Abstract
Background:
Continuous subcutaneous insulin infusion (CSII) is used less for type 2 than for type 1 diabetes because of inconsistencies in evidence of effectiveness. We reviewed published guidelines on intensive insulin therapy in type 2 diabetes to assess whether updating of guidance is needed with respect to evidence used and recommendations for CSII in diabetes management.
Methods:
A literature review was performed to identify published national and international guidelines on type 2 diabetes management. Searches were performed using PubMed, Cochrane Library, and Embase databases, and websites of national health care agencies, reimbursement agencies, and professional associations. Searches were limited to articles published in English between 2004 and 2014 and 1666 unique hits were identified, of which 22 were reviewed following screening.
Results:
Only 6 of the 22 guidelines identified from North and South America, Western Europe, Greece, and Israel provided specific recommendations on intensive insulin therapy and the role of CSII, and only 1 provided information on the grade of evidence supporting recommendations. Quality appraisal based on the AGREE II tool suggested that published guidelines may have limitations in terms of search methodology and evidence grading, and findings were of mixed rigor and clarity. Only 3 guidelines described the population for whom CSII may be appropriate.
Conclusions:
Guidelines need to improve the evidence base, rigor, clarity, and grading of evidence associated with recommendations on intensive insulin therapy in type 2 diabetes. Future updates may benefit from considering recent evidence on the efficacy of CSII in poorly controlled patients on MDI.
The management of type 2 diabetes mellitus remains a significant health care challenge. Despite evidence from landmark clinical trials and recommendations in published guidance, glycemic control is often poor for patients in clinical practice and insulin therapy can be delayed or is not optimized. 1 Barriers to the initiation and intensification of insulin therapy include the known side effects, such as weight gain and increased risk of hypoglycemia, concerns over the complexity of the treatment regimen, and fear of injections.2-5 The implications of reluctance to initiate and intensify insulin therapy have been described in the primary care setting.6,7 Analyses suggest that at least 40% of individuals initiating insulin failed to achieve target glycated hemoglobin (HbA1c) levels of ≤7.5% (58 mmol/mol), with average time-to-insulin initiation >11.5 years from diagnosis, suggesting that patients frequently fail to meet glycemic control targets with insulin therapy.
Intensification of the insulin regimen to control hyperglycemia is traditionally achieved in type 2 diabetes with basal-bolus insulin therapy (short-acting prandial insulin, together with long-acting insulin injections such as glargine or detemir), also known as multiple daily injections (MDI) of insulin. However, MDI regimens can be overwhelming and unpopular with a large number of patients, and despite combination with metformin, many patients still fail to achieve good glycemic control. Comorbidities such as obesity and hypertension are very common and weight gain associated with high insulin doses can make it even more difficult to achieve good glycemic control. 8
Although there are considerable clinical trial data in populations with type 1 diabetes with elevated HbA1c and/or disabling hypoglycemia on MDI that supports the benefits of continuous subcutaneous insulin infusion (CSII, insulin pump therapy) as an alternative intensified insulin regimen, far fewer clinical investigations of CSII have been reported in type 2 diabetes.9,10 A 2014 consensus statement from the American Association of Clinical Endocrinologists (AACE) and the American College of Endocrinology (ACE) listed 13 published studies of CSII in populations with type 2 diabetes. 9 Results from these studies were mixed in terms of demonstrating a reduction in HbA1c, hypoglycemia rates or weight gain, and limited by relatively small patient numbers and different trial designs, so the that no consistent body of evidence emerged on the efficacy of CSII in type 2 diabetes.
Similarly, a recent review that examined the evidence base for CSII in type 2 diabetes 11 included data from 4 randomized controlled trials (RCTs)12-15 and 5 open-label observational studies16-20 (2 prospective and 3 retrospective). Two RCTs showed a relative HbA1c benefit with CSII versus MDI in obese patients with poor baseline glycemic control and high insulin requirements,14,15 but the other 2 trials showed no clear benefit for CSII.12,13 The observational studies, which may possibly be more representative of the patients treated in routine clinical practice than volunteers in RCTs, also suggested that there are benefits for CSII in type 2 diabetes patients with poor glycemic control and/or severe insulin resistance, though again many of these studies were small-scale.
