Abstract
Background:
Insulin treatment is an essential hormone replacement therapy for the survival of people with type 1 diabetes and is often used for treatment in type 2 diabetes, particularly as the disease progresses. Advances in insulin therapy have been made since its discovery, including production of human insulin and development of insulin analogs with improved efficacy and safety profiles. The different types of available insulin formulations allow health care professionals to personalize treatment to an individual’s needs. Generally, insulin requires parenteral administration via subcutaneous injection owing to very low oral bioavailability.
Methods:
This article reviews the human, technological, economical, and regulatory factors affecting the performance of insulin pens and the relationship between them. Opportunities and challenges that insulin pen injections may encounter in the future are also considered.
Results:
Insulin delivery devices, together with other factors, influence dose accuracy, convenience, and quality of life, contributing to easier medication administration with high efficacy and safety. For patients, ease of use, fast and accurate drug delivery, and painless injection are the most valuable features of an insulin injection device. For manufacturers, technological feasibility and economic viability also need to be considered when developing injection devices.
Conclusion:
Insulin pen injectors are generally preferred over vial and syringe, although access may be limited in some health care systems. Insulin pen injectors can adapt to different insulin regimens and formulations and have the potential to acquire dosing data in real time.
Introduction
An estimated 537 million individuals worldwide are currently living with diagnosed diabetes, with 150 to 200 million people reliant on insulin replacement treatment for their health.1,2 Since the discovery of insulin 100 years ago and the first subcutaneous administration of the peptide hormone extracts from bovine pancreas, considerable improvements have been made across all aspects of insulin therapy. 3 The improvements over the years have helped to reduce the treatment burden for individuals and to improve the process of taking medication. In developed countries, a wide range of insulin formulations are available to health care professionals (HCPs) and people with type 1 or type 2 diabetes. This can help with personalizing the treatment regimen to an individual’s needs, thereby optimizing disease management. Of the 60 peptide drugs approved in the United States, insulin therapy was the first peptide therapeutic used in humans. 4 Despite research efforts, most human insulin and insulin analogs, being of peptide nature and, therefore, destroyed in the gastrointestinal tract, have prohibitively low bioavailability when given orally.5 -7 Although pulmonary administration improves the bioavailability compared with oral administration, it remains considerably lower than that with subcutaneous administration.8,9 Therefore, injection devices, such as insulin syringes, pen injectors, or insulin pumps, are still the most widely used and effective ways to deliver insulins to the subcutaneous space and subsequent distribution to the insulin receptors in various tissues.
Depending on the type of diabetes and the best personalized treatment for the person with diabetes, insulin replacement regimens typically consist of basal insulin (long-acting insulin) and/or bolus insulin (fast-acting, mealtime insulin), as well as correction insulin doses when needed. These are typically delivered by vial and syringe, pen injectors, or insulin pumps. 10 The number of insulin injections per day varies greatly between people with diabetes from once per day to multiple injections per day with administration happening both at home and outside home.
Basal insulin analogs deliver the constant level of insulin required to maintain stable glucose levels overnight and between meals. First-generation long-acting insulin analogs exhibit relatively short half-lives, suitable for once-daily dosing.11 -16 Basal insulins with long half-lives, suitable for once-weekly dosing, are under development by Eli Lilly (basal insulin Fc) and Novo Nordisk (insulin icodec).
Unlike basal insulins, bolus insulin analogs have a quick onset and a short duration of action and are designed to address the spike in blood glucose that occurs following a meal. These include rapid-acting insulins (eg, insulin lispro, insulin aspart, and insulin glulisine)17 -19 and ultrarapid insulins (faster insulin aspart and ultrarapid insulin lispro).20,21 Dual-action insulin formulations (a premixed mixture of bolus and basal insulins) have been developed to ease the treatment burden (fewer injections) for people who can use a fixed ratio combination.
