Abstract
Background:
Very few patient-reported outcomes have been published in regard to opinions of individuals with type 1 diabetes concerning adjunctive therapy. The aim of this subanalysis was to qualitatively and quantitatively assess the thoughts and experiences of participants with type 1 diabetes who have used low doses of empagliflozin as an adjunct to hybrid closed-loop therapy.
Methods:
Semi-structured interviews were performed with adult participants who completed a double-blinded, crossover, randomized controlled trial using low-dose empagliflozin as an adjunct to hybrid closed-loop therapy. Participant experiences were captured through qualitative and quantitative methods. A descriptive analysis was performed using a qualitative approach; attitudes toward relevant topics were extracted from interview transcripts.
Results:
Twenty-four participants were interviewed; 15 (63%) perceived differences between interventions despite blinding, due to glycemic control or side effects. Advantages that arose were better glycemic control, in particular postprandially, requiring less insulin, and ease of use. Disadvantages were thought to be adverse effects, increased incidence of hypoglycemia, and increased pill burden. Thirteen (54%) participants were interested in using low-dose empagliflozin beyond the study.
Conclusions:
Many participants had positive experiences with low-dose empagliflozin as an adjunct to the hybrid closed-loop therapy. A dedicated study with unblinding would be beneficial to better characterize patient-reported outcomes.
Keywords
Introduction
Despite the known benefits of glycemic control in type 1 diabetes (T1D), a minority of those with T1D are able to achieve these goals with intensive insulin therapy.1 -4 One way to improve glycemic control is through the use of adjunctive therapies, such as sodium-glucose linked cotransporter (SGLT) inhibitors. SGLT inhibitors in routine insulin therapy have demonstrated reductions in glycated hemoglobin (HbA1c), total daily insulin requirements, and weight but unfortunately have also demonstrated an increased risk of diabetic ketoacidosis. 5 Hybrid closed-loop therapy is another innovation in the management of T1D, which has been shown to reduce HbA1c and hypoglycemia, while increasing time in the target range and treatment satisfaction.6,7 Unfortunately, not all those with T1D who use the hybrid closed-loop therapy achieve target HbA1c or percentage of time in the target range between 3.9 and 10 mmol/L.6,8,9 Short studies, however, have shown that SGLT inhibitors as an adjunct to this technology can increase time in the target range with reduced insulin requirements and without increasing hypoglycemia.10 -12
Few studies have published patient-reported outcomes with SGLT inhibitor use in T1D; the inTandem studies demonstrated reduced diabetes distress and increased treatment satisfaction with sotagliflozin through validated questionnaires, while treatment satisfaction scores increased with dapagliflozin use in the Dapagliflozin Evaluation in Patients With Inadequately Controlled Type 1 Diabetes (DEPICT) trials.13,14 However, there are no qualitative data on perceptions and experiences concerning adjunctive therapies in those with T1D.
There have been limitations in worldwide use of SGLT inhibitors as an adjunct in T1D, predominantly due to the concern of diabetic ketoacidosis. Various guidelines and subanalyses have been formulated to mitigate this risk and to guide off-label use, which is of growing interest to many patients and practitioners.15,16 The interest of those with T1D in adjunctive therapies and their unaddressed concerns may help to guide the importance of adjunctive therapy use and authorization in research and clinical practice.
We recently conducted a randomized controlled trial comparing empagliflozin 2.5 mg and 5 mg with placebo as an adjunct to a research-based hybrid closed-loop system in adults with T1D and suboptimal control. 17 In our trial, empagliflozin at both doses increased time in the target range by 11 to 12 percentage points compared with placebo. The objective of this subanalysis was to capture participant experiences during the study, in particular, their views on using low-dose empagliflozin as an adjunct to the hybrid closed-loop therapy.