Inconsistency and relative weakness in the evidence base for CSII in type 2 diabetes has led to several national guideline committees not recommending insulin pump therapy as a treatment option in type 2 diabetes, for example the widely quoted CSII guidelines from the UK National Institute for Health and Care Excellence (NICE). 21 However, a recent large-scale RCT of CSII versus MDI in a selected population of type 2 diabetes patients uncontrolled on MDI (the OpT2mise trial 22 ) has provided new, more robust evidence of the efficacy of CSII that may question whether an update of guidelines for the appropriate use of CSII in type 2 diabetes is now needed.
The OpT2mise study was a multicenter, multinational RCT that enrolled patients with type 2 diabetes and poor glycemic control despite MDI therapy. After a 2-month run-in period aimed at optimizing control on MDI, 331 patients who had continued poor control (HbA1c 8.0-12.0% [64-108 mmol/mol]) and insulin doses of 0.7-1.8 units/kg were randomly allocated to receive either CSII or to continue MDI with long- (glargine or detemir) and short-acting (aspart, lispro, or glulisine) insulin analogs. After 6 months of follow-up, mean ± SD HbA1c had decreased from a baseline of 9% (75 mmol/mol) in both groups by 1.1 ± 1.2% (12 ± 13 mmol/mol) on CSII and 0.4 ± 1.1% (4 ± 12 mmol/mol) in the MDI group, corresponding to a difference in HbA1c of −0.7% (95% CI −0.9 to −0.4%; –8 mmol/mol [–10 to −4 mmol/mol], P < .0001) between treatment groups. Mean ± SD insulin doses were also lower on CSII therapy: 97 ± 56 versus 122 ± 68 units, CSII versus MDI (P < .0001), with no significant difference in bodyweight change between the 2 groups (1.5 ± 3.5 versus 1.1 ± 3.6 kg, p = 0.322). Safety profiles were comparable between CSII and MDI. Publication after 12 months of follow-up has reported both the sustainability and reproducibility of effects of CSII on glycemic control. 23 After the 6-month randomization phase of the study, patients in the MDI group were switched to CSII therapy for the 6-month continuation phase. The improvement in HbA1c was maintained in the CSII group over the continuation phase, while switched patients showed a reduction in HbA1c such that there was no significant difference between groups at the 12-month time point. Daily insulin doses in CSII group remained stable over the continuation period and doses in the switched group decreased by 19%, such that daily insulin doses were equivalent in both groups at 12 months. There were no differences in weight gain and safety profiles remained comparable between groups after 12 months of follow-up.
Since the OpT2mise trial was the first large study to demonstrate that CSII is more effective than MDI at reducing HbA1c in people with poorly controlled type 2 diabetes, it is timely to discuss whether this new level of clinical evidence should influence and inform future guidelines for the management of the disease. The aim of the present study was to review the current international guidance on intensive insulin therapy and CSII in type 2 diabetes, and the evidence on which it is based, and to consider what, if any, additional evidence might be needed for CSII to be recommended as a routine treatment in selected people with type 2 diabetes.
Research Design and Methods
Study Design
A literature review to identify published national and international guidelines on the management of type 2 diabetes. The review was designed to specifically identify guidance on intensive insulin therapy and CSII, the evidence cited to justify guidance and the evidence grading given (if any) in each publication.
