Abstract
Closed-loop insulin delivery systems are fast becoming the standard of care in the management of type 1 diabetes and have led to significant improvements in diabetes management. Nevertheless, there is still room for improvement for the closed-loop systems to optimize treatment and meet target glycemic control. Adjunct treatments have been introduced as an alternative method to insulin-only treatment methods to overcome diabetes treatment challenges and improve clinical and patient reported outcomes during closed-loop treatment. The adjunct treatment agents mostly consist of medications that are already approved for type 2 diabetes treatment and aim to complete the missing physiologic factors, such as the entero-endocrine system, that regulate glycemia in addition to insulin. This paper will review many of these adjunct therapies, including the basic mechanisms of action, potential benefits, side effects, and the evidence supporting their use during closed-loop treatment.
Introduction
Over 2 decades ago, the Diabetes Control and Complications trial clearly demonstrated that improved glycemic control reduces the risk of microvascular and macrovascular complications in type 1 diabetes (T1D). 1 In line with increased technology use, the Advanced Technologies and Treatments for Diabetes (ATTD) consensus in 2019 recommends time in range per day for individuals with T1D be greater than 70% (16 hours and 48 minutes) and time below target glucose range be less than 4% (1 hour). 2 Despite this knowledge and continued advancements with diabetes technology, the attainment of optimal glycemic control in T1D remains elusive. Closed-loop (CL) automated insulin delivery systems are now clinically in use and have revolutionized the management of T1D. Studies comparing CL insulin delivery systems with conventional pump therapy show that CL systems are associated with a greater percentage time in range (TIR) and a reduction in hypoglycemia while improving sleep, ability to exercise, and quality of life measures.3-5 Despite these tremendous advancements, glycemic control achieved with CL systems is not perfect, particularly during the postprandial period. While the underlying reason for postprandial hyperglycemia is multifactorial, the key factor is the slow absorption and action of the pre-prandial insulin bolus that fails to mimic endogenous insulin action. Moreover, current treatment methods do not fully address the dysregulation of other glucoregulatory hormones that is commonly observed in people with T1D. The failure of glucagon suppression in the immediate postprandial period, insufficient secretion of amylin which is co-expressed and co-secreted with insulin by the pancreatic β-cells, dysregulation of glucagon-like-peptide-1 (GLP-1) and gastric inhibitory peptide (GIP) which are secreted by the L cells of the small intestine are some of the elements that contribute to suboptimal postprandial glycemic control for people with T1D. Additionally, in contrast to common perception that individuals with T1D are mostly lean, we now know that the rates of overweight and obesity in T1D equal those of the general population. Obesity is an independent risk factor for microvascular and macrovascular complications with risk compounded in individuals with T1D. 6
For all these reasons, adjunct treatments to insulin treatment during CL have been introduced as an alternative method to optimize glycemic control and address additional treatment challenges during T1D treatment. The goals of adjunct therapy include helping individuals with T1D achieve glycemic targets by increase in target TIR and lowering the risk of hypoglycemia, controlling weight gain and related complications, and improving quality of life.
Also, addition of certain adjunctive therapies may offer additional benefits such as renal protection or cardiovascular disease reduction. This review presents the physiologic basis and the supporting data for a host of adjunct therapies (Table 1) that have been investigated to optimize CL glycemic control and discusses the potential benefits and adverse effects, including the treatment burden placed on individuals with T1D and caregivers with the addition of an adjunct treatment.
Adjunctive Treatments Evaluated in Closed-Loop Trials.