Methods
Study Design and Participants
The research design of the trial has previously been reported. 17 In short, we performed a three-way, randomized, crossover, double-blind trial comparing empagliflozin (Boehringer Ingelheim, Germany) at 2.5 mg and 5 mg daily with placebo, while using the McGill Artificial Pancreas, a research-based hybrid closed-loop therapy involving the iPancreas closed-loop system 18 (Oregon Health & Science University, Portland, OR) comprised of the Dexcom G6 CGM (Dexcom, San Diego, CA), a study pump (a noncommercial t:slim TAP3; Tandem Diabetes Care, San Diego, CA), and a study phone with an application that encompasses the McGill dosing algorithm.19,20 More detail on this algorithm is discussed previously. 17 Participants were 18 years of age or older with T1D who had at least three months of experience on pump therapy, and who did not achieve ≥70% time in the target range (3.9-10 mmol/L) after two weeks on our McGill Artificial Pancreas or their commercial hybrid closed-loop system. Informed consent was obtained before the initiation of study procedures. The trial was conducted at the Clinique Médicale Hygea (an affiliate of the McGill University Health Center) in Montréal, QC, Canada, with approval through Advarra (the local research ethics board) and Health Canada. The trial was also registered (NCT04450563).
Interviews
A subanalysis was performed for participants who completed the trial. One week after the last intervention, a semi-structured interview was conducted to assess participant experiences using the study drugs and devices, as well as their overall experiences in the trial; Supplemental Table S1 lists the interview questions and structure in English. Interview questions were developed from and based on another study within our research group that was codeveloped with a psychologist. There were no piloted interviews for these specific questions before their use as the analysis was in itself exploratory. Interviews took place remotely through secure teleconference because of the COVID-19 pandemic and were conducted in English or French by a researcher conducting the trial. Although drugs were blinded, possible effects of the study drug were elucidated by identifying differences between interventions and the run-in.
Transcription and Data Analysis
Interviews were audio-recorded and later transcribed verbatim by one of the researchers (M.-R.P.). A descriptive analysis was performed using a qualitative approach to assess participant perceptions of the study devices and study drugs, as well as some discussion around general adjunctive therapy use in T1D. A priori codes informed by the interview guide were used to organize responses. Transcripts were then coded by two research personnel (M.-R.P. and M.O.) with the a priori codes and subsequently with secondary, tertiary, and quaternary codes (listed finally as codes, categories, and themes) using an inductive-deductive approach via an encoding software program (MAXQDA, version 22.1.1, VERBI, Berlin, Germany). Review of agreement in coding between researchers was performed for every six participants, to assess for consistency and reconcile discrepancies. A review of final themes between both researchers and by one of the subinvestigators (A.H.) of the trial was conducted. This is similar to other exploratory qualitative analyses using semi-structured interviews in the field of diabetes technology and in other qualitative research.21 -24
Topics, themes, and preferences were described as proportions to quantitatively enhance details in recurring topics. Given the exploratory nature of the data, no further statistical analyses were performed.
Results
Twenty-four adults with T1D completed the end-of-study interviews. The mean (standard deviation [SD]) age was 33 (14) years, with duration of diabetes being 21 (13) years, and mean (SD) HbA1c of 8.1% (0.5%), that is, 65 mmol/mol (6 mmol/mol). Of all, 50% were male, and 46% preferred to conduct the interview in French. More participant characteristics are displayed in Supplemental Table S2. Supplemental Table S3 demonstrates the frequency of codes identified throughout the interviews.
When participants were asked to contrast interventions, 15 (63%) could feel there were differences between interventions despite blinding, due to glycemic effects (9/15, 60%) or side effects (6/15, 40%). Although interviewers did not ask participants to identify any of the study drugs, 11 participants voluntarily made comments during the interview concerning the identification of the study drugs as they discussed differences between interventions. Ten out of 11 participants (91%) identified study drugs correctly when verified after unblinding. Note that the unblinded identity of certain interventions are revealed within the text in parentheses for comparison, but these identities were not known to interviewees or interviewers at the time of the recording.
Perceived benefits of empagliflozin, as per participants, were those that were experienced in comparison to their usual therapy or to the run-in phase, when there were no study drugs administered. The common benefits voiced by participants were improved glycemia, requiring less insulin, and lack of side effects or disruption in usual habits (Table 1). Glycemic benefits were not only more time spent with a normal blood glucose level but also the benefit of experiencing less overt hyperglycemia. As one of the participants described, “I found . . . in Intervention 2 [empagliflozin 5 mg], . . . there my blood sugars were beautiful. No matter what I did, they stayed really nice” (P022, translated from French).
Themes Related to the Study Drugs and Adjunctive Therapies in End-of-Study Interview (N = 24).