Data Sources and Searches
Literature searches were designed using high-level Medical Subject Headings (MeSH) terms. The search string used for the PubMed database was: ((((“Diabetes Mellitus, Type 2”[Mesh]) AND (“Guidelines as Topic”[Mesh] OR “Guideline” [Publication Type] OR “Standard of Care”[Mesh] OR Guidelin*[tiab] OR Recommendation*[tiab] OR (“health”[tiab] AND “technology”[tiab] AND “assessment”[tiab]) OR “health technology assessment”[tiab])) NOT (“clinical trial”[Publication Type] OR “editorial”[Publication Type] OR “letter”[Publication Type] OR “meta analysis”[Publication Type] OR “randomized controlled trial”[Publication Type])) AND (USA[tiab] OR (United[tiab] and states[tiab]) OR (North[tiab] and America[tiab]) OR America[tiab] OR American[tiab] OR Canada[tiab] OR Canadian[tiab] OR Europe[tiab] OR European[tiab] OR England[tiab] OR English[tiab] or UK[tiab] OR (United[tiab] and Kingdom[tiab]) OR Britain[tiab] OR British[tiab] OR France[tiab] OR French[tiab] OR Germany[tiab] OR German[tiab] OR Italy[tiab] OR Italian[tiab] OR Spain[tiab] OR Spanish[tiab] OR Greece[tiab] OR Greek[tiab] OR Israel[tiab] or Israeli[tiab] OR USA[au] OR (United[au] and states[au]) OR (North[au] and America[au]) OR America[au] OR American[au] OR Canada[au] or Canadian[au] OR Europe[au] OR European[au] OR England[au] OR English[au] OR UK[au] OR (United[au] and Kingdom[au]) OR Britain[au] OR British[au] or France[au] or French[au] OR Germany[au] OR German[au] OR Italy[au] OR Italian[au] OR Spain[au] OR Spanish[au] OR Greece[au] OR Greek[au] OR Israel[au] OR Israeli[au])) AND (“2004/09/30”[PDat] : “2014/09/30”[PDat] AND English[lang]). Search syntax was adapted as required for searches in the Embase and Cochrane Library databases. Hand searches were also performed of the websites of national health care agencies, reimbursement agencies and professional associations in the same geographies. Searches were performed in September 2014 and all hits were screened for inclusion in the review by 2 researchers with discrepancies resolved by discussion.
Study Selection
