Abstract
The Biomarkers for the Diagnosis of Heart Failure in Diabetes webinar was hosted by Diabetes Technology Society on September 20, 2023, with the objective to review current evidence and management practices of biomarker screening for heart failure in people with diabetes. The webinar discussed (1) the four stages of heart failure, (2) diabetes and heart failure, (3) natriuretic peptide and troponin for diagnosing heart failure in diabetes, (4) emerging composite and investigational biomarkers for diagnosing heart failure, and (5) prevention of heart failure progression. Experts in heart failure from the fields of clinical chemistry, cardiology, and diabetology presented data about the importance of screening for heart failure as an often-unnoticed complication of people with type 1 and type 2 diabetes.
Introduction
Moderators
Alexander Fleming, MD
Kinexum, Harpers Ferry, WV
David Sacks M.B., Ch.B., FRCPath
National Institutes of Health, Bethesda, MD
Heart failure (HF) is a common complication of people with diabetes. 1 With the poor prognosis of HF, identification and prevention from progression of this cardiovascular disease (CVD) must be made a priority in people with both type 1 diabetes (T1D) and type 2 diabetes (T2D) and a priority for women, because women are at a greater risk for HF following a diagnosis of diabetes than men. 2 However, HF may be difficult to diagnose because a person with diabetes often presents with asymptomatic HF lacking signs of hypertension, coronary artery disease, valvular disease, and other cardiac risk factors. 3
Early identification and initiation of treatment to delay or prevent progression of preclinical HF to clinical HF are important public health objectives. The natriuretic peptides, B-type natriuretic peptide (BNP) and N-terminal pro-B-type natriuretic peptide (NT-proBNP), have the strongest evidence demonstrating their sensitivity and specificity to diagnose earlier stages of HF and predict risk for progression to later stages of HF.4,5
However, new biomarkers are now being recognized and tested, including cardiac troponins, soluble ST2, galectin-3, and growth differentiation factor-15 (GDF-15). 6 These biomarkers may help inform treatment and diagnosis beyond the “gold standard” natriuretic peptide biomarkers. More investigation must be done to elucidate the role of these novel compounds either in isolation or in combination as part of a composite risk score in early screening and prognosis decisions, especially when it comes to treating HF in people at high risk of HF, including people with diabetes.
Session 1: The Four Stages of HF
Speaker
James L. Januzzi Jr., MD, FACC, FESC
Cardiology Division, Massachusetts General Hospital; Hutter Family Professor of Medicine, Harvard Medical School; Director, Heart Failure and Biomarker Trials, Baim Institute for Clinical Research; Boston, MA
Heart failure is a complex clinical syndrome with symptoms and signs that can be caused by any structural or functional impairment of ventricular filling or ejection of blood. In many patients, cardiac structural abnormalities, such as left ventricular chamber dilatation and ventricular systolic and diastolic dysfunction, may be present
Heart failure is a major complication of diabetes mellitus whose incidence and prevalence are rising inexorably. A better understanding of the way HF presents and progresses will inform better care for affected individuals.
The Universal Definition of Heart Failure 7 has established a useful rubric by which HF may be categorized, as presented in Figure 1.
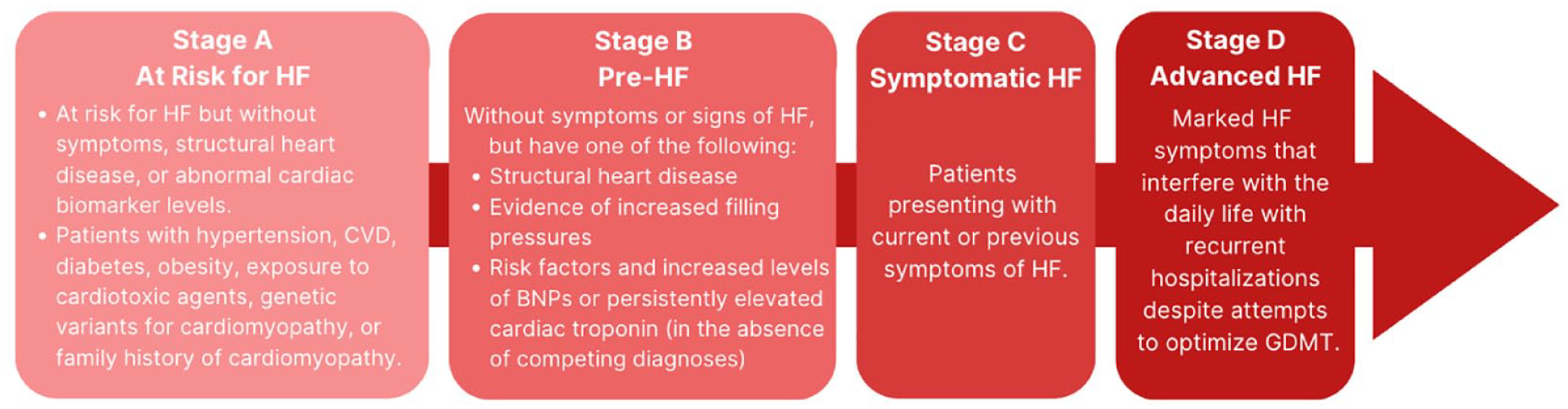
The Universal Definition of Heart Failure.
