Abstract
Background:
A basal bolus insulin regimen requires multiple daily insulin injections, which might discourage patient adherence. As a potential solution, a mealtime insulin-delivery system—a 3-day wearable bolus-only patch—was designed to manually administer mealtime insulin discreetly by actuating buttons through clothing, without the need for multiple needle sticks.
Method:
Extensive functional testing of the patch included dose accuracy (from initial fill of the device to empty), pressure-vacuum leak testing, last-dose lockout and occlusion detection (safety alert features that lock the dosing buttons when no insulin is delivered), assessments of insulin drug stability, toxicological risk (including chemical testing), and system biocompatibility.
Results:
Dosing accuracy was 2 units ±10% (with U-100 insulin) over a range of environmental conditions, with ≥95% reliability and confidence. The fluid seal performance and the safety alert features performed with ≥95% reliability and ≥95% confidence. The system met acceptable standards for insulin (U-100 lispro and aspart) stability for its intended 3-day use, in addition to the operational requirements. The toxicological risk assessment and demonstrated biocompatibility suggested that the patch is safe for human use.
Conclusions:
Benchtop performance showed that the bolus-only patch is a safe, accurate, and reliable device for mealtime insulin delivery.
Keywords
Achieving a glycated hemoglobin (HbA1c) target of ≤7% leads to fewer micro- and macrovascular complications in people with type 1 and type 2 diabetes (T1D and T2D).1-5 A recent meta-analysis found that the HbA1c target is most likely achieved with intensive insulin therapy. 6 Currently marketed insulin-delivery devices (syringes and pens) do not adequately address the barriers to the initiation and management of clinically appropriate dosing regimens. 7 Poor adherence to insulin dosing has been reported in up to 40% of long-term and newly starting insulin users with T2D. 8 In a survey of 502 insulin users, over half of respondents intentionally omitted insulin doses because of interference with daily activities, injection pain, and embarrassment. 9 In addition, forgotten insulin supplies often lead to missed insulin doses. 10 Another study demonstrated that 51% of people felt insulin therapy would restrict their lives, which led to a lower adherence to treatment regimens. 11 Patients also report changing their life to avoid extra injections. 12 The use of a syringe is known to pose problems for the elderly and those with visual impairments.13,14 Thus, there is a need for an insulin-delivery device that can reduce usage barriers associated with currently available devices.
With the goal of making bolus insulin delivery easier and more discreet for people with diabetes, Calibra Medical (Wayne, PA) developed a 3-day wearable, bolus-only, mealtime insulin-delivery system. This report describes the benchtop performance of this system, including laboratory verification test data. Validation testing involved simulated use studies on the cognitive and behavioral aspects required for the safe use of the patch in people with diabetes and has been described elsewhere. 15
Methods
The effect of environmental factors on device performance was evaluated using 186 devices. Dose accuracy, fluid seal verification, occlusion detection (OCCD), and last-dose lockout (LDLO) were tested. After sterilization, per American National Standards Institute/the Association for Advancement of Medical Instruments/International Organization for Standardization (ISO) 11135-1:2007 16 and climate and transport preconditioning per American Society for Testing Materials (ASTM) D4169-DC 13, 17 the devices were divided into groups of 46 to be tested under 4 different environmental conditions: hot operation, cold operation, hot soak, and cold soak (Table 1). These groups were further divided into 100-unit (n = 19), 200-unit (n = 19), and 220-unit (n = 8) subgroups, representing minimum, nominal, and maximum fill conditions. The 220-unit group ensures a design margin beyond the nominal 200-unit fill.
Test Sample Preconditioning and Environmental Requirements.
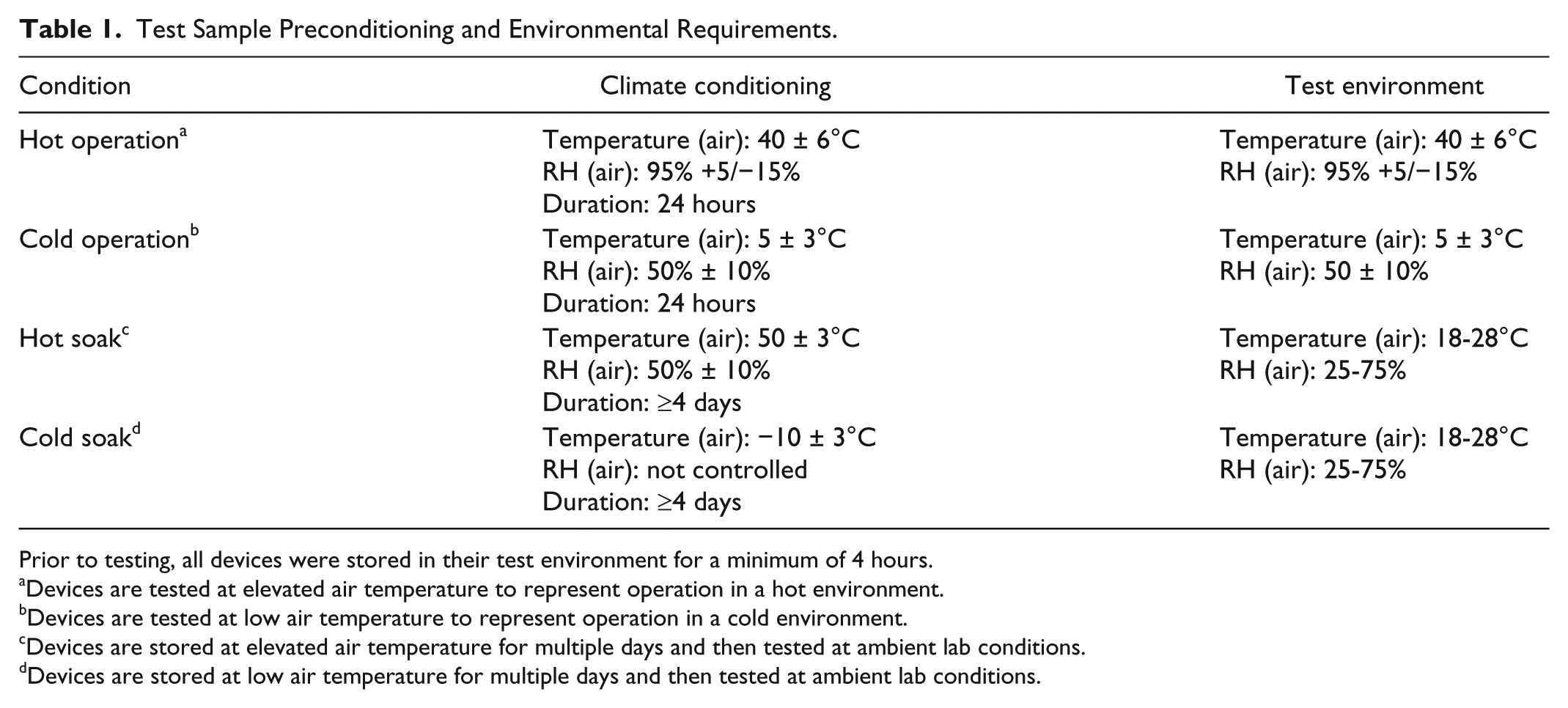
Prior to testing, all devices were stored in their test environment for a minimum of 4 hours.
