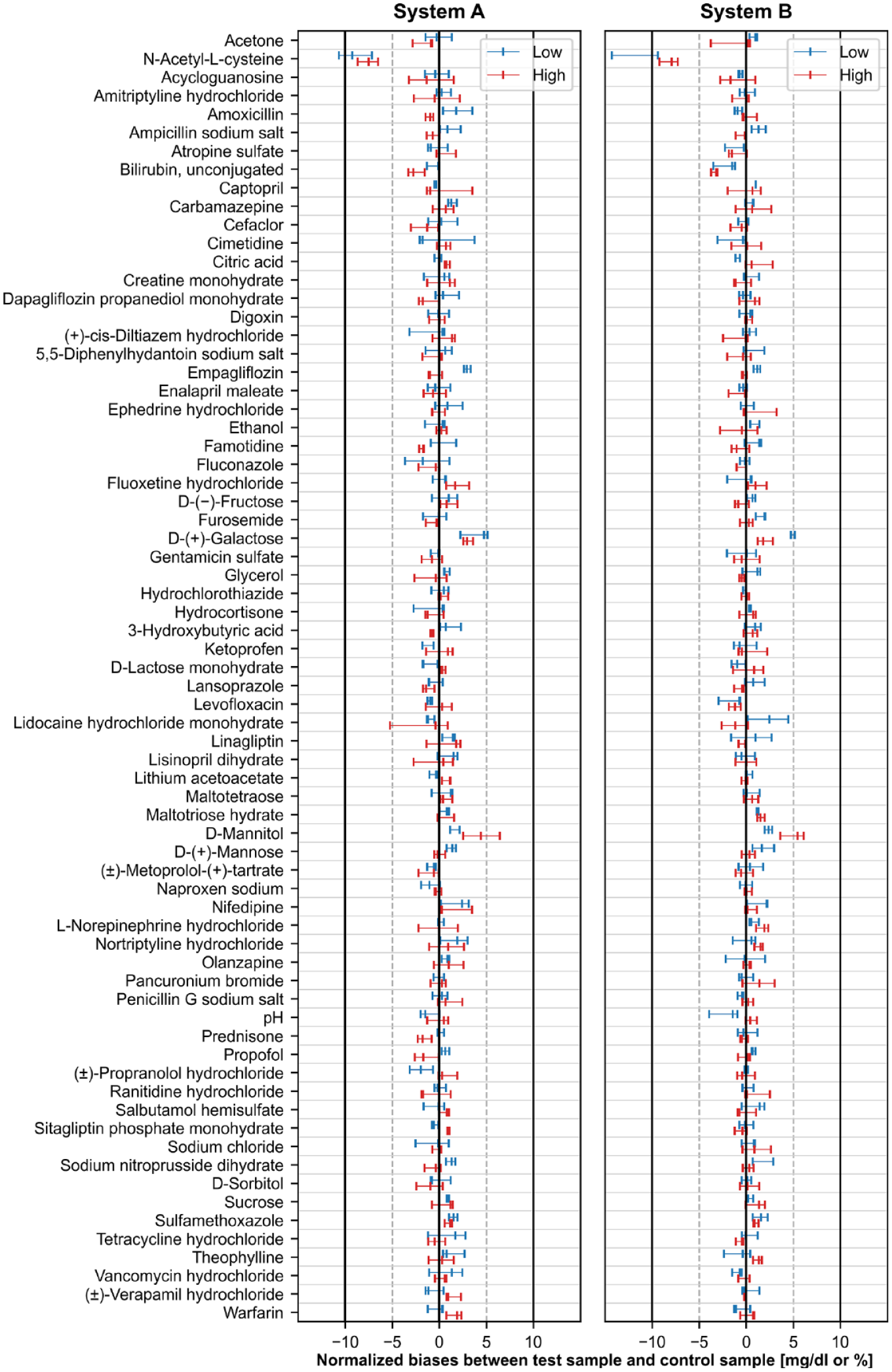
Normalized biases obtained for 70 potential interferents evaluated with systems A and B. The range of normalized biases is given for glucose concentrations ≤100 mg/dL (Low) and >100 mg/dL (High) as blue and red horizontal lines, respectively, with vertical lines representing the location of the normalized biases for the three evaluated reagent system lots. Black solid lines indicate the defined acceptance limits of 10 mg/dL or 10% at glucose concentrations ≤100 mg/dL or >100 mg/dL, respectively. The dashed gray lines represent normalized biases of ±5 mg/dL or ±5%.