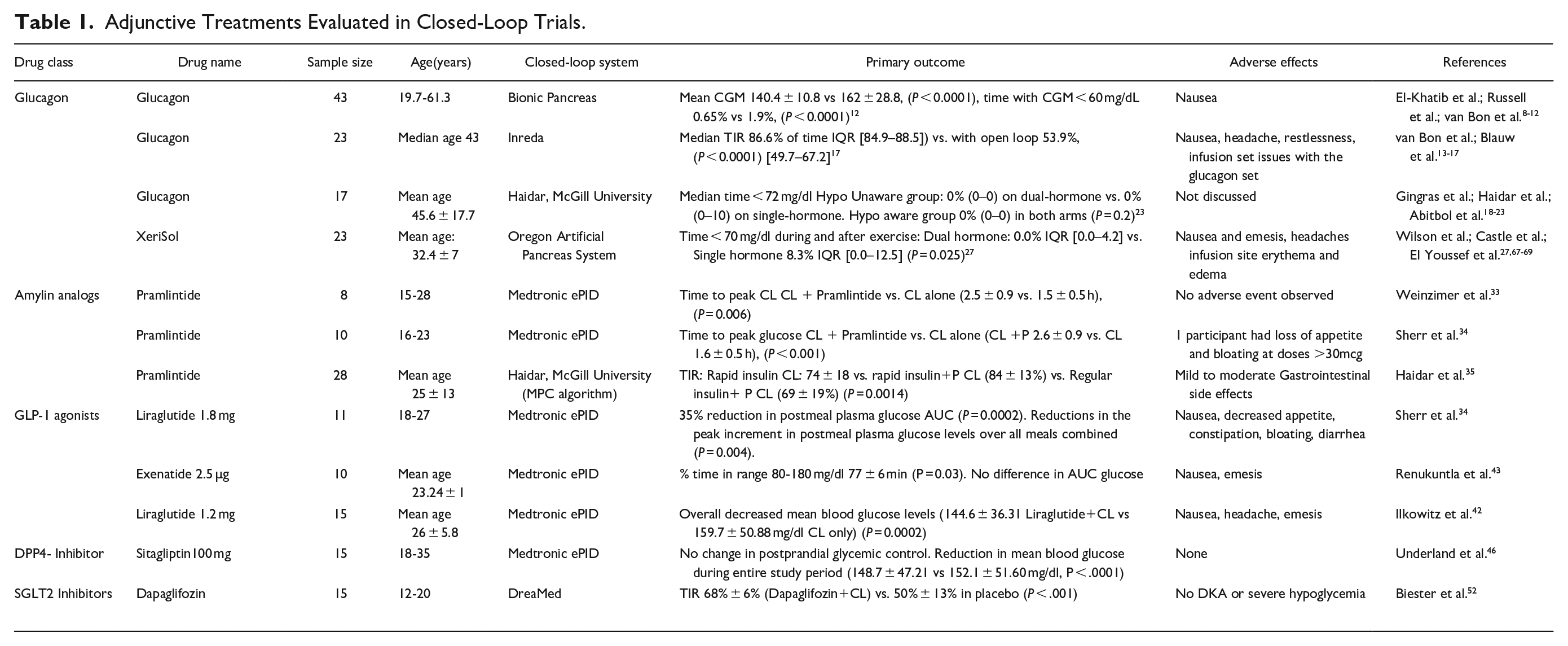
Adjunct Treatments
Glucagon
Glucagon, as a counter-regulatory hormone, has a significant impact on glucoregulation through its role in gluconeogenesis and glycogenolysis. In T1D, the loss of insulin signaling via β-cells leads to glucagon suppression, in turn causing hypoglycemia in the setting of exogenous hyperinsulinemia. In contrast, inappropriately elevated glucagon levels result in exaggerated hyperglycemia during the postprandial phase due to dysregulation of glucagon release. 7 Although insulin only CL systems can decrease the risk of both hypo and hyperglycemia, the fear of hypoglycemia remains a significant challenge in tight glucose control due to late onset of insulin action as well as slow clearance. The use of bihormonal CL systems with glucagon infusion as an adjunct to insulin infusion has been explored to improve glycemic control and further reduce hypoglycemia by mimicking a more physiologic approach in the management of T1D. Bihormonal systems deliver subcutaneous glucagon automatically via a pump from an infusion site that is separate from the insulin infusion site when the glucose value based on the readings from a Continuous Glucose Monitoring (CGM) system is predicted to be low is or is low.