Devices are tested at elevated air temperature to represent operation in a hot environment.
Devices are tested at low air temperature to represent operation in a cold environment.
Devices are stored at elevated air temperature for multiple days and then tested at ambient lab conditions.
Devices are stored at low air temperature for multiple days and then tested at ambient lab conditions.
Mealtime Insulin-Delivery System
The mealtime insulin-delivery system (Calibra Medical, Wayne, PA, USA) has been classified as a new US Food and Drug Administration (FDA) product code OPP, under 21 CFR 880.5725; approved for use with rapid-acting insulins, U-100 lispro and aspart. The system is labeled for subcutaneous delivery of bolus insulin in adults (>21 years) requiring mealtime insulin.
The system includes a patch, inserter, fill syringe, and needle (Figure 1). The patch consists of an insulin reservoir, a cannula connected by a mechanical pump, and a valve. Sequenced actuation of the pump and valve occurs when the user presses the buttons on the patch to deliver the insulin dose from the reservoir through the cannula into the body (Figure 2). The mechanical timing ensures that the reservoir feature is never directly connected to the user. The insulin reservoir can be filled with up to 200 units of rapid-acting insulin, using the included fill syringe and needle. The patch requires priming to remove air bubbles from the reservoir and fill the internal fluid path with insulin. The inserter is then used to place the patch on the body, inserting the introducer needle and passing the cannula through the skin into the subcutaneous tissue. The needle is removed once the cannula is in place, leaving the patch adhered comfortably to the user’s skin.
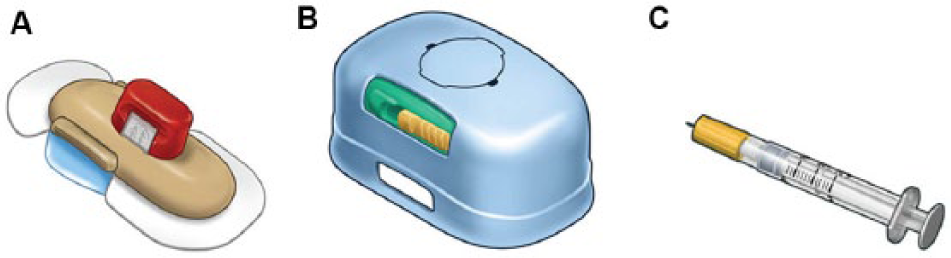
Mealtime insulin-delivery system. The patch is preassembled with a guide for the fill syringe (blue cap) and an introducer needle (red cap) for the cannula (A). The inserter (designed for reuse) places the patch on the body and inserts the needle and cannula (a soft polymer tube) through the skin into the subcutaneous tissue (B). The sterile fill syringe and needle are specially designed for filling the insulin reservoir of the patch (C).
