Abstract
Globally, health care workers (HCWs) are at a high risk of occupational exposure to needlestick injuries (NSIs). Needlestick injuries not only are associated with an increased risk of infections caused by bloodborne pathogens but are also a primary source of emotional distress and job burnout for HCWs and patients. Insulin injection–related NSIs are common among HCWs working in hospitals in the Asia-Pacific (APAC) region and impose a significant burden. Insulin pen needles have a high risk of transmitting infections (at both the patient-end and cartridge end of the sharp) after use. Recapping a needle after administering an insulin injection poses a major risk to HCWs. Currently, several safety-engineered needle devices (SENDs) are available with active or passive safety mechanisms. Passive insulin safety pen needles with dual-ended protection and automatic recapping capabilities have resulted in a significant drop in accidental punctures to HCWs while administering insulin to patients with diabetes. In this article, we have reviewed the burden and common causes of NSIs with insulin injections among HCWs in the APAC region. We have discussed current approaches to address the issues associated with NSIs and the benefits of introducing SENDs in health care settings, including long-term care facilities, nursing homes, and home care settings where patients may require assisted insulin injections. This review also summarizes key strategies/recommendations to prevent NSIs in HCWs and patients with diabetes in the APAC region.
Introduction
Needlestick injuries (NSIs) are one of the most important occupational hazards and pose a significant risk to health care workers (HCWs) globally. As per the Centers for Disease Control and Prevention (CDC), an estimated 385 000 cases of NSIs occur annually among HCWs in hospitals in the United States. 1 Furthermore, the European Agency for Safety and Health at Work reports around 1 million cases of NSIs among HCWs in Europe each year. 2 Nurses, surgeons, and emergency room workers are at high risk of NSIs.3-6 More than 20 types of infectious diseases, including hepatitis B (HBV), hepatitis C (HCV), and human immunodeficiency virus (HIV), have been reported to be transmitted via contaminated needles due to NSIs.3,7-9 The World Health Organization (WHO) estimated that among 35 million HCWs, around 3 million experience percutaneous exposures to bloodborne pathogens each year (HBV: 2 million; HCV: 0.9 million; and HIV: 170 000). 10 Bouya et al 3 evaluated the prevalence of NSIs among HCWs (87 studies, N = 50 916 HCWs) in 31 countries across the globe. The study reported the 1-year global pooled prevalence of NSIs among HCWs as 44.5% (95% confidence interval [CI]: 35.7-53.2), with the highest prevalence of NSIs in Southeast Asia (58.2%; 95% CI: 36.7-379.8). Hypodermic needles were reported as the most frequent cause of NSIs, with a prevalence of 55.1%, followed by intravenous (IV) cannula (23%). 3
Insulin needles are responsible for approximately 20% of all syringe-related NSIs. 11 According to a study in France, NSIs with injection pens accounted for 39% of NSIs associated with subcutaneous (SC) injection. 12 Kiss et al 13 reported that insulin pens were the most frequent cause of NSI (40.4%) among nursing personnel in nursing homes. This was due to unsafe needle-handling practices (routine handling [52.6%] and needle recapping [28.1%]). 13 Studies have shown that insulin injection–related NSIs are prevalent among HCWs working in hospitals in the Asia-Pacific (APAC) region and impose a significant burden.14-17 Kuppusamy et al 14 reported the occurrence of NSIs among staff nurses (100 nurse respondents) in a private hospital in Singapore. The study identified a higher number of NSIs in the medical/surgical department (59%), with a greater proportion of diabetes patients requiring insulin injections, followed by the accident and emergency department (38%), and operating theaters (3%). 14 Insulin injection pens–related injury was the most frequently noted injury among medical staff in China, accounting for nearly 26% of all sharp injuries. 16 Zhao et al 15 reported the incidence of NSIs involving injection pens as 139.5 per 1000 nurses per year in China. The use of insulin injection pens caused 10.2 NSIs per 100 000 injections. 15 Around 3.2% of nurses reported having HBV infections and 0.9% of nurses reported having HCV infections as a consequence of NSIs. 15 The study recommended precautionary efforts for NSIs, including the adoption of injection devices with superior safety features, as the total direct cost per insulin injection-related NSI (costs of lab tests, post-exposure prophylaxis [PEP], and infection treatment) was estimated to range between ¥1884 and ¥2389 ($279–$354). 15
Emotional exhaustion and stress have been reported after NSIs. Wang et al 18 highlighted a significant correlation between emotional exhaustion, depersonalization, and sharps injury (P < .001). NSIs can cause significant financial burden and reputational damage to the organization and HCWs. Costs associated with NSIs may include consultation charges, laboratory analysis, PEP, and hepatitis B booster vaccinations.19,20 In this article, we have reviewed the burden and common causes of NSIs with insulin injections among HCWs in the APAC region. We have discussed current approaches to address the issues associated with NSIs and the benefits of introducing safety-engineered needle devices (SENDs) over conventional devices in health care settings. This review also summarizes key strategies/recommendations to prevent NSIs in HCWs and patients with diabetes in the APAC region.
