Abstract
An increasing number of patients with type 2 diabetes mellitus (T2DM) use insulin pumps. The first insulin pumps especially designed for patients with T2DM have recently become available. However, national guidelines do not primarily recommend the use of continuous subcutaneous insulin infusion (CSII) for this patient group. The effectiveness of CSII in T2DM has not yet been convincingly demonstrated, despite some positive evidence. An overview and an assessment of various studies to date will be given. T2DM is a heterogeneous disease with a substantial phenotypic variability; therefore, it is difficult to provide general conclusions about the effectiveness of CSII in T2DM therapy. The pump types, characteristics, and associated barriers may play a relevant role for therapy outcome. Most advanced functions like various bolus dosages offered by conventional insulin pumps are not needed for T2DM treatment and complicate the device handling for this subject group. Additionally, new technologies like increased connectivity, advanced software features, and interoperability are currently becoming available representing further barriers. The implementation of this technological progress might be a benefit for pumps for T2DM as well. However, these have not been sufficiently examined either and increased security challenges due to integrated peripheral components should not be neglected in terms of a sound cybersecurity. Pump features and handling for patients with T2DM should be as easy as possible, indicating a need for insulin pumps specially designed for patients with T2DM. However, it has to be investigated if pumps designed for T2DM are more effective than other intensified insulin regimens.
In patients with type 1 diabetes mellitus (T1DM), insulin therapy by means of continuous subcutaneous insulin infusion (CSII) with an insulin pump is a well-established therapeutic option. There is a large body of evidence showing beneficial effects of CSII in patients with T1DM, because it mimics the physiological situation by combining a (quasi) continuous insulin infusion rate to cover the basal insulin requirements with additional bolus deliveries to cover prandial insulin requirements and corrections of high glucose values. 1
Insulin therapy in patients with type 2 diabetes mellitus (T2DM) can be managed by different treatment strategies such as basal insulin only, twice-daily premixed insulin, basal-bolus therapy (by means of multiple daily injections [MDI] regimens), or CSII.2,3
However, national guidelines do not primarily recommend the use of CSII for this patient group. It is rather classified as a special option in case a given patient does not achieve his therapeutic goal by using MDI.2 -4
Nevertheless, benefits of CSII that have primarily been proven in patients with T1DM might also be applicable to patients with T2DM. Thus, the number of patients with T2DM using an insulin pump is increasing 5 and first insulin pumps especially designed for patients with T2DM have become available. 6
Currently, several reviews have summarized the results of clinical trials assessing the outcome of CSII in patients with T2DM. Some reviews could not detect beneficial effects of CSII therapy for patients with T2DM 7 or concluded similar outcomes of CSII and MDI.8 -11 Other reviews, however, reported better glycemic control in people with long-standing diabetes and poor glycemic control at baseline through CSII compared with MDI.12 -14 Since most trials do not show a consistent body of evidence, this conclusion mainly refers to the results of the OpT2mise study. 15 This first and as of yet only large-scale randomized trial about CSII in T2DM demonstrated superior effectiveness of CSII in people with poorly controlled T2DM compared with MDI. 15
Clinical Evidence for CSII in Patients With T2DM
When looking at recent trials evaluating the efficacy of CSII in patients with T2DM, these trials differ considerably with respect to study design, methods, used insulin pumps, subject population, and therefore in study quality and maybe also outcomes. Table 1 lists studies on CSII in T2DM published between 2003 and 2018 and summarizes the main characteristics and outcomes.
Studies on CSII in T2DM Published Between 2003 and 2018.