Discussion
In this study, an interference screening test based on the requirements of ISO 15197:2013 was performed for two BGMSs, one POCT system and one BGMS for self-testing, evaluating a subset of 70 potential interferents expected to be common in people with diabetes.
This screening test revealed no interference effect with 69 of the 70 potential interferents for both systems, when applying the predefined acceptance criteria of normalized biases within ±10 mg/dL at glucose concentrations <100 mg/dL and within ±10% at glucose concentrations ≥100 mg/dL. Normalized biases of 66 and 67 potential interferents even fell within tighter intervals of ±5 mg/dL or ±5% at glucose concentrations ≤100 mg/dL or >100 mg/dL, respectively, for BGMSs A and B.
The interference testing procedure used in this study is an in-vitro test intended for screening purposes, and therefore limitations have to be considered. For example, the properties of chemical compounds used in the study to prepare the test sample may differ from those of the compound naturally occurring in blood samples, for example, after ingestion of a drug. 2 Furthermore, interference may not be detected with the investigated concentrations of glucose and potential interferent, as interference effects may offset at certain constellations of interferent and measurand concentrations. 2 Thus, results obtained using samples generated in-vitro cannot completely substitute an evaluation of patient samples.
The testing protocol used in this study refers to the requirements of ISO 15197:2013, which is intended for BGMSs for self-testing and is therefore not directly applicable to POCT systems. However, as respective requirements for POCT systems do not exist and as the used procedures are based on the CLSI guideline EP07 which addresses laboratory measurement procedures in general, using the identical procedures for the evaluation of BGMSs for self-testing and for POCT systems seems reasonable. This also applies to the acceptance criteria, as POCT systems should fulfill at least the performance requirements stipulated for self-testing systems.
According to ISO 15197 and CLSI EP07, interference effects are determined as differences or relative differences between mean glucose concentrations obtained for test samples and corresponding control samples. In this evaluation, a modified calculation was used by determining a normalized bias, with the aim to compensate for differences in glucose concentrations between test samples and control samples which are not attributable to the respective potential interferent. This may be especially beneficial regarding glycolysis effects, as measurements on test samples and control samples have to be performed sequentially, and thus glycolysis may lead to lower glucose concentrations in the last measured sample.
The potential interferents investigated in this study were selected based on their relevance in people with diabetes and include common drugs and metabolites. Medication frequently found in people with diabetes, including antidiabetic agents, medicines used for the treatment of cardiovascular, gastrointestinal, or infectious diseases, as well as psychotropic and analgesic medicines were considered. A recent cohort study of 1023 people with type 1 or type 2 diabetes showed that at least approximately 20% of participants used medications from these groups. This underlines the importance to ensure that such substances do not impact BG measurements. 17
Among the potential interferents evaluated in this study, only N-Acetyl-L-cysteine, a substance used as mucolytic agent and as antidote for acetaminophen intoxications, caused interference based on the defined acceptance criteria. Interference effects caused by N-Acetyl-L-cysteine are reported to also occur with other BGMSs and with different laboratory assays.18-22 However, the interference effect observed in this evaluation was only limited, as it was restricted to the tested low glucose concentration range and to one of the three evaluated reagent system lots for systems A and B, respectively. Thus, in summary, both tested BGMSs were found to be robust with respect to the evaluated potential interferents. Extensive evaluations of potential interfering factors performed by manufacturers can make an important contribution to ensure reliability of BGMSs.
Footnotes
Acknowledgements
The authors would like to thank Natalie Volz and Jessica Kaifel for their expert performance of the laboratory work, and the study participants for the provision of the required blood samples.
Abbreviations
BG, blood glucose; BGMS, blood glucose monitoring system; CAS, chemical abstracts service; CLSI, Clinical and Laboratory Standards Institute; FDA, U.S. Food and Drug Administration; ISO, International Organization of Standardization; IVDR, In-Vitro Diagnostic Medical Device Regulation; POCT, point-of-care testing; Rili-BÄK, Richtlinie der Bundesärztekammer zur Qualitätssicherung laboratoriumsmedizinischer Untersuchungen.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is general manager and medical director of the Institute for Diabetes Technology (Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany) that carries out clinical studies, for example, with medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IfDT have received research support, speakers’ honoraria, or consulting fees in the last 3 years from Abbott, Ascensia, Berlin Chemie, Boydsense, Dexcom, Lilly, Metronom, Medtronic, Menarini, MySugr, Novo Nordisk, PharmaSens, Roche, Sanofi, Terumo. AB, SP, JM, NJ, MT, SS, and CH are employees of IfDT at the time of study conduct. TS, TA, and MS are employees of Terumo Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study and medical writing was supported by TERUMO Europe NV.