Concentrated formulations of insulins such as insulin degludec U-200 and human regular insulin U-500 are also available. 22 These concentrated insulins allow reduced injection volumes in people who require high doses of insulin.23,24
Administration of insulins requires an injection device that has high dose accuracy, owing to insulin’s potency, onset of action, and effect on blood glucose, and that is easy to use, owing to the high injection frequency.
The insulin regimen is just one factor contributing to successful diabetes management in a multifactorial and complex process also involving education, lifestyle modifications, psychosocial wellbeing aspects, health capital, and glucose monitoring.10,25,26 One of the major limiting factors in obtaining a good glycemic control, which is crucial in the management of diabetes, is hypoglycemia (and the fear of hypoglycemia).27 -29 Furthermore, the definition of success in diabetes management may be different for individuals with diabetes, HCPs, and society in general. In addition to relevant treatment outcomes, differences in preferences and expectations between individuals with diabetes, HCPs, and society could play an important role in the overall treatment and management of diabetes.
For people who require insulin to manage diabetes, the selection of insulin formulation is only one part of the story (Figure 1): A careful choice of delivery device can influence the perception of treatment success through reducing the complexity of dosing, as well as improving convenience, quality of life, and treatment outcomes via improved consumption of medication.30,31 Injection depth is a key parameter because it affects pain levels and the pharmacokinetic and pharmacodynamic profiles of insulins. Injection into subcutaneous tissue is preferred because it is the least painful and provides the most consistent insulin absorption.32 -35 While pen injectors are more accurate and easier to use than vial and syringe, they are also more expensive. 36 Although vial and syringe are still widely used in the United States, data from 2020 suggest a shift toward predominant use of pen injectors. 37 Nowadays, pen injectors are mature, highly refined, standard products, and many models are available. 38 The option selected for each individual should ideally be based upon a personalized assessment of the individual’s needs. However, in reality, factors such as the health care system, health care plan, socioeconomic status, cultural factors, and availability in individual countries will affect the selection of an insulin delivery device. A successful insulin delivery system must achieve the right balance between human desirability, technological feasibility, and economic viability (Figure 2) and may vary by country owing to economic and societal factors. 39 Consequently, a patient-centric approach is critical for the successful design and use of diabetes injectable therapy devices.40,41 From the perspectives of people with diabetes and HCPs, an injection device should be easy to learn/teach how to use, be easy to use, deliver fast and painless injections, be able to deliver the needed dose in one injection, and have low variability in dose delivery.42 -45
The insulin injection device plays a crucial role in the success of diabetes management.
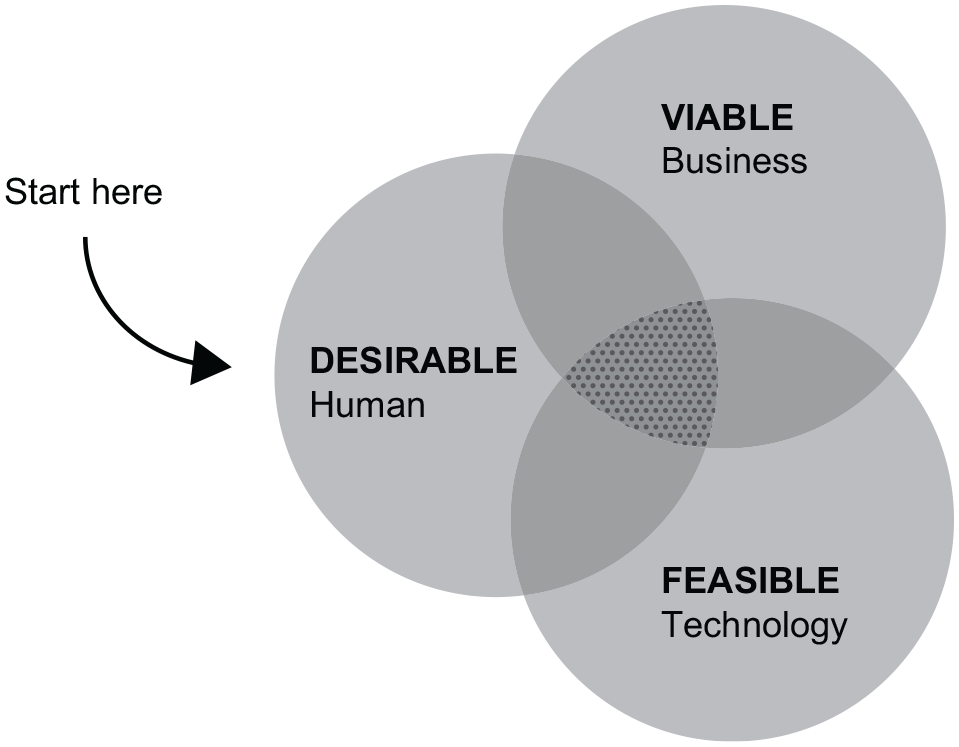
Human-centered design thinking for selecting insulin delivery approaches. Adapted from IDEO U (https://www.ideou.com/pages/design-thinking).