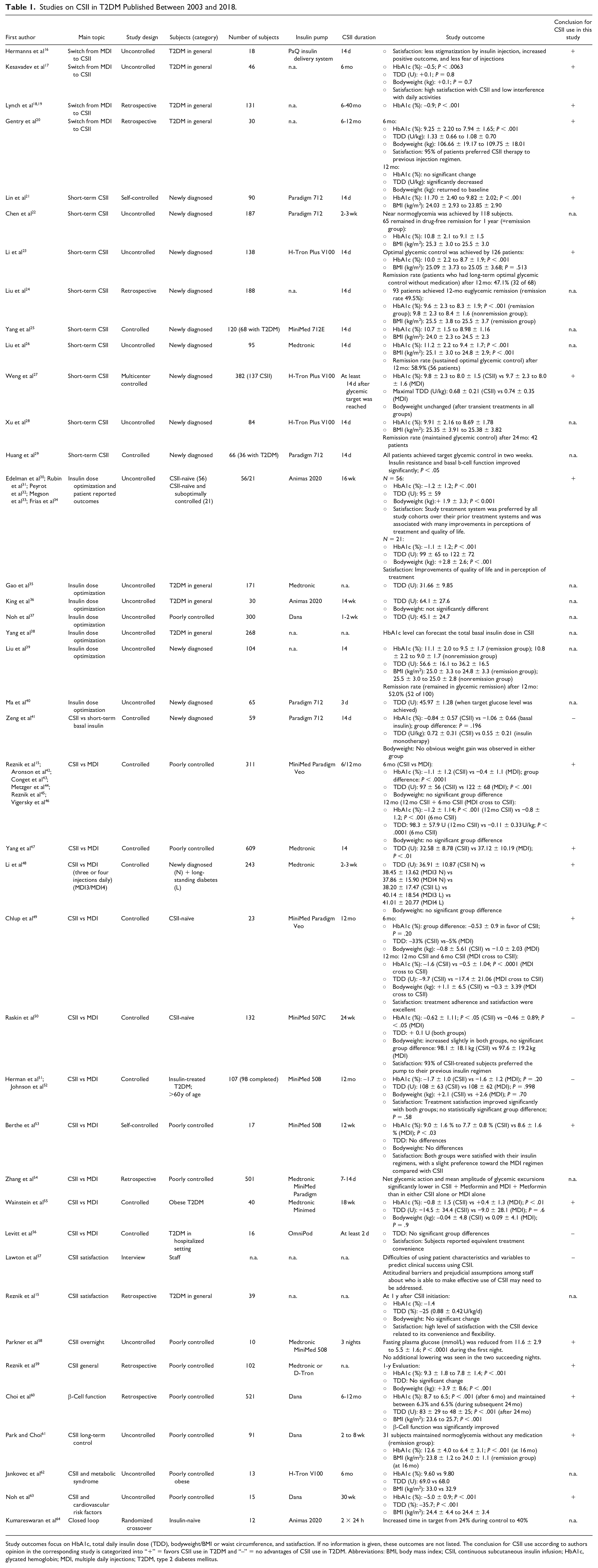
Study outcomes focus on HbA1c, total daily insulin dose (TDD), bodyweight/BMI or waist circumference, and satisfaction. If no information is given, these outcomes are not listed. The conclusion for CSII use according to authors opinion in the corresponding study is categorized into “+” = favors CSII use in T2DM and “–” = no advantages of CSII use in T2DM. Abbreviations: BMI, body mass index; CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin; MDI, multiple daily injections; T2DM, type 2 diabetes mellitus.
The trials had different objectives, as they covered change from MDI to CSII, short-term CSII in newly diagnosed patients with T2DM, CSII in hospitalized settings, and overnight-only CSII. Subject groups included newly diagnosed, poorly controlled, CSII- or insulin-naïve patients with T2DM, and patients with T2DM in general. Additionally, the number of subjects recruited for these studies ranged from 10 to 609 subjects, and the CSII intervention was between one day and five years. In most trials, the insulin pumps used were not particularly designed for T2DM (see Table 2).
Insulin Pump Characteristics Used in Clinical Trials Assessing the Effect of CSII in Patients With T2DM.