Heart failure is a major complication of diabetes mellitus, whose incidence and prevalence are progressively rising. When a person with diabetes develops HF, their risk for death is more than twice that of a person without diabetes. Thus, it is important to recognize the risk of HF, its early manifestations, and its later establishment at the earliest possible time to more assiduously treat affected patients. The Universal Definition of Heart Failure has established a useful rubric by which HF may be categorized. Knowledge of the four stages of HF will help clinicians understand the treatment steps necessary for patients. These four stages of HF include the following:
Stage A: This stage is considered the “at risk” stage and includes persons who are at risk of HF from medical conditions that increase the risk for the diagnosis. This includes hypertension or coronary artery disease. Every person with diabetes mellitus has stage A HF.
Stage B: This stage is also known as “pre-heart failure” because there is cardiac dysfunction; however, symptoms are not present. Stage B is also defined by the presence of abnormal circulating concentrations of prognostic cardiac biomarkers, including natriuretic peptides. Patients in this stage may notice reduced exercise capacity, but do not have overt HF and are at considerable risk for progressing to the symptomatic stages of the diagnosis.
Stage C: This stage includes persons with previous or present symptoms of HF. It represents a high-risk stage for death.
Stage D: This stage includes persons with advanced, end-stage HF.
Session 2: Diabetes and HF
Speaker
Rodica Pop-Busui, MD, PhD
University of Michigan, Ann Arbor, MI
Early diagnosis of HF could enable targeted treatment to prevent adverse outcomes.
Measurement of natriuretic peptides on at least a yearly basis is recommended to identify the presence of stage B HF and to determine risk for progression to symptomatic HF based on validated cutoff values (eg, NT-proBNP > 125 pg/mL) and based on population data and/or clinical trials.
The identification of an abnormal natriuretic peptide concentration should be part of individualized, team-based management decision plans.
Implementation of these guidelines at the point of care should be prioritized to reduce the risk for progression to more advanced stages, hospitalizations, and death.
The American Diabetes Association (ADA) consensus report on HF in diabetes was developed as a result of a request from the at-large diabetes care provider community. 5 It outlines the epidemiological evidence of the high prevalence and incidence of HF in diabetes and provides stepwise guidance to practitioners on the best approaches for screening and diagnosing HF in individuals with diabetes with the goal of ensuring access to optimal, evidence-based management and prevent progression to later stages for all. Both T1D and T2D increase the risk of developing HF, and HF may be the first presentation of CVD in many individuals with diabetes, particularly in women. 5 A person with established diabetes (particularly in the presence of other risk factors) should be considered in stage A HF, and many people with diabetes have stage B HF. Early diagnosis of HF enables targeted treatment to prevent progression of disease and other adverse outcomes. However, HF in individuals with diabetes is frequently underdiagnosed. Beyond hyperglycemia, multiple risk factors and unregulated metabolic pathways cause structural and functional changes to the heart, as presented in Figure 2.
