Abstract
Self-monitoring of blood glucose is a key aspect of diabetes management. Depending on the technology used, however, various substances can jeopardize the reliability of the measurements and precipitate complications with potentially life-threatening consequences when blood glucose was deemed well-controlled. As such, it is important for all involved to be aware of those factors. Officially suggested procedures for testing and alternatives have each their own advantages and limitations, and interferences may be found beyond the substances to be tested provided by the various pertinent institutions. This article reviews these pros and cons and illustrates how interference testing beyond established standards contributes to patient safety. Once identified, interfering substances are included in product labeling and healthcare professionals and users need to be trained to be aware of these risks.
Why Interference Testing?
Self-monitoring of blood glucose (SMBG) is the most widely used method for measuring glucose worldwide. It is an important part of diabetes management for people with diabetes (PwD). For PwD treated with insulin, it is a critical part for determining the correct insulin dose. Even for PwD not on insulin, it can help understanding effects of food, medication, intercurrent illness, and lifestyle changes, and help to assess the effectiveness of antidiabetic therapy, permitting PwD and healthcare professionals (HCPs) to personalize diabetes management and avoiding therapeutic inertia.1 -9
Self-monitoring of blood glucose can thus help to improve long-term glycemic control and/or avoid acute hypo- and hyperglycemic events. However, a falsely high reading can lead to a missed hypoglycemic event or even induce one, due to incorrect insulin dosing. Conversely, a false low reading can mask or lead to a hyperglycemic event. Both types of events can lead to poor outcomes, ranging from increased likelihood of comorbidities 10 to death. 11 Therefore, accurate measurement of the glucose concentration is important.
However, the accuracy of a result does not only depend on the general accuracy of the measurement technology used but can be confounded by various interfering substances. 11 Given that people with type 2 diabetes are prescribed an average of 8.4 different drugs in Germany 12 and 4.1 in the United States, 13 polypharmacy is particularly common among PwD. This both increases the number of potentially present interfering substances and the likelihood that a drug will be replaced by novel therapeutic options at some point.
Knowledge of what substances can interfere with glucose measurement is thus critical to assess how reliable a measurement result is.
What Is Interference, Anyway?
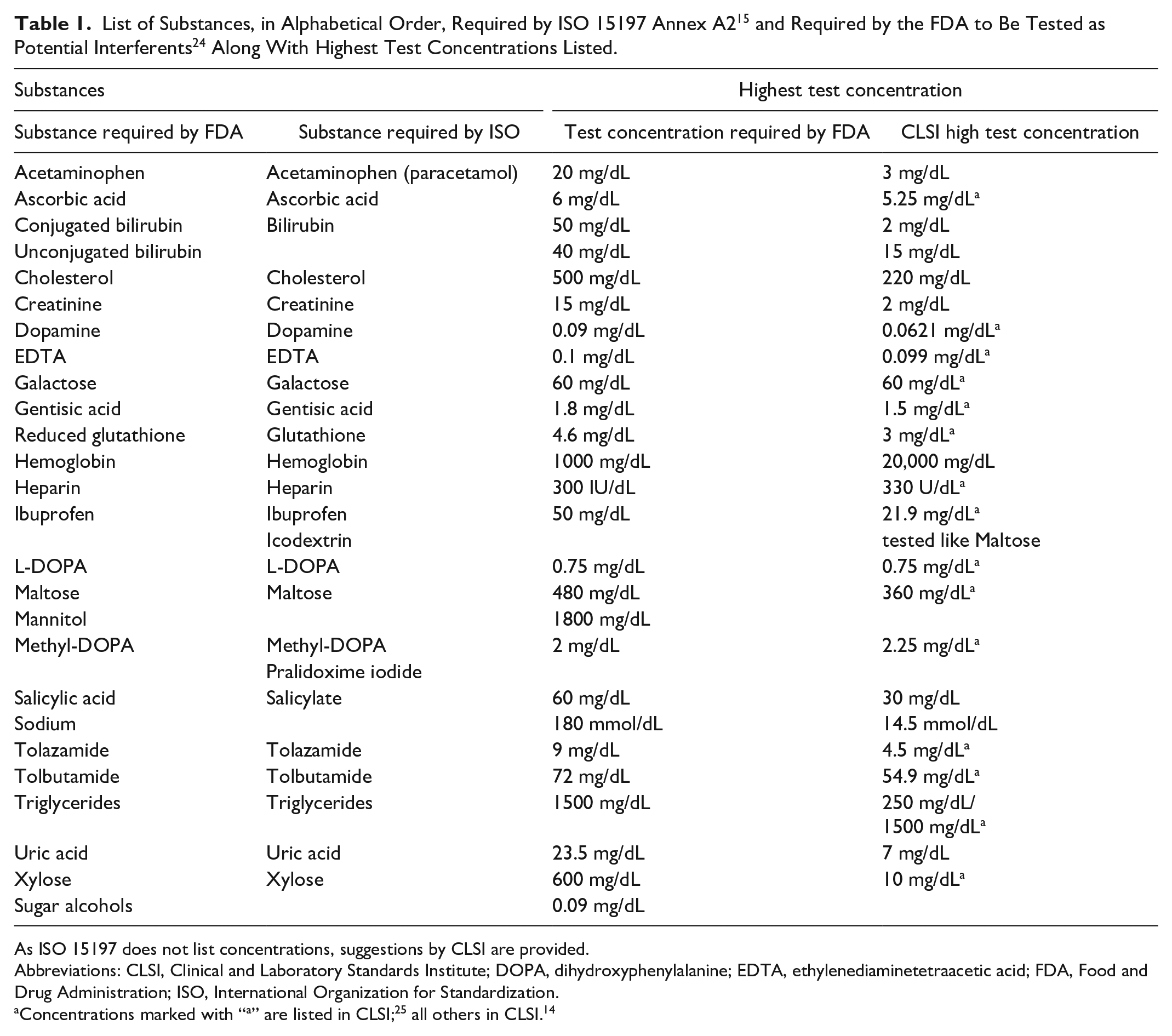