Abbreviations: CSII, continuous subcutaneous insulin infusion; DM, diabetes mellitus; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
In the OpT2mise study, a two-month run-in period with a standardized titration protocol (including basal and bolus adjustments) for dose optimization was scheduled, before patients with continued poor glucose control were allocated randomly to the CSII group or MDI group. 15 In the CSII group, a basal rate with 50% of the total daily insulin dose was initiated. The significant glycemic improvements observed after six months of CSII were also prevailing during the next six-month follow-up and were reproduced by the MDI group that switched to CSII after the first six months. 42
In other trials, a run-in period before starting CSII was performed as well; however, the duration of such periods was highly variable.16,26,29,41,49,53 In most trials initiating CSII, a basal insulin delivery of 40%-60% of the total daily insulin dose was chosen.26,27,29,47,56 In some studies, patients stopped previous antidiabetic therapy options when CSII therapy was initiated,31,33,40,52 whereas others used previous therapy options like oral antidiabetic drugs additionally. 15
There was a high variation in the extent of education programs the patients underwent during the different trials. CSII therapy requires users and health care professionals to fully understand how to use the given insulin pump and provided features; therefore, education programs and intensive training providing this knowledge are essential for insulin pump use.65,66 Lack of education may lead to inappropriate and thereby insufficient use of the insulin pump features increasing the risk of complications like ketosis or severe hypoglycemia.65,66
One study assessed the effect of an insulin pump particularly intended for patients with T2DM (PAQ, CeQur). 16 An introducing publication reported reduced barriers to insulin treatment with the use of PAQ, particularly because it takes less time for managing insulin injections, an easier injection of the right amount of insulin at the right time, and undisturbed flexibility. 16 Another article evaluated the use of a patch pump (Omnipod, Insulet Corp) in patients with T2DM as well. 56 The use of that patch pump in combination with continuous glucose monitoring (CGM) in an inpatient setting did not provide superior glucose control over basal-bolus MDI therapy. Furthermore, the inpatient pump initiation was described as technically challenging and time consuming.
Patch Pumps Compared With Traditional Pumps
Currently, several insulin pumps are available, varying regarding handling, material, design, functions, and costs. In general, one can distinguish between conventional durable insulin pumps and patch pumps. Compared to conventional durable insulin pumps with visible insulin infusion sets (IIS), patch pumps are usually attached directly to the skin 67 and typically deliver insulin through a short cannula from the pump to the subcutaneous tissue without an external tubing. 68 Consequently, they are usually smaller and more discreet. While some are full-featured, there are also simplified devices, which primarily are intended as insulin pen replacements. 6 Most patch pumps have disposable components or are completely disposable. 6 A few have prefilled cartridges, but most require manual filling of the insulin reservoir. Patch pumps can deliver either basal or bolus insulin or both. The variety of patch pumps shall reflect the varying patient requirements (eg, different types of diabetes). 67
Compared with conventional insulin pumps, patch pumps show advantages like being smaller and lighter, no tubing, and no device that has to be carried in the pocket or somewhere else, but there are also limitations like more waste.
Insulin Pumps Designed for T2DM
Current insulin pump systems for patients with T1DM feature a lot of advanced functions, which are usually not necessary in the treatment of patients with T2DM and might rather complicate device handling for this subject group. A variety of fine-tuning features for bolus dosages according to carbohydrate intake, for example, or multiple basal rates may not be used by most patients with T2DM.12,14,16 Furthermore, the complex educational approach for pump use required for the advanced functions of current pump system might contribute to underutilization of insulin pumps in patients with T2DM 69 and deter potential patients from using CSII. 14 In addition, costs for less complex pumps are lower.