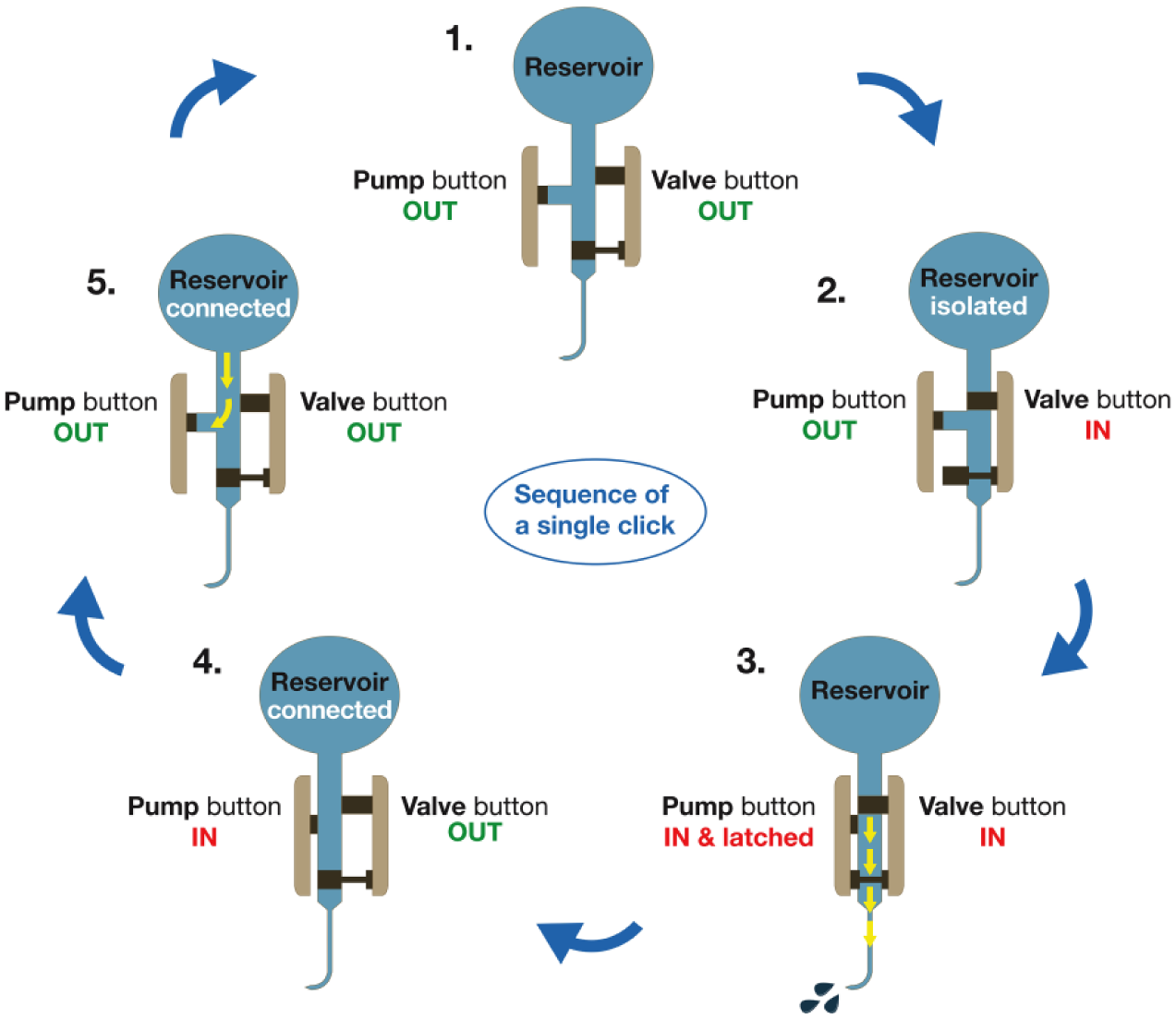
Mechanism by which the mealtime insulin-delivery device delivers a single 2-unit dose of insulin. 1. At the start, both the pump and valve button are in the “OUT” position. 2. Upon squeezing the buttons, the valve button depresses first, disconnecting the insulin reservoir and opening the fluid pathway to the cannula. 3. Only then, the pump button depresses, delivering a 2-unit dose. 4. Upon releasing the buttons, the valve button returns into the “OUT” position, closing the pathway to the cannula and reconnecting the insulin reservoir. 5. Last, the pump button releases and simultaneously draws the next dose from the insulin reservoir. This sequence is repeated for each subsequent dosing.
Each patch can be worn on the body for up to 3 consecutive days (Figure 3). “Change by” (date) stickers help users to remember when their patch requires replacing. The patch is completely mechanical, without a display, memory, or controller, and does not require batteries. It delivers 2 units of rapid-acting insulin with each simultaneous squeeze of the 2 independent buttons located on opposite sides of the patch, ensuring that no insulin is delivered if 1 button is pressed accidentally (Figure 3). Squeezing 2 buttons simultaneously provides the user with direct tactile feedback and an audible “click” sound indicating delivery of a 2-unit dose of insulin. The buttons release in preparation for the next dose of insulin.
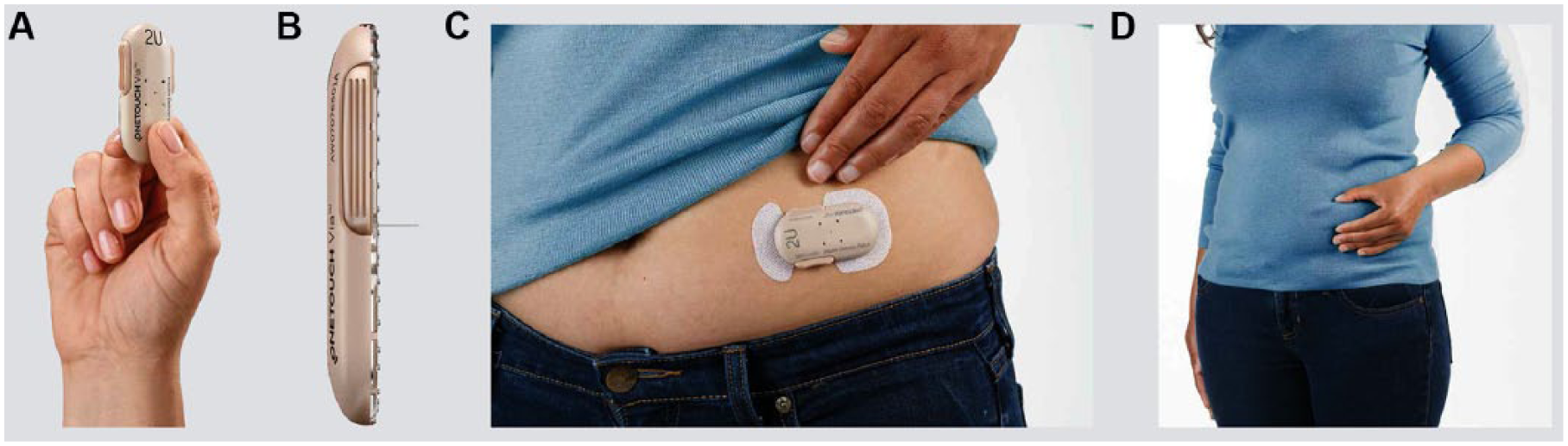
Mealtime insulin-delivery system (Calibra Medical, Wayne, PA, USA). The patch size is 65 × 35 × 8 mm (A). Mealtime insulin is delivered via a soft cannula (B). The patch is worn on the abdomen for up to 3 days (C). Mealtime insulin can be dosed through clothing by actuating the buttons on both sides of the patch (D).
Rapid-Acting Insulins
For design verification and mechanical testing, the rapid-acting insulins U-100 lispro and aspart were purchased from local pharmacies.
Data Analysis
Attribute data were evaluated as “pass” or “fail” and variable data were analyzed to assess the upper and lower bound reliability and confidence for each test specification (Minitab 16, tolerance interval function), requiring ≥95% reliability and ≥95% confidence to pass the test.