Searches were limited to articles published in English between 2004 and the search date. We excluded guidelines for the management of type 1 diabetes using intensified insulin regimens, including CSII. We also excluded guidelines pertaining to the management of pregnant type 1 and type 2 diabetes patients.
Data Extraction
A total of 1666 unique hits were identified by the searches of the publication databases (Figure 1). During screening, 1481 references were excluded as they were not publications containing guidance or recommendations on the management of diabetes. Of the remaining 185 articles, 80 did not contain any information on the management of type 2 diabetes and a further 52 were excluded as they did not contain relevant information on intensive insulin therapy, leaving 53 publications for full text review. Hand searches identified an additional 4 articles and therefore full text review was performed on 57 guideline publications, leading to the identification of 22 guidelines with information directly relevant to intensive insulin therapy in type 2 diabetes, as summarized in Table 1.
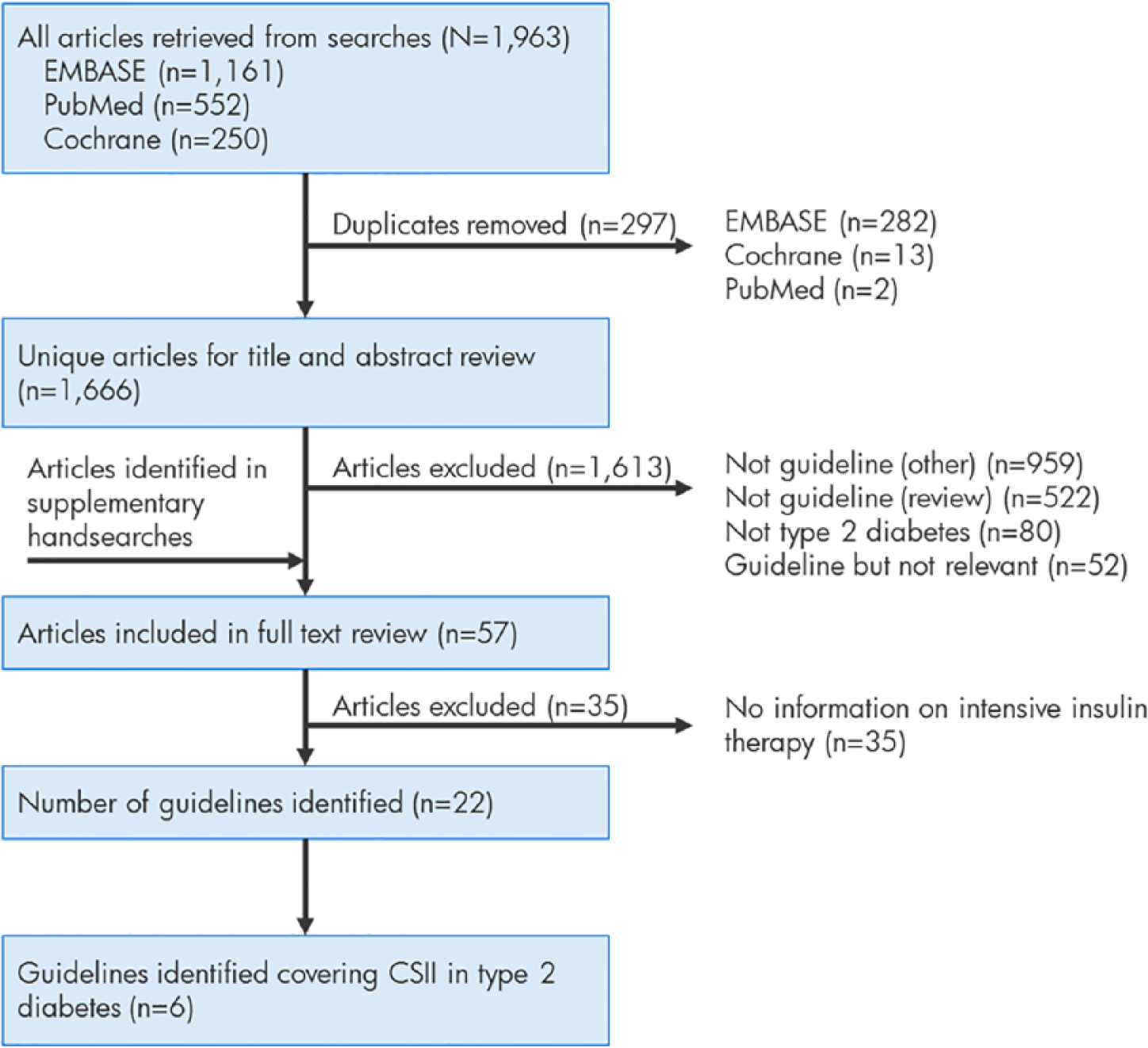
Flow diagram of literature database searches and screening.
Summary of Published Guidelines on Intensive Insulin Therapy in Type 2 Diabetes.
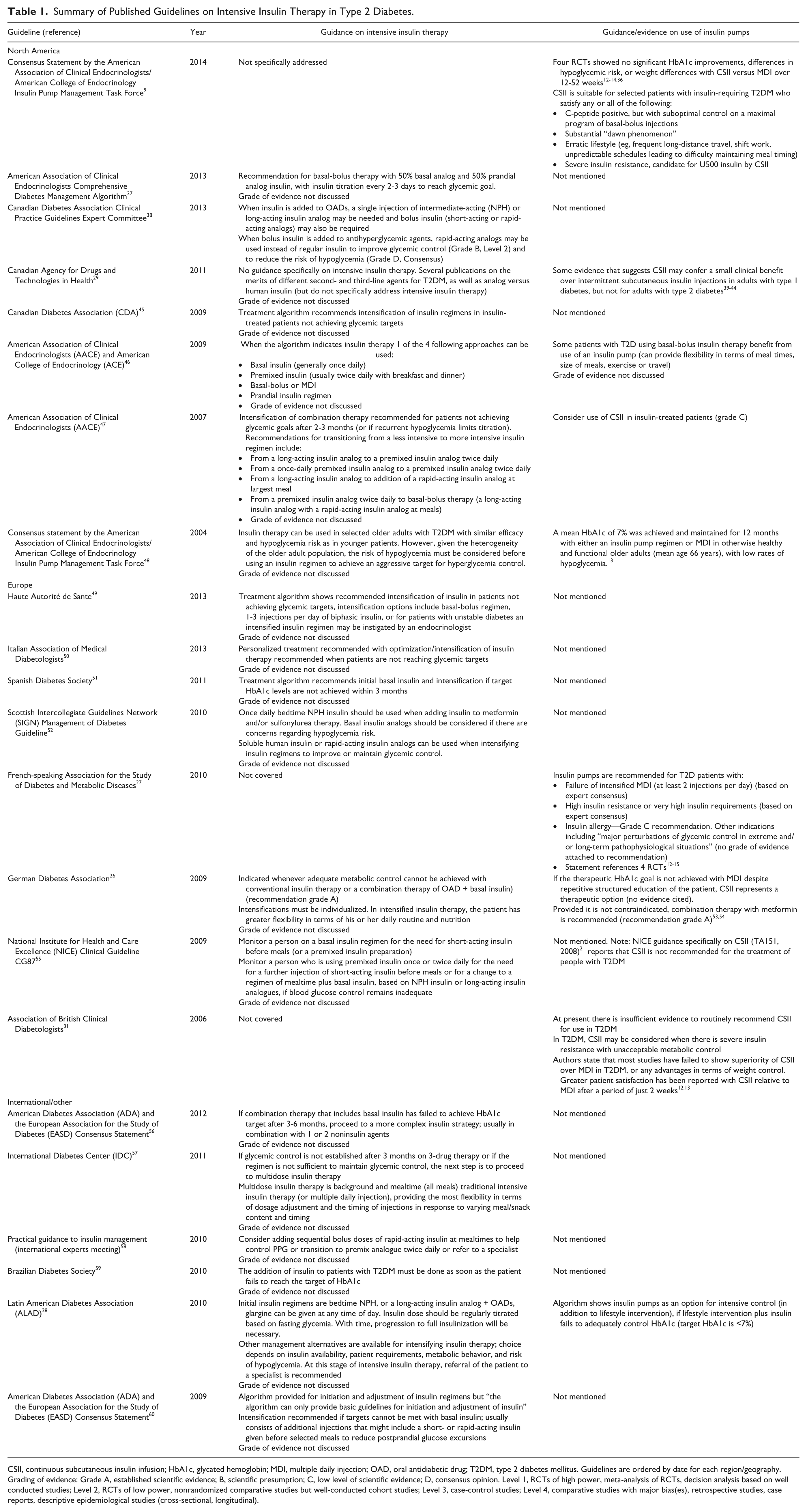
CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin; MDI, multiple daily injection; OAD, oral antidiabetic drug; T2DM, type 2 diabetes mellitus. Guidelines are ordered by date for each region/geography. Grading of evidence: Grade A, established scientific evidence; B, scientific presumption; C, low level of scientific evidence; D, consensus opinion. Level 1, RCTs of high power, meta-analysis of RCTs, decision analysis based on well conducted studies; Level 2, RCTs of low power, nonrandomized comparative studies but well-conducted cohort studies; Level 3, case-control studies; Level 4, comparative studies with major bias(es), retrospective studies, case reports, descriptive epidemiological studies (cross-sectional, longitudinal).
Quality Assessment
Quality and reporting of guidelines was assessed using the AGREE II recommendations 24 with scoring for search methods, evidence grading, rigor, clarity and conflict of interest.
Data Synthesis and Analysis