Common Causes of NSIs Among HCWs
Mengistu and Tolera 21 examined various risk factors associated with the occurrence of NSIs among HCWs in developing nations (Malaysia, Iran, India, Nigeria, Ethiopia, Pakistan, Tanzania, and Egypt). Female gender, recapping of needles, heavy workload, and failure to adopt precautions were identified as the potential risk factors linked with the high prevalence of NSIs. 21 The incidence of NSIs among HCWs in the APAC region has been reported to be associated with age, history of related training/education, failure to observe precautions, and number of shifts per month.22-24 A cross-sectional questionnaire survey among hospital nurses in South Korea highlighted that the occurrence of NSIs was significantly associated with work experience and emotional exhaustion during work. 25 Rampal et al 26 studied the common causes of NSIs among HCWs in Serdang Hospital, Malaysia. NSIs occurred when HCWs forcefully recapped or manually tried to disassemble the needle or sharp device. 26 The majority (66.1%) of the HCWs assumed that needles should be recapped after use and only 52.5% of the HCWs had knowledge about needleless safety devices. 26 The incidence of NSIs among HCWs administering injections to patients with diabetes or blood draws with lancets (during blood glucose monitoring) is just as high or higher than in other departments or wards. 27 Misalignment of the angle of injection and poor visualization are key causes of NSIs with the use of insulin pens (IPs). 28 NSIs could happen during the injection if the HCW does not maintain a 90º angle to the patient’s body. 28
Concerns Related to NSIs Among HCWs
Physical Effects and the Risk of Transmission of Infections
Fingersticks and injections administered to patients with diabetes present a risk of blood exposure to the injector as well as other HCWs. 27 The risk of transmission of infections after NSIs may be affected by several key factors, such as the depth of injury, type of sharp used, chances of infection, injury into an artery or vein with the contaminated needle, and body fluid inoculated at the time of injury. 29 Occupational exposure to contaminated sharps is responsible for 37% to 39% of the worldwide cases of HBV and HCV infections in HCWs, which impact the quality of life. 19
