Abstract
Background:
Continuous subcutaneous insulin infusion (CSII) use in adults with type 1 diabetes offers psychosocial and clinical benefits, but little is known about its impact on such outcomes in the type 2 diabetes (T2D) population. To address this gap, we conducted a quasi-experimental prospective study to assess psychosocial, glycemic, and behavioral changes over six months in T2D adults on multiple daily injections (MDI) who were interested in starting Omnipod DASH, comparing those who did versus did not start on it.
Methods:
In total, 458 adults with T2D completed baseline questionnaires assessing psychosocial dimensions (eg, diabetes distress), clinical metrics (eg, HbA1c [glycosylated hemoglobin]), and behavioral measures (eg, missed mealtime boluses). Six months later, 220 (48.0%) completed the same questionnaire again. To examine differences in outcomes over time between those who began CSII (n = 176) versus those who remained on MDI (n = 44), a latent change score approach was used.
Results:
The CSII users reported greater gains than MDI users on all major psychosocial metrics, including overall well-being (P < .001) diabetes distress (P < .001), perceived T2D impact on quality of life (P = .003), and hypoglycemic worries and concerns (P < .001). The CSII users similarly reported a larger decline in HbA1c than MDI users (P < .05) and greater declines in two critical self-care behaviors: number of missed mealtime boluses (P < .001) and number of days of perceived overeating (P = .001).
Conclusions:
The introduction of CSII (Omnipod DASH) in T2D adults can contribute to significant psychosocial, glycemic, and behavioral benefits, indicating that broader use of CSII in the T2D population may be of value.
Introduction
Insulin pump therapy, or CSII (continuous subcutaneous insulin infusion), has been shown to enhance glycemic outcomes in adults with type 2 diabetes (T2D),1,2 but such benefits can only be maintained if users opt to persist with their CSII tools and engage them in an effective manner over time. Users are more likely to do so when they recognize that such tools are helping them to achieve tangible and personally important goals, which include positive changes in psychosocial outcomes, especially quality of life (QOL), as well as glycemic outcomes.
While observational evidence as well as randomized controlled trials (RCT) indicate that the use of stand-alone CSII (ie, not sensor augmented) in adults with type 1 diabetes (T1D) contributes to improvements in key psychosocial outcomes,3-7 little is known about the impact of CSII on such outcomes in the T2D population. While several small single-arm trials and retrospective observational studies have pointed to potential QOL benefits,8,9 only two RCTs to date have directly compared psychosocial outcomes between CSII and multiple daily injections (MDI) in T2D populations. The first RCT, using the Diabetes Quality of Life Clinical Trial Questionnaire and the 36-Item Short Form Survey (SF-36), found no significant difference in QOL change between the CSII (MiniMed 508) and MDI groups. 10 The second RCT found that CSII (Calibra PAQ MEAL, delivering mealtime insulin only) versus MDI use led to a significantly greater (though modest) rise on several subscales of the Diabetes Specific Quality of Life Scale (in particular, feeling less constrained by diet). 11
While newer generations of multifunction, stand-alone CSII technology have been introduced over the past decade, it is noteworthy that no RCT or prospective study has yet to examine the impact of any of these new systems on critical psychosocial outcomes in T2D populations. In particular, full-featured tubeless pumps (ie, the Omnipod DASH) have not been evaluated. Furthermore, key psychosocial elements that are now recognized as central to the personal experience of insulin users (eg, diabetes distress, hypoglycemic fears, and worries) have yet to be examined in prospective CSII trials for either T2D or T1D group. In sum, we suspect that by focusing on current CSII technology and including more patient-relevant psychosocial measures, we may discover that CSII contributes to clinically relevant psychosocial benefits, as well as glycemic and behavioral benefits, for T2D adults over time.
To address this supposition, we conducted a quasi-experimental prospective study to assess psychosocial, glycemic, and behavioral changes over a six-month period in MDI-using T2D adults who expressed an active interest in starting CSII. Outcomes were compared between those who started and remained on CSII over the six-month period versus those who never started CSII and remained on MDI. Our primary hypothesis was that the CSII group would evidence significantly greater psychosocial benefits than the group remaining on MDI; secondarily, we hypothesized that there would be a parallel improvement in glycemic and behavioral outcomes.
Participants and Methods
Participants
Adults with T2D currently using MDI who contacted Insulet Corporation about starting a tubeless insulin pump (Omnipod DASH Insulin Management System) were invited to participate in the independent online survey study being conducted by the Behavioral Diabetes Institute (BDI). Inclusion criteria were: T2D duration ≥5 years, age ≥25 years, MDI use ≥1 year, no prior CSII experience, and actively planning, or hoping, to start on CSII in the immediate future. A total of 1426 adults expressed to the Insulet staff an initial interest in participating and provided their contact information; Insulet staff then forwarded by secure email this information to the BDI research team.