We tabulated information from guidelines according to recommendations given on intensive insulin therapy and CSII, and evidence cited and graded, and then performed a qualitative analysis of the data.
Results
Review of the 22 published guidelines identified by the literature review indicated a paucity of prescriptive recommendations on intensive insulin therapy for the management of type 2 diabetes (Table 1). Eight guidelines were identified for North America, 8 for Europe, 5 were multi- or international, and 1 was for Brazil. Only 6 of the 22 guidelines provided specific recommendations on the types and combinations of insulin to be used and, of these, only 1 provided information on the grade of evidence that supported recommendations (the Canadian Diabetes Association Clinical Practice Guidelines Expert Committee report 25 ).
In terms of recommendations pertaining to CSII in patients with type 2 diabetes, only 6 guidelines were identified (that had not been superseded by updated versions). Arguably the most detailed recommendations on CSII were made in the consensus statement by the AACE/ACE Insulin Pump Management Task Force, where it was recommended that CSII is suitable for selected patients with insulin-requiring type 2 diabetes who satisfy any or all of the following: C-peptide positive, but with suboptimal control on a maximal program of basal-bolus injections; a substantial “dawn phenomenon”; erratic lifestyle (eg, frequent long-distance travel, shift work, unpredictable schedules leading to difficulty maintaining meal timing); or severe insulin resistance (candidate for U500 insulin by CSII). 9
A recommendation that CSII may be appropriate for those failing MDI (or basal-bolus therapy) was also made in guidelines from the German Diabetes Association, 26 the French-speaking Association for the Study of Diabetes and Metabolic Disease (FASDMD), 27 and the Latin American Diabetes Association. 28 The FASDMD guidelines also included recommendations for CSII in the following populations: high insulin resistance or very high insulin requirements; insulin allergy; or other indications including major perturbations of glycemic control in extreme and/or long-term pathophysiological situations.