Psychological Effects
The risk of emotional and psychological adverse effects is high after NSIs. Matsubara et al 30 revealed that HCWs who experienced NSIs in the past 6 months had significantly higher anxiety scores than those who did not experience it (P = .004) at tertiary hospitals in the Lao People Democratic Republic. In addition, 42.7% of HCWs were fearful of needles and sharp devices in the subsequent 2 weeks than at the time of the interview. 30 Another study from the United States involving 400 nurses caring patients with diabetes mentioned that 2 weeks after NSIs, 60.1% of nurses were fearful of using needled devices and 41.8% of nurses felt anxious, depressed, or stressed. 31 Disposable syringes were responsible for 80% of the NSI events, and in 50% of the injuries, the device was equipped with a safety feature that either was ineffective (85.5%) or malfunctioned (3.6%-9.1%). 30 Consequently, nurses missed 77 days of work. 31
Financial and Reputational Impact
Direct costs include testing the source and exposed HCW, in addition to post-exposure medical visits and treatment (prophylaxis). Indirect costs include lost productivity due to time off from work, litigations, and exposure consequences such as emotional distress and anxiety. Mannocci et al 32 reported that the overall median of the means of costs for managing an NSI was $747 (mean of means: $861; a range of means: $199–1691). Another study at National University Hospital, Singapore, estimated that the annualized economic burden ranges from $109 800 to $563 152 due to 244 NSIs in 5957 HCWs in 2014. 20 Kunishima et al 33 estimated the annual economic burden of in-hospital NSIs (testing and treatment) among Japanese HCWs as $302 million based on an average cost per NSI of $577 and 525 000 NSI cases. Costs due to each NSI in the United States are estimated to range from $71 to $4838. 34
Current Approaches to Address the Issues Associated With NSIs