Procedures
Interested subjects received an email invitation to the study from the BDI research team, explaining that it was being conducted in collaboration between Insulet and the BDI, survey responses would be kept anonymous, and participation was voluntary. The invitation explained that there would be two surveys to be completed for most participants, one prior to starting CSII (baseline) and one six months hence. They then accessed an online link to complete seven screening items, and if eligible, an informed consent document and the baseline survey. At completion of the baseline survey, participants received a $25 electronic gift card.
Participants who completed the baseline survey were invited to complete a second survey six months later if (1) they had been actively using the Omnipod DASH for ≥4 months (thus comprising the CSII group) or (2) they had never started CSII, despite their initial interest, and remained on MDI (the MDI group). A $50 gift card was provided after completion of the six-month survey. Note that all data were entered into a central database using a HIPAA-protected server, with no linkages to personal health information or personal identifiers. The research protocol was approved by Ethical and Independent Review Services, a community-based Institutional Review Board.
Measures
The first section of the survey consisted of items to determine key sample characteristics, including demographic information, diabetes history, and critical clinical metrics. These included age, gender, race/ethnicity, education, annual income, years since diagnosis, last self-reported clinic HbA1c (within the last three months only), body mass index (BMI; self-reported weight and height), number of severe hypoglycemic episodes (ie, requiring the assistance of another) in the last month, current mode of glucose monitoring, and hypoglycemia awareness (via the single-item Gold questionnaire). 12
To assess relevant psychosocial dimensions, the second section of the survey included the eight-item Type 2 Diabetes Distress Assessment System (T2-DDAS) Core scale, 13 which assesses overall diabetes distress; the seven-item DAWN2 Impact of Diabetes Profile (DIDP), 14 which assesses the perceived impact of diabetes on QOL; and the 14-item Hypoglycemic Attitudes and Behavior Scale (HABS), which assesses worries and fears related to hypoglycemia. 15 Also included was the World Health Organization-5 (WHO-5), which assesses overall QOL and well-being. 16 Six new items were developed to capture how limited or restricted by T2D the individual might feel (eg, “I don’t feel as free to live my life the way I want,” “I feel limited about what I should and shouldn’t eat”). Each item was scored on a five-point, Likert scale, ranging from “strongly disagree,” “disagree,” “neutral,” “agree,” and “strongly agree.” Finally, device satisfaction was assessed with the 12-item Insulin Device Satisfaction Survey (IDSS). 17
The third section consisted of a short series of items which we constructed (ie, not a validated scale) to assess diabetes self-management behaviors. Participants estimated how many days over the past week (0-7) they had engaged in moderate or strenuous exercise, had overeaten (“on how many days do you feel that you ate more than you probably should have?”), had missed mealtime bolus injections, and—at baseline only—had missed basal insulin injections. While additional questions about diet behavior could have been valuable, we were concerned about the overall length of the survey instrument; therefore, we included only a single item on this subject (number of days when overeating occurred), which is recognized as a common barrier to T2D self-management and glycemic control. 18 Of note, we did not have access to participants’ pump data, which is why assessment of missed boluses was limited to self-report.
The baseline and six-month surveys included the same measures, except the baseline survey had several additional items that were not expected to change or were no longer relevant at six months (eg, sociodemographics, missed basal insulin injections).
Data Analysis
Data were analyzed using IBM SPSS Statistics 28 and Mplus Version 8.7. Descriptive statistics were computed for all variables, which were examined for outliers and normality of distribution. To assess potential bias due to missingness at six months, independent samples t-tests examined differences in key sample characteristics or survey measures between participants who completed the six-month survey and those who did not. In addition, prior to testing our main hypotheses, independent samples t-tests were conducted to identify any differences in the same baseline characteristics between the CSII and MDI groups at baseline.
To test our main hypotheses concerning differences in outcomes over time between the CSII and MDI groups, we used a latent change score approach to create error-free estimates of change in variables of interest from baseline to six months later. 19 This maximum likelihood–based approach makes use of all available data (even from participants who only completed the baseline survey) and thus is well-suited to handle missing data across time. Separate models were run for all clinical indices, psychosocial dimensions, and behavioral measures (20 total, one per outcome variable). In each model, an error-free latent estimate of change in the outcome variable from baseline to six months was created and then treated as a dependent variable within the same structural equation model. The focal independent variable was group (CSII = 1, MDI = 0), which was entered as a predictor of latent change over time. Covariates included race/ethnicity (non-Hispanic white = 1, others = 0), gender (male = 1, female = 0), and age in all models. Baseline levels of the outcome variable were allowed to covary with its latent change score.