Here, we provide a narrative review of the key factors affecting the performance of insulin pen injector technology according to our experience and expertise.
Factors Affecting the Performance of Insulin Pen Injector Technologies
Human Factors
To develop a successful insulin pen injector, the users’ needs as well as their environments must remain a central focus throughout, from conception to market launch and beyond.40,41 Thus, the first step should be to gain a deep understanding of an individual’s needs regarding their disease management in general and their insulin administration devices in particular. 39 Results of a survey of 504 people with type 2 diabetes in the United States or the United Kingdom identified confidence in administering the correct dose (59.5% of participants), ease of selecting the correct dose (53.2% of participants), overall ease of use (47.4% of participants), and frequency of injections (44.2% of participants) as the most important features that would determine their choice of an injection device (selected from 17 attributes). 43 These results confirmed previous reports indicating that receiving the correct insulin dose is the primary concern of individuals using insulin pen injectors46,47 and that the likelihood of people using a device increases with their perception of its ease of use.45,48,49
Other critical aspects to consider while designing an injection device are people’s concerns, such as fear of needles and pain associated with injections; side effects of the drug; risk of injection-site reactions; and the people’s physical condition (eg, their dexterity, vision, and hearing), which may deteriorate with age.47,50,51 Any of these factors, if not addressed appropriately, may lead to reduced adherence to a prescribed medication regimen and eventually to poor treatment outcomes.30,52 Various studies have demonstrated that the increased injection comfort of pen injectors compared with vial and syringe was associated with improved adherence to a prescribed medication regimen and improved outcomes.53,54 The prevalence of fear of injections in individuals treated with insulin has been reported to range from 6% to 43%, as assessed using the Diabetes Fear of Injecting and Self-Testing Questionnaire. 55 Additionally, the fear of needles and anticipated pain related to injection was reported by 35% to 53% of insulin-naive people with diabetes.56,57 However, it is important to differentiate between a fear of needles and needle phobia, which is seen in very few people. People with needle phobia may experience severe anxiety leading to a panic attack, and this may lead to avoidance of medical care (including seeing a doctor).
The risk of an injection-site reaction, including lipoatrophy, lipohypertrophy, allergy, skin infections, and abscess formation, is another concern for individuals with diabetes.58 -60 These dermatological complications are most often associated with a poor injection technique and repeated use of the same injection area.61,62 In addition, priming the pen injector after attaching a new needle is key to ensuring that the pen injector is in good working order and to removing potential air bubbles. However, it has been reported that only 57.4% of people with diabetes using a pen injector primed the pen and needle as recommended by the manufacturer. 63 These issues could be addressed with improved and repeated patient education and easy-to-use pen injectors. Procedures that are cumbersome or implemented to prevent occasional events, such as too much air in an insulin pen injector, are often subject to omission. Ideally such procedures should be made easier or removed by design.