Besides the patch pump PAQ by CeQur, the simplified bolus patch pump Simplicity by CeQur (formerly known as PAQ Meal by CeQur, OneTouch VIA by Johnson & Johnson, or Finesse by Calibra Medical) and the patch pump V-Go by Valeritas are particularly intended for patients with T2DM (see Table 3). 6 The V-Go is a wearable basal-bolus insulin patch pump without requiring any programming. It is available in three models delivering constant basal infusion rates of 20, 30, or 40 U per day. Additionally, a mealtime dosing of prandial insulin in 2 U increments is possible. The system can be used for one day and is approved for U-100 insulin. The PAQ is a three-day patch pump holding up to 330 U of U-100 insulin, providing seven different predefined basal rates and boluses in 2 U increments. The patch pump enables a continuous basal insulin delivery using different constant basal rates, and insulin boluses can be delivered by pressing the button on the pump. There is no need to inject insulin with a syringe or pen. For bolus deliveries only, the Simplicity bolus patch pump usable for three days is available. The V-Go and Simplicity are fully disposable and work mechanically; there is no controller or display for pump use, representing simple pen replacements.6,70
Insulin Pumps Designed Specifically for Patients With T2DM.
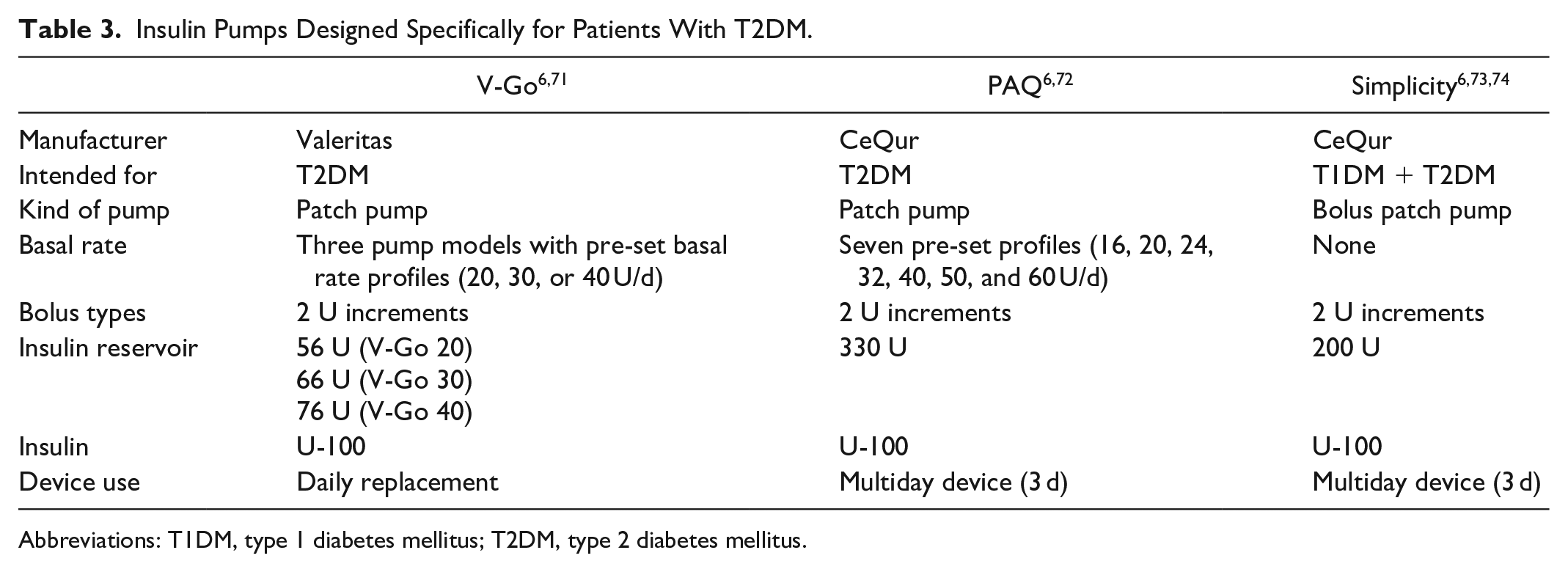
Abbreviations: T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
The PAQ is composed of a disposable insulin reservoir and a reusable messenger communicating its battery life and when the reservoir has to be changed. 72
Software for Insulin Pumps
Insulin pumps represent software-intensive medical devices. 75 Most complex pumps include software that presents past basal and bolus doses as well as additional information that may assist patients and clinicians in diabetes management. 76 Software can be used for dosage control, providing display output or coordinating functions of various components within the pump as well. 77 As further complex functions like dosage calculation based on automatic or manual integrated glucose values and carbohydrate intake have become available, also safety functions become increasingly relevant.77,78
It has to be noted that due to the warrantied life span of an insulin pump, typically most pumps use unchanged software for at least four years and no process for updating the pumps is given during this period. 79 Tandem Diabetes Care designed a platform for the t:slim insulin pump to update the device software by user. Thus, prior to the end of the four-year period, users can update their software enabling the incorporation of new features as they become available for improved diabetes management.