Dose Accuracy Verification
The specification for dose accuracy of each 2-unit dose is ±10% with 95% confidence and reliability over the dosing range of the device. Dose accuracy calculates a tolerance interval for the 4 combined environmental conditions based on the confidence and reliability. This tolerance interval must fall within the limits of the dose accuracy specification. A total of 186 devices were tested for dose accuracy using a computer-interfacing control box connected to a pneumatically operated fixture for clicking the device and a calibrated semi-microbalance for weighing each dose delivered. After the initial setup, the system was semiautomated, requiring only that the operator loaded the device into the fixture, selected a data-storage location on the linked computer, and ran the LabVIEW (National Instruments) dose accuracy program. The program executed the test and gathered data by actuating the device buttons and subsequently weighing each dose until the device was empty and the insulin-delivery buttons locked; the volume was calculated from the measured weight of each dose. Dose accuracy was measured from initial fill of the device to empty, excluding the first and last doses locking out the patch.
Seal Verification
Fluidic seal closure was tested by injecting air into the device to generate positive pressure (1.25 ± 0.25 psi) and then drawing the air out to generate a vacuum (−1.25 ± 0.25 psi) while monitoring for leaks over 20 seconds. The sealed fluid paths were accessed at the cannula opening and from the fill septum. Testing the device from these 2 locations, with the buttons first in the “in” and then in the “out” position, allowed access to all fluid-seal surfaces.
Alert/Alarm Feature Verification
The OCCD alerts the user that no insulin has been delivered when pressure increases in the patch during dosing due to a cannula occlusion. The insulin-delivery buttons will be locked in the fully depressed position, indicating that there is an occlusion. To test the OCCD feature, devices were pressurized from the cannula tip in increments of 2 psi until the buttons locked when actuated. To meet its requirement, the OCCD feature must lock the buttons at a pressure range of 10-40 psi, which is representative of a cannula occlusion.
The LDLO feature has been designed to lock the buttons in the fully depressed position after the last full dose of insulin is delivered. The LDLO is triggered by a low-pressure/empty drug reservoir. In the LDLO test, dose accuracy for devices was analyzed until the insulin-delivery buttons locked, excluding the first and last doses. Data for the dose prior to the lockout dose were statistically analyzed to confirm that 2 units ±10% of insulin was delivered with ≥95% reliability and confidence.
Insulin Stability and Chemical Testing
For insulin-stability tests, the devices were filled with insulin lispro or aspart and kept at 37 ± 2°C with gentle agitation to simulate conditions under wear. After 0, 3, or 6 days, the insulin was removed from the device and prepared for drug product analysis. Control samples of each insulin were left in their original container and incubated under the same conditions. Both insulins were tested in triplicate with 3 devices per time point. For testing, all insulin was extracted from the device through the flow path by pressing the buttons. Days 0 and 3 were tested to ensure drug stability and safety in the device over its approved wear time; an additional analysis was done at Day 6 to ensure a margin of safety (MOS) as per FDA regulations.
The stability of insulins lispro and aspart was tested following US Pharmacopeia (USP) tests and monographs, or validated WuXi AppTec (St. Paul, MN, and Marietta, GA) methods.18 -27 Standards used for insulin-stability testing are listed in detail in Table 2.
Summary of Chemical and Insulin Stability.
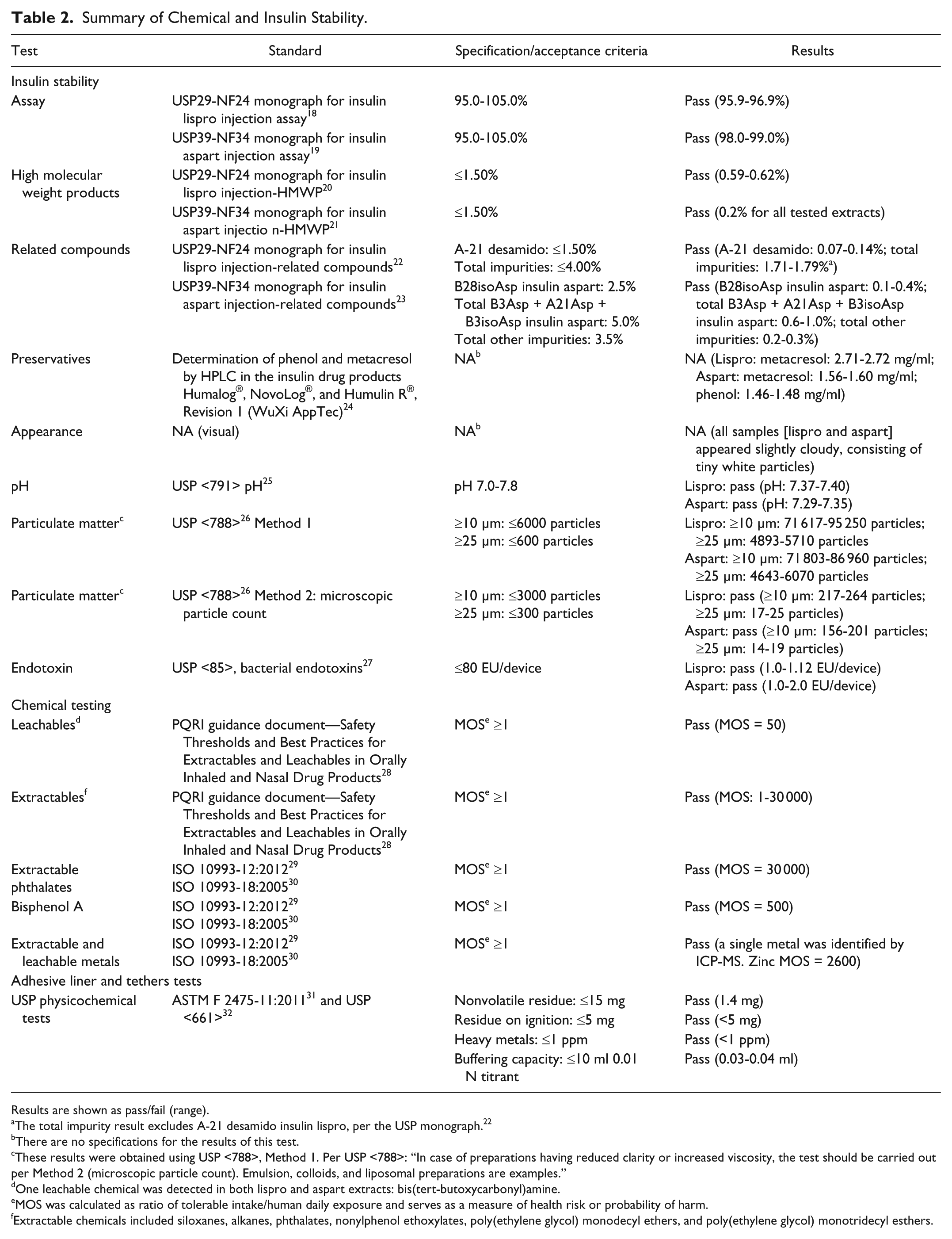
Results are shown as pass/fail (range).