The occurrence of NSIs can be broadly linked to (1) engineering factors, (2) organizational/administrative factors including the availability of supplies and policies for reporting, (3) behavioral factors/work practices notably needle recapping and disposal-related issues, and (4) level of training and protection of HCWs.22-24,26,35 The WHO has identified a hierarchy of controls to reduce the risk of NSIs. Figure 1 lists the hierarchy of control measures to reduce the risk of NSIs in hospital settings in the order of priority. 36 These include (1) elimination of hazards, (2) engineering controls, (3) administrative controls, (4) work practice controls, and (5) use of personal protective equipment. 36 Wittmann A developed a standardized risk-assessment matrix for medical sharps injuries. This can help identify the probable risks of bloodborne virus transmission associated with devices, level of sharps safety required, and appropriate preventive measures (Figure 2). 37 Directive 2010/32/EU requires that formal risk assessments are performed for all activities where medical sharps are used and that wherever there is a risk of injury or infection, it must be eliminated using appropriate training, improved working, and the introduction of safety-engineered medical devices. 37
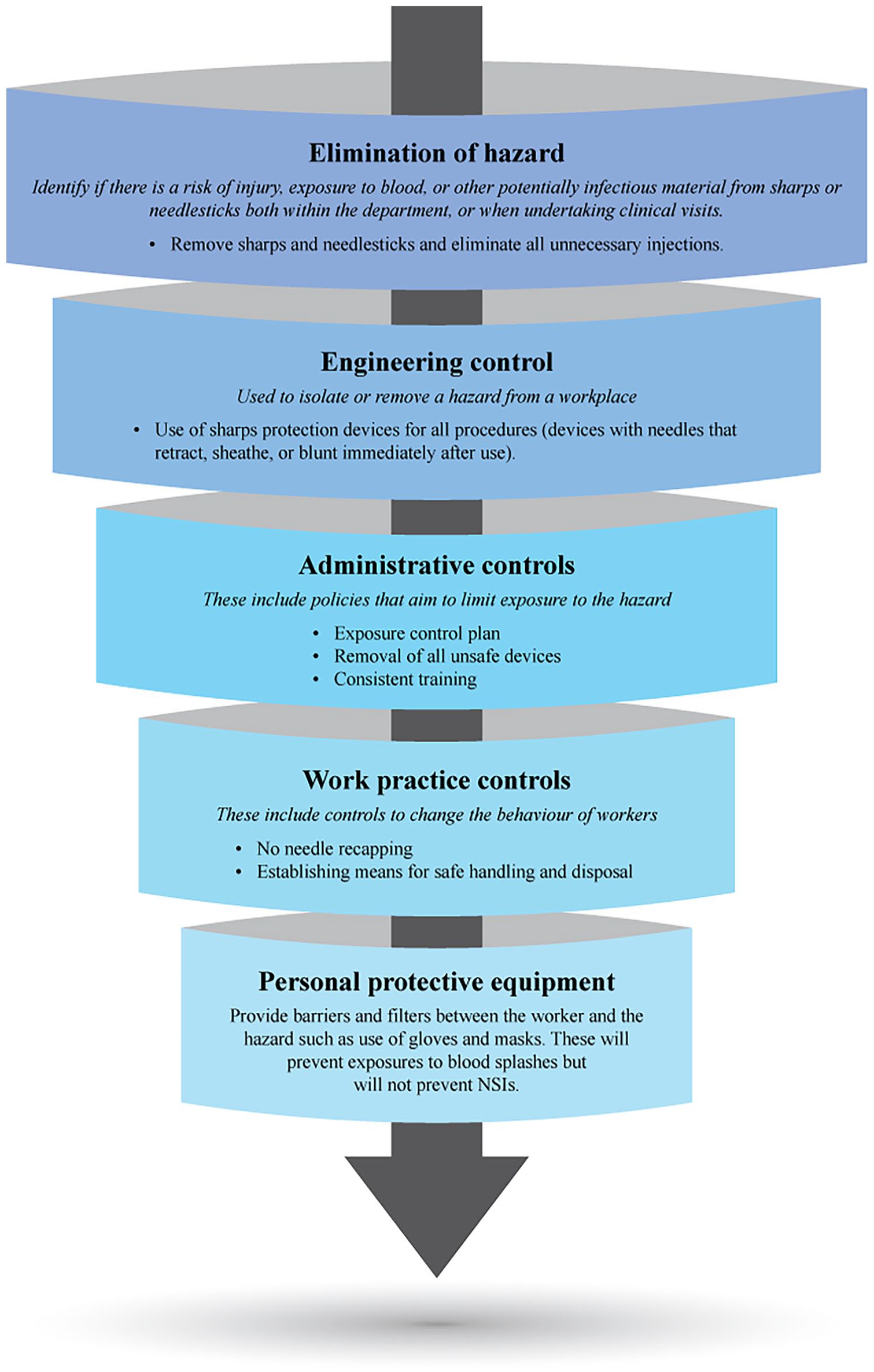
Hierarchy of controls to reduce the risk of NSIs (order of priority: highest on the top). Abbreviations: NSIs, needlestick injuries; WHO, World Health Organization.
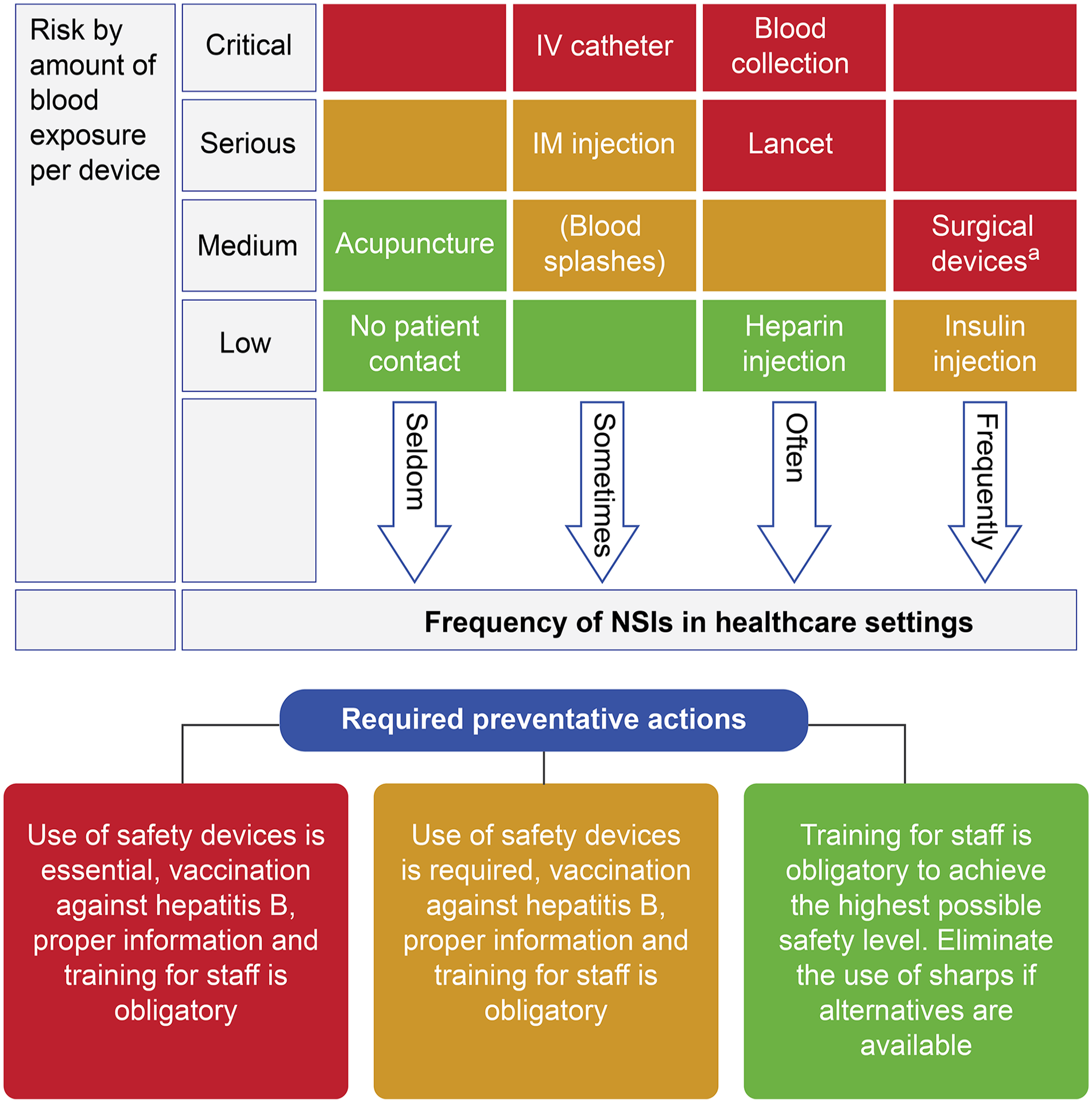
Risk-assessment matrix and analysis. Abbreviations: IV, intravenous; IM, intramuscular; NSIs, needlestick injuries.
United States was the first nation to enact the Needlestick Safety and Prevention Act (NSPA) in 2001 to reduce the incidence of NSIs among frontline HCWs. 38 The act emphasized evaluation and implementation of safety-engineered devices, including sharps with engineered sharps injury protection and needleless systems. The law required the updation of the exposure control plan and maintenance of a sharp injury log documenting the different types of devices causing injuries. 