Abstract
Background:
Impaired renal function is a well-known risk factor of cardiovascular disease, but its relation to heart failure in individuals with type 1 diabetes has been sparsely studied. The aim of our study was to quantify the risk increase for development of heart failure with decreasing kidney function in individuals with type 1 diabetes.
Methods:
Three equations were used to calculate eGFR (estimated glomerular filtration rate) for individuals with T1D and no known heart failure in the Swedish National Diabetes Registry. Proportional hazards regression models were constructed to evaluate the association between eGFR and hospitalization for heart failure (HF).
Results:
Among 13 781 individuals (mean age 41.1 [SD 13.3] years at baseline), 330 (2.4%) were hospitalized for HF over median follow-up of 7.0 years. Renal function was normal (eGFR > 90 mL/min/1.73 m2) in 67% of individuals according to the Cockcroft-Gault formula, compared to 51% and 41% according to the Chronic Kidney Disease Epidemiology (CKD-EPI) and Modification of Diet in Renal Disease (MDRD) formulas. For eGFR 45-60 ml/min/1.73 m2, hazard ratios (HRs) for hospitalization (reference >90 mL/min/1.73 m2) were 3.18 (95% CI 2.17, 4.65), 2.12 (1.16, 3.08), and 2.44 (1.69, 3.55) using the Cockcroft-Gault, MDRD, and CKD-EPI formulas. With eGFR <30 ml/min/1.73 m2 there was a HR of 3.78 (2.15, 5.91), 3.44 (2.14, 5.51), and 3.51 (2.21, 5.51) compared to normal kidney function (>90 mL/min/1.73 m2).
Conclusions:
In individuals with T1D, risk of hospitalization for heart failure was over 2 times greater at eGFR 45-60 ml/min/1.73 m2 and more than 3 times greater at eGFR <30 ml/min/1.73 m2 when compared to normal eGFR.
Heart failure (HF) is associated with a poor prognosis, hospital admission, and increasing prevalence, especially among younger people. 1 In individuals with type 1 diabetes, risk of hospital admission due to HF is higher compared with the general population.2,3 Therefore, identifying risk factors in individuals with type 1 diabetes may improve the prognosis for this high risk population.
Renal function, as determined by glomerular filtration rate (GFR), has been shown to be an independent risk factor for cardiovascular disease (CVD) in the general population and individuals with type 2 diabetes.4-6 But there are no large studies among individuals with type 1 diabetes. Several methods for determining GFR are available. 7 However, logistical constraints prohibit the use of the most precise methods, such as 24-hour creatinine clearance or isotopic GFR in clinical practice. Instead, several equations based on creatinine clearance have been developed to estimate GFR (eGFR), most commonly, the Cockcroft-Gault 8 and Modification of Diet in Renal Disease (MDRD) formulas. 9 A recently described equation, Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI), 10 has been validated in population studies and is potentially more reliable than the MDRD in individuals with type 1 diabetes.11,12
Present guidelines 13 recommend using MDRD or CKD-EPI over the Cockcroft-Gault formula based on data 14 comparing both to the gold standard 51Cr-EDTA clearance, which showed MDRD to be more accurate in diagnosing moderate and severe renal failure than Cockcroft-Gault. In the current study we evaluated the risk of hospitalization for HF in individuals with type 1 diabetes related to eGFR calculated with the Cockcroft-Gault, MDRD, and CKD-EPI formulas.
Subjects and Methods
Data were obtained from the Swedish National Diabetes Registry (NDR), a nationwide quality-assurance instrument for diabetes care, in conjunction with outcomes data from the Swedish hospital discharge and cause-specific death registries. A cohort of individuals 18 years or older who had type 1 diabetes mellitus and no known HF and in the NDR between January 1998, and December 2003 was identified (N = 20 985). This cohort has previously been described.2,15 From 1998 to 2001, creatinine data were available for less than 1% of these individuals. However, 65% of individuals in 2002 and 81% in 2003 had at least 1 creatinine value registered. For the present study, we selected individuals from the original cohort with at least 1 creatinine measurement (N = 14 006), with the time of first measurement used as the baseline. Of these individuals, 280 had no measurements for HbA1c, blood pressure, or body mass index (BMI) and were excluded. Another 8 individuals were excluded for having extreme creatinine values (<20 or >800 ml/min/1.73 m2). Therefore, the final study cohort consisted of 13 718 individuals who were followed from the first creatinine measurement until hospitalization for HF, defined as a principal or contributory discharge diagnosis with an International Classification of Diseases 10 (ICD-10) code of I50, death from any cause, or the end of 2009. The diagnosis of HF in the Swedish Hospital Discharge Registry has been validated, showing 95% accuracy for primary diagnoses and 82% irrespective of diagnosis position. 16 In total, there were 330 cases of HF during follow-up.