The total impurity result excludes A-21 desamido insulin lispro, per the USP monograph. 22
There are no specifications for the results of this test.
These results were obtained using USP <788>, Method 1. Per USP <788>: “In case of preparations having reduced clarity or increased viscosity, the test should be carried out per Method 2 (microscopic particle count). Emulsion, colloids, and liposomal preparations are examples.”
One leachable chemical was detected in both lispro and aspart extracts: bis(tert-butoxycarbonyl)amine.
MOS was calculated as ratio of tolerable intake/human daily exposure and serves as a measure of health risk or probability of harm.
Extractable chemicals included siloxanes, alkanes, phthalates, nonylphenol ethoxylates, poly(ethylene glycol) monodecyl ethers, and poly(ethylene glycol) monotridecyl esthers.
In addition, a toxicological risk assessment for all extracts was done per current ISO guidelines28-30 to assess potential health risks posed by extractable and leachable chemicals from the patch. Device extractions for chemical tests were performed under various chemical conditions, solvent polarities, and pH (ethanol, isopropyl alcohol/water, 5% HNO3, acetonitrile, and water) and analyzed by gas chromatography-mass spectrometry (MS), liquid chromatography-MS, and inductively coupled plasma (ICP)-MS in accordance with ISO 10993-12:2012 29 and ISO 10993-18:2005. 30 For the toxicological risk assessment, 33 chemicals of potential concern were identified, tolerable intake (TI [µg/kg/day]) was established, the human daily exposure (HDE [µg/kg/day]) representative of worst-case exposure was estimated, and MOS calculated (Table 2).
All testing was performed at approved laboratories (WuXi AppTec, St. Paul, MN, Marietta, GA, and Nelson Laboratories, Inc, Salt Lake City, UT).
Adhesive Liner and Tethers
Chemical packaging tests on the adhesive liner and tether parts were done following ASTM F 2475-11 31 and USP <661> 32 for level 2 packaging materials where physicochemical tests are required. The adhesive liner was tested for buffering capacity and heavy metals, and the tethers for buffering capacity, heavy metals, nonvolatile residue, and residue on ignition (Table 2).
Biocompatibility
A biological safety evaluation of the materials used in the inserter, patch, syringe, adhesive liner, and tethers was conducted by Nelson Laboratories, Inc. (Salt Lake City, UT) and biocompatibility tests in accordance with the corresponding sections of ISO 1099334-42 were conducted by WuXi AppTec (Table 3).
Summary of Biocompatibility Testing.
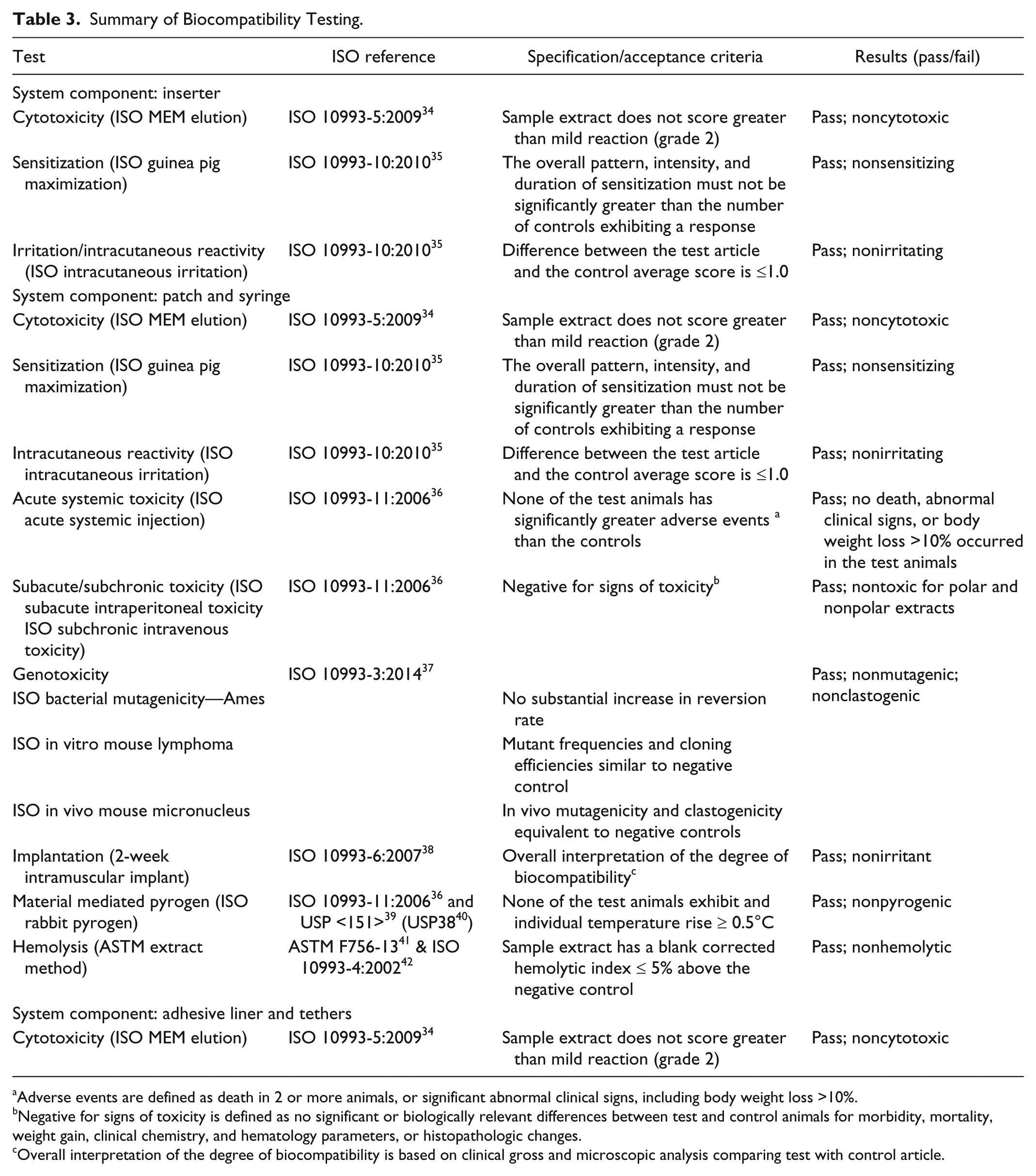
Adverse events are defined as death in 2 or more animals, or significant abnormal clinical signs, including body weight loss >10%.