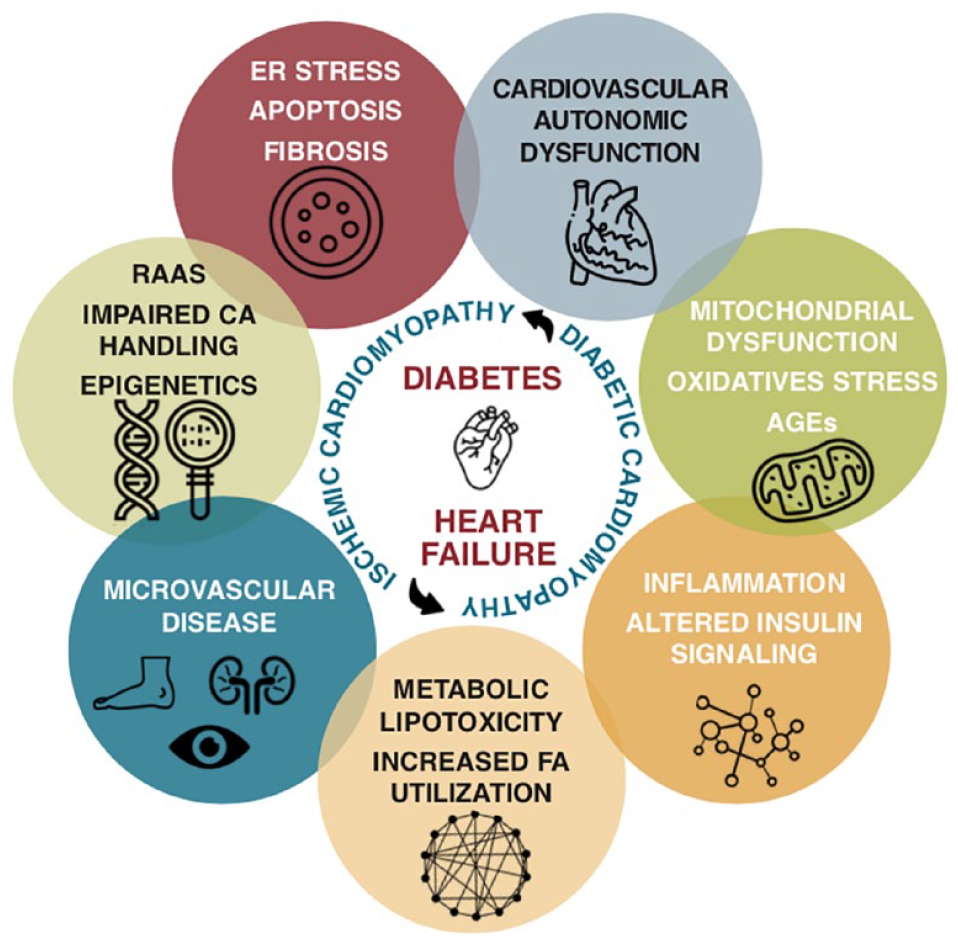
Structural and functional changes in the heart of people with diabetes.
Among individuals with diabetes, measurement of a natriuretic peptide on at least a yearly basis is recommended to identify possible presence of stage B HF and to predict the risk for progression to more advanced, symptomatic stages of HF. The same steps should be included for women with diabetes and individuals with T1D. 5 The management decisions that follow identification of an abnormal natriuretic peptide concentration should be individualized and might include further diagnostic studies, use of guideline-directed medical therapy (GDMT), and/or therapies with proven usefulness to prevent HF events, avoidance of treatments that increase HF risk, and a team-based approach, as presented in Figure 3. Concerted efforts are needed to implement these guidelines at the point of care. 8
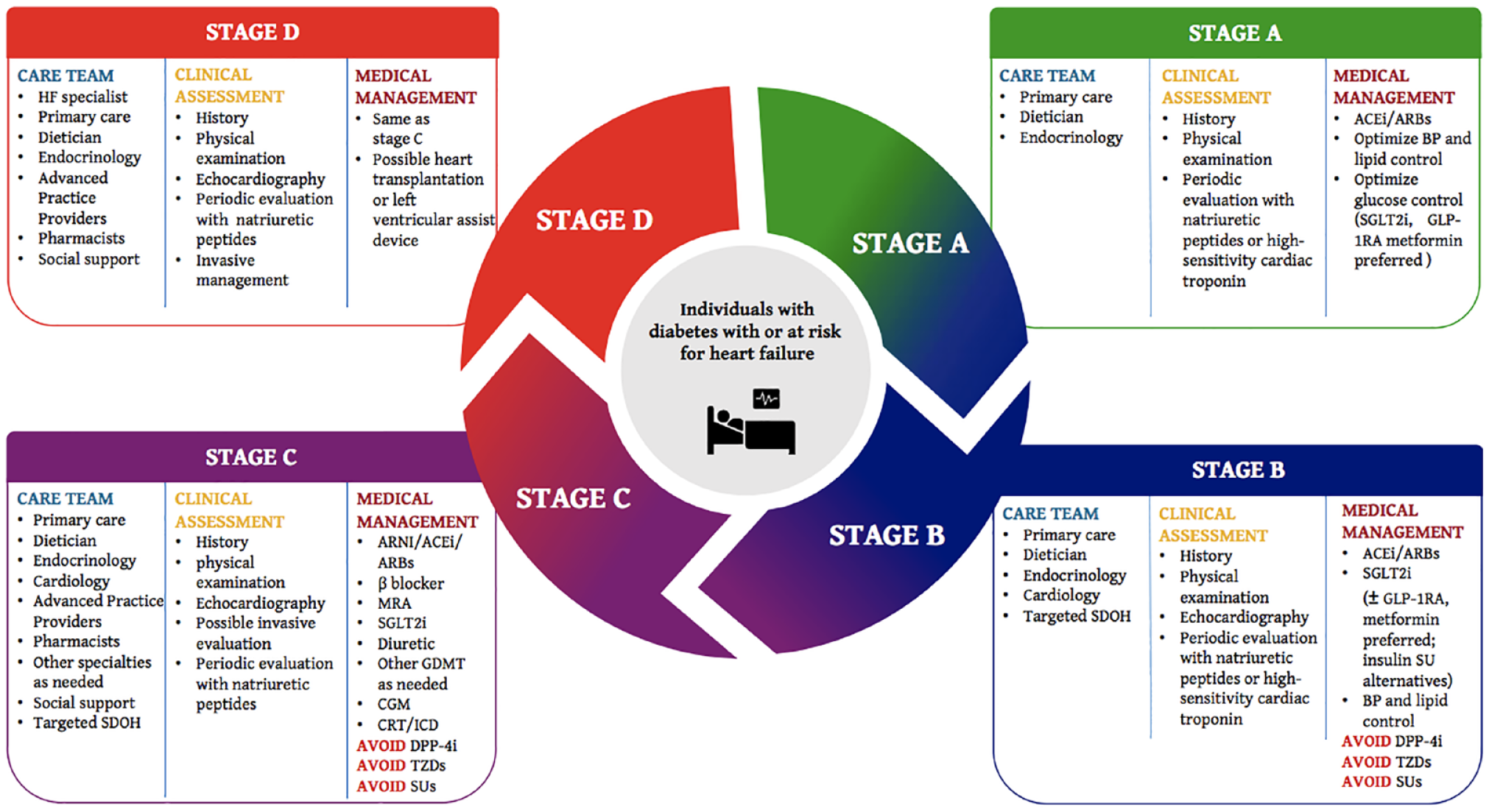
Multidisciplinary personalized care for individuals with heart failure and diabetes.
Session 3: Natriuretic Peptide and Troponin for Diagnosing HF in Diabetes
Speaker
Ibrahim A. Hashim, MSc, PhD
University of Texas Southwestern, Dallas, TX, USA
B-type natriuretic peptide is derived from a prohormone (proBNP) which contains 108 amino acids. Circulating proBNP is cleaved into two parts, including the biologically active 32-amino acid hormone BNP, which is the C-terminal fragment, and the biologically inactive 76-amino acid substance, which is the N-terminal fragment and is called NT-proBNP.
Several assays are available for the measurement of BNP and NT-proBNP. The latter is available from two manufacturers which affords some degree of standardization not present for BNP.
Variable cutoffs for acute and nonacute settings with clear and gray zones are in use. N-terminal pro-B-type natriuretic peptide has age-related cutoffs compared with those for BNP for all ages. Normal concentration of NT-proBNP is below 125 pg/mL for adults <75 years and below 450 pg/mL for adults ≥75 years.
Highly sensitive assays for troponin T and troponin I are in use with values reported in whole numbers in ng/L. Levels > 6 to 15 ng/L indicate cardiac injury.