38 Between 2010 and 2014, many countries across North America, Europe, and East Asia adopted needle safety legislation that mandated health care facilities to provide safety-engineered devices to reduce the risk of NSIs among HCWs. 39 Ou et al 39 compared the incidence rates of NSIs between legislated (Italy, Canada, Poland, USA, Taiwan) and unlegislated (Thailand, Hong Kong, Korea, Saudi Arabia) countries after the adoption of NSPA. The risk of NSIs among HCWs in legislated countries decreased by 22% after 3 to 6 years of legislative enactment. In contrast, the incidence rate of NSIs decreased by 8.3% from 2005 to 2010 and by 16.2% from 2011 to 2015 in Thailand and Korea. 39
Evaluation of the Efficacy of SENDs in Preventing NSIs in Patients With Diabetes
Safety-engineered injection pens with both front- and back-end needle protection have been introduced in many countries across the globe. 15 Active SENDs require deliberate activation of the safety features during device use, whereas passive SENDs require no additional steps by the user (HCW or patient) to activate the safety mechanism. 29 Passive SENDs are of 2 types: (1) semiautomatic SEND, which withdraws the needle into the barrel of the syringe with a push button and (2) automatic SEND, which covers the needle bevel with a safety shield after use. 40 The passive, dual-ended safety technology of IP needles is superior to traditional active safety features for syringes. The dual-ended protection of the passive SEND is intended to cover the portion of the needle in contact with the patient and the back end that penetrates the rubber tip of the pen. 41 When used appropriately, automatic passive SEND eliminate the risk of exposure to bloodborne pathogens because the needle is not exposed outside of the patient’s posture. 40 The Workshop on Injection Safety in Endocrinology consensus group identified the key features of a SEND to reduce the occurrence of NSIs in patients with diabetes (Table 1). 27
Key Features of SENDs as Per WISE Consensus Group Recommendations for Patients With Diabetes.

Source: Strauss. 27
Abbreviations: SENDs, safety-engineered needle devices; WISE, workshop on injection safety in endocrinology; NSIs, needlestick injuries.