The Clinical and Laboratory Standards Institute (CLSI) defines an interference as “a cause of medically significant difference in the measured test result due to the effect of another component or property of the sample.” 14 What constitutes an acceptable degree of interference varies by medical implications. For blood glucose (BG) measurements, the International Organization for Standardization (ISO) standard 15197:2013 and the European harmonized version EN ISO 15197:2015 define the limit for concentrations <5.55 mmol/L (<100 mg/dL) as a mean difference of 0.55 mmol/L (10 mg/dL) and for higher concentrations of 10% 15 (Figure 1). Interferences exceeding these limits must be labeled.
Area of allowed deviation (in blue) of a glucose measurement result from the result of a designated comparison method as per ISO 15197:2013. 15 For glucose concentrations below 100 mg/dL, the acceptable difference is 10 mg/dL on average. For higher concentrations, the acceptable difference is 10% of that concentration on average. (Note that this is lower than the limits for overall system accuracy requested by ISO 15197:2013, which are at 15 mg/dL or 15%, respectively, for 95% of all measurements). Abbreviation: ISO, International Organization for Standardization.
Any measurement technique can be subject to interference, based simply on the fact that manufacturers design and verify their systems under an expected range of clinical and environmental parameters. Within these parameters, a thorough risk management system is applied during product development to ensure that safety and performance are maintained for most circumstances the product will encounter in the real world. However, these safeguards do not preclude all scenarios where an inaccurate result may be provided by the system.
Interfering factors can be categorized into two groups: Endogenous factors, that is, various physiological characteristics, such as hematocrit, triglycerides, or bilirubin, and exogenous factors, such as vitamin C, acetaminophen, or other drugs, or temperature or altitude outside the verified range of operation. Endogenous factors can be the consequence of various physiological (eg, pregnancy) or pathological conditions, such as particular metabolites or changes in the composition or properties of the sample (eg, a pathological hematocrit, low or high O2 tension). Exogenous factors can be substances introduced during patient treatment, such as drugs, anticoagulants, and parenteral food, or substances ingested by the patient on their own initiative—drugs of abuse, nutritional supplements, or various foods or drinks. They may also include substances added during sample preparation, be it consciously, such as anticoagulants, preservatives, or stabilizers, or inadvertently, as contaminants, be it from hand creams, soap, glove powder, or macroscopic objects, such as stoppers, and so on. In addition, storage and ambient conditions may also change the chemical and physical properties of the sample to a state that interferes with measurements.