Three guidelines on type 2 diabetes management were identified where CSII was not recommended for the general type 2 diabetes population. The Canadian Agency for Drugs and Technologies in Health (CADTH) reported (2011) that “there is some evidence that suggests continuous insulin infusion by ambulatory pumps may confer a small clinical benefit over intermittent subcutaneous insulin injections in adults with type 1 diabetes, but not for adults with type 2 diabetes.” The Association of British Clinical Diabetologists (ABCD) indicated (2006) that there is insufficient evidence to routinely recommend CSII for use in type 2 diabetes, but acknowledged that CSII may be considered when there is severe insulin resistance with unacceptable metabolic control. The NICE Guidance on CSII (2008) also indicates that CSII is not recommended for the treatment of type 2 diabetes. 21
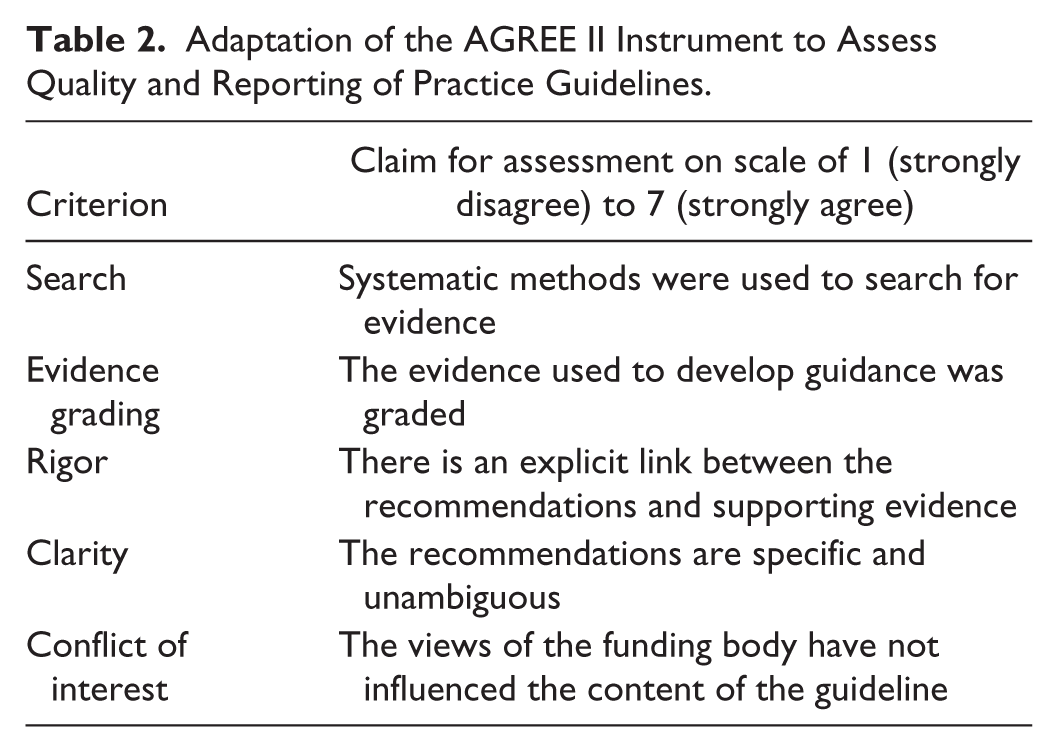
To assess the evidence base on which the CSII recommendations in the published guidance was based, an appraisal was performed using the relevant criteria from the AGREE II tool, designed to assess the quality and reporting of practice guidelines. 24 Five criteria were scored on a scale of 1 (strongly disagree) to 7 (strongly agree) covering the following themes (Table 2): search (systematic methods were used to search for evidence); evidence grading (the evidence used to develop guidance was graded); rigor (there is an explicit link between the recommendations and supporting evidence); clarity (the recommendations are specific and unambiguous); and conflict of interest (the views of the funding body have not influenced the content of the guideline). In addition, a total score was calculated by summing the scores for each of these 5 criteria.
Adaptation of the AGREE II Instrument to Assess Quality and Reporting of Practice Guidelines.