A different analytic approach was used for one binary outcome variable: having at least one severe hypoglycemic episode in past month. We used multiple logistic regression with the same focal independent variable (group) and covariates (race, gender, age) to predict residualized change in severe hypoglycemic episodes from baseline to six months. That is, the binary dependent variable was severe hypoglycemic episodes at six months while controlling for severe hypoglycemic episodes at baseline.
Finally, to better understand the nature of change in the variables over the six-month period and to contextualize the results of the main analyses, we then examined bivariate change within each group (CSII vs MDI) via paired samples t tests. Two-tailed significance tests using an alpha = .05 were conducted for all models.
Results
Sample Characteristics
Of the initial pool of 1426 participants, 572 did not respond to the invitation to participate, 384 completed the screener and were found to be ineligible, and 12 completed the screener but declined to participate. The remaining 458 adults were confirmed to be eligible, provided informed consent to participate, and completed the baseline survey, thus comprising the final study sample. At follow-up, 172 did not respond to the six-month survey invitation, 66 responded but were ineligible, and 220 (48.0%) completed the six-month survey. As shown in Table 1, no significant differences in key sample characteristics or baseline survey measures were observed between those who did versus did not complete the six-month survey.
Key Sample Characteristics and Baseline Variables, No. (%).
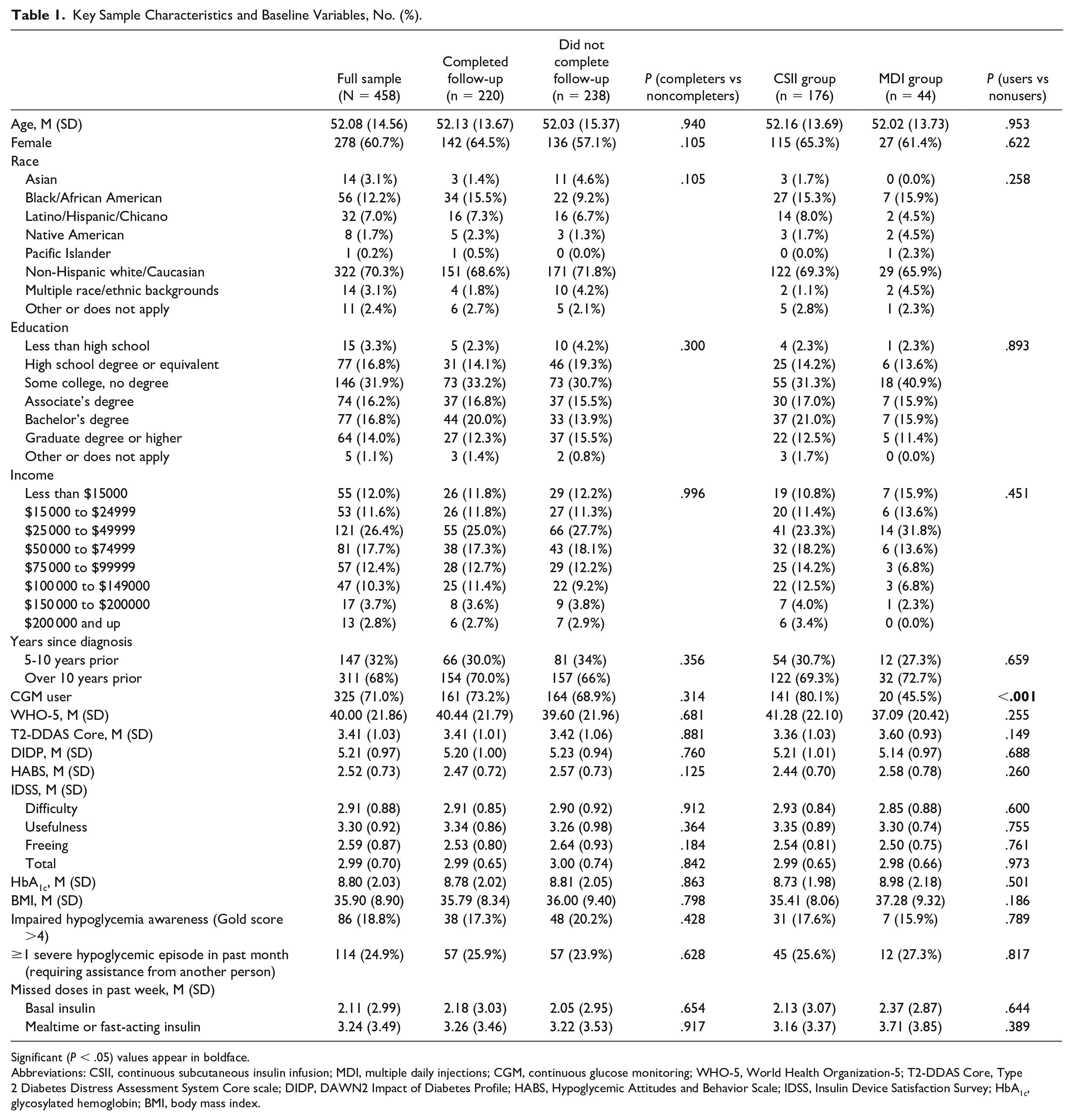
Significant (P < .05) values appear in boldface.