Type 1 diabetes mellitus is classified in the NDR as treatment with insulin only and age of onset 30 years or younger. This definition has been validated to be correct in 97% of cases. 17 This study was approved by the regional ethics review board at the University of Gothenburg (Gothenburg, Sweden), and all participants provided verbal informed consent to be included in the NDR.
Construction of Covariates
For HbA1c, systolic and diastolic blood pressure, and BMI, the mean of all available values until the first creatinine measurement was used as that covariates baseline value. Smoking and blood pressure medication at study start was categorized as none (all values were “no”), yes (≥1 “yes” value), or unknown (no information).
Baseline variables for atrial fibrillation (I48), valve disease (I05-I09 and I34-I36), myocardial infarction (I21), and HF (I50) were obtained from the Swedish Inpatient Register and included as 0-1-variables, with 1 indicating that the event occurred before study start. Information on myocardial infarction was available beginning in 1987, and valvular disease and atrial fibrillation beginning in 1997.
The Cockcroft-Gault, MDRD, and CKD-EPI formulas were used to calculate eGFR.8-10 For each, a categorical variable was constructed based on GFR thresholds for chronic kidney disease. 18 The lowest category, GFR <15 mL/min/1.73 m2, was merged with GFR 15-30 mL/min/1.73 m2, due to a minimal number of observations. The final categories were <30, 30-45, 45-60, 60-90, and ≥90 mL/min/1.73 m2.
Statistical Analysis
The association between eGFR and HF was investigated using proportional hazard (Cox) regression. For each eGFR formula, a model was constructed, adjusted for age, sex, and diabetes duration (model A), and also for HbA1c, BMI, diastolic and systolic blood pressure, smoking, and history of myocardial infarction, stroke, valvular disease, atrial fibrillation, and blood pressure medication (model B, or the main model), with all covariates defined at study start. Hazard ratios for eGFR were used to describe the relative increase in risk of having an HF hospitalization event in each eGFR category, with ≥90 mL/min/1.73 m2 used as the reference or normal category.
The predictive ability of each eGFR formula was evaluated via systematic pairwise comparisons of nested models using likelihood ratio tests. In other words, the proportional hazard model containing 2 eGFR variables, A and B, was compared against the models containing only A and only B, respectively, with all other covariates being equal. If the A+B model had a significantly (P < .05) better fit than the model with only B, then variable A was interpreted as contributing significantly over B. Likewise, if the A+B model showed better fit than the model with only A, then variable B contributed significantly over A. If A contributed significantly over B, but not vice versa, then A was considered a stronger predictor than B. If both tests were significant the variables could not be ranked.
Results
Characteristics of the Cohort
In 13 718 individuals with type 1 diabetes, mean (± standard deviation) age was 41.1 (13.3) years at baseline and 55.1% were men. Mean HbA1c was 65 (13) mmol/mol (8.1 (1.2) %) and mean duration of diabetes was 25.5 (13.9) years. Mean baseline creatinine level was 88.7 µmol/L. There were 327 individuals (2.4%) with a previous myocardial infarction. Baseline characteristics for the cohort are shown in Table 1.
Characteristics of 13 718 Individuals With Type 1 Diabetes at First Registration of Creatinine 1998-2003.