Besides classical insulin pump software, mobile apps including bolus calculators and feedback of insulin delivery amount and timing may support treatment adherence, as well. 80 But on the other hand, having too much information to handle might discourage some patients with T2DM that typically prefer an easy management. Thus, in the previously described patch pumps intended for patients with T2DM mainly no software or remote controller is integrated.
Interoperability and Automated Insulin Delivery Systems
In addition to insulin pumps, diverse other technological devices like blood glucose meters or CGM systems are available for patients with diabetes. Combining CSII and CGM in a sensor-augmented pump (SAP) therapy provides the pump with current glucose information that can be used for insulin adjustments. A study with hospitalized patients with T2DM demonstrated a significant reduction in the time to achieve glucose targets and to decrease exposures to hypo-/hyperglycemia using a SAP compared with MDI therapy. 81
As the devices like CGM and blood glucose meters provide data as well, an integrated analysis of all these devices would provide a complete picture of all therapy-related data. This requires, however, the possibility to connect these devices and promote exchange.
The US Food and Drug Administration (FDA) encourages manufacturers to establish devices that allow communication with a variety of other devices and also to make use of shared information by its new device interoperability pathway. In 2019, two insulin pumps with interoperable technology were cleared to market. The tubeless Omnipod DASH and the Tandem Diabetes Care t:slim X2 insulin pump were referred as alternate controller-enabled insulin pumps (ACE pumps).82 -84 These pumps enable performing integrated diabetes therapy with different components based on individual device preferences.
Recently, the FDA also authorized the first interoperable, automated insulin dosing controller: the Tandem Diabetes Care Control-IQ.82,83 Connected to an ACE pump and an integrated CGM, the glycemic controller automatically adjusts basal insulin deliveries. Thus, after the approval of the integrated automated insulin delivery (AID) system MiniMed 670G, the controller represents the first stand-alone interoperable automated glycemic controller.
The possibility to connect other devices is currently not given by simple patch pumps specifically intended for patients with T2DM. However, the use of AID systems may simplify treatment for both patient and caregivers in inpatient settings. An improved time in range without increased risk of hypoglycemia when using an AID system compared to usual care has already been demonstrated in inpatients with T2DM. 85 Additionally, high system acceptability was reported and most patients were pleased to have managed their glucose control autonomously. 85
However, incorrect calculations or delays of insulin delivery, for example, due to loss of communication represent risks associated with the use of interoperable devices. Thus, the FDA is establishing special controls to provide a safety and effectiveness assurance for such devices.82,83
Cybersecurity of Insulin Pumps and AID Systems
The use of modern diabetes devices, in particular if interoperable, is associated with increased security challenges. The peripheral component risk is increasing, because devices like computers and mobile phones are increasingly integrated into insulin pump systems as well. 86 Many insulin pumps and their corresponding remote control deliver or receive their information, like dosing commands or glucose information, via Bluetooth. In addition, device software integrity leads to more complex systems causing potential cybersecurity vulnerabilities. Thus, a secure information flow is needed to maintain confidentiality, integrity, and availability of the personal diabetes information.86,87 Recently the first recall of a diabetes device because of cybersecurity vulnerabilities was reported in the United States. 88 The risk of hackers connecting to the insulin pump and changing its settings was described.