Interfering substances themselves can lead to a measurement bias (measurement error caused by the interfering substance) in a patient, depending on their concentration in the blood. This also means that the bias changes as the concentration changes, which can lead to erroneously misinterpreting a change in interfering substance concentration as a change in the concentration of the substance to be measured. For BG measurement, a variety of conditions and interferences affect the accuracy of the result, depending on the chemistry used and the individual measurement technology.
How Interference Happens in SMBG
The SMBG test strips contain enzymes, which react with glucose, a mediator, and a coenzyme. Electrodes to elicit a signal for the readout may also be present directly in the reaction and, for strips using a photometric readout, an indicator for color formation is included. Interference depends on the combination of the enzyme and the coenzyme. A variety of glucose oxidases (GODs) and glucose dehydrogenases (GDHs) from various biological sources, together with one of several coenzymes (GOD with Flavin adenine dinucleotide [FAD] as coenzyme, or three different types of GDH designated according to their respective coenzyme, FAD, pyrroloquinoline quinone [PQQ], or nicotinamide adenine dinucleotide [NAD]), are used in SMBG systems. Modification of the protein chain of the enzyme has led to SMBG systems with higher glucose specificity, in particular regarding other monosaccharides.16 -19 The enzyme itself is generally responsible for the specificity of the reaction. The mediator mentioned will receive electrons from the coenzyme and react with either the electrode or the indicator dye used. Every component can theoretically interact with substances included in the sample beyond glucose in a manner introducing bias.
One example in which awareness of interfering substances is critical is newborns with galactosemia. Lactose from milk is broken down into glucose and galactose, with the latter normally being further metabolized into glucose. In about one in 40,000 to 100,000 births, an autosomally recessive mutation leads to a loss of one or the other enzyme involved in that second step. This leads to toxic accumulation of galactose in the body. In addition, due to the failed metabolization, hypoglycemia can manifest. However, galactose is also an interfering substance for several enzymatic systems used to measure BG, leading to elevated measurements that can mask hypoglycemia. Even if the toxic levels of galactose are dealt with, overlooking the interference by galactose and failure to address the hypoglycemia in time may lead to life-threatening complications and developmental impairment for the neonate. 20
How to Test for Interference
The ISO 15197 standard expects interference testing to happen for at least two glucose levels, one between 2.8 and 5.5 mmol/L (50-100 mg/dL) and the other between 13.9 and 19.4 mmol/L (250-350 mg/dL). For each of these, a base pool with defined glucose concentration needs to be prepared. The ISO 15197 standard recommends comparing readings of paired blood samples, one spiked with the suspected interfering substance and the other only with the corresponding volume of the diluent used for the interfering substance. The samples should then be measured twice with an accurate comparison method. Subsequently, BG measurements should be made with 10 test strips each from three different lots, followed again by two measurements with the accurate comparison method. The latter step is to verify that the BG concentration in the sample has not changed over time.
The resulting values are to be compared with each other and the comparison value. If they differ by more than 10 mg/dL for glucose concentrations below 100 mg/dL or more than 10% above that concentration, they qualify as an interfering substance. (The allowed error for interference is smaller than the total error allowed as other factors, such as imprecision and bias, are also contributing to the measurement error.) In such a case, ISO 15197 requires a dose–response assessment using five different concentrations of that substance achieved by diluting the sample with the highest concentration of the interfering substance with the control sample. Blood glucose should be measured again with 10 strips from three different lots each. With this, the concentration of the interfering substance at which the bias surpasses the acceptable level should be determined—if necessary and appropriate by linear regression analysis.
The CLSI suggests a similar procedure, as well as starting with three times the maximal expected therapeutic concentration of interferents for the detection of a potentially introduced bias.
However, this method can only assess potential interferents directly and not metabolites—especially unknown ones—of drugs or other substances. This approach also only tests individual substances. It is, however, possible that individual substances remain below the 10 mg/dL or 10% cutoff, whereas their combination may well be above that threshold.
Therefore, the CLSI also suggests a method using actual samples from patients being treated with a certain drug. These samples are, on the one hand, measured with the test method and the results compared with measurements with a comparison method expected to be resistant to the potential interferents. Similar methods using real-world data from post-market surveillance have been used for various systems to complement data from ISO 15197 interference studies.21,22
People with diabetes are often multimorbid and are treated with multiple drugs. The described method can detect interferences only arising out of synergistic effects caused by polypharmacy. It will also detect interferences caused by unknown metabolites and uses actual plasma concentrations based on real bioavailability rather than theoretical calculations. However, it has considerable limitations: pinpointing the actual cause of the bias can be complex or impossible as correlation does not prove causation and the actual factor may even be an endogenous metabolite rather than a factor of a drug used in treatment. It may also be challenging to recruit enough samples to allow for statistically robust analysis—especially for newer substances and for determining dose–response relationships. Finally, large, worst-case concentrations may not be present in the patient pool, which poses the risk of overlooking interferences that, while not necessarily frequent, affect particularly vulnerable patients. Both approaches are thus complementary and together provide maximum information about interfering drugs.