Abbreviations: CSII, continuous subcutaneous insulin infusion; MDI, multiple daily injections; CGM, continuous glucose monitoring; WHO-5, World Health Organization-5; T2-DDAS Core, Type 2 Diabetes Distress Assessment System Core scale; DIDP, DAWN2 Impact of Diabetes Profile; HABS, Hypoglycemic Attitudes and Behavior Scale; IDSS, Insulin Device Satisfaction Survey; HbA1c, glycosylated hemoglobin; BMI, body mass index.
Table 1 shows sample characteristics and descriptive statistics for the baseline survey measures. Of the final study sample (N = 458), the majority were women (60.7%), non-Hispanic white (70.3%), and 50.0% reported an annual household income <$50 000. The mean age at baseline was 52.1 (±14.6) years. The majority (68%) noted that they had been living with T2D for more than 10 years. At baseline, the mean BMI was 35.9 (±8.9) and mean HbA1c was 8.8% (±2.0). Though continuous glucose monitoring (CGM) use was common (71.0% endorsed), 24.9% reported at least one severe hypoglycemic episode within the past month and 18.8% indicated impaired hypoglycemic awareness (Gold score >4). In the past week, the majority (53.6%) reported missing >1 basal injection and 67.9% reported missing >1 mealtime injection. On average, at baseline, overall well-being was poor, with mean WHO-5 = 40.0 (±21.9). Clinically significant levels of diabetes distress were also common, with mean T2-DDAS Core = 3.4 (±1.0).
At six months, most respondents (n = 176; 80%) had started CSII, while a smaller number (n = 44; 20%) remained on MDI. Table 1 shows results from independent samples t-tests of baseline differences between the CSII and MDI groups. Results failed to reveal any significant differences in baseline characteristics, clinical metrics (eg, HbA1c), psychosocial dimensions, or behavioral measures between participants who had started CSII versus those who had not (ie, remained on MDI), with one exception: CGM use was significantly more common among those who then began CSII (80.1%) than among those who continued on MDI (45.5%; P < .001).
Psychosocial Outcomes
Results of the primary analyses, as seen in Table 2A, show that group (CSII vs MDI) emerged as a significant predictor of change in nearly all psychosocial dimensions. Specifically, CSII users reported significantly greater improvement than the continuing MDI users on all major psychosocial metrics over the six-month study period. These included larger increases in overall well-being (WHO-5, P < .001) and greater reductions in diabetes distress (T2-DDAS Core; P < .001), perceived negative impact of diabetes on QOL (DIDP; P = .003), and hypoglycemic worries and concerns (HABS; P < .001). Of the six new items focusing on the degree to which the individual’s lifestyle was feeling limited or restricted by T2D, the CSII group reported significantly greater reductions (indicating that they now felt less restricted) than the MDI group in five of the six items; these included too much time spent thinking about T2D, inability to be spontaneous in one’s life, and feeling limited regarding exercise and food (in all five cases, P < .001). In parallel, CSII users demonstrated significantly greater improvement in overall satisfaction with their insulin delivery system than MDI users (IDSS; P < .001). This was evident across all three IDSS subscales, including “Usefulness,” Difficulty,” and “Freeing” (in all cases, P < .001).
Latent Change Score Model Results.