Thus, a sound cybersecurity should be established to prevent unauthorized agents from hacking diabetes devices.87,89 The Diabetes Technology Society developed the DTSec Cybersecurity standards for diabetes device cybersecurity containing performance and assurance requirements.89,90 Furthermore, in course of the do-it-yourself (DIY) movement, the risk that hacked data and software have any safety issues is high as well. 87 However, maybe the device interoperability pathway by the FDA will enable more secure options for using AID systems the DIY community could benefit from.
As long as simple patch pumps intended for T2DM do not have interfaces, cybersecurity is not a risk in these devices.
Specific Needs in T2DM
To summarize, insulin pumps exhibit numerous aspects that must be taken into account in the selection of an insulin pump. Additionally, T2DM is a heterogeneous disease with a substantial phenotypic variability, which also can lead to misclassifications as described by de Lusignan et al. 91 The conflicting efficacy of CSII reported by different trials may be at least partly due to different characteristics of the patients studied. For example, there are varying baseline characteristics regarding glycemic control, duration of T2DM, age of patients, or insulin requirements. Thus, to assess the efficacy of CSII in patients with T2DM, a more personalized therapy approach may be helpful.
Newly Diagnosed Patients With T2DM
In most trials of the use of CSII in patients with newly diagnosed T2DM, CSII was only used for short, one-to-three-week treatment courses. An early intervention of insulin therapy currently is not recommended in guidelines but described by several articles. Subjects were included when they never received an antihyperglycemic therapy before the study. It was shown that the early intervention reverses β-cell deficiency13,29 and improves glycemic control. In studies focusing on long-term outcomes it was shown that patients sustained optimal glycemic control, increased insulin response, or had stable HbA1c levels in long-term follow-up.22,26,27 Thus, early CSII intervention appears to be an effective therapy in newly diagnosed patients with T2DM. For short-term usage of CSII, the use of simple and affordable patch pumps should be adequate in T2DM.
Poorly Glucose Controlled Patients With T2DM
As reported in some studies, CSII is an effective alternative for patients with T2DM failing glycemic control with other types of antidiabetic therapy.15,53,60,63 Insulin pumps are a good option especially for people not achieving adequate glycemic control with MDI, for example, due to repeated omission of insulin delivery, potentially caused by fear of daily injections, 16 irregular insulin injections, or missing insulin delivery devices, 13 because the burden of transporting insulin and its delivery device is reduced with a wearable insulin pump.
Elderly and Care-Dependent Patients
Many patients with T2DM are elderly and around 90% of people with T2DM are associated with multiple comorbidities, 92 complicating the effective use of an insulin pump. Simple insulin pumps designed for patients with T2DM as reported above intend to simplify pump use compared with advanced pumps, thus reducing the level of technical skills and cognitive abilities required by the user. In some patient groups the involvement of other people supporting diabetes management may be advisable. Especially in elderly people and those with cognitive impairment, the handling of an insulin pump by the users themselves is difficult, 13 and the handling by a caretaker would also be easier. Automatically delivered constant basal rates may be helpful in case of cognitive impairment. In addition, a supervision of insulin delivery using stored delivery data could simplify diabetes therapy. In these cases, concepts of “share the care” may be useful.
Many caregivers only have limited access to glucose data leading to limited assistance in diabetes management. 93 Thus devices with CGM connectivity, enabling AID, may be helpful in this subgroup relieving users and caregivers from manual insulin administration and protecting users from unrecognized episodes of dysglycemia. 94 Furthermore an enhanced interoperability would make data between medical devices more readily available by a greater diversity of data sharing points. 93 To this end, a pan-European consortium started the project CLOSE (Automated Glucose Control at Home for People with Chronic Disease) to develop integrated AID solutions tailored to the needs of people with T2DM. 94
Barriers and Solutions to Insulin Pump Use in T2DM
Despite numerous benefits that insulin pumps may exhibit, there are still barriers that discourage from broader use in T2DM. Safety aspects, whether with regard to malfunction, mishandling, or cybersecurity, have to be addressed for insulin pumps intended for T2DM. Every unintended insulin delivery has the potential to endanger the user’s life or at least to lead to unsatisfactory therapy outcomes. 75 Thus, in the selection of an insulin pump several factors must be taken into account.