Natriuretic peptide BNP (amino acids 77-108) and NT-proBNP (amino acids 1-76) are produced following cleavage of the 108-amino acid pro-BNP. They are produced by myocardial cells in response to volume overload. They induce natriuresis and vasodilation and inhibit the renin-angiotensin-aldosterone system (RAAS), and both effects lead to reduced volume overload. 9 Their synthesis and release into the circulating are surrogate markers of intracardiac volume and increased filling pressure, and as such, they are markers of congestive HF. Several assays are commercially available for the measurement of BNP and NT-proBNP and several cutoff values have been proposed for detection of HF ranging from 35 to 50 pg/mL for BNP and from 100 to 125 pg/mL for NT-proBNP. 10 The clearance of the low-molecular-weight NT-proBNP depends on renal function, and thus, age-related cutoffs are in use. 11 A normal concentration of NT-proBNP is below 125 pg/mL for adults <75 years and below 450 pg/mL for adults ≥75 years as presented in Figure 4. Several laboratory-based and point-of-care assays are commercially available. The assays are calibrated against synthetic NT-proBNP and BNP by mass. However, because NT-proBNP availability is restricted to certain manufacturers (Roche and QuidelOrtho), this restriction affords some degree of standardization that supports commutability and transferability of results. This is not necessarily the case for BNP, which is available by many manufacturers, and consequently, values vary significantly between different manufacturers’ systems. High-sensitivity troponin T and I assays are commercially available for detecting increased levels of these biomarkers, which if elevated, although suggestive of HF, can indicate myocardial injury through any mechanism.12,13 About 10% to 20% of patients with diabetes are at risk of developing HF and that periodic measurement of BNP or NT-proBNP has been suggested for early detection and diagnosis of HF among people with diabetes.
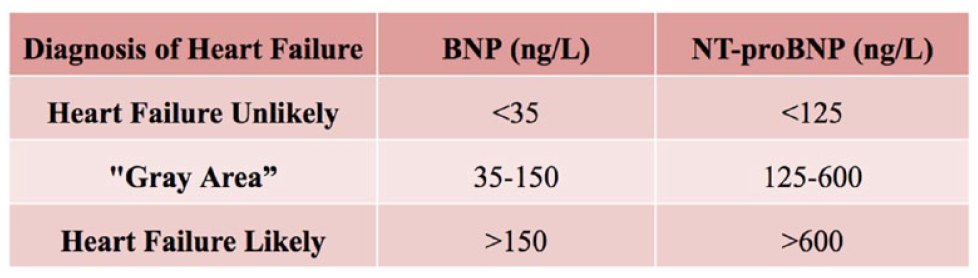
Natriuretic peptide cutoff values for the diagnosis of heart failure.
Session 4: Emerging Composite and Investigational Biomarkers for Diagnosing HF
Speaker
Alan H.B. Wu, PhD
University of California, San Francisco, San Francisco, USA
While the natriuretic peptides are the established biomarkers for diagnosis of risk stratification for HF, they are not as useful for monitoring therapeutic success.
Additional laboratory tests are needed to assess risks for adverse events for patients with HF.
Novel HF biomarkers include serum galectin-3, soluble ST2, and GDF, which are not increased in volume overload and therefore they have low biological variation.
The natriuretic peptides (BNP and NT-proBNP) are established biomarkers used for the diagnosis and short-term risk stratification for patients suffering from acute heart failure (AHF). The high sensitivity and specificity of NT-proBNP are illustrated in Figure 5. The concentrations of these peptides in the blood are increased in response to circulatory volume overload and left ventricular remodeling. Their value in monitoring the progress of anti-HF medications, however, has been debated. 15 Changes in a patient’s fluid status can produce variability in test results. As such, the weekly and monthly biological variation is high, and large changes in serial test results, that is, reference change value (RCV) > 100%, are required to achieve statistical differences. 16 Serum galectin-3 and soluble ST2 are novel HF biomarkers whose change in blood levels is more influenced by cardiac remodeling rather than excess fluid accumulation, and the RCVs for these tests are considerably lower than those for BNP and NT-proBNP. Galectin-3 is a lectin that participates in cell proliferation, host defense, and fibrosis. Soluble ST2 is a decoy receptor that blocks the cardioprotective effects of interleukin-33, resulting in cardiac hypertrophy and fibrosis. 17 While these tests are not as useful as the natriuretic peptides for diagnosis of acute decompensated HF, they provide a more accurate assessment of HF treatment. 18 When these biomarkers are increased in serum, they indicate disease progression, prompting physicians to change their management approach to avoid adverse cardiac events and rehospitalization. Growth differentiation factor-15 is a cytokine that is released following stress. In patients suffering from acute myocardial infarction, increases in GDF-15 are correlated to a high likelihood of a patient developing AHF. Like galectin-3 and ST2, GDF-15 has a low RCV. 19 Assays for these tests are not available within hospital or emergency department laboratories, but instead, from national reference laboratories.