Negative for signs of toxicity is defined as no significant or biologically relevant differences between test and control animals for morbidity, mortality, weight gain, clinical chemistry, and hematology parameters, or histopathologic changes.
Overall interpretation of the degree of biocompatibility is based on clinical gross and microscopic analysis comparing test with control article.
Results
Dose Accuracy Verification
Dosing accuracy of the patch was well within the specified criteria of ±10% per 2-unit dose administration with ≥95% reliability and confidence (Figure 4). Dosing accuracy is maintained over the device’s dosing range for a 200-unit fill (Figure 4). Dose-accuracy requirements were met under all environmental conditions (Table 4) that included 12 737 data points. The first dose of a new 2-unit patch is nominally 1.7 units, as 0.3 units are used to fill the cannula after the insertion needle is removed. The last dose of the patch when the LDLO alarm is activated might also be <2 units.
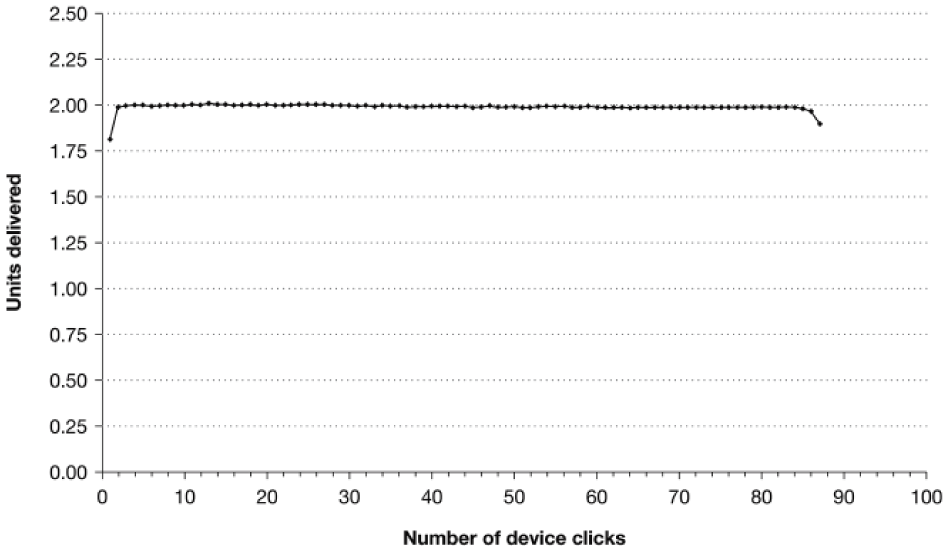
Representative dose accuracy of a sample patch. Example of dose accuracy of a single mealtime insulin-delivery-system patch filled with NovoLog® insulin in the ambient laboratory environment after one 24-hour cold-soak condition. Dose accuracy of the patch delivering bolus insulin in 2-unit increments is shown. When a patch is filled, 0.3 units will be needed to fill the dead space in the cannula after the insertion needle is removed. Thus, the first dose will be 1.7 units. Similarly, the final dose will be short, as approximately 0.3 units will be left in the cannula.
Summary of Dose Accuracy Testing.