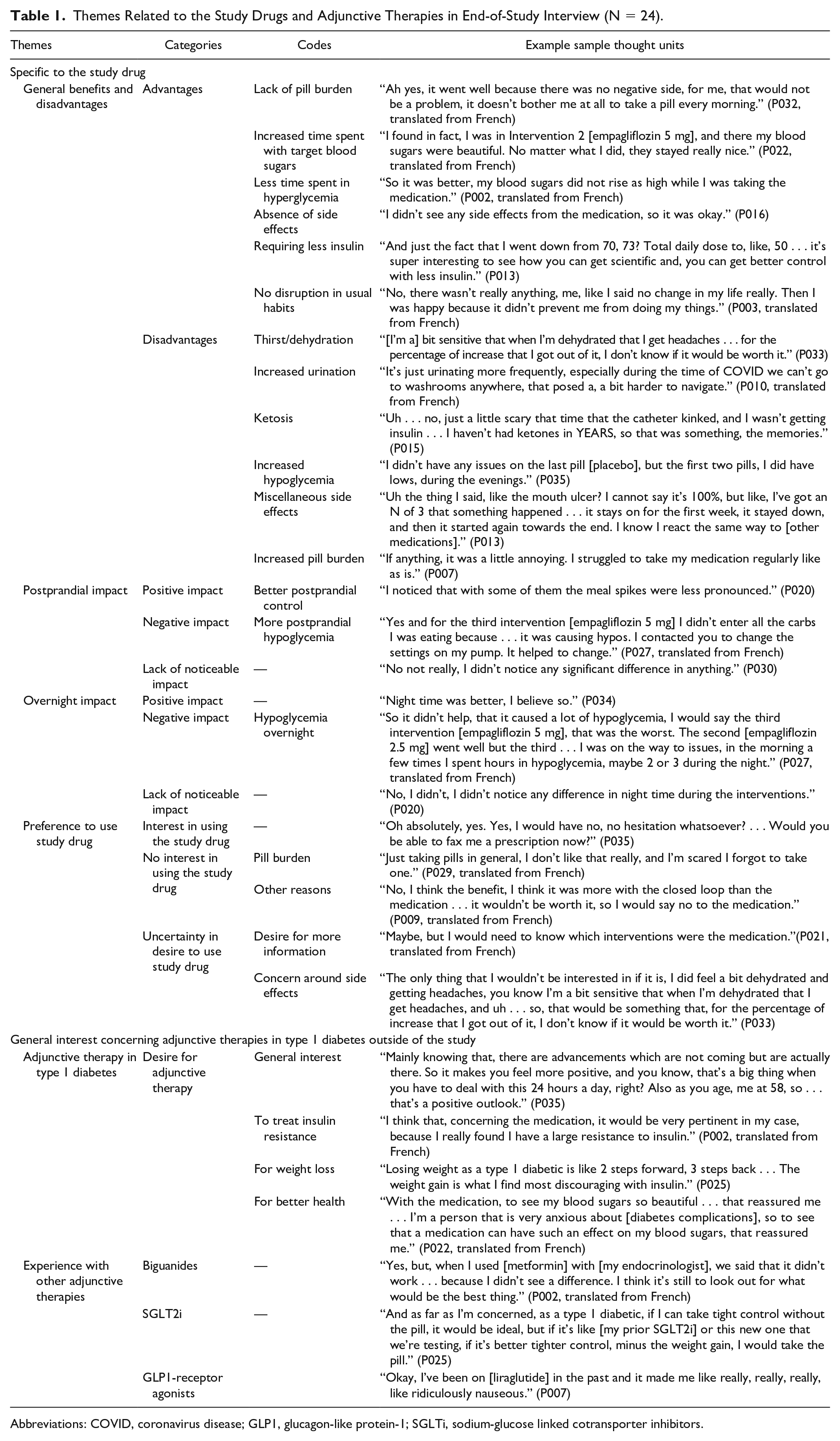
Abbreviations: COVID, coronavirus disease; GLP1, glucagon-like protein-1; SGLTi, sodium-glucose linked cotransporter inhibitors.
