Abstract
The global burden of diabetes continues to rise, resulting in a concomitant increase in the global demand for insulin replacement therapies. Subcutaneous insulin administration using pen injectors is one of the most common insulin delivery methods; these devices are often preferred over vial and syringe administration by individuals with diabetes owing to their convenience and ease of use, and by clinicians owing to enhanced user adherence and high dose accuracy. However, prefilled insulin pen injectors are commonly made of plastic and many are disposable; consequently, both their production and use are associated with a high carbon footprint and substantial medical waste through poor recycling infrastructures, posing a growing environmental concern. Historically, producing durable insulin pen injectors required high-quality metal components, which are more environmentally taxing despite being reusable. Conversely, plastic has a lower environmental footprint per unit but conventional plastic pen injectors have lacked durability, leading to greater consumption overall. Therefore, reusable insulin pen injectors that can be used repeatedly for several years, while also being easy to use and accurate, are essential for sustainable diabetes management. Novo Nordisk has developed several insulin pen injectors, such as NovoPen (NP) 4 (a reusable pen part of the NP family), FlexTouch (a disposable pen) and, more recently, DuraTouch (DT; a reusable pen). Here, we describe the technical attributes of DT using other pen injectors manufactured by Novo Nordisk as contextual comparators to illustrate how DT may meet users’ functional needs, while helping to reduce the environmental burden associated with the use of insulin pen injectors.
Introduction
Diabetes affects an estimated 540 million individuals worldwide as of 2025, with type 2 diabetes comprising more than 90% of cases.1,2 By 2050, the global prevalence is projected to reach 853 million. 1 Globally, an estimated 150 to 200 million individuals with diabetes rely on insulin replacement therapy. 3
Subcutaneous administration methods, including vials and syringes, pen injectors and continuous subcutaneous insulin infusion (CSII) pumps remain the most widely used and effective methods for insulin delivery.4-8 While alternative insulin administration routes, such as inhaled insulin, are available and have demonstrated clinical efficacy, their uptake remains limited.6,9,10 Moreover, despite increasing use of CSII pumps over the years, particularly in individuals with type 1 diabetes, insulin pen injectors are widely used owing to their affordability, ease of use and low technical burden, although access may vary depending on the health care system, insurance coverage and individual needs. 11 In some regions, limited affordability and access to appropriate delivery devices remain barriers to optimize diabetes care, highlighting the value of cost-effective, reusable options that accommodate the needs of diverse users.12,13 Compared with vial and syringe administration, pen injectors offer greater dosing accuracy, reduced injection pain and improved ease of use, flexibility and convenience, all of which can contribute to better treatment adherence.4,5,14,15 This is of clinical importance because suboptimal adherence to insulin therapy is associated with poorer glycemic outcomes, increased risk of developing diabetes-related complications and greater health care resource utilization. 16 Improving insulin pen injector usability may help to alleviate comfort-related barriers and support better long-term clinical outcomes 16 ; however, the need for multiple daily injections can still pose a burden, particularly for individuals who struggle with injection force or handling pen injector devices. 17
To enhance usability and support adherence, insulin pen injectors have undergone continuous development, incorporating features such as non-extending dose buttons for easier handling, larger dose displays for improved readability and lower injection force requirements to reduce injection discomfort.17,18 Ensuring that insulin pen injectors are intuitive, comfortable to use and adaptable to individuals with varying needs and different insulin regimens is essential for optimizing glycemic management and treatment outcomes.8,14 Importantly, these insulin pen injector features may support treatment adherence among individuals with dexterity limitations, visual impairments or cognitive challenges, highlighting the role of insulin pen injector design in supporting inclusive diabetes care.17,18
Beyond usability, the environmental impact of insulin pen injectors is an emerging concern, particularly regarding the waste generated by disposable pen injectors. Given the rising prevalence of diabetes, efforts to minimize the environmental burden of insulin pen injectors have become a global priority. 12 Insulin pen injectors can be broadly categorized as either disposable or reusable. 19 Disposable pen injectors come prefilled with insulin and are used multiple times, typically over the course of a week, before being discarded, contributing to plastic waste and carbon emissions. In contrast, reusable pen injectors are designed for long-term use with replaceable insulin cartridges, reducing medical waste (eg, volume of discarded disposable pen injectors versus discarded cartridges) and the environmental impact. 17 With the number of insulin users continuing to grow worldwide,1,2 the increasing demand for insulin pen injectors poses a long-term sustainability challenge.20,21
Novo Nordisk introduced its first insulin pen injector in 1985 and has since developed a range of reusable and prefilled disposable insulin pen injectors with various attributes designed to meet the diverse needs of users. 18 Among these, the reusable NovoPen (NP) 4 and the disposable FlexTouch (FT) serve as key predecessors to DuraTouch (DT), a reusable insulin pen injector developed to balance usability with sustainability. In this commentary, we focus on these three Novo Nordisk insulin pen injectors, summarizing their key attributes and highlighting how DT compares with its predecessors in terms of usability while also describing its potential impact on sustainability.