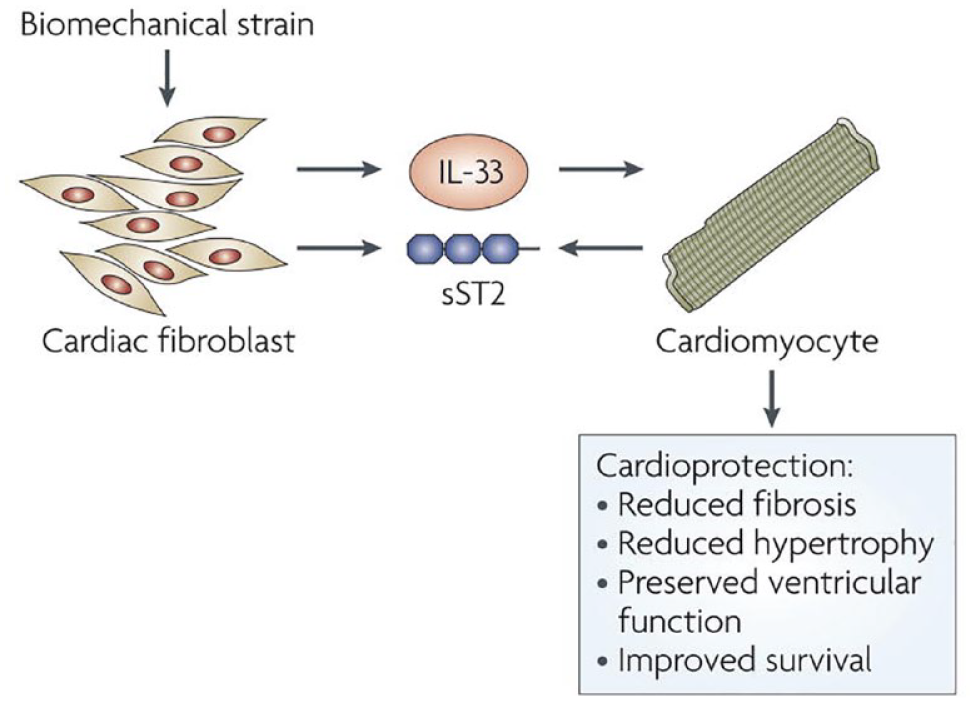
ST2/IL-33 as a cardioprotective system. Biomedical strain onto cardiac fibroblast releases IL-33 which provides cardio protection. Soluble ST2 acts as a decoy binding to IL-33 making its physiologic action unavailable.
Session 5: Prevention of HF Progression (Including Case Studies)
Speaker
Ambarish Pandey, MD
University of Texas Southwestern, Dallas, TX, USA
Early identification of patients with diabetes at the highest risk of HF may allow for interventions to mitigate HF risk.
Clinical risk scores, including the Thrombolysis in Myocardial Infarction-Heart Failure (TIMI-HF) risk score and the weight (body mass index), age, hypertension, creatinine, high-density lipoprotein cholesterol, diabetes control (fasting plasma glucose), electrocardiogram QRS duration, myocardial infarction, and coronary artery bypass grafting (WATCH-DM) risk score and cardiovascular biomarkers, including NT-proBNP and high-sensitivity cardiac troponin T testing, can be used for risk stratification.
Patients at elevated risk may benefit from sodium glucose cotransporter 2 inhibitor (SGLT2i) therapy, strict blood pressure (BP) control with angiotensin-converting enzyme inhibitor (ACEi)/angiotensin II receptor blocker (ARB) therapy, management of possible comorbid chronic kidney disease, and possible cardiologist referral.