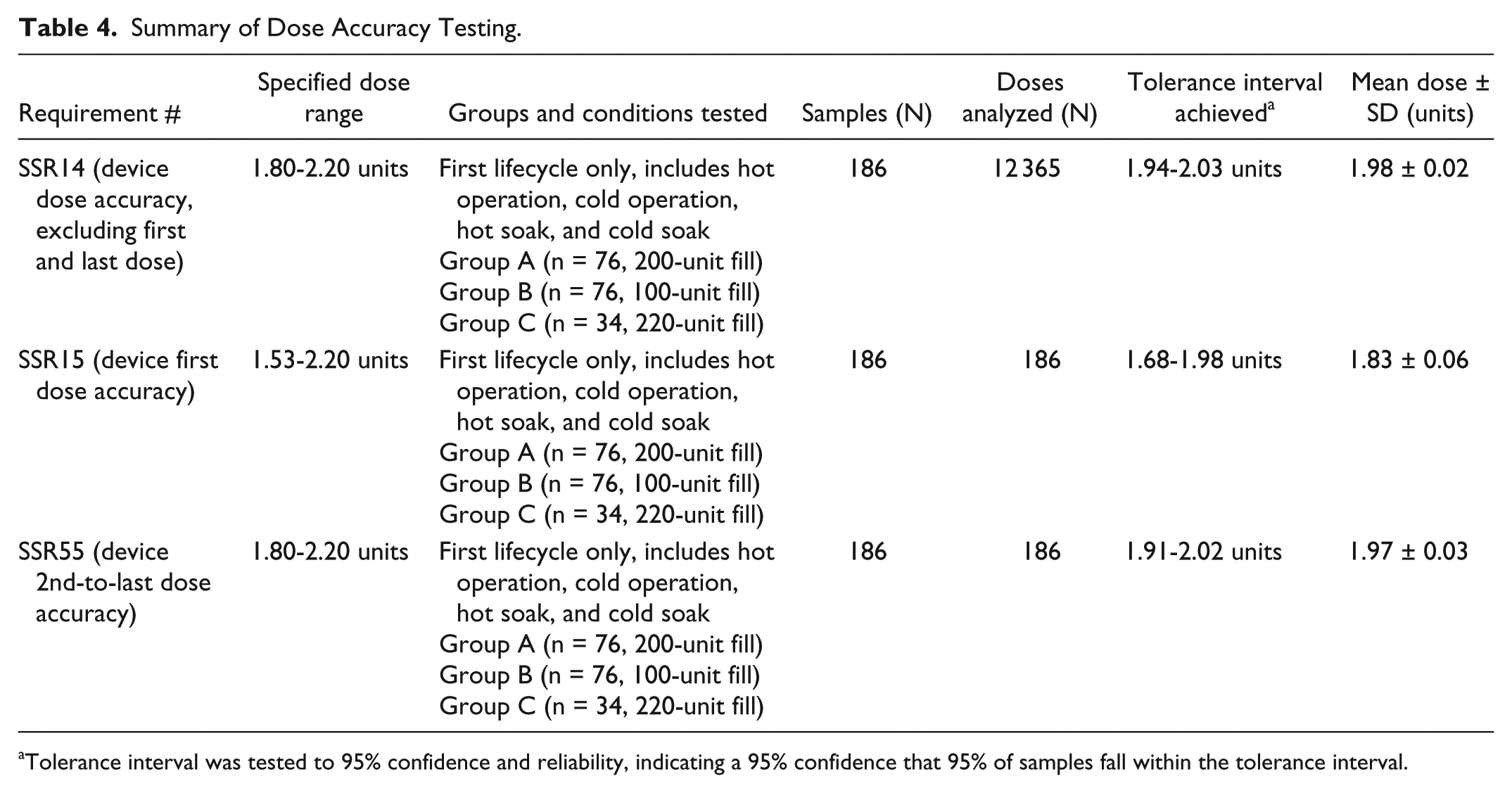
Tolerance interval was tested to 95% confidence and reliability, indicating a 95% confidence that 95% of samples fall within the tolerance interval.
Under all environmental conditions, the device delivered a penultimate dose prior to the lockout dose that passed the tolerance interval analysis for 2 units ±10% with ≥95% reliability and confidence.
Seal Verification
All devices (186/186) passed pressure-vacuum leak testing under all environmental conditions, with fluidic seal performance reaching ≥95% reliability and confidence.
Alert Feature Verification
The patch met the requirements for OCCD under all environmental conditions. All devices tested (186/186) locked the buttons at >10 psi and <40 psi, with ≥95% reliability and confidence.
All devices tested (186/186) achieved LDLO during the lockout dose under all environmental conditions, demonstrating ≥95% reliability and confidence.
Insulin Stability and Chemical Testing
Insulin stability acceptance criteria and results for the required tests at Day 6 are listed in Table 2. Both rapid-acting insulins met all the specifications of identity, strength, purity, stability, and chemical composition for up to 6 days in the patch.
No changes in pH were detected in any of the extracts at any time point measured, suggesting chemical and physical stability of the insulins.
Preservatives such as phenol and metacresol act as antimicrobial and stabilizing agents in the insulin formulations, and were detected in the tested samples at all time points.
Related compounds such as A-21 desamido, B28isoAsp insulin aspart, total B3Asp+A21Asp+B3isoAsp insulin aspart were well below the allowed threshold.
A visual control of the appearance of the extracted insulin samples showed that all samples, for both lispro and aspart, appeared slightly cloudy due to tiny white particles. Particulate matter in the insulin extracts from the devices was determined by microscopic counting. 26 All samples passed the criteria for parenteral drug preparations, with the number of particles at Day 6 not exceeding 264 particles ≥10 μm in size or 25 particles ≥25 μm in size per container. In addition, extracts for both insulins tested well below the required threshold of ≤1.5% for high-molecular-weight particles (HMWP) at all 3 time points.
Bacterial endotoxins can cause febrile reactions. Therefore, the devices were tested for bacterial endotoxins per USP <85>. 27 All tested extracts showed ≤2 endotoxin units (EU) per device, indicating the devices were safe for use.
For the toxicological risk assessment, the TI:HDE ratio (available dose) of leachable and extractable substances was used to calculate the MOS. 33 For adult exposure to the device, all extractable and leachable chemicals identified possessed calculated values for MOS ≥1, indicating a negligible toxicological risk (Table 2).
Adhesive Liner and Tethers
The adhesive liner and tethers passed specifications for cytotoxicity and physicochemical testing as defined in ASTM F 2475-11:2011 31 and USP <661> 32 and are thus considered noncytotoxic. Tests and results are listed in Table 2.
Biocompatibility
No hazards were identified in the biocompatibility assessment based on the chemical tests of the device materials; therefore, biocompatibility of the system was tested as outlined in ISO 10993. All materials that come in contact with the patient met defined requirements. Tests and results are detailed in Table 3.
Together, results from available toxicity data, calculated MOS, and demonstrated biocompatibility indicate that the use of the patch is safe and that, even with prolonged contact (≥30 days), the likelihood that extractable and leachable chemicals from the patch would produce unacceptable health risks in the adult population is negligible.