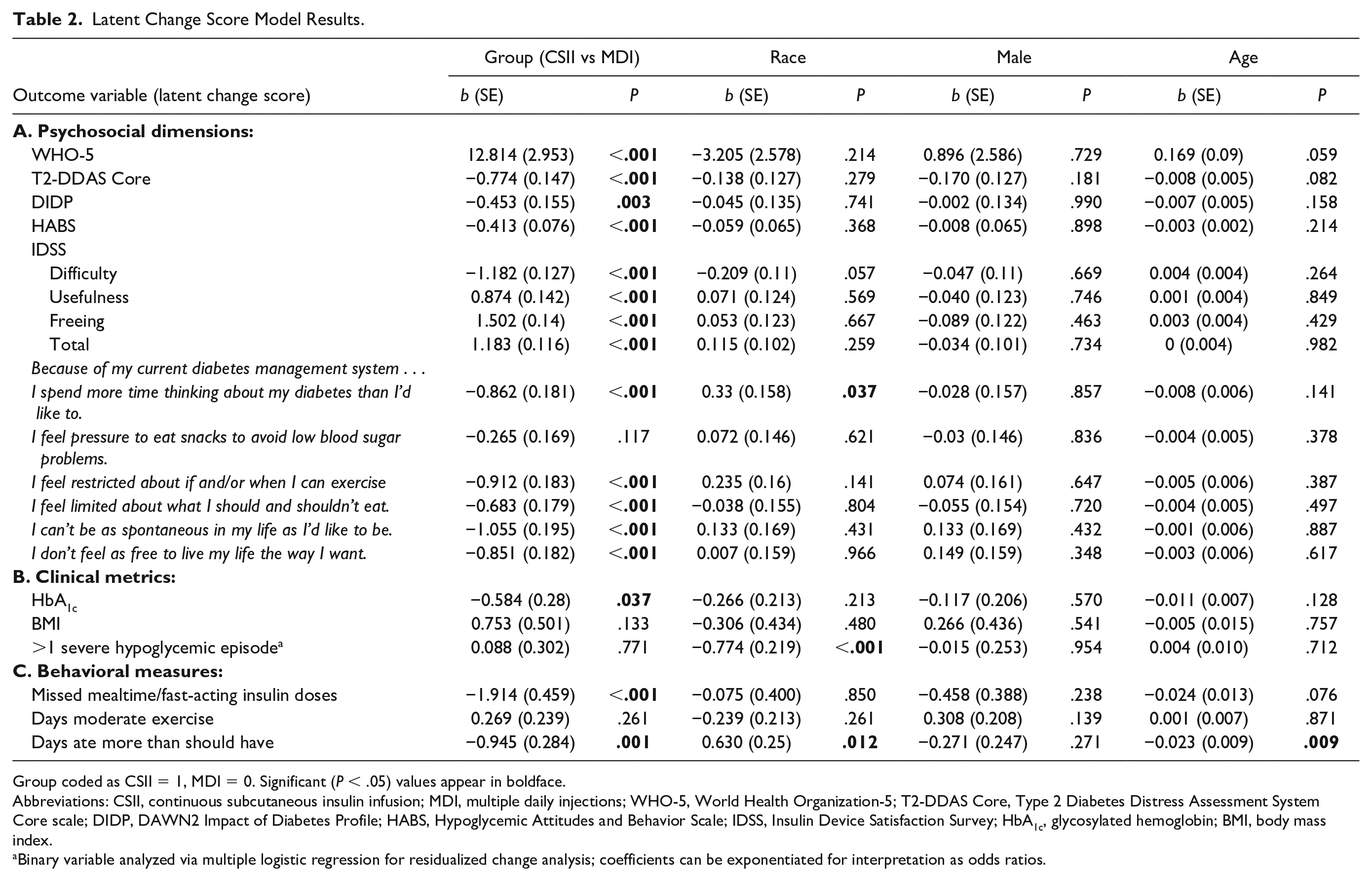
Group coded as CSII = 1, MDI = 0. Significant (P < .05) values appear in boldface.
Abbreviations: CSII, continuous subcutaneous insulin infusion; MDI, multiple daily injections; WHO-5, World Health Organization-5; T2-DDAS Core, Type 2 Diabetes Distress Assessment System Core scale; DIDP, DAWN2 Impact of Diabetes Profile; HABS, Hypoglycemic Attitudes and Behavior Scale; IDSS, Insulin Device Satisfaction Survey; HbA1c, glycosylated hemoglobin; BMI, body mass index.
Binary variable analyzed via multiple logistic regression for residualized change analysis; coefficients can be exponentiated for interpretation as odds ratios.
Table 3A shows paired samples t-test results examining bivariate change in psychosocial dimensions over time within each group. Of note and in line with the findings reviewed above, across all of the psychosocial dimensions (with one exception), we found that significant gains were only observed in the CSII group (in all cases, P < .005). The one exception was diabetes distress, for which a significant, though modest, improvement was also observed in the MDI group (P < .05).