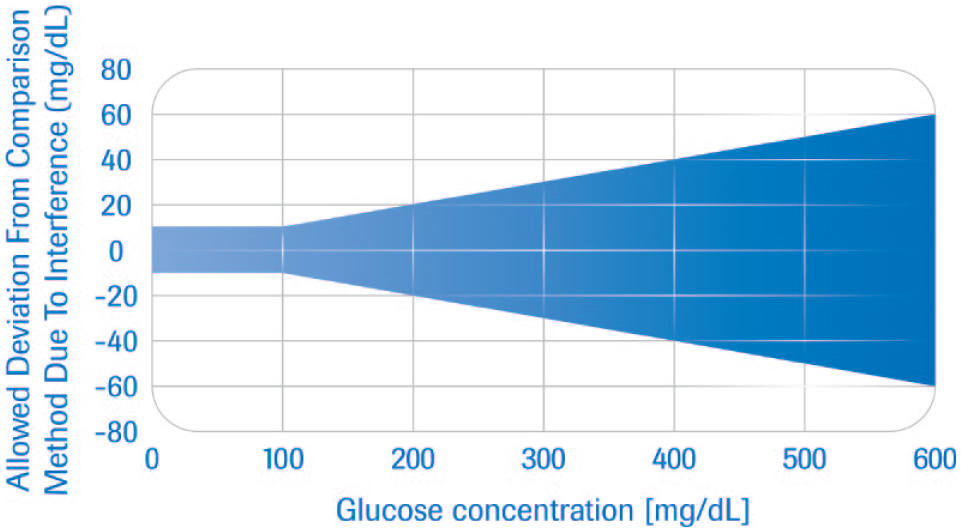
The ISO 15197 standard additionally categorizes the overall accuracy of systems, including the interference, semiquantitatively in terms of consequences of the divergence from the actual result, depending on the actual BG concentration and the degree of bias (Figure 2). This method is based on the Parkes Consensus Error grid. 23
Grading of the clinical risk of deviations of glucose measurements by error grid analysis as per International Organization for Standardization 15197:2013 15 (Parkes Consensus Error grid for type 1 diabetes). Deviations are categorized based on clinical relevance in regions ranging from A (no clinical consequence) over B (altered clinical action, but no change of outcome), C (altered clinical action likely to affect outcome), D (altered clinical action that could pose significant medical risk), to E (altered clinical action that could have dangerous consequences). Abbreviation: BG, blood glucose.
Any interference thus detected should be labeled in the product information for BG measurement systems, including at which concentration the introduced bias surpasses the acceptable limits. As the bias is a function of the concentration of the interfering substance, without knowledge of that concentration, it is impossible to determine the actual bias and thus correct the measurement value. Therefore, it is best to simply consider measurements in the presence of interfering substances above the threshold concentration as generally unreliable and unsafe for insulin dosing.
What Substances Should Be Tested?
All of the considerations of the previous section, however, only apply once a decision has been made as to what substances to test for interference. In this domain, deciding the initial candidates to test is straightforward, as ISO 15197 provides a list of substances to test as a minimum in Annex A2 (Table 1). In addition to that list, a risk analysis must be performed depending on the methodology used.
As ISO 15197 does not list concentrations, suggestions by CLSI are provided.
Abbreviations: CLSI, Clinical and Laboratory Standards Institute; DOPA, dihydroxyphenylalanine; EDTA, ethylenediaminetetraacetic acid; FDA, Food and Drug Administration; ISO, International Organization for Standardization.
The Food and Drug Administration (FDA) list largely agrees with ISO 15197 24 (Table 1) and specifies concentrations for testing many of them. As mentioned, this approach does not cover interferents from combined drugs.