Discussion
The mealtime insulin-delivery system met all the acceptance criteria as outlined in the Product Quality Research Institute (PQRI) guidance and ISO 10993 standards. Results from the different environmental conditions indicate that the insulin in the patch is stable for the intended 3-day period of use and remains at an operational temperature at 5-40°C. The patch, while worn on the body, may be exposed to various environments from hot and humid to freezing cold and is water resistant for showering, bathing, and swimming, meeting an IPX7 water-resistant rating (data not shown). When nonoperational, the patch should be stored at 10-25°C and should be at room temperature when filled with insulin immediately prior to application. It is not refillable after application, nor once it has been removed.
Maintenance of glycemic control is affected by accuracy of insulin dosing. The patch consistently delivered an accurate 2-unit dose (±10%) of insulin with each actuation of the delivery buttons. Actuation of both buttons is required to deliver a full 2-unit dose of insulin and, therefore, partial dosing cannot occur. Both the tactile feel of the buttons (over or under clothing) being depressed as well as an audible click ensures the user that a 2-unit dose was delivered. The OCCD safety feature on the patch alerts the user if no insulin is delivered. User validation 43 demonstrated equivalent mean daily blood glucose while using the patch compared with using a pen/syringe.
The patch was designed to overcome many of the design drawbacks of insulin pens, and syringes and vials, for accurate bolus dosing. It has been shown that 20% of adult users do not self-administer the correct dose of insulin with pens or syringes. 44 Pens must be prepared correctly prior to use to ensure accurate dosing—a step often omitted by patients. 45 In addition, pens and syringes require the patient to hold the needle in place in the subcutaneous tissue during injection for accurate insulin delivery. Since the patch cannula is already in the subcutaneous tissue for the duration of wear, the user only needs to actuate the device the appropriate number of times to ensure the proper dose has been given. For a 3-day wear, the patch provides on average 20 units of insulin per meal with a 200-unit fill. With the patch, a reminder card on dose counting is provided to help the user with the counting technique recommended for insulin administration. Patient usability studies have shown the effectiveness of this approach to minimizing potential errors in dose counting. 15 Participants (including 50% age ≥55 years) across 90 simulated trials (4, 12, and 18 units) with a number of potential distractions successfully administered the correct dose 100% of the time (data on file). Insulin pumps with electronic controls can deliver a more accurate dose of insulin than injection devices such as syringes and pens. 46 However, the low pressure in these systems makes them susceptible to cannula/tubing blockages. The patch has a built-in alert feature to inform the patient of a possible blockage by locking the insulin-delivery buttons.
The patch has been designed to alleviate some of the concerns of people with diabetes regarding the delivery of bolus insulin. Compared with a pen or syringe and vial, the patch does not require people to stick themselves with a needle multiple times daily and thereby alleviates the need to plan their day around injections. Because patients wear the patch on their body, they do not need to carry any additional insulin-delivery supplies. In comparison with pumps, the patch is smaller, easier to use, more discreet, and costs less. In addition, the pump requires switching from multiple daily injection therapy to continuous subcutaneous insulin delivery. The patch does not provide any basal insulin therapy option and patients who would benefit from basal insulin treatment would need an additional daily injection or use a pump instead.
This performance study did not assess objective glucose control or perceived clinical efficacy. However, in people with T1D or T2D, the patch demonstrated comparable efficacy to a pen or syringe and vial, and was associated with significant higher device satisfaction and improved quality of life. 43 In addition, the safety profile of this patch has been shown to be similar to currently available injection devices for mealtime insulin, including insertion site reactions for irritation (erythema or papular reaction), edema, or ecchymosis. 43 Provider-related barriers to initiating mealtime insulin can derive from providers’ concerns about their patients’ abilities or willingness to adopt an intensive insulin therapy regimen. 47 In a feasibility study, health care providers preferred the patch over pens or syringes for their patients, 48 which may aid in decreasing such barriers. The health care provider should assess the patient’s suitability to wear the patch and use the patch accurately and safely.
Conclusion
Under all environmental conditions assessed, the patch passed pressure-vacuum leak testing and OCCD testing, and achieved LDLO, with ≥95% reliability and confidence. Materials used for the patch passed insulin stability and chemical testing as well as biocompatibility testing, providing objective evidence that the patch is reliable for the intended users and environments. This reliability is supported by efficacy and safety evidence from a clinical trial. 43
The patch offers accurate dosing, an alert feature when no insulin is delivered, and discreet use in public places (with actuation of the device possible through clothing). This simple, wearable device may improve adherence to an insulin regimen involving multiple bolus doses, especially for people with T2D who initiate insulin later in life. 48 In light of the rising diabetes epidemic, solutions that enable glycemic control improvement have the potential to contribute to better long-term outcomes and reduce costs of care.49 -52
Footnotes
Acknowledgements
Editorial support in the preparation of this manuscript was provided by Martina Fuchsberger, PhD, of Excerpta Medica, funded by Calibra Medical.
Abbreviations
ASTM, American Society for Testing Materials; CFR, Code of Federal Regulations; EU, endotoxin units; FDA, US Food and Drug Administration; HbA1c, glycated hemoglobin; HDE, human daily exposure; HMWP, high-molecular-weight particles; HPLC, high-performance liquid chromatography; ICP-MS, inductively coupled plasma mass spectrometry; ISO, International Organization for Standardization; LC-MS, liquid chromatography-mass spectrometry; LDLO, last-dose lockout; MEM, minimum essential medium; MOS, margin of safety; NA, not applicable; NF, National Formulary; OCCD, occlusion detection; PQRI, Product Quality Research Institute; RH, relative humidity; SD, standard deviation; SSR, subsystem requirement; T1D, type 1 diabetes; T2D, type 2 diabetes; TI, tolerable intake; U, units; USP, US Pharmacopeia.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DMD, BJC, NSM, JHN, and JEN are full-time employees of Calibra Medical. TMH, BC, and PAM are former full-time employees of Calibra Medical. AT has received contracting fees from Calibra Medical. NM is a full-time employee of LifeScan, LLC. MHT has received consulting fees from Calibra Medical.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Calibra Medical. The authors received no financial support for the development of this manuscript.