Postprandial glycemia, in particular, was perceived to be affected by using a study drug while on the hybrid closed-loop therapy, in comparison to the run-in. One participant explained,
“I think fewer peaks? Like I feel in the first intervention [empagliflozin 5 mg], my peaks after meals weren’t as big and for the next [placebo] there were big peaks but then we readjusted the insulin.” (P007)
The use of less insulin was of interest, in particular because some participants discussed their struggles with insulin response and increasing dose requirements in their usual care. All were aware of empagliflozin’s mechanism of action from the informed consent process. The separation of blood glucose control with insulin use, however, with the use of this drug, was impressionable. As one participant explained, “It’s super interesting to see how you can get scientific and, you can get better control with less insulin” (P013).
Perceived disadvantages of the study drug included more episodes of hypoglycemia (particularly postprandially), urination and thirst, and other adverse effects (Table 1). As mentioned, increased urination was a known and explained mechanism of action of empagliflozin that participants were aware of; therefore, participants discussed their heightened awareness of this effect during the trial.
Mean morning ketone levels were not significantly increased in empagliflozin arms compared with placebo
17
; however, the daily surveillance of ketone levels performed in this study is more than what is done in routine practice. Some participants had negative experiences with ketosis due to catheter malfunction. One interviewee commented,
“It happened once during the last week of intervention 3 [empagliflozin 5 mg], that . . . my ketones went up . . . That part is a little bit of black magic, I don’t get how it works, and what to do to control it, but I guess . . . I would figure it out if I’d be on the meds for a bit longer.” (P013)
Pill burden was interpreted differently among participants. Some participants reported no issue taking a pill daily, whereas others felt this was a nuisance. These perceptions were shared by both those with and without other prescribed oral medications.
Of the 23 participants who discussed their interest in empagliflozin, 13 (57%) were interested in its use outside of the study. Those unsure (4/23, 17%) or not interested (6/23, 26%) expressed concerns with pill burden, side effects, and preference to see the final results of the trial. One participant expressed,
“No, I think the benefit . . . was more with the closed loop than the medication . . . It wouldn’t be worth it, so I would say no to the medication.” (P009, translated from French)
Adjunctive therapy was viewed favorably by many participants. Eleven (46%) participants expressed a general desire for any type of adjunctive therapy outside the study. Specific reasons included better health (including but not exclusive to glycemic control), reducing insulin resistance, and weight loss (Table 1). As one participant described,
“Losing weight as a type 1 diabetic is like 2 steps forward, 3 steps back . . . The weight gain is what I find most discouraging with insulin.” (P025)
Some participants had already used adjunctive therapies in the past (Table 1). These included biguanides, glucagon-like protein-1 (GLP1)-receptor agonists, and other SGLT2 inhibitors, with participants having prior negative experiences with biguanides and GLP1-receptor agonists due to a lack of efficacy or an increase in adverse effects.
“When I used [biguanides] with [my endocrinologist], we said that it didn’t work . . . because I didn’t see a difference. I think it’s still to look out for what would be the best thing . . . I think that, concerning the [study drug], it would be very pertinent in my case, because I really found I have a large resistance to insulin.” (P002, translated from French)
For some, the study experience triggered discussion with their health care provider concerning the use of adjunctive therapy (Supplemental Table S4). Some participants became interested in its off-label use outside of the study and were willing to discuss the utility of the drug with their health care provider. One participant jokingly stated, when asked if they would be interested in empagliflozin use, “Oh absolutely, yes. Yes, I would have no, no hesitation whatsoever? . . . Would you be able to fax me a prescription now?” (P035).
Although it was not a focus of the study, some participants discussed how the hybrid closed-loop therapy or adjunctive study drugs impacted physical activity; feelings were mixed (Supplemental Table S5). Feelings ranged from increased confidence with exercise due to reduced hypoglycemia or hyperglycemia with the overall study regimen to fear of hypoglycemia attributed to the study drug and to lack of comfort pertaining to specific aspects of the devices (eg, preference for a tubeless insulin pump).
Themes related to participants’ experiences with the hybrid closed-loop system and being in a clinical trial reflect what is previously published in the literature (Supplemental Table S4).21,25,26 Participants’positive and negative effects of being in a clinical trial included the benefit of increased support and the burden of increased preoccupation with their diabetes.