Paired Samples T-test of Change Over Time Within Groups.
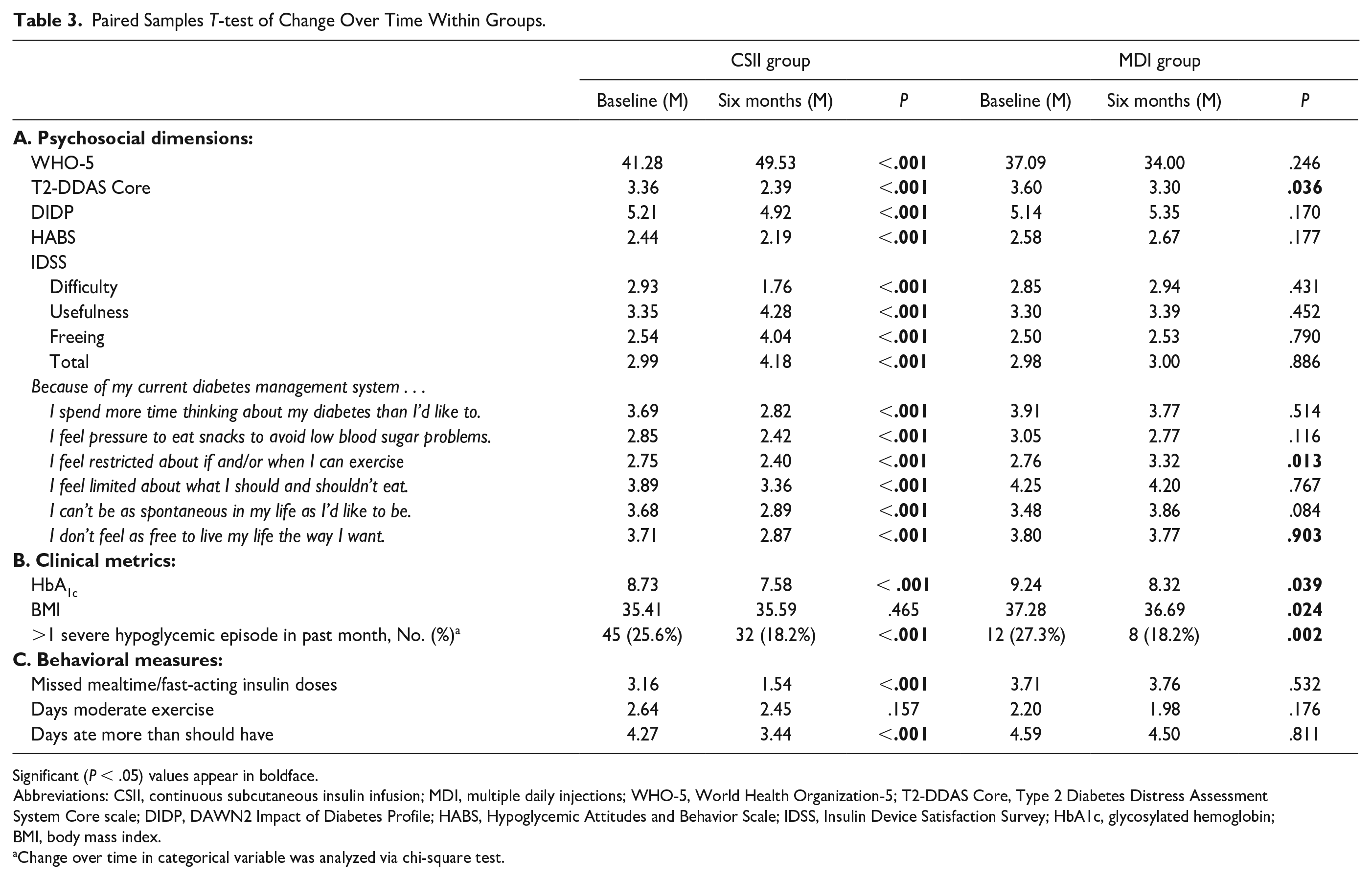
Significant (P < .05) values appear in boldface.
Abbreviations: CSII, continuous subcutaneous insulin infusion; MDI, multiple daily injections; WHO-5, World Health Organization-5; T2-DDAS Core, Type 2 Diabetes Distress Assessment System Core scale; DIDP, DAWN2 Impact of Diabetes Profile; HABS, Hypoglycemic Attitudes and Behavior Scale; IDSS, Insulin Device Satisfaction Survey; HbA1c, glycosylated hemoglobin; BMI, body mass index.
Change over time in categorical variable was analyzed via chi-square test.