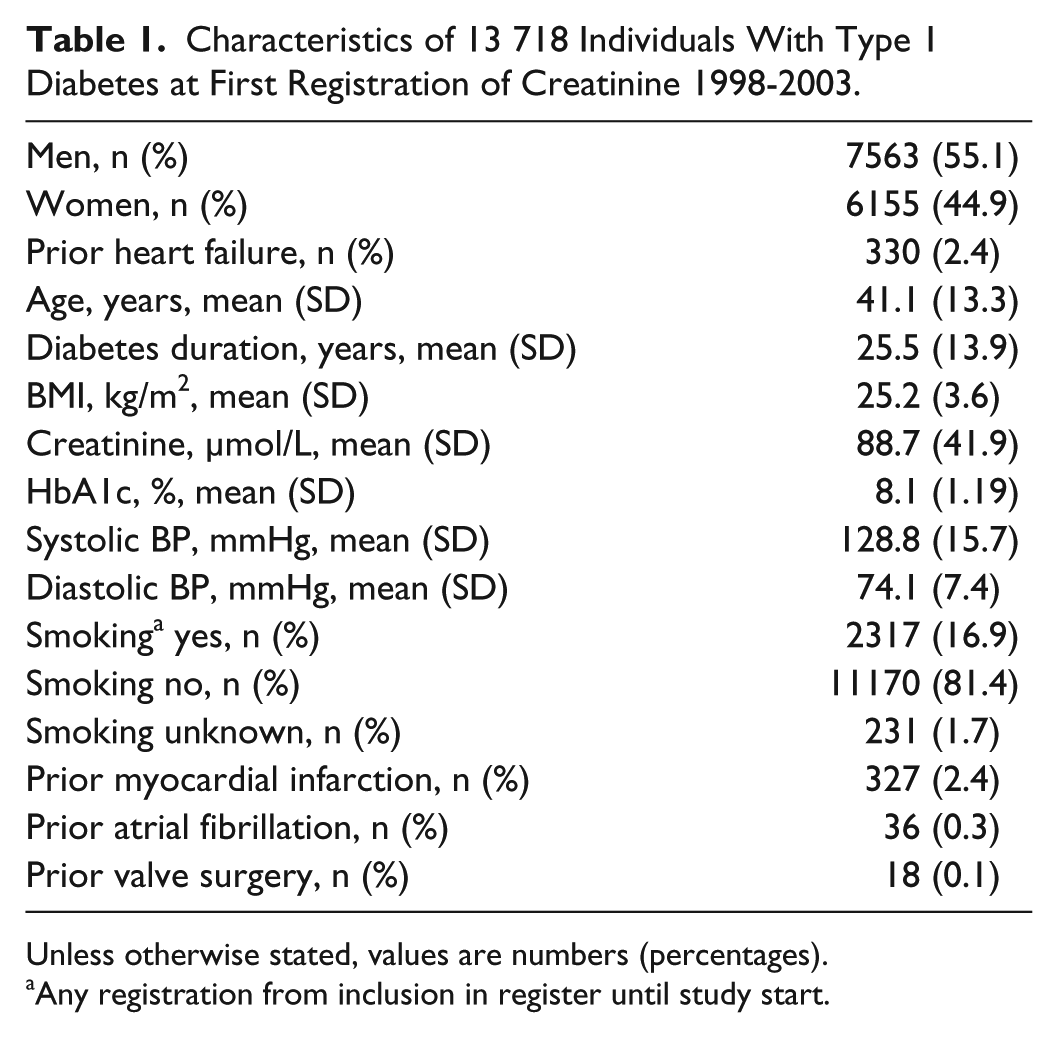
Unless otherwise stated, values are numbers (percentages).
Any registration from inclusion in register until study start.