Education programs and intensive trainings providing basic knowledge are essential for insulin pump use.65,66 Furthermore, for most patients with T2DM many features offered by modern insulin pumps for CSII therapy are unnecessary and additionally associated with increased safety risks. Thus, a simplified pump may be sufficient for most12,14,16 and complex educational requirements for pump and software features are not needed with such simplified pumps. Especially for older people the adaption to technology is difficult leading to reluctance against insulin pumps. 6 In T2DM multiple comorbidities may complicate the effective use of an insulin pump as well. The user interface should be easy to read with buttons being big enough to manage the pump (or the handheld in case the pump has one) easily, even in the presence of comorbidities of T2DM like visual impairments and limited motoric skills. Insulin pumps without IIS and prefilled cartridges could facilitate the handling as well.
Traditional insulin pumps contain IIS with external tubing that can cause risks and user problems. IIS are regarded as the “Achilles heel of CSII” and one of the main reasons for discontinuing CSII. In addition to infusion site reactions, insufficient insulin delivery, for example, due to clogging, air bubbles, or kinking of the tubing represent the main issues with IIS.67,95 As patch pumps do not have an external tubing, there might be an advantage considering IIS issues. 67 IIS usually have to be replaced every two to three days. However, in practice an IIS is often used for several days or even longer. To investigate the effects of an IIS use longer than recommended, clinical trials are currently performed. Furthermore, novel insulin infusion catheters were designed to guarantee functionality in case of kinking or occlusion. 96
Skin problems like inflammations or even allergic contact dermatitis, due to either the insertion or the adhesive fixing the IIS or the patch pump, are increasingly reported. To prevent skin reactions many patients use protective tapes or glues under the IIS/patch pump.97,98
In contrast to patients with T1DM, insulin-resistant patients with T2DM require larger amounts of insulin per day. 99 For many insulin pumps, the maximal bolus dose is limited, which may preclude adequate bolus administrations for patients with T2DM. Moreover, due to the use of high doses of U-100 insulin, more frequent insulin reservoir changes are needed in such patients. Reservoirs with sufficient volume, avoiding frequent reservoir changes, and the possibility to use concentrated insulin would simplify device handling for patients with T2DM, providing an important aspect for improved device acceptance by the patients. Second-generation ultra-long-acting highly concentrated basal insulins like the FDA-approved insulins Glargine U300 or Degludec U200 show a greater duration of action, reduced glucose variability, and fewer glucose peaks compared to other long-acting insulins. However, adverse events as reported for the risk of overdosing as well as the cost effectiveness need to be taken into consideration.100,101 The described concentrated basal insulins are currently available in disposable pen devices 100 for MDI but not for CSII therapy.