Several investigators have been working toward the commercialization of a glucagon and insulin bi-hormonal CL system. A group at the Boston University and Massachusetts General Hospital has been testing a glucagon-insulin bi-hormonal bionic pancreas system since 2010 in various settings by mostly crossover studies using a Model Predictive Control (MPC) for insulin and a proportional integral derivative (PID) controller for glucagon delivery. In their first human study, the system achieved an average blood glucose of 140 mg/dl with no hypoglycemia episode that required intervention (n = 6 participants) in the setting of high carbohydrate intake, missed premeal bolus, and variable insulin kinetics. Five participants with lower insulin lispro absorption had at least one episode of non-severe hypoglycemia requiring treatment. 8 Their following studies were conducted in a relatively wide range of participants9-11 including a recent outpatient randomized clinical trial during which the bihormonal automated insulin delivery system achieved an average CGM reading of 140.4 ± 10.8 mg/dL vs. 162 ± 28.8 mg/dL and 0.66% vs. 1.9% for time with hypoglycemia when compared to conventional diabetes treatment. In the bionic pancreas period, the mean total daily insulin dose (TDD) was more than TDD during the convention treatment arm (0·66 U/kg per day vs 0·62 U/kg per day; P = .014). 12
Concurrently, several other groups, including Oregon University, Inreda in The Netherlands, and McGill University, have been conducting studies using glucagon-insulin bi-hormonal CL systems. Inreda, following their initial studies13-16 tested their fully automated CL bihormonal system in a 2-week outpatient, randomized, crossover trial compared with 2-weeks of usual care in 23 adults with T1D. Median TIR was increased from (53.9% [49.7–67.2] to 86.6% of time [84.9–88.5]) safely with a reduction in hypoglycemia from 2.0% (0.7–3.6) to 0.4% (0.1–0.8). 17 TDD was higher during closed loop (41.4 units/day) compared with usual care (40.1 units/day; P = .0017). Inreda’s system, intended to obtain CE marking, includes 2 transmitters for glucose readings and a wearable device to integrate the CGM, accelerometer, control algorithm, insulin pump, and glucagon pump. This fully automated CL system does not require manual meal boluses or exercise announcements.
Haidar and his colleagues at McGill University have conducted several clinical trials testing the safety and efficacy of their bihormonal CL system.18-22 Its overnight use led to 79-94 % TIR 72-180 mg/dl) and 0-2.4% time with hypoglycemia ( < 72 mg/dl). 21 Their 60 hour use of a bihormonal CL system resulted in 3% with time < 72 mg/dl and 79% TIR (72-180 mg/dl). 22 In this study, premeal bolus was based on the meal carbohydrate content. Their most recent publication provides an assessment of their overnight bihormonal artificial pancreas system to reduce nocturnal hypoglycemia by targeting participants with hypoglycemia awareness. Both single and bihormonal systems showed no significant difference in preventing nocturnal hypoglycemia in individuals with hypoglycemia unawareness. 23
In most of the studies testing insulin and glucagon CL systems, the glucagon requires a separate reservoir, infusion set, and a pump. The glucagon cartridge/reservoir and infusion sets need to be replaced every 24 hours due to the instability of available glucagon preparations and potential cytotoxic amyloid fibril formation. This task places a large burden on individuals with T1D.24,25 Development of innovative glucagon formulations with improved stability and longer half-life opened up new avenues for the glucagon-insulin bi-hormonal systems. Dasiglucagon (Zealand Pharma, Denmark), a stable glucagon analog in a ready-to-use aqueous solution, has been tested in the iLet bihormonal system. The preliminary results from the phase 2 study revealed superior blood glucose control with the bi-hormonal system compared to the insulin only system during the study (mean sensor glucose 139 ± 11 vs. 149 ± 13 mg/dl, P = .004 respectively) while the percentage of time with CGM glucose < 54 mg/dl was nominally reduced (0.2%, IQR 0-0.4 bi-hormonal system vs. 0.6%, IQR 0.2-1.1% insulin only, P = .11). 26
The Oregon University CL group used stable glucagon (XeriSol glucagon:Xeris Pharmaceuticals, Chicago, IL) in their 76 hour, randomized, crossover, outpatient study that showed an increase in the TIR during the bihormonal CL system use compared to Predictive Low Glucose Suspend (PLGS) (7.5%) with some increase in hyperglycemia compared to a single hormone CL system (28.2 vs. 25.1%). 27 Insulin TDD was not different among groups. 27 The most common side effect of glucagon has been nausea that resulted in 4 out of 23 study participants withdrawing from the study.