eGFR Levels in the Cohort and Events of Heart Failure
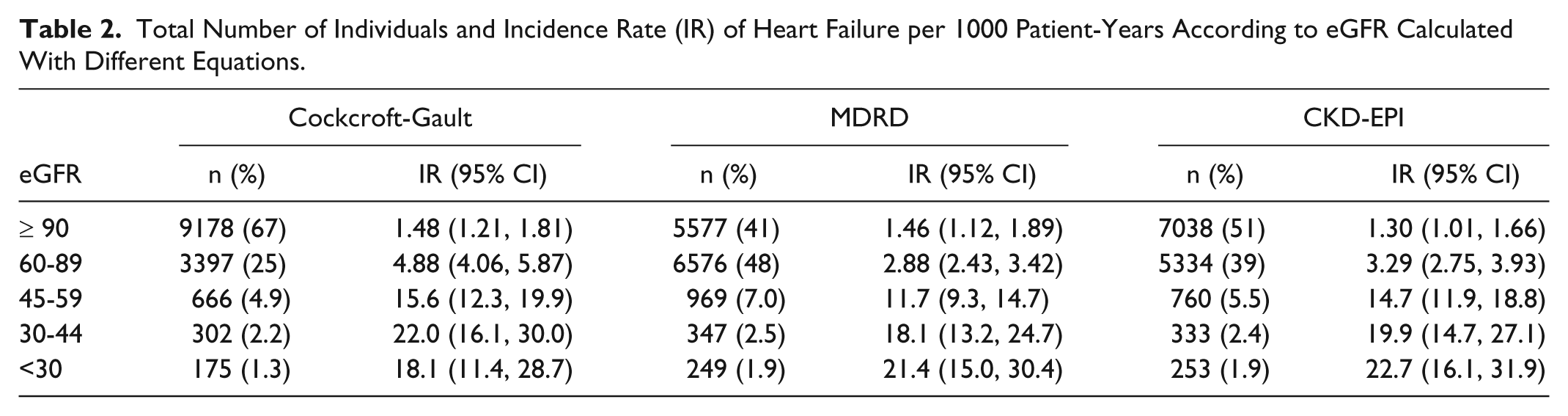
Mean eGFR at baseline according to Cockcroft-Gault was 105.9 (36.0) ml/min/1.73 m2, and 89.2 (23.1) ml/min/1.73 m2 and 86.2 (24.9) ml/min/1.73 m2 by the CKD-EPI and MDRD equations, respectively. According to Cockcroft-Gault, 67% had normal eGFR, compared to 51% according to CKD-EPI and 41% according to MDRD. Severely decreased eGFR or kidney failure (<30 mL/min/1.73 m2) was prevalent in 1.3% using Cockcroft-Gault and 1.9% for both CKD-EPI and MDRD.
Among all individuals, 330 (2.4%) were admitted with a primary or secondary diagnosis (ICD-10 code of I50 as a principal or contributory discharge diagnosis) of HF during a median follow-up of 7 years. The incidence of HF increased with decreasing eGFR, except as calculated by Cockcroft-Gault where the incidence decreased slightly in the <30 mL/min/1.73 m2 category. Table 2 contains incidence rates of HF per 1000 patient-years by eGFR category for each equation.
Total Number of Individuals and Incidence Rate (IR) of Heart Failure per 1000 Patient-Years According to eGFR Calculated With Different Equations.
Relative Risks of Heart Failure in Relation to eGFR
Decreasing eGFR was strongly associated with the development of HF in the proportional hazard model adjusted for age, sex, and diabetes duration (model A). Regardless of formula, risk was 2-fold or greater when eGFR was <60 ml/min/1.73 m2 compared to the reference category. Hazard ratios (HR) increased with decreasing eGFR for all formulas (Table 3).
Hazard Ratios of Hospitalization for Heart Failure for Each of the 3 eGFR Variables.