In a large, multicenter, observational study conducted in 61 hospitals in France, the passive, fully automatic SEND was associated with lower NSI incidence rates (0.06 NSIs per 100 000 devices [95% CI: 0.01–0.11]) than active devices with the semiautomatic safety feature (1.18 NSIs per 100 000 devices [95% CI: 0.85–1.51]), and manually activated protective sliding (5.20 NSIs per 100 000 devices [95% CI: 4.61–5.78]) or toppling shield (2.94 NSIs per 100 000 devices [95% CI: 2.35–3.53]). 42 The study also reported that NSIs with injection needles and/or syringes amounted to 2.71 NSIs per 100 000 devices vs. 0 NSIs per 100,000 IPs with safety needles. 42 Similar results were observed in Japanese HCWs, wherein the introduction of safety-engineered pen needles for insulin injection decreased NSIs from 0.33 to 0.20 cases per month. 43 Yakushiji et al 44 highlighted that IP needles with automatic recapping capabilities resulted in a significant drop in the incidence of inadvertent punctures while administering insulin to patients with diabetes in Japan (P < .001).
Injection of insulin into the SC tissue is recommended for optimal absorption. 45 In hospital settings, IV insulin may be ordered. Intravenous infusion is preferred in critical care (medical/surgical), perioperative inpatient settings, and patients whose glycemia is poorly controlled with SC insulin. 46 During SC insulin therapy, accidental intramuscular (IM) injections may increase pain and/or adversely affect glucose control in patients with diabetes. 45 This can be managed by lifting a skin fold and/or the use of shorter needles. The lifting of a skin fold puts the HCW at risk of a “through-and-through” NSI, especially when a longer needle (12.7 mm) is used. 27 An evaluation of skin thickness and SC adipose layer thickness by ultrasound was conducted by Gibney et al 45 in adult patients with diabetes with a range of adiposity (sites: arm, thigh, buttocks, and abdomen) and diverse demographic characteristics (Caucasian, Asian, Black, and Hispanic). The mean skin thickness ranged from 1.87 mm in the thigh to 2.41 mm in the buttocks of the subjects. The mean SC adipose layer thickness ranged from 10.35 mm in the thigh to 15.45 mm in the buttocks. 45 The study estimated that, with 5-mm needles, >98% of 90° insertions were SC. 45 In fact, the use of a shorter length, for instance, a 4-mm needle (90° to the skin surface, with no skin pinch up), was estimated to deliver insulin subcutaneously in patients with diabetes more than 99.5% of the time. 45 The usage of 6-mm and 8-mm needles resulted in >5% and 15% IM delivery, respectively. 45 In 2015, experts from 54 countries at the Forum for Injection Technique and Therapy: Expert Recommendations workshop formulated “New Insulin Delivery Recommendations” for the management of patients with diabetes. 47 A key recommendation was that the shortest needles (4-mm pen and 6-mm syringe needles) are safe, effective, and less painful and should be preferred in all patients with diabetes. 47
There is a scarcity of studies that assess the economic benefits of introducing IP needles with automatic dual-ended safety technology in hospital settings from an Asian perspective. Glenngård and Persson 48 assessed the potential benefits of introducing a safety device with the needle and syringe in Swedish hospitals. The study highlighted the benefits of introducing safety devices in terms of economic cost offsets (investigation and treatment of NSIs). 48 In a prospective 9-month (SANITHY) study to assess the cost-benefit of IP devices and safety needles as compared to vial/syringes in hospitalized patients requiring insulin therapy in a general hospital in Northern Italy, IPs had a higher median direct cost than traditional syringes (43 vs. 18 €/patient [~$50 vs. $21]; P < .0001) and safety syringes (21.5 €/patient [~$25], P < .0001). 49 Nevertheless, when indirect and injury costs were taken into consideration, the estimated savings for using IP with dual-ended safety needles over traditional syringes were significantly as high as 32 €/patient (45.8 vs. 77.6 €/patient [~$53 vs. $90]; P < .0001). 49 Ermakova and Taylor 50 performed a budget impact analysis of introducing 5-mm, dual-ended safety pen needles for insulin injections in hospital settings (estimated 74 141 insulin injections for a 500-bed hospital) relative to long, single-ended safety pen needles. The total annual cost of insulin injections with long, single-ended safety pen needles was estimated to be $58 066 (device acquisition: $22 242; NSIs: $5165; hypoglycemia: $30 659). 50 The model estimated a total annual cost of $36 970 to be associated with 5-mm, dual-ended safety pen needles, which represents cost savings of $21 096. 50
Strategies to Prevent NSIs in Patients With Diabetes and HCWs