The guidelines scored poorly on the search criteria and none of current published guidelines/consensus statements identified by the literature review were based on a systematic review of the literature (Table 3). In general, the guidelines also scored poorly on evidence grading, with only 1 guideline attempting to grade evidence appropriately (FASDMD). 27 Results were mixed, in terms of both rigor and clarity, with only 3 out of 6 guidelines attempting to describe the population with type 2 diabetes for whom for CSII therapy would be most appropriate. Conflict of interest was not identified in any of the guidelines reviewed, although an influence could not be ruled out based on the information provided in the Canadian Agency for Drugs and Technologies in Health Report. 29 Total scores ranged between 13 and 25 (out of possible total of 35), which may reflect limitations in the literature search methodology for the guidelines. None of the published guidelines captured the full body of published evidence on CSII in populations with type 2 diabetes, contributing to limitations in evidence grading, rigor and clarity.
Quality Appraisal of Evidence Supporting Current Guidelines on the Use of CSII in the Management of Type 2 Diabetes.
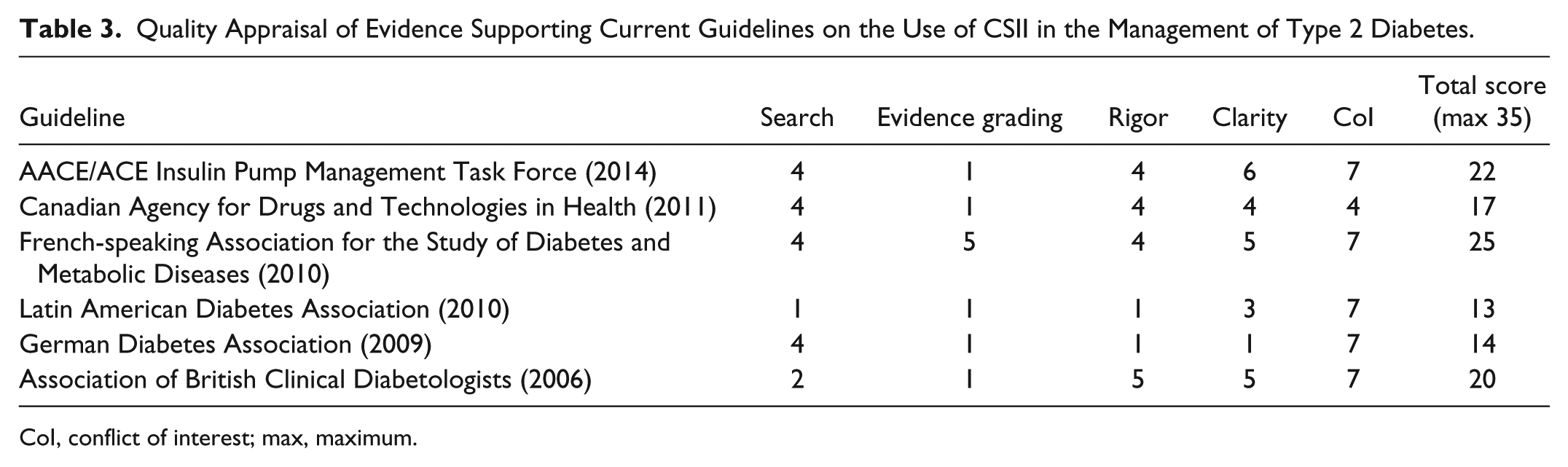
CoI, conflict of interest; max, maximum.
Discussion
Our literature review identified 22 guidelines or consensus statements with information on intensive insulin therapy in type 2 diabetes published between 2004 and 2014. Of these, only 6 provided recommendations on the role of CSII in modern clinical practice and the guidance provided was equivocal and not based on the full body of available evidence.
Most recently and after the literature search was conducted, Inzucchi et al published an update to the ADA/EASD Position Statement on the management of hyperglycemia in patients with type 2 diabetes. 30 This statement and others we have reviewed (for example, the AACE guidance 9 ) emphasize the need to individualize treatment targets and strategies, with a focus on patient-centered care, shared decision making, and the role of clinician judgment over and above prescriptive treatment recommendations. But, as with other guidelines, the ADA/EASD statement makes little mention of insulin pump therapy, indicating only that CSII is a less commonly used and more costly alternative to MDI and that, for patients with latent autoimmune diabetes of adulthood (LADA), a basal-bolus approach or an insulin pump would represent the optimal approach to therapy. 30 The evidence base for discussing the role of CSII in type 2 diabetes is not made clear in these guidelines, nor is any evidence grading provided for the statements made.