Comparing Key Attributes of Insulin Pen Injectors
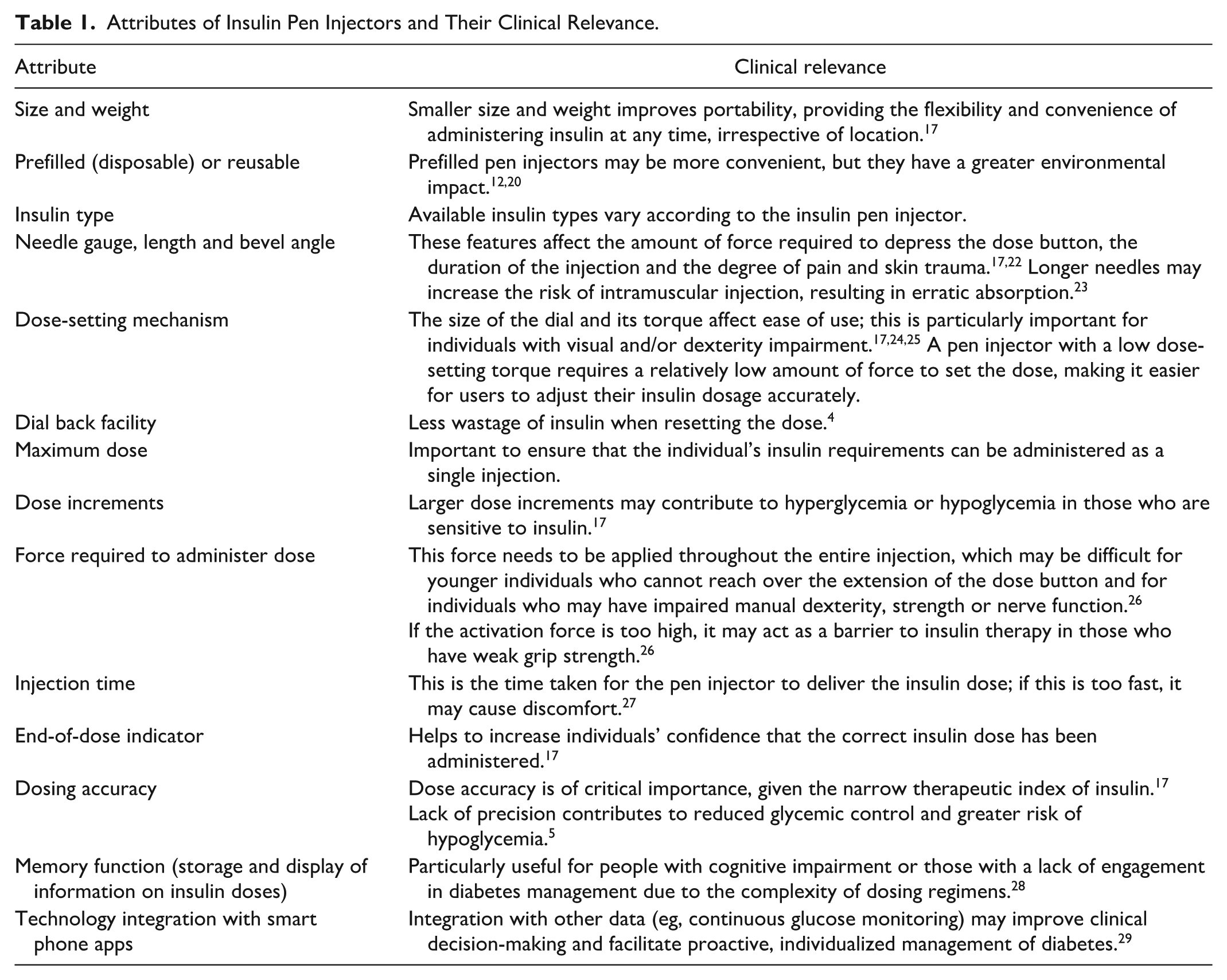
Design and functionality of insulin pen injectors play a critical role in ensuring ease of use, dosing accuracy and adherence to insulin therapy. The key attributes of insulin pen injectors and their clinical relevance are summarized in Table 1.