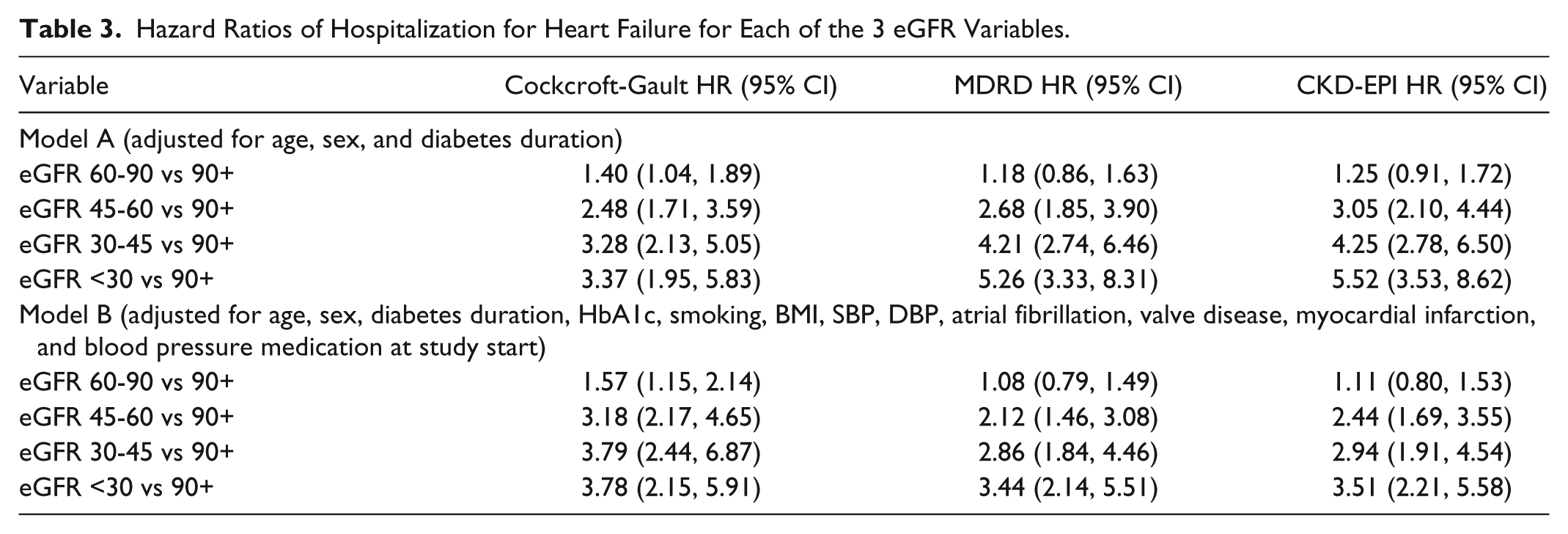
In model B, the relationship between eGFR and hospitalization for HF remained statistically significant (Table 3). Decreasing eGFR from 90 to 59 mL/min/1.73 m2 resulted in increasing risk of clinical HF, which ranged from 2.12 to 3.18 depending on the formula used. For both CKD-EPI and MDRD, there was a stepwise increase in HR for clinical HF with decreasing eGFR, while the HR was numerically lower in the <30 mL/min/1.73 m2 category according to Cockcroft-Gault compared with the 30-45 mL/min/1.73 m2 category. For all 3 formulas, the relative risk of hospitalization for HF was at least 3-fold greater in the <30 ml/min/1.73 m2 category compared to the reference (Table 3).
Predictive Power of Hospitalization for HF for Various eGFR Measures
Results of pairwise comparisons between eGFR formulas with likelihood ratio tests of their predictive ability for HF hospitalization are presented in Table 4. For both CKD-EPI and MDRD, there was a significant improvement in overall model fit when included on top of Cockcroft-Gault. In contrast, Cockcroft-Gault added no significant increase in overall model fit when compared to CKD-EPI but did when compared to MDRD (P = .042), implying that these 2 measures add different information to the model and cannot clearly be ranked in terms of prognostic value.
Pairwise Test of eGFR Variables for Information Gain When Added to Each Other.

Discussion
In this first population-based study of the risk of hospitalization for HF in relation to eGFR among individuals with type 1 diabetes, comprising 13 781 individuals, reduced eGFR strongly predicted future hospitalization for HF. Irrespective of eGFR measure used (Cockcroft-Gault, CKD-EPI, or MDRD) the risk was at least 2 times greater in individuals with moderately reduced eGFR (45-60 ml/min/1.73 m2). In individuals with severely reduced eGFR (<30 ml/min/1.73 m2) the risk of future hospitalization for HF was 3 times greater than that for individuals categorized as having normal renal function (>90 ml/min/1.73 m2).