To summarize, although more insulin only CL systems are increasingly being used in the management of T1D with well-proven treatment effect in real-life settings,28-30 bihormonal systems could reduce the average glucose level and achieve lower glucose targets without increasing hypoglycemia, specifically following meals and exercise as the next generation CL systems. Long term and unsupervised clinical trials on the use of bihormonal systems are needed to prove their efficacy, safety, and the impact on patient reported outcomes before they become standard treatment.
Pramlintide
Amylin is co-secreted with insulin from pancreatic beta cells in response to blood glucose rise typically after meals. It slows gastric emptying, suppresses glucagon secretion, and promotes satiety, thereby controlling the postprandial rise in blood glucose. The autoimmune destruction of β-cells leading to T1D not only affects insulin synthesis but also impairs amylin secretion and release from the beta cell. The paradoxical, inappropriate, postprandial hyperglucagonemia resulting in immediate postprandial hyperglycemia has been attributed to impaired amylin secretion for people with T1D. Pramlintide, an analog of amylin, is an FDA-approved injectable drug both for T1D and type 2 diabetes (T2D) and is proven to reduce postprandial hyperglycemia and increase satiety. 31 Despite its FDA approval since 2005, Pramlintide use has not been widely adopted due to the added burden of 3 to 4 daily injections or a separate pump and infusion set due to its incompatibility with insulin. Co-formulation of pramlintide and insulin at a fixed ratio during pump treatment has been examined as a practical approach to minimize the number of injections. 32 Pramlintide adjunct treatment has been tested in the CL systems by various investigators. A short term, inpatient study published in 2012 showed a significant delayed time to peak blood glucose (2.5 h vs. 1.5 h) and reduced the peak (88 ± 42 vs. 113 ± 32 mg/dL) using an older version of the Medtronic CL system with a PID algorithm plus 30 mg premeal injections of pramlintide with no premeal announcement. Average glucose values were decreased from 152 ± 47 mg/dl in CL alone to 143 ± 44; during the use of CL plus pramlintide, while there was no significant change in the control group. 33 Premeal insulin levels were significantly higher during CL plus pramlintide (21.7 vs. 17.0 mU/mL). 33 Following this initial study, a higher dose (60 mg/meal) of pramlintide over 3-4 weeks was used in 10 participants over a 24 hour period which delayed the time to peak plasma glucose excursion: (CL 1.6 ± 0.5 h vs. CL with pramlintide 2.6 ± 0.9 h) and reduced the postprandial incremental plasma glucose AUC. 34 Insulin TDD was reduced by 9% following pramlintide treatment. 34
Haidar et al. 35 tested a bihormonal CL system that delivers pramlintide and insulin, with pramlintide delivered in a glucose-responsive, basal-bolus manner at a fixed ratio relative to insulin. Twenty-eight adults with T1D completed this 24 hour inpatient randomized crossover trial. They compared 3 different variations of their original artificial pancreas system to test the pramlintide intervention: rapid insulin-alone, rapid insulin-and-pramlintide and regular insulin-and-pramlintide. The rapid insulin and-pramlintide system increased the TIR from 74% ± 18% to 84% ± 13%, mostly due to increase in daytime TIR without an increase in hypoglycemia and severe gastrointestinal side effects. Insulin dosing was higher in the regular insulin-and-pramlintide group. One of the limitations of pramlintide use is having a separate pump and infusion set due to incompatibility. Thus, having a stable coformulation of pramlintide and rapid insulin is expected to have a dual benefit of reducing the postprandial glucose excursions and decreases the burden on individuals with T1D. 36 Investigational insulin-pramlintide co-formulations in a stable mixture have already demonstrated a 95% reduction in the first hour post-meal glycemic excursions (mean ± SD, ΔAUC blood glucose 0-1 h:1.4 ± 9.9 mg*h/dL co-formulation vs. 43.5 ± 15.3 mg*h/dL insulin only; P < 0.0001) during clinical trials. In addition, the co-formulation treatment has shown favorable post-prandial blood glucose control over insulin only and insulin and pramlintide separate injection treatments [delta maximum post-meal blood glucose 87.0 ± 35.5 mg/dL co-formulation, 109.2 ± 31.1 mg/dL insulin only (P = .0133), 109.4 ± 44.3 mg/dL human insulin and pramlintide separate injection (P = .0357). 37 Future clinical trials are underway to assess co-formulation treatment on glycemic control and weight loss for people with T1D. 38