Attributes of Insulin Pen Injectors and Their Clinical Relevance.
Several surveys conducted among individuals with diabetes showed that insulin pen injectors should be simple to use, easy to learn and teach, and provide fast injections with minimal pain.30-33 In addition, studies have highlighted the importance of accurate and reliable insulin delivery, as well as minimal physical effort during dose administration.17,24,25,28,30,33 The force required to administer a dose is another key factor because excessive force may discourage proper use of insulin pen injectors. 24 Furthermore, the dose-setting mechanism, dial torque and injection time influence usability, particularly for individuals requiring high-dose insulin regimens.17,25 To ensure consistent insulin delivery and usability, international regulatory bodies have established strict performance criteria. 23 The International Organization for Standardization (ISO) 11608-1 defines requirements for needle-based injection systems, including insulin pen injectors, covering aspects such as dose accuracy across different conditions and mechanical robustness. 23 Beyond usability, the choice of insulin pen injector should be tailored to individual needs, considering self-management capabilities, insulin type and dose requirements. 17 A well-designed insulin pen injector should accommodate a range of user preferences, ensuring ease of use across diverse populations. 17 For example, the Novo Nordisk insulin pen injectors differ in design and have varying attributes that influence their suitability for different users. 18 Insulin pen injector design differences, when integrated as part of a tailored treatment approach, can influence clinical outcomes by affecting how confidently and consistently individuals administer their insulin doses. 18 Choosing a device suited to an individual’s dexterity, vision and dosing needs may support individuals with achieving glycemic control.34,35
In addition to the Novo Nordisk reusable pen injectors, other reusable insulin pen injectors are available, including AllStar (Sanofi, Paris, France), TouStar (Sanofi) and HumaPen (Eli Lilly, Indianapolis, USA), which offer alternative design features, for example, differing maximum doses, total capacity and device mass, and may meet different user needs.4,36 While detailed data for comparative assessments with DT were not available for these devices at the time of writing, their availability highlights the range of reusable insulin pen injector options currently on the market.
NP 4
NP 4 (Novo Nordisk A/S, Bagsvaerd, Denmark), introduced in 2005, is a widely used reusable pen injector comprising a disposable insulin cartridge and a syringe in a single device, capable of administering insulin doses up to 60 U in 1 U increments (Figure 1a).19,37 It was designed to incorporate a number of features intended to improve the user experience compared with that of earlier pen injectors in the NP family, including a larger (more than four times) dose scale to improve ease of reading, a reduced injection force (~50%), compatibility with a range of needles (eg, NovoFine [NF] and NovoTwist [NT] single-use needles) and an audible end-of-dose click. 38 The dosing accuracy of NP 4 was evaluated across a range of conditions, including different insulin doses, temperature extremes, high humidity and mechanical stress. 39 In addition, its long-term accuracy was tested over 5475 simulated injections, equivalent to three daily injections for five years. 39 Across all conditions, NP 4 fulfilled the ISO 11608-1 requirements for needle-based injection systems, demonstrating consistent dosing accuracy (Table 2),23,39 and remains compliant with the current standard.