Although the CLSI does list a series of substances as well, these are not specific for BG. Nonetheless, its list in CLSI EP07 Appendix A (Supplemental Table A1) 14 includes many of the substances listed above and suggests concentrations for testing that are not always the same as those listed by the FDA. Furthermore, potential interfering substances and concentrations to test them at are listed in the Supplement EP37. 25 The reasons for the choice of the respective concentrations are not always clear.
This illustrates that requirements are currently not harmonized. In addition, with the lists having been created at some point in the past, when new drugs are introduced to the market, manufacturers have to do a risk assessment whether these need to be tested. Both for diabetes treatment and for frequent comorbidities, new therapeutic compounds are constantly being introduced. A prime example is sodium glucose linked transporter–2 (SGLT-2) inhibitors that are not on either list but have become a common drug in the treatment of PwD. Thus, to ensure safety, manufacturers need to be vigilant about new developments and should continuously test new candidates for interfering substances and inform HCPs and end users of any new findings.
The potential sources of relevant candidates include reports or information requests from HCPs or PwD as well as literature on new drugs and new therapeutic approaches not only for diabetes treatment itself but also for the prevalent comorbidities. A continuous literature surveillance program, aided by pertinent databases such as one maintained by Wiley for the American Association for Clinical Chemistry (AACC) 26 based on the works by Young, 27 Young and Friedman, 28 and Narayanan et al 29 is very much needed.
Avoiding interference is better than labeling interference as it makes using the system safer for HCP and PwD. Once candidates have been identified, they should be included in any further blood glucose measurement system development at an early stage. It may well be feasible to avoid or compensate for interferences from the beginning by redesigning the test to maximize reliability of SMBG. Finding interferences ex post can only be the second-best option as it implies circumstances under which the patient cannot trust the BG readings.
How Far Should This Be Taken?
The scope this can reach is illustrated by the fact that, for example, Roche Diabetes Care is testing more than 200 substances for interference with BG measurements with their systems to minimize the risk of bias being introduced and is continuously expanding that list (cf. supplemental data online). One new addition, the SGLT-2 inhibitors mentioned above, was tested on the newest models of Roche BG monitoring systems and found not to be interfering. 30 This removes any lingering doubt as to the validity of BG results for patients under that therapy. On the contrary, N-acetyl cysteine and ceftriaxone were found to interfere with some BG monitoring systems. This allowed for the optimization of the measurement method of subsequent BG monitoring systems to avoid such interference from the start. Similarly, for other manufacturers, interfering substances not required by either ISO 15197 or the FDA have occasionally been reported, such as glibenclamide. 31 This illustrates that testing along official guidelines alone is not enough to ensure patient safety in a constantly changing therapeutic environment. To ensure that patients with diabetes can trust their BG readings, interference testing needs to be comprehensive and constantly expanded.
In the past years, continuous glucose monitoring (CGM) has become an important tool for diabetes management. Interference in CGM is even more complex, 32 in particular because concentrations in the interstitial fluid can be quite different from blood concentrations. More research on interference testing in this context is urgently needed.
“You can only find what you are looking for” and an interference not tested for is potentially an interference missed.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221140420 – Supplemental material for Drug Interference in Self-Monitoring of Blood Glucose and the Impact on Patient Safety: We Can Only Guard Against What We Are Looking for
Supplemental material, sj-docx-1-dst-10.1177_19322968221140420 for Drug Interference in Self-Monitoring of Blood Glucose and the Impact on Patient Safety: We Can Only Guard Against What We Are Looking for by Oliver Hauss, Rolf Hinzmann and Brian Huffman in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
AACC, American Association for Clinical Chemistry; BG, blood glucose; CGM, continuous glucose monitoring; CLSI, Clinical and Laboratory Standards Institute; DOPA, dihydroxyphenylalanine; EDTA, ethylenediaminetetraacetic acid; FAD, flavin adenine dinucleotide; FDA, Food and Drug Administration; GOD, glucose oxidase; GDH, glucose dehydrogenase; HCP, healthcare professional; ISO, International Organization for Standardization; NAD, nicotinamide adenine dinucleotide; PQQ, pyrroloquinoline quinone; PwD, people with diabetes; SGLT-2, sodium glucose linked transporter–2; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RH and BH are employees of Roche Diabetes Care. As proprietor of Dr. Hauss Training & Consulting, OH is providing consulting, training, and medical writing services. No other potential conflicts of interest relevant to this article could be identified.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Writing of this article was funded by Roche Diabetes Care GmbH, Mannheim, Germany.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.