GLP-1 Agonists
GLP-1 is an incretin hormone produced in the L cells of the small intestine in response to food intake, and it increases endogenous glucose dependent insulin secretion, slows gastric emptying, promotes satiety, and decreases postprandial glucagon secretion. GLP-1 is rapidly degraded by the enzyme dipeptidyl-peptidase-4 (DPP-4) and has a very short half-life.
Synthetic GLP-1 receptor agonists are resistant to degradation by DPP-4 and have a longer half-life, facilitating clinical use. 39 This class of drugs has shown a lot of promise in the management of T2D given their role in weight loss, and in 2019 the GLP-1 agonist Liraglutide was approved by the FDA for use in children ≥12 years of age with T2D.40,41 The rising incidence of obesity in individuals with T1D has necessitated evaluation of new treatments to control weight gain in people with T1D given the added burden of obesity and insulin resistance. This in turn has led to the investigation of the use of GLP-1 analogs as an adjuvant therapy in T1D for the dual purpose of weight control and postprandial blood glucose control.
A study of 11 participants of ages 18-27 year with T1D compared postprandial glucose excursions before and after treatment with 1.8 mg liraglutide in addition to CL therapy. Their results showed no decrease in time-to-peak blood glucose with a significant 22% reduction in peak postprandial glucose, a 39% decrease in AUC, a reduction in total daily insulin dose by 26% (P = .05) and a weight loss of 3.2 ± 1.8 kg (P = .003) after 4 weeks of liraglutide therapy. 34 Liraglutide adjunct treatment during CL treatment was examined by another research group using a slightly lower dose at 1.2 mg liraglutide in 15 adult participants with T1D. Mean blood glucose levels and 2 hour post-breakfast and post-lunch blood glucose AUC were significantly reduced by liraglutide adjunct treatment compared to CL alone (P = .0002 and P < .05 respectively). The incidence of hypoglycemia did not differ between liraglutide + CL and CL alone treatment arms (P = .83, Fisher’s exact test). 42
Another study conducted in 10 adult participants with T1D requiring a 27-hour inpatient admission on 3 occasions, 2-4 weeks apart, entailed CL treatment with insulin alone, exenatide 2.5 µg +insulin or pramlintide 30 µg+insulin in random order. Pramlintide and exenatide were given prior to lunch and dinner which were standardized meals of 60 grams of carbohydrates. The exenatide adjunct treatment was significantly better in attenuating postprandial hyperglycemia compared to CL insulin only treatment (s < 0.03) while there was no significant improvement in post-prandial blood glucose levels for the pramlintide adjunct treatment vs. CL alone (P > 0.05). 43
With the exception of oral semaglutide, GLP-1 agonists have to be administered as a subcutaneous injection. This class of drugs can cause gastrointestinal side effects in a dose-dependent manner and should not be used in individuals with a personal or family history of medullary thyroid cancer or multiple endocrine neoplasia 2A or 2B. As expected, side effects reported in the CL trials were mainly gastrointestinal.34,42,43 Overall, while there are potential advantages to the adjuvant use of GLP-1 agonists with CL, it is not clear if the benefits justify the additional burden and potential side effects. It would be ideal to identify specific patient groups that might benefit from the GLP-1 analog treatment option with future clinical trials.