To provide some context to these findings, we took a closer look at two key results: the observed change in diabetes distress and overall well-being. We examined the proportion of individuals whose T2-DDAS Core score was below the threshold for elevated diabetes distress (score < 2, indicating absence of significant distress) at baseline and six months. In the CSII group, 14% were below the elevated distress threshold at baseline, but at six months this number had grown to 39%. In the MDI group, 6.8% were below the threshold at baseline, and at six months this number remained relatively low at 11.4%. For the WHO-5, a score >50 is considered a healthy/satisfactory level of well-being. In the CSII group, only 34.7% were in this range at baseline, but six months later this number had grown to 50.6%. In the MDI group, 27.3% had healthy/ satisfactory levels of well-being at baseline, but by six months this number had dwindled to 20.5%.
Clinical Outcomes
Primary results for the clinical metrics (HbA1c, BMI, hypoglycemia) are shown in Table 2B. First, glycemic improvement over the study period was significantly greater among CSII users than MDI users (P < .05, Table 2B). Bivariate tests showed that significant drops in HbA1c were observed in both the CSII and MDI groups (Table 3B). Specifically, mean HbA1c reduction was 1.2% (±1.7) in the CSII group (P < .001) and 0.8% (±1.6) in the MDI group (P < .05). Regarding change in BMI, no significant group differences were apparent (Table 2B), though in bivariate analyses, the MDI group evidenced a significant, though modest, drop (P < .05, Table 3B). Finally, while bivariate analyses showed that the proportion of individuals who experienced ≥1 severe hypoglycemic episode in the past month dropped significantly in both the CSII (P < .001) and MDI groups (P = .002; Table 3B), group (CSII vs MDI) did not emerge as a significant predictor in the multiple logistic regression model (Table 2B).
Behavioral Outcomes
Results of the primary analyses of behavioral measures are shown in Table 2C. First, latent change model results showed a significantly greater drop in missed mealtime boluses per week in the CSII group than the MDI group (P < .001). Specifically, CSII users decreased from a mean of 3.2 missed doses per week to 1.5 missed doses per week, whereas continued MDI users remained unchanged at 3.7 missed doses per week (Table 3C). Similarly, the CSII group evidenced a significantly greater drop over time in days per week of perceived overeating than the MDI group (P = .001). In both cases, bivariate analyses (Table 3C) showed significant improvement only among CSII users (Ps < .001), with no apparent change among MDI users (Ps > .5). Finally, group did not significantly predict change in days per week of at least moderate exercise (P > .2, Table 2C), and did not evidence significant bivariate change over time in either group (Table 3C).
Of note, the observed pattern of psychosocial, clinical, and behavioral results was adjusted for gender, age, or ethnicity (Table 2A-C). Further analyses found that no changes in the results were apparent after additional adjustment for CGM use (data not shown).
Discussion
The introduction of CSII (Omnipod DASH) in this sample of insulin-using adults with T2D was associated with broad improvement in psychosocial outcomes as well as glycemic and behavioral outcomes after six months, compared with those who were similarly interested in starting CSII, but never did so and remained on MDI. Participants in the CSII group evidenced a significantly greater rise in overall well-being; a greater drop in diabetes distress, the perceived negative impact of T2D on QOL, and hypoglycemic worries and concerns; a larger diminution in the sense of being constrained or restricted in one’s lifestyle by T2D; and a significantly larger rise in satisfaction with their insulin delivery system than the MDI group. Among the most robust effects was that the proportion of participants below the threshold for elevated diabetes distress (T2-DDAS Core < 2) in the CSII group more than doubled over time, from 14% to 39%, while the proportion below the threshold in MDI group remained at a low level over the six-month period (6.8%–11.4%). We also observed a substantial increase from baseline to six months in the proportion of CSII participants achieving satisfactory overall well-being (WHO-5 score > 50), from 35% to 51%, whereas the proportion of MDI participants reaching this same level dropped from baseline (27%) to six months (21%).
The Omnipod group similarly reported a significantly larger decline in HbA1c than the MDI group as well as greater declines in two critical self-care behaviors: number of missed mealtime boluses and number of days of perceived overeating. It is particularly noteworthy that the frequency of missed mealtime boluses from baseline to six months dropped by more than half in the Omnipod group (from 3.2 to 1.5 events/week), whereas the MDI group was unchanged (rising slightly from 3.7 to 3.8 events/week). Collectively, these data are consistent with the hypothesis that CSII use contributes to meaningful gains in diabetes-specific and general QOL in addition to glycemic control in adults with T2D.