Discussion
In this subanalysis of our randomized controlled trial, despite blinding, we were able to characterize participants’ experiences throughout this short study concerning low-dose empagliflozin as an adjunct to the hybrid closed-loop therapy. Not only were many able to appreciate differences between interventions, but for the majority, the study was a positive experience. However, it is important to highlight that participants who did not sense differences between interventions still had objective benefits in glycemic control with empagliflozin compared with placebo.
This subanalysis demonstrated that adjunctive therapy is of interest to the T1D community, with benefits extending beyond glycemic control. These findings are similar to those from a prior study that used quantitative survey based on the data from sotagliflozin in T1D which demonstrated that weight reduction was an unmet need in T1D respondents, with the majority interested in adjunctive therapy. 27 SGLT inhibitors are not the only adjunctive agents to improve the quality of life, as a pilot study using metformin 28 and larger studies using liraglutide29,30 showed improved treatment satisfaction in questionnaires. However, none of these studies reviewed adjunctive therapy in the context of hybrid closed-loop therapy, which is a growing field given the increasing popularity of commercial systems.31,32
Pharmacotherapies used adjunctively in T1D, such as SGLT inhibitors and GLP-1 receptor agonists, are extensively used in type 2 diabetes because of their cardiac and renal protective properties and their weight loss benefits. 33 Unfortunately, the landmark trials assessing the use of SGLT inhibitors in individuals with and without type 2 diabetes excluded those with T1D; therefore, there are no targeted trials assessing cardiac or renal endpoints with SGLT inhibitor use in this population.34 -38 This is similar to the weight loss trials assessing the use of GLP-1 receptor agonists.39,40 These therapeutic benefits, weight loss in particular, are sought after by those with T1D; therefore, recording patients’ thoughts and experiences on this matter can help orient research.
This subanalysis had several limitations. The analysis was an exploration of qualitative methods with some quantification but was not a dedicated qualitative protocol; a larger qualitative study exploring adjunctive therapies in T1D would be best to explore core themes surrounding this topic. Selection bias was also a limitation; participants demonstrated enough competency to be on SGLT inhibitors, as well as interest in inclusion into a study using adjunctive therapies. Because convenience sampling was used, conversations were limited by the design of the main study. The use of the same researcher from the study performing the interviews may have also introduced bias; however, from our interviews, the support throughout the study was felt to be positive (Supplemental Table S4). Blinding was also a limitation as participants were not certain which intervention was the active drug and which drug contained the higher dose. The short duration of each intervention also limited participants’ abilities to assess the risks and benefits of empagliflozin. In particular, a study of longer duration may have been required to see incidents of ketosis or diabetic ketoacidosis, as was seen in a similar open-label trial using empagliflozin 5 mg as an adjunct to automated insulin delivery, where one case of ketoacidosis was observed. 11 Dedicated future qualitative studies would thus be warranted.
Conclusion
Most adults with suboptimally controlled T1D who used blinded low-dose empagliflozin as an adjunct to the hybrid closed-loop therapy had a positive experience. Adjunctive pharmacotherapy is of interest to those with T1D; longer, unblinded studies are required to further characterize this interest and its potential positive effects.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231176302 – Supplemental material for Participant Experiences of Low-Dose Empagliflozin Use as Adjunct Therapy to Hybrid Closed Loop: Findings From a Randomized Controlled Trial
Supplemental material, sj-docx-1-dst-10.1177_19322968231176302 for Participant Experiences of Low-Dose Empagliflozin Use as Adjunct Therapy to Hybrid Closed Loop: Findings From a Randomized Controlled Trial by Melissa-Rosina Pasqua, Madison Odabassian, Michael A. Tsoukas and Ahmad Haidar in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
GLP1, glucagon-like protein-1; HbA1c, glycated hemoglobin; SD, standard deviation; SGLT, sodium-glucose linked cotransporter; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.H. received research support and material support from Eli Lilly, Dexcom, Tandem, Adocia, AgaMatrix, YpsoMed, and Medtronic; consulting fees from Eli Lilly; IP acquisition fees from Eli Lilly and YpsoMed; and has pending patents in the diabetes technology field. M.A.T. received research support from AgaMatrix and speaker honoraria from Eli Lilly, Novo Nordisk, Boehringher Ingelheim, Janssen, and AstraZeneca. No other authors have conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was supported by the National Institute of Health (1DP3DK106930). The source of funding had no role in the study’s design, data collection, outcome interpretation, or manuscript writing.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