DPP-4 Inhibitors
DPP-4 inhibitors are a related class of oral diabetes drugs that inhibit the enzyme DPP-4. DPP-4 enzyme deactivates a variety of bioactive peptides, including GLP-1 and GIP. DPP-4 inhibitors increase GLP-1 and GIP levels that in turn inhibit glucagon release and decrease gastric emptying with a subsequent decrease in blood glucose. 44 However, DPP-4 inhibitors have a modest effect on GLP-1 levels and activity compared with GLP-1 receptor agonists. DPP-4 inhibitor adjunct therapy has not shown any improvement in glycemic control when used with insulin treatment for people with T1D, as summarized by a meta-analysis of pooled data from 5 randomized clinical trials and did not improve post-prandial blood glucose levels during CL.45,46 DPP-4 inhibitors are generally well tolerated in the short term and the commonly reported side effects include headache, nasopharyngitis, upper respiratory tract infection and a slight increased risk of gastrointestinal side effects. 47 In summary, results from clinical trials suggest that the clinical utility of DPP 4 inhibitor adjunct treatment for T1D is limited.
Sodium-Glucose Co-Transporter (SGLT) Inhibitors
SGLTs are a group of proteins that play important roles in glucose transport. SGLT2 mediate the reabsorption of 90% of glucose from the kidney proximal tubule into the blood circulation. SGLT2 inhibitors are an oral class of drugs that lower plasma glucose levels by blocking proximal tubular reabsorption of filtered glucose and promoting renal excretion of glucose. 48 Their glucose lowering effect is insulin independent, therefore they are expected to lower total insulin dose given that much of the glucose is excreted from urine without the need for insulin coverage. This group of drugs is now increasingly being used in the treatment of T2D given their added benefit of weight reduction and improvement in long term cardiovascular outcomes. SGLT inhibitors are shown to lower HbA1c, weight, and blood pressure without an increase in hypoglycemia in individuals with T1D and could have a renal protective action for those with albuminuria. 49 However, SGLT2 inhibitors have a range of potential adverse effects including genito-urinary tract infections, hypotension, risk of bone fractures and risk of diabetic ketoacidosis which is a particular concern for their use in T1D. 50 SGLT inhibitors are approved as an adjunct treatment in Europe and Japan and not yet approved as an adjunct treatment for T1D in the US. SGLT2 inhibitors have also been evaluated as part of triple drug adjunct treatments, which have become a possibility with the hope that combination of multiple adjunct drugs might exponentially increase the favorable clinical outcomes. A small scale clinical study was conducted to examine the impact of the combination of insulin, dapaglifozin, and liraglutide on glycemic control and weight control. Thirty participants with T1D on insulin and liraglutide were randomized to receive either 10 mg of dapaglifozin or placebo. Thirty participants with T1D on insulin and liraglutide were randomized to receive either 10 mg of dapaglifozin or placebo during a clinical study. The study participants in the dapaglifozin group had a 0.66% reduction in HbA1c (P < .01 vs placebo) and a 1.9 kg weight loss (P < .05 vs placebo). The study results also showed an increase in glucagon and ketogenic markers (ß-hydroxybutyrate and acetoacetate) with dapaglifozin treatment highlighting the importance of monitoring for ketosis during SGLT2 adjunct treatment. 51
As for CL studies, a single-center, double-blind, randomized, placebo-controlled, crossover trial studied the SGLT2 inhibitor dapaglifozin in 30 adolescents and young adults (median age 17 years) with T1D on a CL insulin delivery system. Participants were on the DreaMed Glucositter CL algorithm using a Medtronic pump and sensor and received either 2 doses of 10 mg of dapaglifozin or placebo over 24 hours with 2 unannounced mixed meal tests 6 hours apart. Their results showed that TIR was significantly higher in the dapaglifozin group compared with placebo (68% ± 6% vs. 50% ± 13%; P < .001) with no abnormal serum beta hydroxy butyrate values detected and without significant difference in the occurrence of hypoglycemia. 52