The use of different formulas for eGFR led to disparate results in terms of renal function category and ability to predict hospitalization for HF. Although population-based studies relating eGFR to hospitalization for HF to our knowledge are absent among individuals with diabetes, several other studies have focused on eGFR as a risk factor for CVD and mortality in the general population. These studies have generally used a composite endpoint of ischemic heart disease, congestive HF, and stroke. In 1 of the largest (N = 1 120 295) studies in the general population, Go et al 6 found an 180% increase in risk of CVD among individuals with eGFR <29 mL/min/1.73 m2 (MDRD) compared with >60 mL/min/1.73 m2. A smaller study of only individuals with type 2 diabetes 5 , also with a CVD endpoint, found an approximate 3-fold risk increase among individuals with eGFR <29 mL/min/1.73 m2 (MDRD) compared with >90 mL/min/1.73 m2. Comparable risk increases were also seen by Manjunath et al, 19 for de novo atherosclerotic CVD, with an approximate 2.5-fold risk increase between individuals with eGFR 30-59 mL/min/1.73 m2 (MDRD) and >90 mL/min/1.73 m2.
The main differences between the Cockcroft-Gault, CKD-EPI, and MDRD equations were that Cockcroft-Gault tended to classify a greater proportion of individuals to either normal renal function (>90 ml/min/1.73 m2) or advanced renal impairment (<30 ml/min/1.73 m2).
Michels et al 20 compared Cockcroft-Gault, CKD-EPI, and MDRD with a gold standard GFR measurement, 125I-iothalamate, and found that CKD-EPI best estimated GFR, although its accuracy was close to that of MDRD. Thus, it is possible that the greater predictive ability of CKD-EPI than Cockcroft-Gault is explained by more accurate classification of renal function. Although our results should be interpreted with caution, they support the use of CKD-EPI over Cockcroft-Gault in clinical practice. However, since there was no clear difference between CKD-EPI and MDRD, the latter of which is commonly used today, we cannot conclude which formula is the most predictive of HF hospitalization.
It should also be noted that the predictive power of various eGFR formulas cannot be determined by comparing hazard ratios, since each formula categorizes individuals into different eGFR groups. This was also apparent in the current study, since the reference group substantially differed by eGFR formula with respect to individuals who were included in the group.
Nevertheless, our results support the use of eGFR to indicate an increased risk for clinical HF in everyday practice, although it remains unknown whether impaired kidney function, manifested as decreased eGFR, is only a marker for microvascular disease or if it in fact increases the risk of developing HF. As understanding of the risk factors for HF among individuals with type 1 diabetes improves, it may be prudent to screen for early signs of HF by cardiac ultrasound in certain subgroups. Individuals with decreased eGFR, regardless of equation used, may be 1 subgroup. Others may include those with known risk factors for HF hospitalization as demonstrated in previous work of individuals with type 1 diabetes, including older age, poor glycemic control, hypertension, smoking, obesity, and longer diabetes duration.2,15,21
Strengths of our study include the large number of individuals and that our models were adjusted for several important risk factors. A weakness is that we relied on hospitalization for HF as an outcome rather than an assessment of cardiac function. Another possible limitation is the accuracy of creatinine values derived from different laboratories over time. Although standardization programs in Sweden have been initiated since many years, there are still, both in Sweden and globally, possible improvements to be performed in this field. 22 These methodological aspects are likely to have minimal effects, if any, on the main conclusions of this article regarding renal function as a risk factor of hospitalization for HF in individuals with type 1 diabetes. However, our results regarding which eGFR formula is most predictive of hospitalization for HF should be interpreted with caution due to these limitations, and need to be confirmed in other studies.
Conclusion
In individuals with T1D impaired renal function estimated by eGFR based on creatinine strongly predicts future hospitalization for HF. Risk of hospitalization for HF was over 2 times greater at eGFR 45-60 ml/min/1.73 m2, increasing with lower eGFR, to more than 3 times greater at eGFR < 30 ml/min/1.73 m2).
Footnotes
Acknowledgements
We want to thank all data collecting clinicians and staff at the National Diabetes Registry. Editorial assistance was provided by Joseph W. Murphy.
Abbreviations
CKD-EPI, Chronic Kidney Disease Epidemiology formula; CVD, cardiovascular disease; eGFR, estimated glomerular filtration rate; HF, heart failure; MDRD, Modification of Diet in Renal Disease formula; NDR, Swedish National Diabetes Registry; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was financed by grants from the Swedish state (ALF-agreement), the Swedish Society of Medicine, Novo Nordisk Foundation, and the Health & Medical Care Committee of the Regional Executive Board, Region Västra Götaland, Sweden.