The perception of insulin treatment and insulin pen injector is also affected by people’s ethnic affiliations, and influencing factors include cultural beliefs and values, religion, and health literacy.64 -67 A study in the United Kingdom found that ethnicity was a factor in the initiation of insulin therapy, for example, people from South Asia were considered more likely to be influenced by negative experiences of insulin therapy within community networks. 65 A study in Pakistan among a largely Muslim community identified that insulin could interfere with religious obligation and that the fear of hypoglycemia during the Ramadan may be particularly distressing and result in apprehension regarding insulin use. 67 In addition to insulin treatment, among East Asian American populations originating from China, Korea, Japan, and Taiwan, the required specialized diet needed for the management of type 2 diabetes is perceived to disrupt social harmony and make East Asian Americans feel burdensome to others. 68 These cultural and socioeconomic barriers need to be addressed by HCPs when developing a diabetes management plan. 69
Overall, offering individuals living with diabetes well-designed pen injectors and visual instructions for use could provide an experience of safety and ease of use that empowers them to take the next step in their treatment, help improve medication taking, and ultimately lead to improved outcomes and quality of life.
Technological Factors
To optimize insulin therapy and improve medication consumption as prescribed, pen injections must be as accurate, easy, painless, and free of discomfort as possible. Owing to the narrow therapeutic index of insulins, the accuracy of insulin pen injectors is of critical importance. 70 Indeed, delivering a smaller-than-intended dose may result in hyperglycemia, while delivering a dose greater than that intended may lead to hypoglycemia. Moreover, the daily dose of insulin needed for each person can vary greatly, depending on body weight and insulin sensitivity.71 -74 Over the past 35 years, technological innovations have enabled the design of accurate pen injectors with small, single-use needles and low manual injection force to improve the overall injection experience for individuals, 75 with improved accuracy and reproducibility of the delivery of insulin doses, including small doses, compared with vial and syringe.76 -79 Asakura et al 36 reported that using a pen injector was more accurate than vial and syringe regardless of HCP experience in insulin therapy. Approved insulin pen injectors fulfill the regulatory minimum requirements for dosing accuracy.80 -85
The introduction of shorter and thinner single-use needles for pen injectors has reduced the level of pain experienced by people with diabetes during insulin injections compared with traditional vial and syringe. 86 Many factors, including the injection technique, design of the injection device, injection area (abdomen, thigh, or arm), and needle visibility, influence pain perception. One of the most important elements to consider from a pain perspective is the needle design. First, the length of the needle should be long enough to ensure insulin delivery beyond the dermis and avoid intradermal administration and, at the same time, be short enough to avoid intramuscular injections. Both intradermal and intramuscular injections have shown increased risk of hypoglycemia compared with subcutaneous injection due to differences in insulin pharmacokinetics.87,88 While skin thickness is mostly similar across populations (regardless of age, sex, and body mass index [BMI]), subcutaneous thickness varies with age, sex, BMI, and injection site, with thicker subcutaneous tissues observed in truncal sites (abdomen and buttocks) than in limbs (thighs and arms). The risk of intramuscular injections of insulin is greater in slimmer, younger people, male patients, and in those using limbs instead of truncal injection sites. 