(a) NP 4, (b) FT, and (c) DT pen injectors.
Key Attributes of DT, FT and NP 4.
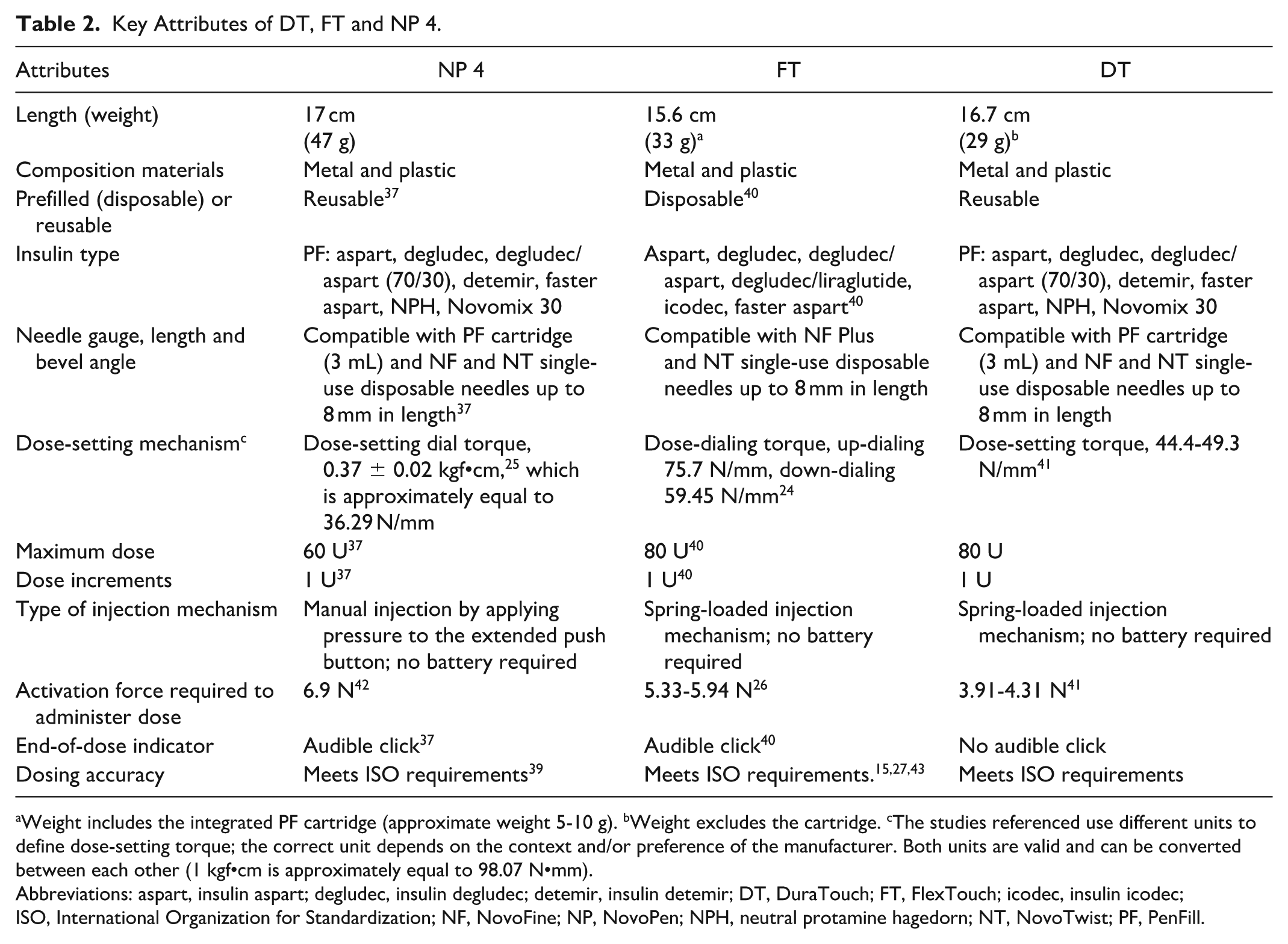
Weight includes the integrated PF cartridge (approximate weight 5-10 g). bWeight excludes the cartridge. cThe studies referenced use different units to define dose-setting torque; the correct unit depends on the context and/or preference of the manufacturer. Both units are valid and can be converted between each other (1 kgf•cm is approximately equal to 98.07 N•mm).