Pens are the most widespread devices for insulin delivery; however, the lack of insulin dose logging represented a shortcoming for using an insulin pen. In 2017 the first smart pen that records insulin dose amount and timing and transmits the information to a mobile app was approved by FDA. 80 Thus, also modern smart insulin pens enable access to important clinical data and may represent a useful option for people with less complex insulin regimens. However, the number of subcutaneous injections is reduced when using simple patch pumps. 92
In most countries, insulin pumps are not reimbursed by the health care system for patients with T2DM. According to the US Medicare decision memo, for all people with T2DM, including insulin-requiring, insulin pumps will be denied as not medically necessary. 102
To conclude, pump features and handling for patients with T2DM should be as easy as possible, indicating a need for insulin pumps specially designed for patients with T2DM. A summary of the preferable characteristics of an insulin pump intended for patients with T2DM is provided in Table 4. In several studies, good glycemic control was achieved with only one or two basal insulin infusion rates per day in patients with T2DM.30,36,58,70 A simple to use patch pump with a maximum number of two basal rates 16 and one standard bolus type should be sufficient for most patients with T2DM therapy. Furthermore, basal rates do not need to be changed as frequently as in insulin pumps for T1DM. A prefilled insulin reservoir 14 volume of 3mL, with the possibility to use higher concentrated insulin should be available. Moreover, for hospital and retirement homes, the opportunity of CGM connectivity enabling AID would be preferable. Additionally, cost effectiveness is a topic that should not be neglected. An affordable insulin pump for T2DM therapy may increase insulin pump usage in this patient group considerably, which might, in turn, help alleviating the current lack of evidence by providing real-world data.
Preferable Characteristics of an Insulin Pump Intended for Patients With T2DM.

Abbreviations: CGM, continuous glucose monitoring; T2DM, type 2 diabetes mellitus.
Conclusions and Necessary Future Research
In patients with T1DM, CSII is a widely used therapeutic option. However, guidelines for the treatment of patients with T2DM classify the use of an insulin pump as a special option only, limiting the usage of pumps to those patients who do not achieve their therapeutic goals by using other therapeutic options. Studies evaluating the effect of insulin pump use in patients with T2DM report different outcomes; this might be influenced by the assessed patient group, pump type used, CSII duration, and other methodological aspects.
T2DM is a heterogeneous disease with a substantial phenotypic variability; therefore, it is difficult to provide general conclusions about the effectiveness of CSII in patients with T2DM. In specific subject groups, like poorly controlled patients, the use of an insulin pump is probably more effective than MDI therapy. The pump types and characteristics used for patients with T2DM play a relevant role for the therapy outcome as well. Most of the advanced insulin pump features offered by modern conventional insulin pumps are not needed for the treatment of patients with T2DM and complicate the device handling for this subject group. Additionally, increased security challenges, especially due to increasingly integrated peripheral components, should not be neglected in terms of a sound cybersecurity.
These barriers point out that pump features and handling for patients with T2DM should be as easy as possible indicating the need for insulin pumps specially designed for patients with T2DM. Randomized controlled studies should be conducted to assess whether (patch) pumps specifically designed for T2DM are more effective than other intensified insulin regimens by appropriate studies. Study designs should reflect the realistic use in the respective population and consider handling aspects. A sufficiently long run-in period (at least two months) and follow-up period (at least six months) should be ensured.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is general manager of the IfDT (Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Dexcom, LifeScan, Menarini Diagnostics, Metronom Health, Novo Nordisk, PharmaSens, Roche, Sanofi, Sensile, and Ypsomed. DW and SB are employees of the IfDT. LH is a consultant for a number of companies that develop novel diagnostic and therapeutic options for diabetes treatment; for example, Roche Diabetes Care, Becton Dickinson, Lifecare, and Berlin-Chemie. LH is shareholder of the Profil Institut für Stoffwechselforschung, Neuss, Germany and ProSciento, San Diego, USA. BK received research funding from Roche Diabetes Care Deutschland GmbH. OS has acted as member of advisory boards and/or given lectures under support from Abbott, Astra Zeneca, Bayer Healthcare, Boehringer-Ingelheim, Eli Lilly, Medtronic, Novartis, Roche Diagnostics, and Sanofi; OS is CEO and founder of Sciarc GmbH. UG is an employee of Roche Diabetes Care GmbH, Mannheim, Germany. RZ has received speaker’s honoraria and/or served on advisory boards from/of Abbott, Ascensia, AstraZeneca, BerlinChemie, Dexcom, Lilly, Novo Nordisk, and Roche Diabetes Care.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors received support for writing this article from Roche Diabetes Care, Mannheim, Germany.