88 Although the shortest needles are sufficient to traverse the skin and enter the subcutaneous fat, longer needles are associated with an increased risk of intramuscular injections. 88 Improvements in needle design have helped to reduce the dexterity requirement and the risk of intramuscular injection, especially in children, whose subcutaneous tissue is thinner than that of adults.89,90 Second, the thickness of the needle should also be carefully considered because a needle that is too thin will require a high injection force and a needle that is too thick may cause bleeding and pain.91,92 Finally, the sharpness of the needle (bevel angle) affects the penetration force and, therefore, pain sensation, with sharper needles causing less pain to the patient. 87
The viscosity of insulin formulations can also affect the performance of pen injectors. Although most insulins have a viscosity close to that of water at 20°C, 93 this increases at lower temperatures (eg, refrigerator temperature). The viscosity can also be affected by the insulin concentration. These potential increases in viscosity would require a higher injection force for delivery and would lead to an increased injection time. Storage conditions are also important for the integrity of the formulation and the performance of the injection device. 94 For example, improper storage of disposable insulin pens with the needle attached may cause air buildup, which could increase the injection time and, consequently, could affect the dose of insulin delivered. 63 Storage with the needle attached can also cause evaporation and aggregate formation that eventually may lead to needle clogging. Once in use, the pen injector should be kept at room temperature (in general, but dependent upon the specific pen injector type, at <30°C and for no longer than 30 days) to avoid the formation of air bubbles, which may occur during expansion and contraction of the injection device in response to a temperature change. 95 Furthermore, insulins would degrade if stored for too long or exposed to high temperatures or direct sunlight, which would reduce the effectiveness of the treatment. 95
The need to remember what the most recent dose was and when it was injected is another aspect of the burden associated with insulin therapy. The introduction of a memory function was the first step toward developing a “smart” insulin pen that helped improve dosage tracking.38,96 A study by Adolfsson et al 96 demonstrated that the use of a pen injector with a memory function was associated with a decrease in the number of missed injections. The first smart insulin pen reached the market in 2017. Smart pens automatically transfer details of the time and dose of their most recent insulin injection to a smart phone and can, for example, calculate the next dose required depending upon which apps use the dosing data, thus reducing the number of dosing errors and improving glycemic control. 5 Another feature of smart pens with the potential to greatly improve diabetes management is their ability to record data about injection times in relation to meals and total dose received. This wealth of information, collected automatically without adding any major burden to the user, will be invaluable in helping HCPs to use shared decision-making, provide personalized treatment plans, and, ultimately, optimize insulin therapy.32,97 -99 Adolfsson et al 32 were the first to report the effect of the use of a connected smart pen on clinical outcomes in people with type 1 diabetes: The use of a smart pen in conjunction with continuous glucose monitoring seemed to lead to fewer missed bolus doses and increases in duration in range than during the period before introducing the device. 32
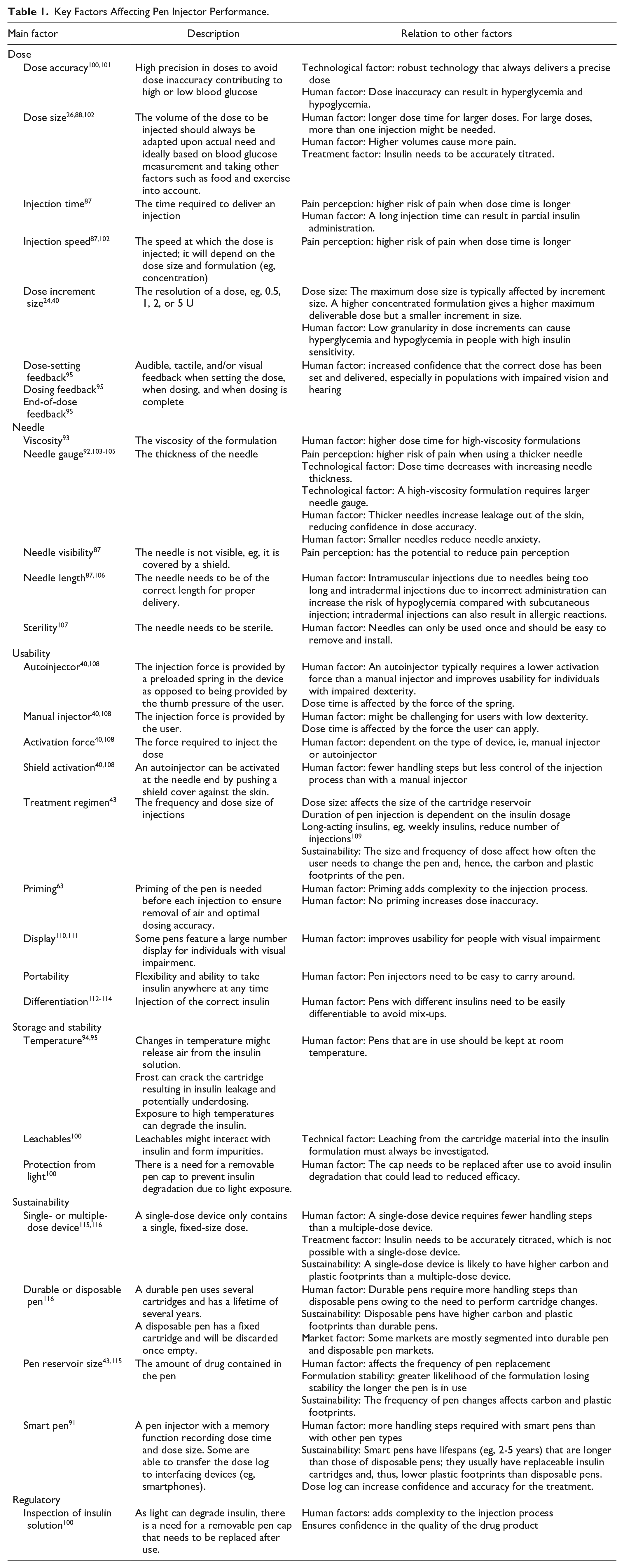