Abbreviations: aspart, insulin aspart; degludec, insulin degludec; detemir, insulin detemir; DT, DuraTouch; FT, FlexTouch; icodec, insulin icodec; ISO, International Organization for Standardization; NF, NovoFine; NP, NovoPen; NPH, neutral protamine hagedorn; NT, NovoTwist; PF, PenFill.
FT
FT (Novo Nordisk A/S, Bagsvaerd, Denmark) (Figure 1b), introduced in 2011, is a disposable insulin pen injector designed to enhance user-friendliness. It delivers insulin doses up to 80 U in 1 U increments. 40 It shares several attributes with NP 4, such as ease of handling, compatibility with a range of needles (eg, NF and NT single-use needles) and accurate dosing, as well as an audible end-of-dose click. In addition, FT incorporates a non-extending dose button, reducing the required injection force, a design particularly beneficial for individuals who might struggle with the force needed to use traditional insulin pen injectors.15,19,26 FT uses an internal spring mechanism to preload the activation force when the dose is set, which is released when the dose button is pressed. 26 Testing showed that the activation force remained low (5-6 N) across different insulin formulations, needle specifications and temperatures. 26 Dosing accuracy was evaluated under ISO 11608-1 guidelines, similar to NP 4, demonstrating precision across low (1 U), mid-range (40 U) and high (80 U) doses within the acceptable limits (Table 2).15,23,27,43
DT
DT (Novo Nordisk A/S, Bagsvaerd, Denmark) (Figure 1c) is a reusable insulin pen injector that was recently designed to combine and optimize several user-friendly design features from FT and NP 4. The key attributes of DT, NP 4 and FT are summarized and compared in Table 2.
DT can deliver insulin doses up to 80 U (in incremental steps of 1 U), which is in line with the dosing enabled by FT but in contrast to NP 4, which has an upper dose limit of 60 U. 37 This higher dosing capacity may help to address the need for higher insulin doses. DT is also compatible with a range of needles (eg, NF and NT single-use needles). The dosing accuracy and robustness of DT were evaluated in line with ISO 11608-1 23 across a range of insulin doses, environmental conditions and storage conditions demonstrating compliance with ISO-defined acceptance limits. Reported values were either better than or comparable to those previously obtained for NP 4 39 and FT.15,27,41,43 The ability to accommodate higher insulin doses without the need for multiple injections or complex dose splitting may allow for more seamless integration into an individual’s daily routine, thereby potentially reducing treatment burden and supporting adherence.
Beyond offering consistently high dose accuracy, DT incorporates several design enhancements aimed at improving usability. Building upon the features of NP 4 and FT, DT incorporates an easy-to-read dose scale and a non-extending dose button that requires a lower activation force (mean activation force range: 3.9-4.3 N) than NP 4 (mean activation force 6.9 N). 42 Further assessments were conducted to evaluate user handling after DT was exposed to various storage conditions. These assessments examined factors such as dose-setting torque, hold force and injection time to ensure consistent performance. Across all tested conditions, DT maintained stable operation, with no substantial deviations in performance parameters, suggesting that the device remains easy to use even after exposure to environmental stressors. The low activation force and stable dose-setting mechanism may be particularly beneficial for individuals with manual dexterity challenges. A handling and usability study that assessed 100 participants with and without prior injector pen experience reported that 85% of participants found DT very easy or extremely easy to use, and 87% of participants found DT easy or very easy to learn how to use. 44 These findings suggest that DT may be well accepted by users with varying levels of injector pen injector experience, potentially facilitating ease of adoption and supporting user confidence in insulin administration.