Tarigan et al 51 assessed the effectiveness of 3 different prevention interventions for NSI prevention: (1) training alone, (2) safety-engineered devices alone, and (3) a combination of training and safety-engineered devices. While training intervention can reduce the risk of NSIs (34%), the combination of safety training and the use of safety-engineered devices (62%) can offer greater protective effects. 51 The CDC emphasizes the use of broad-based, multicomponent prevention approaches to reduce NSIs among HCWs and patients. The recommended components include education on optimal injection techniques, use of SENDs, appropriate sharps disposal, and adequate staff-to-patient ratios. 52 Attention must be paid to the proper use of active safety devices in terms of activation and adequate training should be ensured. 27 The habit of slanting the injection needle overly should be circumvented. 53 The CDC guidelines also emphasize the importance of a “safety culture” while recognizing the need for institution-specific occupational health and safety programs. 52 Steps should be taken in standardizing supplies to include SENDs and fostering awareness of best practices in sharps handling and disposal. Economic considerations are of utmost importance when initiating any change in clinical practices. It is important to revise educational approaches to include interactive learning opportunities and skill validation for newly hired HCWs and patients. Studies imply that the preliminary purchase costs of safety injection devices may be greater than conventional ones, but the substantial reductions in NSI incidence and other complications offset (reduced risk of hypoglycemia), and can often recover, these costs. Standardization of SC insulin administration practices and products, strategic partnerships, in-house cost-benefit analysis, education/training, and implementation and audit of correct injection procedures are some of the strategies that can be employed to increase the acceptance of safety-engineered needles among HCWs and hospitals. Figure 3 provides a summary of multicomponent prevention approaches for NSIs and key steps to improve the acceptance of SENDs among HCWs and patients with diabetes.27,29,52,54,55

Multicomponent prevention approaches and key steps in improving the acceptance of SENDs among HCWs and patients with diabetes. Abbreviations: SENDs, safety-engineered needle devices; HCWs, health care workers; NSIs, needlestick injuries.
Conclusion
Evidence from this review emphasizes that the most effective approach for the prevention of NSIs with insulin injections is through administrative and engineering controls. Continuous education on the prevention of NSIs and strict adherence to occupational health and safety policies and protocols, along with the introduction of SENDs may yield significant results. Widespread use of SENDs in health care facilities in the APAC region would require legal enforcement. Mandating the use of SENDs and the adoption of preventive regulatory policies can reduce the risk of NSIs among HCWs. Though the cost of SENDs can be a drawback for some facilities in the APAC region, this may be offset by reduced training requirements and cost savings associated with fewer NSIs and reduced time off work. Lastly, more studies need to be conducted that assess the economic benefits of IP needles with automatic, dual-ended safety technology in hospital settings (acute and long-term diabetes care) from an Asian perspective.
Footnotes
Acknowledgements
Excellent medical writing and editorial assistance was provided by BioQuest Solutions Pte. Ltd, funded by BD.
Abbreviations
APAC, Asia-Pacific; CDC, Centers for Disease Control and Prevention; CI, confidence interval; EU, European Union; HBV, hepatitis B; HCV, hepatitis C; HCWs, health care workers; HIV, human immunodeficiency virus; IM, intramuscular; IP, insulin pen; IV, intravenous; NSI, needlestick injuries; NSPA, needlestick safety and prevention act; PEP, post-exposure prophylaxis; SC, subcutaneous; SEND, safety-engineered needle device; WHO, World Health Organization; WISE, workshop on injection safety in endocrinology.
Author Contributions
Mafauzy Mohamed and Nikhil Tandon: Conceptualization and original draft writing and reviewing. Youngsoon Kim, Irene Kopp, Nagaaki Tanaka, Hiroshige Mikamo, Kevin Friedman, and Shailendra Bajpai: Reviewing and editing of manuscript. All authors provided critical feedback and contributed to data acquisition and interpretation of data and approved the final version for submission.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Shailendra Bajpai and Kevin Friedman are employees and stockholders of BD/embecta (formerly BD Diabetes Care), manufacturer of a range of safety-engineered devices. No other potential conflict of interest relevant to this article was reported by the rest of the authors.