Importantly, each feature of modern insulin pen injectors addresses a mix of user, legislative, and/or manufacturer needs and must be balanced against the needs of the target population and environment. Features cannot be evaluated and optimized in isolation because they affect each other. For example, a thin needle might reduce pain perception but will increase the injection time and, hence, might reduce ease of use (Table 1). To address the needs of specific user groups, such as the pediatric population, some insulin pen injectors offer a dose increment of as low as 0.5 U, with a reduced injection force needed and a shorter design.40,117,118 Some devices also incorporate audible clicks to confirm dose delivery 40 and help to increase individuals’ confidence that the correct insulin dose has been administered. Some pen injectors have been designed for people requiring large doses of insulin and are able to deliver concentrated insulin analogs at up to 300 U per injection in increments of 5 U of insulin.22,119 A pen injector has also been developed for U-500 insulin to address an observed increase in medication errors which may have contributed to underutilization of the U-500 insulin therapy. 119 This innovation also removes the need for the potentially confusing manual dose conversions needed for concentrated insulins.
Key Factors Affecting Pen Injector Performance.
Another innovation addressing the needs of people with impaired dexterity is spring-loaded mechanisms to reduce the activation force required for dose delivery.40,108 Some pen injectors also feature a large number display for people with visual impairment, which is common in people with diabetes.110,111
Regulatory Requirements
The design of pen injectors is highly regulated. Various government bodies, such as the US Food and Drug Administration, set out requirements for pen injectors. 100 Manufacturers are also required to follow industry standards like ISO 11608 and ISO 60601. For instance, the dose-accuracy requirements from the standards mandate extensive testing to demonstrate that the delivered dose is the same as the set dose and that multidose pen injectors accurately deliver each randomly set dose. Other requirements for pen injectors include biocompatibility testing, which should consider all components of the injection device and the formulation being delivered, and the potential for the presence of leachables or extractable device materials in the insulin formulation or the adsorption of the product onto injector components. Other regulations relate to shelf-life stability and expiration date, the impact of environmental conditions such as extreme temperatures on the device and the formulation, sterilization, usability testing, and the ability to visually inspect the insulin. 100 Smart pens containing electronic components must be tested in different environmental conditions (eg, warm and humid environments) to demonstrate safe and correct performance under such conditions.
Commercial Factors
Although the patient is the key factor when designing and developing an insulin injection device, commercial factors cannot be ignored. These include considerations from the perspectives of users, manufacturers, and payers. To improve diabetes management, an injection device must be accessible, affordable, and cost-effective. Thus, the use of pen injectors varies significantly between countries, for example, because of reimbursement policies and accessibility. For example, while the use of pen devices is high in Europe (80%-90%), it remains lower in the United States (approximately 60%) and in developing countries (approximately 17%).37,107 Interestingly, although more costly than vial and syringe, 120 pen injectors have been shown to reduce health care costs in people requiring insulin therapy owing to fewer hypoglycemic episodes than with vial and syringe.121,122 This poses the question of the overall cost-effectiveness associated with the use of pen injectors from a payer’s perspective.