Affordability and accessibility are also considerations for global adoption of insulin pen injectors, particularly in low- and middle-income countries. 13 While cost structures and prescribing practices vary across health care systems, UK-based research has shown that reusable pen injectors are generally cost-neutral or less expensive over time than prefilled pen injectors due to their cartridge-based design.20,21 These features, along with the usability and sustainability benefits (discussed below) may support broader access and scalability across diverse health care settings.12,21
Sustainability and Environmental Impact of Insulin Pen Injectors: Potential Role in Sustainability
The rising global prevalence of diabetes and growing use of disposable delivery devices for diabetes medications have contributed to the environmental burden of medical waste, particularly from insulin pen injectors. 12 It has been estimated that hundreds of millions of individuals with diabetes use disposable delivery devices daily, generating large volumes of medical waste globally. 12 For example, in Bosnia and Herzegovina alone, an estimated 3.2 million insulin pen injectors were discarded in 2020, generating over 600 tons of waste. 45 As insulin pen injector use continues to rise, reducing medical waste is a growing priority, particularly because regulatory bodies in Europe and the United States are introducing sustainability policies for medical devices. 12 In the United Kingdom, health care contributes to approximately 5% of the national carbon footprint, with 20% of the footprint of the National Health Service (NHS) arising from medicines and chemicals, and 10% from medical equipment. 21 From a clinical standpoint, choosing an insulin pen injector that balances usability with sustainability may support better adherence, reduce environmental burden and align with health care system priorities.
A key factor influencing the environmental impact of insulin pen injectors is device reusability. Reusable pen injectors help to reduce plastic waste and carbon emissions by limiting the disposal frequency of the device itself, although cartridges still require regular replacement.17,20 Historically, the production of durable reusable insulin pen injectors has relied on high-quality metal components to ensure long-term performance, but this comes with a higher environmental cost due to the energy-intensive nature of metal manufacturing. 46 In contrast, while plastic has a lower environmental footprint per unit of material, conventional plastic insulin pen injectors have typically been less durable and used in much higher quantities. 18 This is exemplified by the NP family, which has featured durable metal components since its introduction in 1985. DT was developed to overcome this trade-off by using recyclable plastic in a robust design that maintains long-term usability and offers the combined benefits of durability and reduced environmental impact. 41
While some disposable pen injectors can be recycled, the success of such efforts depends on manufacturer-provided composition details and access to specialized recycling programs. 12 In recent years, pharmaceutical companies have introduced recycling initiatives to reduce the environmental impact of disposable pen injectors, but these efforts remain limited to a small number of countries.47,48 Expanding these initiatives and transitioning to reusable insulin pen injectors are essential for reducing the environmental footprint of insulin therapy. This aligns with broader sustainability goals outlined by the NHS and supported by the Sustainable Markets Initiative, which highlight the importance of cross-industry collaboration and product-level interventions to decarbonize care pathways, including those for diabetes. 21 Carbon emission reduction is important for both environmental sustainability and the protection of population health, including that of individuals with diabetes. 21
An analysis of UK prescribing data from 2021 to 2023 investigated the potential waste reduction from switching to reusable insulin pen injectors. 20 In a life cycle assessment (LCA) analysis, reusable insulin pen injectors were assumed to be used for three years based on manufacturer warranties and environmental impacts were calculated across a range of daily insulin doses (20-100 U). 20 It was estimated that, as well as reducing annual NHS insulin prescribing costs, switching to reusable pen injectors would save 67.6 tons of plastic waste and 406.2 tons of CO2 equivalent. 20 Growing awareness among individuals with diabetes regarding the environmental impact of insulin delivery devices may further accelerate the adoption of reusable pen injectors as a more sustainable alternative to disposable options. 12 Reusable insulin pen injector use, along with responsible prescribing and waste-reduction strategies, has been identified as a practical and impactful intervention for lowering carbon emissions in diabetes care. 20 Health care professionals and systems are increasingly encouraged to consider environmental sustainability when making prescribing decisions, with reusable insulin pen injectors recognized as an actionable step toward greener diabetes care. 21
