Abstract

Insulin pump therapy is an integral part of type 1 diabetes management. The COVID-19 pandemic posed challenges to the usual insulin pump initiation process, prompting increased reliance on virtual training. In this study, we evaluated 12-month glycemic and safety outcomes of virtual versus in-person training.
In an academic diabetes clinic, we examined insulin pump initiations in 25 sequential patients who underwent virtual training between May 2020 and February 2021 and in 15 patients who received in-person training prior to May 2020. Patients met with a diabetes educator for approximately 4 sessions via either audio-visual teleconference (virtual training group) or in-person (routine training group). All patients used a continuous glucose monitor (CGM) and none had used an insulin pump prior to the study period. We compared CGM glycemic metrics at baseline, 3, 6, 9, and 12 months after pump initiation. We used Shapiro-Wilk test to determine the distribution of glycemic outcome, medians and interquartile ranges to describe each outcome, and Wilcoxon signed rank test to assess statistical differences between the groups.
Both groups were predominantly white (77.5%), non-Hispanic (87.5%), male (60.0%), with type 1 diabetes (82.5%). The average age was 45.8 years. 80% of the virtual and 73% of the routine training group used Tandem t:slim X2 insulin pump with Control-IQ Technology. Average time to pump start was faster in virtual group (50 vs 80 days). Baseline time in range (TIR) (BG 70-180 mg/dL) was 61.0% and 65.8% in the virtual and routine groups, and 71.0% and 78.8% at 12 months. Baseline time in hypoglycemia (BG < 70 mg/dL) was 1.0% and 0.5% in virtual and routine groups, and 0.4% and 0.6% at 12 months. Baseline time in hyperglycemia (BG > 180 mg/dL) was 33.9% and 34.1% in the virtual and routine groups, and 20.0% and 20.9% at 12 months. There was no statistically significant difference in glycemic outcomes between the groups at baseline or at 12 months (see Figure 1). One case of diabetic ketoacidosis (DKA) occurred in both groups after pump initiation but neither resulted in hospitalization.
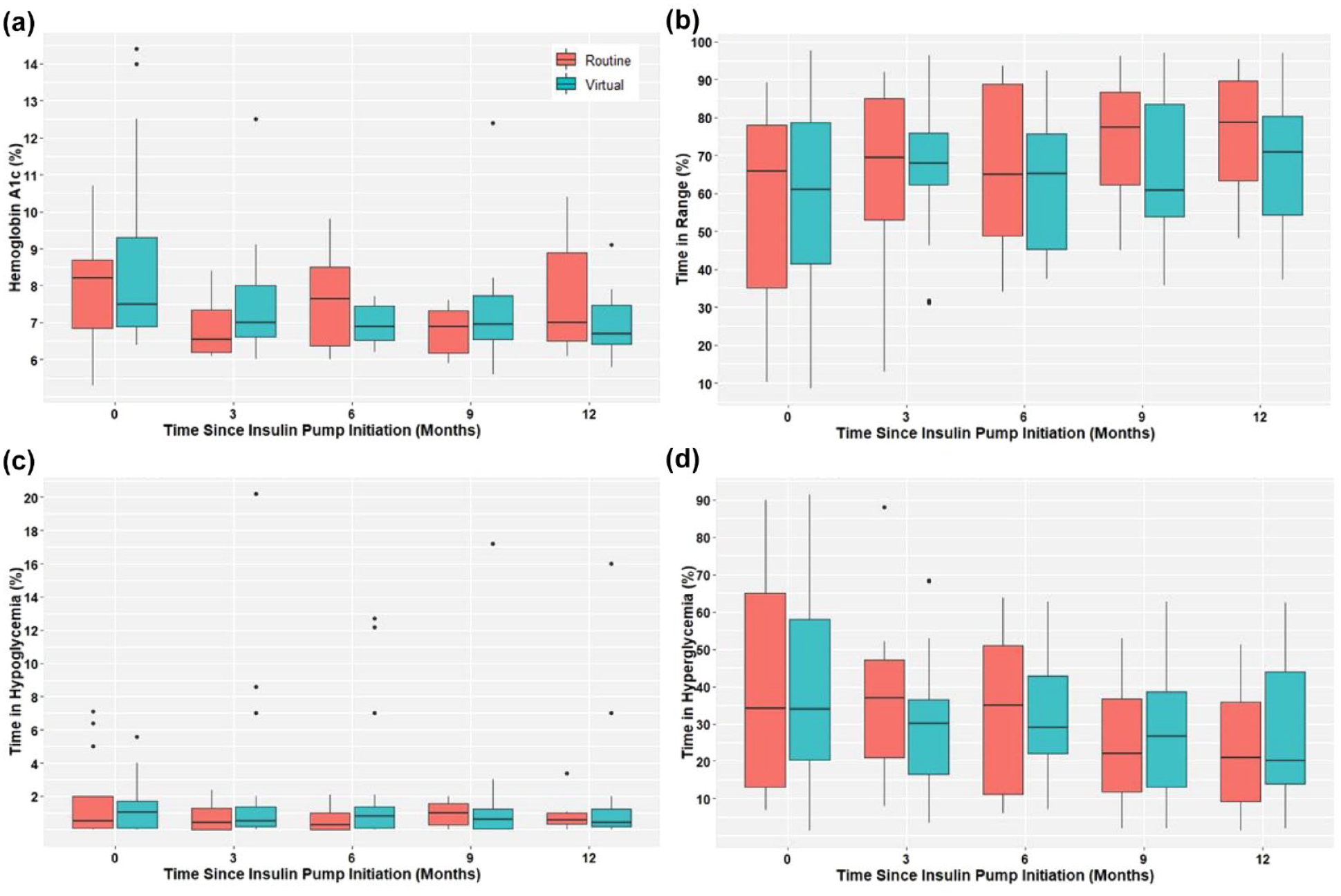
Comparing glycemic control outcomes at baseline, 3 month, 6 months, 9 months, and 12 months after insulin pump initiation between routine training group and virtual pump training group. (a) Hemoglobin A1c levels in routine versus virtual insulin pump initiations. (b) Time in range (70-180 mg/dL) in routine versus virtual insulin pump initiations. (c) Time in hypoglycemia (<70 mg/dL) in routine versus virtual insulin pump initiations. (c) Time in hyperglycemia (>180 mg/dL) in routine versus virtual insulin pump initiations.
Our study showed that patients who underwent virtual pump training achieved improvement in average blood glucose, TIR, and time in hyperglycemia without worsening hypoglycemia, similar to the routine training group. Safety data were comparable between the groups. In previous studies, Pinsker et al 1 found significantly higher TIR (72% vs 67%, P < .01) in the virtual compared with in-person training group who started Tandem t:slim pump 6 months prior. Vigersky et al 2 found that TIR was 68% in the virtual and 70% in the in-person training group 1 month after patients started MiniMed 670G.
Our study is unique as it included pump-naïve patients who likely require more intensive training. The study also followed patient outcomes for a more extended period-of-time (12 months) and included specific safety outcomes, namely, DKA events post pump initiation.
Our study demonstrates that timely initiation of pump therapy can be accomplished via virtual training without increased rates of hypo- or hyperglycemia. Larger prospective studies are needed to confirm these findings.
Footnotes
Abbreviations
BG, blood glucose; CGM, continuous glucose monitor; DKA, diabetic ketoacidosis; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