Diabetes is a major risk factor for the development of HF. The comorbid combination of diabetes and HF is associated with a poor prognosis. Early identification of high-risk individuals is key to the prevention of HF among patients with diabetes. This is especially relevant as treatment with SGLT2is has been shown to lower the risk of hospitalization or death due to incident HF. 20 However, these therapies are costly and underutilized. Moreover, SGLT2i therapies have a high number needed to treat at the lower end of the HF risk spectrum. Thus, there is a need to better risk stratify patients with diabetes to administer preventive therapies to those at highest risk. HF risk scores have been developed specifically for patients with DM. One such score is the TIMI-HF risk score for diabetes mellitus, developed in the Saxagliptin Assessment of Vascular Outcomes Recorded TIMI and Dapagliflozin Effect on CardiovascuLAR Events trial cohorts. The TIMI risk score assessed the risk of HF hospitalization. However, prevalent HF is a key risk factor for the risk score and thus, the score is not optimal for identification of incident HF risk. Alternatively, the WATCH-DM risk score, presented in Figure 6, predicts incident HF among patients with diabetes and is derived from the Action to Control Cardiovascular Risk in Diabetes trial and Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial DM cohorts and utilizes many commonly available data elements. 21 Heart failure risk scores can potentially be implemented at a system level to aid in HF risk stratification. 22 Finally, cardiac biomarkers, including NT-proBNP and high-sensitivity cardiac troponin, are strong predictors of HF risk, and natriuretic peptide biomarker-based screening is recommended among patients at risk of developing HF, such as anyone with diabetes.4,5 Importantly, patients may have abnormal biomarkers even in the presence of a normal echocardiogram. 23 Among patients with elevated risk of HF, treatment should include SGLT2i therapy, aggressive risk factor management (including hypertension and kidney disease), and possibly referral to a cardiologist. 24
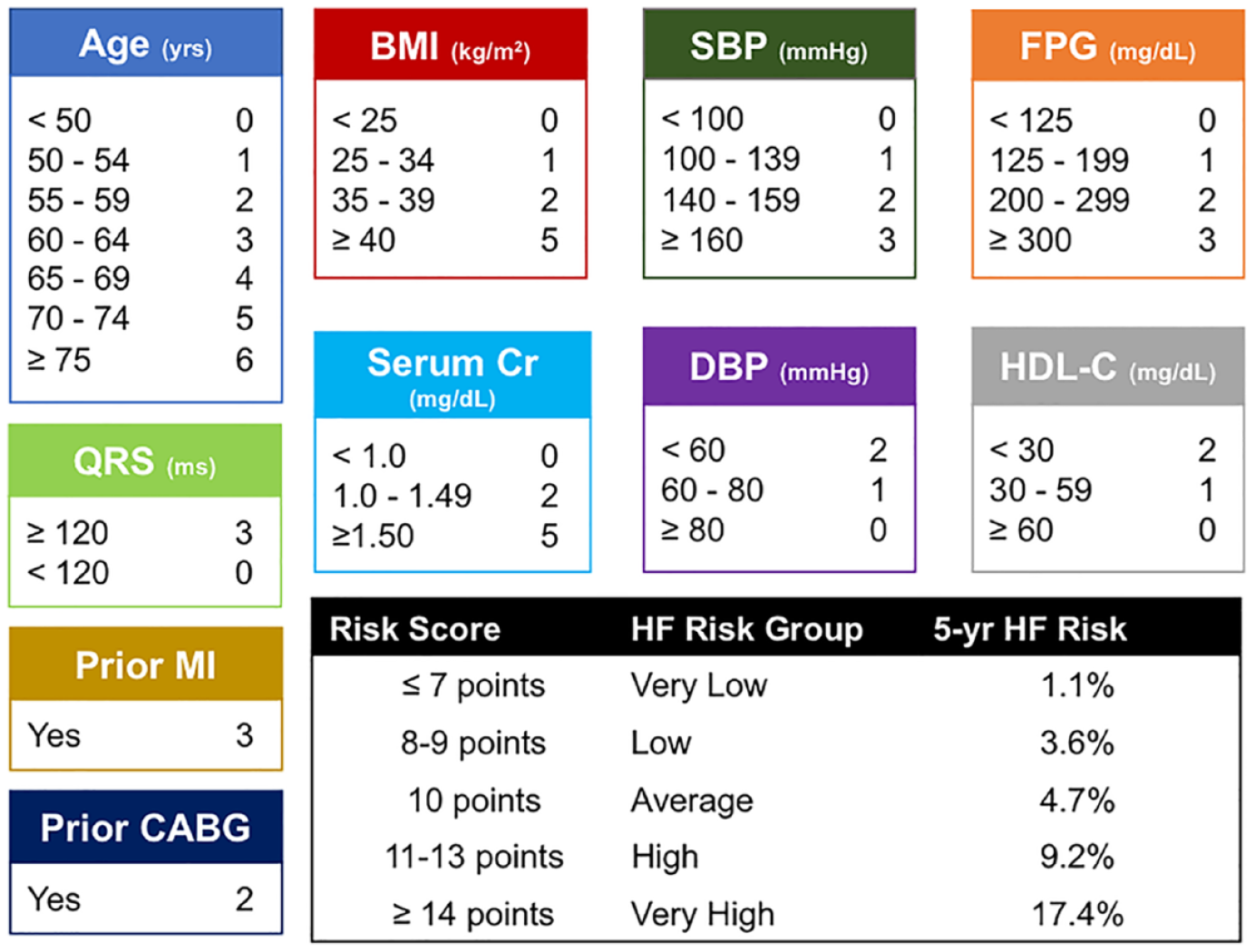
WATCH-DM risk factor score defined as weight (BMI), age, hypertension, CR, HDL-C, diabetes control (FPG) and QRS duration, MI, and CABG.