DT was designed with sustainability in mind, offering a longer in-use time (up to two years) compared with disposable alternatives, but a shorter in-use time than NP 4, which has an in-use time of up to five years. 49 Compared with NP 4, DT is primarily made from well-characterized plastic compounds, selected for their durability and recyclability, reflecting a trade-off between a shorter in-use time of DT versus NP 4 and a lower environmental impact. An LCA, conducted by Novo Nordisk using previously described methods,20,41 was used to assess the environmental and carbon footprint of DT. The analysis was performed in accordance with ISO 14040:2006 and ISO 14044:2006 guidance for pharmaceutical products and medical devices and was based on a defined daily dose of 40 U of insulin and an assumed device use time of two years. As DT was not in full-scale production at the time of assessment, proxy data from FT (collected in 2022) were used for energy consumption and distribution estimates. 41 It should be noted that the environmental impact of reusable versus prefilled insulin pen injectors depends on contextual factors such as return rates, usage duration and disposal methods. 20 The LCA estimated that the total carbon footprint of DT is 0.538 kg of CO2 equivalents per pen injector over its full life cycle in the European Union, 0.953 kg of CO2 equivalents per device in the United States and 0.482 kg of CO2 equivalents per device in Japan. 41 The primary contributors to this footprint are raw materials and distribution, which together account for over 80% of total emissions. 41 The plastic footprint for DT, including Penfill (PF) consumption with NF needles was 0.12 kg of plastic per year excluding packaging and 0.47 kg of plastic per year including packaging; the footprint for DT excluding PF consumption was 0.012 kg of plastic per year. 41 This is lower than the plastic footprint for FT, which was 1.01 kg of plastic per year excluding packaging and 1.32 kg of plastic per year including packaging.
An LCA study assessed NP 4, including the PF cartridge and needle, estimating a carbon footprint of approximately 6 to 12 kg of CO2 equivalents per year, or 0.017 to 0.033 kg of CO2 equivalents per day. 49 The plastic footprint for NP 4 was approximately 0.14 kg of plastic per patient per year, increasing to 0.47 to 0.53 kg when packaging was included. 49 While NP 4 is a durable and reusable device with an in-use time of up to five years, its estimated full device life cycle carbon footprint (over five years) is approximately 12.5 to 16.5 kg of CO2 equivalents per device, 49 which is higher than that of DT (0.482-0.953 kg of CO2 equivalents per device). 41 This higher footprint is likely attributable, in part, to the greater use of metal components in NP 4, which require more energy-intensive manufacturing processes than plastic. Therefore, DT may offer a more balanced solution in terms of both sustainability and usability. As the adoption of reusable insulin pen injectors increase, transitioning to sustainable insulin delivery solutions, such as DT, will likely be beneficial for reducing medical waste and carbon emissions.
Summary
Given the narrow therapeutic range of insulin, precise dosing is critical in terms of optimizing glycemic control and reducing the risk of hypoglycemia. 17 DT has been designed to combine the key user-friendly attributes of FT with the high durability profile of NP 4. DT also delivers consistent and accurate insulin doses, meeting industry standards across its intended dose range under conditions that reflect and exceed those under which it is likely to be used. The pressure required to administer the insulin dose has been further reduced for DT in comparison to that reported for both NP 4 and FT, further increasing ease of use, demonstrated in usability testing, particularly for individuals with poor hand strength or low manual dexterity. As the prevalence of diabetes (and therefore insulin pen injector use) continues to increase worldwide and global environmental considerations become increasingly important, there is greater focus on reducing the environmental impact of devices used to manage diabetes. 21 The introduction of DT may be an important advancement in insulin delivery by integrating usability and sustainability considerations.
Footnotes
Acknowledgements
Medical writing support was provided by Samuel Bestall, PhD, of Oxford PharmaGenesis, Oxford, UK.
Abbreviations
CSII, continuous subcutaneous insulin infusion; ISO, International Organization for Standardization; LCA, life cycle assessment; NHS, National Health Service.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: All authors are employees of and own shares in Novo Nordisk A/S.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Medical writing support was funded by Novo Nordisk A/S.