The strength of our study is that we performed a formal literature search and evaluation of the evidence base underlying practice guidelines on intensive insulin therapy in type 2 diabetes, but it is limited by the fact that the part-funding source also funded the OpT2mise trial of CSII versus MDI, in type 2 diabetes and this may have biased our interpretation of the literature. However, in an effort to maintain transparency, we have made our searches available (on application) to independent scrutiny.
Why is CSII frequently not discussed in guidelines, what are the barriers to incorporating evidence-based recommendations on CSII use into guidelines on type 2 diabetes management and how can we overcome short falls in evidence on CSII? First, notwithstanding new evidence from the OpT2mise trial on the efficacy of CSII in patients poorly controlled on MDI which was published in summer 2014 only a few months before this review was performed, 22 there is a paucity of trials conducted in specific populations where the greatest clinical benefit is likely to be derived (eg, patients with severe insulin resistance and/or continued poor glycemic control on MDI). Some guidelines have noted that there was insufficient evidence found to recommend routine use of CSII within the general type 2 diabetes population,29,31 but they have not considered subgroups where CSII may provide a useful therapeutic option. Additional trials within specific type 2 diabetes populations with clinical problems, such as those with severe insulin resistance and poor control on MDI and early insulin users, may therefore strengthen the evidence base for targeted CSII use in type 2 diabetes. Reanalysis of data from existing trials, such as regression of HbA1c change against baseline covariates such as HbA1c and insulin dosage may also help to identify those who might benefit the most from insulin pump therapy.
Recommendations on CSII use in type 2 diabetes may also be restricted by a lack of cost-effectiveness studies of CSII versus MDI in type 2 diabetes. Recently, Roze et al 32 investigated the cost-effectiveness of CSII versus MDI in type 2 diabetes patients failing to achieve good glycemic control on MDI in the Dutch setting, based on data from the OpT2mise trial; CSII was considered cost-effective for this population of type 2 diabetes patients in this setting. Additional analyses in other countries and groups of patients are now required to establish whether these findings are consistent across different settings and clinical problems. There is also emerging evidence that less complex and costly pumps using simpler insulin infusion regimens may be more suitable for management of type 2 diabetes than the more sophisticated and costly pumps used heretofore for type 1 diabetes. 33 Cost-effectiveness is likely to be considerably improved for these pumps.
In conclusion, we have found that current national and international guidance on intensive insulin therapy in type 2 diabetes seldom mentions whether there is a role for insulin pump therapy in the management of this type of diabetes. When recommendations are made, the evidence is poorly cited, discussed and graded. The OpT2mise clinical trial has provided new evidence that CSII has clinical benefits over MDI in selected patients with type 2 diabetes, notably those who are poorly controlled on MDI. Taken together with the numerous studies showing the high proportion of type 2 diabetic patients failing to achieve satisfactory control on MDI,34,35 this suggests that there may be an important role for a trial of CSII in long-standing type 2 diabetes patients who do not respond to intensification on MDI. We therefore consider that updated guidance on the intensive insulin treatment of type 2 diabetes is needed, with better completeness, rigor, clarity and grading of the evidence on which the guidance is based. Future guidance should explicitly consider the role of insulin pump therapy in selected patients with type 2 diabetes.
Footnotes
Acknowledgements
The authors are grateful to Jayne Smith-Palmer at Ossian Health Economics and Communications for support with the literature review and editorial input. The authors also acknowledge the financial support provided by Medtronic International Trading Sàrl to perform the review.
Abbreviations
AACE, American Association of Clinical Endocrinologists; ACE, American College of Endocrinology; ADA, American Diabetes Association; CSII, continuous subcutaneous insulin infusion; EASD, European Association for the Study of Diabetes; HbA1c, glycated hemoglobin; LADA, latent autoimmune diabetes of adulthood; MDI, multiple daily injections (of insulin); NICE, National Institute for Health and Care Excellence; RCT, randomized controlled trial; SD, standard deviation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: William Valentine is an employee of Ossian Health Economic and Communications, which received financial support from Medtronic International Trading Sàrl to perform the review. Ohad Cohen is an employee of Medtronic International Trading Sàrl, which is a manufacturer of CSII systems.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from Medtronic International Trading Sàrl.