Conclusion
Biomarkers are being increasingly used in the diagnosis, prognosis, and prevention of HF in diabetes. With the results of cross-sectional and interventional trials by cardiologists, and outcome trials by endocrinologists, clinical chemists, and cardiologists, the importance of utilizing biomarkers in the diagnosis, prognosis, and prevention of HF in diabetes has become firmly established. Biomarkers including natriuretic peptides, serum galectin-3, soluble ST2, and GDF-15 have become more widely used to inform management of HF in diabetes. While several laboratory-based and point-of-care assays are commercially available, cutoff values are important to define. 25 It is vital to incorporate patient information, including sex and age to define these values. Guideline-directed medical assessment is essential to monitor the structure and function of the heart in people with diabetes. Implementing current guidelines at point of care is a priority to mitigate the risk of developing asymptomatic HF and then advancing to more severe symptomatic stages, hospitalization, or even death. 26 With continuing validation of various biomarker assays, laboratories will be better able to use biomarkers to improve the diagnosis, staging, and management of HF, especially for people with diabetes.
Footnotes
Acknowledgements
The authors thank Annemarie Sucher-Jones for her expert editorial assistance.
Abbreviations
ACEi, angiotensin-converting enzyme inhibitor; ADA, American Diabetes Association; AHF, acute heart failure; ARB, angiotensin II receptor blocker; BNP, B-type natriuretic peptide; BP, blood pressure; CVD, cardiovascular disease; GDF-15, growth differentiation factor-15; GDMT, guideline-directed medical therapy; HF, heart failure; IL-33, interleukin-33; NT-proBNP, N-terminal pro-B-type natriuretic peptide; pro-BNP, pro-B-type natriuretic peptide; RAAS, renin-angiotensin-aldosterone system; RCV, reference change value; SGLT2i, sodium glucose cotransporter 2 inhibitor; T1D, type 1 diabetes; T2D, type 2 diabetes; TIMI-HF, Thrombolysis in Myocardial Infarction-Heart Failure.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GAF is the Executive Chairman of Kinexum, which advises multiple health product companies, including multiple insulin manufacturers. Relevant device companies advised include Abbott, Biolinq, CMC Magnetics, Diabeloop, Hagar, Know Labs, Modular Medical, SFC Fluidics, and Surf Bio. He was formerly the Group Leader of the Division of Metabolism and Endocrine Drug Products at the US Food and Drug Administration. DBS receives research support from the Intramural Research Program at the National Institutes of Health. JLJ receives grant support from Novartis Pharmaceuticals, Applied Therapeutics, and Innolife; receives consulting income from Abbott Diagnostics, Jana Care, Janssen, Novartis, Prevencio, Quidel, and Roche Diagnostics; and belongs to clinical endpoint committees/data safety monitoring boards for Abbott, AbbVie, Amgen, CVRx, Janssen, MyoKardia, and Takeda. JLJ is also a trustee of the American College of Cardiology. AP has received grant funding (to the institution) from Applied Therapeutics, Gilead Sciences, Ultromics, Myovista, and Roche; has served as a consultant for and/or received honoraria outside of this study as an advisor/consultant for Tricog Health Inc, Lilly USA, Rivus, Cytokinetics, Roche Diagnostics, Sarfez Therapeutics, Edwards Lifesciences, Merck, Bayer, Novo Nordisk, Alleviant, Axon Therapies; and has received nonfinancial support from Pfizer and Merck. AP is also a consultant for Palomarin Inc with stocks compensation and has received research support from the National Institute on Aging GEMSSTAR Grant (1R03AG067960-01), the National Institute on Minority Health and Disparities (R01MD017529), and the National Institute of Heart, Lung, and Blood Institute (R21HL169708). RP-B receives grant support from Novo Nordisk, Lexicon Pharmaceuticals, and Medtronic and received consulting fee from Bayer, Lexicon Pharmaceuticals, Novo Nordisk, Roche. DCK is a consultant for Afon, Better Therapeutics, Integrity, Lifecare, Nevro, Novo, and Thirdwayv. REA, TT, AHBW, and IAH have no disclosures.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The webinar “Biomarkers for the Diagnosis of Heart Failure in Diabetes” and this meeting report were funded by a grant from Roche Diagnostics.