While these findings represent some of the first prospective data pointing to the psychosocial and clinical value of a full-featured, tubeless pump in a T2D population, it is important to note that the Omnipod DASH is not a hybrid closed-loop system. A large proportion of the subject sample was using CGM, but these devices were not integrated with their pump systems. Now that tubeless hybrid closed-loop systems are available (eg, Omnipod 5), we might expect to see similar if not larger improvements in psychosocial outcomes among T2D users once they become approved for use in this population, similar to what has recently been observed in a sample of T1D participants and their families. 20
The psychosocial benefits observed here are seemingly in contrast to the absent or minimal findings seen in prior T2D studies.10,11 We suspect that there are three major factors which may explain this difference. First, the instruments selected for the current study examine psychosocial elements that are recognized as being particularly relevant to the personal experience of insulin users (eg, diabetes distress, hypoglycemic fears, and worries) and are therefore likely to be sensitive to change. Second, a modern, full-featured pump was examined here, in contrast to a much older CSII generation evaluated in one of the prior studies 10 and a novel patch pump in the other. 11 Furthermore, we speculate that the tubeless nature of the Omnipod DASH may have contributed to the observed study benefits, as was suggested by a retrospective report in a large T1D sample. 21 Indeed, we cannot be certain that similar benefits would be observed among users of other modern CSII systems, as the Omnipod DASH remains the only full-featured, stand-alone CSII system that is tubeless. Third, unlike the previous trials where participants were recruited to participate, this was a highly distressed group of participants (eg, more than half of the sample reported severe diabetes distress at baseline) who were actively seeking a treatment change; thus, there was likely greater room for a potential benefit to surface.
One major strength of this study was its quasi-experimental, prospective design, where a pragmatic approach was used to identify MDI-using individuals with T2D who were all initially motivated to start CSII, then compared over a six-month period those who actually started versus those did not and remained on MDI. However, limitations are apparent. This was not an RCT, and it unknown why some initiated CSII while others did not. No pattern of baseline demographic differences between the two groups was apparent, though the CSII group was more likely than the MDI group to be using CGM at baseline. This may suggest that prior comfort with diabetes technology may have been influential in the choice to start CSII. Alternatively, this may point to differences in insurance coverage as a key factor, though no information regarding insurance type or status was collected. Moreover, we are not aware of recently documented rates of CGM use in this particular population, so the representativeness of the rates observed in our sample is unclear.
A second limitation concerns the representativeness of the CSII and MDI groups, since a large fraction of the initial pool of potential participants did not respond to the study invitation; furthermore, of those who were enrolled and did complete the baseline survey, more than half were lost to follow-up (ie, did not complete the six-month survey). There were no significant differences in baseline characteristics between those who did versus did not complete the six-month survey, but it is not known how these two groups may have fared differently over the observation period. For example, perhaps CSII users with minimal psychosocial or clinical benefits tended to not complete the follow-up survey, thus potentially skewing the observed results. Relatedly, due to limited retention at six months, the sample size for the MDI group was quite small (n = 44), which reduces statistical power to detect significant group differences.
Another limitation was that ratings of self-care behavior, including missed boluses, were not derived from a validated instrument. Finally, it must be noted that all behavioral and clinical metrics, including HbA1c, were self-reported, so these results should be viewed with caution. However, of note, several studies have observed close agreement between self-reported and laboratory-assessed HbA1c (r = .84).22,23
Conclusions
In conclusion, this six-month prospective study found that use of a tubeless insulin pump (Omnipod DASH) among T2D adults originally on MDI contributed to significant psychosocial, glycemic and behavioral benefits. This suggests that broader use of CSII in the insulin-using T2D population may be warranted, with the expectation that similar gains could be common. Randomized trials are now needed to replicate, confirm, and extend these findings.
Footnotes
Abbreviations
BDI, body mass index; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DIDP, DAWN2 Impact of Diabetes Profile; HABS, Hypoglycemic Attitudes and Behavior Scale; HbA1c, glycosylated hemoglobin; IDSS, Insulin Device Satisfaction Survey; MDI, multiple daily injections; QOL, quality of life; RCT, randomized controlled trial; SF-36, 36-item short form survey; T1D, type 1 diabetes; T2D, type 2 diabetes; T2-DDAS, Type 2 Diabetes Distress Assessment System; WHO-5, World Health Organization-5.
Authors’ Note
Portions of these findings were presented at the Advanced Technologies and Treatments for Diabetes Annual Conference (February 2023; Berlin, Germany) and at the American Diabetes Association Scientific Sessions (June 2023; San Diego, USA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W.H.P. and E.C.S. have served as consultants for Insulet.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Insulet.