From the manufacturer’s perspective, to be able to provide an injection device that is affordable and of the highest quality, which, therefore, has the greatest benefit for people with diabetes, the device must be cost-effective, scalable, and sustainable.
Finally, the environmental impact of manufacturing and the daily use of insulin pen injectors is an important factor. Although disposable pen injectors are considered by some people with diabetes to be more convenient and user-friendly because they do not require insulin cartridge replacement,38,86 such devices generate a large amount of waste. A study from Bosnia and Herzegovina published in 2020 estimated that 3.2 million plastic pens per year were used in the country, resulting in over 600 tons of waste. 115 As environmental considerations increase in global importance, attention is returning to durable pens as more environmentally friendly alternatives to disposable pens. 116 Reducing the impact of pen injectors on the environment can also be achieved by recycling prefilled pens. 123 In addition, integration of electronic components in modern pen injectors has an environmental cost, which could be acceptable for durable pens but not for disposable pens. For disposable pens, add-on connectivity technology may be the most environmentally friendly solution but may affect the usability of the device. Interestingly, the technology used for the development of the first carbon-emission-free prefilled autoinjector reported in 2020 124 could potentially be used for insulin pen injectors in the future.
Future Perspectives and Conclusions
Remarkable advances have been made to improve the patient experience with insulin injection devices; nonetheless, further progress is on the horizon. Reusable smart pens are relatively new, and it is likely that these will continue to be improved to further alleviate the burden associated with insulin injections. 97 Notably, connected pens and smartphones are enablers of several apps that can help people with diabetes to manage their diabetes on a daily basis. Indeed, personalized treatment and management recommendations could be made based on a large amount of available information, including actual insulin dose, continuous glucose monitoring, food intake, and exercise data. In addition, this would help to reduce decision-making efforts, the risk of errors, and, thus, the burden and stress on the people with diabetes. Future evolutions could also involve the use of artificial intelligence, machine learning, and cloud-based algorithms to provide informed real-time advice to improve diabetes management and, ultimately, quality of life for individuals living with diabetes. The new technologies developed for insulin pen injectors could be applicable to other injectable therapies and vice versa, keeping in mind the specific needs of each target population. As for all new treatments, there is a social and cultural bias that will need to be addressed to ensure that all people with diabetes can receive optimized treatments.
Another promising area of research that has the potential to further reduce the perception of pain associated with insulin injection is the development of ultrathin needles. Li et al 125 have developed an ultrathin needle (120 µm outer diameter) that delivers insulin with minimal invasion of the subcutaneous layer. When tested in healthy pigs, the ultrathin needle resulted in a fivefold smaller puncture area than a 31G needle (261 µm outer diameter). 125
As the needs and requirements of people with diabetes continue to evolve, they will drive the development of new delivery devices (eg, patches, implants, oral devices, inhalers), new insulin formulations (oral bioavailability, glucose-sensitive insulins),87,102,109 the advent of new analogs of insulin (eg, once-weekly basal insulin 126 and oral insulin), or the integration of new functionalities such as continuous glucose monitoring. Whatever the future holds for the landscape of insulin treatment, new technologies will need to address current and future users’ requirements without creating new, unforeseen challenges with no direct benefit to the user, such as the burden to download data and doses or putting the burden of recycling of pens and related material onto the user alone. As such, recycling pen injectors through pharmacies is an option being explored, and alternative materials to traditional plastics (eg, biodegradable plastics) could be used for the development of new pen injectors. Finally, in terms of innovation, the right balance between novel features, cost, and accessibility is integral to the development of new holistic product-service systems for the treatment of diabetes and is also crucial to achieve treatment success.
Footnotes
Abbreviations
BMI, body mass index; HCP, health care professional; U, units.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are full-time employees and shareholders of Novo Nordisk A/S.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing support was provided by Nicolas Bertheleme, PhD, of Oxford PharmaGenesis, Oxford, UK, with financial support from Novo Nordisk A/S.