The role of SGLT2s in potentially reducing the need for carbohydrate counting in CL automated insulin delivery systems has been evaluated. In an open-label, crossover, non-inferiority trial, 30 adult participants with T1D underwent outpatient CL interventions with 3 sequences of strategies for delivering prandial insulin which included carbohydrate counting, simple meal announcement without carbohydrate counting, and no meal announcement. For each sequence of prandial insulin strategies, participants were randomly assigned to receive either 25 mg of empagliflozin or not in a crossover design. Their results showed that the primary analysis of the CL system with no meal announcement and empagliflozin was inferior to the control arm (mean glucose 180.0 ± 28.8 vs. 153.0 ± 27.0 mg/dl; P = .94), and simple meal announcement and empagliflozin was non-inferior (153.0 ± 25.2 mg/dl; P = .003). The use of empagliflozin with carbohydrate counting was associated with lower mean glucose, corresponding to a 14% greater time in target range. Of note, mean fasting ketones levels were higher in participants when on empagliflozin (3.96 ± 3.24 vs. 2.34 ± 1.98 mg/dl; P < .001), but there was no occurrence of ketoacidosis. 53 Overall, strong evidence for the use of SGLT2 inhibitors as an adjunctive therapy to CL glucose control is limited, and the increased risk of ketoacidosis presents a great concern.
In contrast to SGLT2, SGLT1 is predominantly expressed in the small intestine, and inhibition of SGLT1 reduces intestinal glucose absorption. 48 Sotagliflozin, an oral inhibitor of both SGLT1 and 2 has been evaluated as an adjunct to insulin therapy in T1D and has been shown to lower total insulin dose, fasting blood glucose, HbA1c and decrease weight without an increase in severe hypoglycemia.50,54,55 There are currently no studies of SGLT1and 2 inhibitors and CL systems.
Metformin
Metformin is a biguanide that acts by decreasing hepatic glucose output and increasing peripheral insulin-mediated glucose uptake. 56 The rationale for using metformin as an adjunct to insulin treatment has been its potential role in improving insulin resistance without increasing risk of hypoglycemia or weight gain. Metformin has been evaluated as an adjunct therapy in T1D in both adolescents and adults with studies indicating that the addition of metformin reduces the total daily dose of insulin and certain measures of adiposity without significant improvements in HbA1C.57-59 In these studies, participants reported gastrointestinal discomfort as a common adverse event during metformin use. While there are no studies evaluating metformin as an adjunct to CL systems, anecdotal reports indicate that it is being used in the clinical setting to overcome insulin resistance and symptoms of polycystic ovarian syndrome in people with T1D. Formal studies are needed to better evaluate the adjunct use of metformin in specific patient groups during CL treatment.
New Generation Insulin Analogs
Faster acting insulins, that are essential to keep up with dynamic glycemic regulation bring exogenous insulin treatment a step closer to physiologic insulin action. The use of faster-acting insulin analogs may improve glycemic control in closed-loop systems. Faster acting insulin Aspart (Fiasp® (NovoNordisk, Denmark) is one of the first approved new generation insulins. Several closed loop systems were used to test Fiasp including Medtronic 670G, the Medtronic Advanced Hybrid Closed-loop (AHCL) and CamAPS FX.32,60-64 However, the data on how the use of Fiasp in closed-loop impacts glycemic control in the free-living condition is controversial. Eli Lilly’s ultra-rapid insulin (URli) is another ultrarapid insulin that was tested in the Medtronic MiniMed 670G system during a 4-week double-blind, crossover study. Glycemic outcomes, insulin usage, and time in auto mode were similar in both Humalog and URli arms. 65 Ultra-rapid-acting inhaled insulin, or Afrezza (MannKind Corporation, Valencia, CA) can blunt postprandial glucose excursion and decrease late hypoglycemia given its short action profile. In an inpatient, three-way, randomized crossover standardized meal study, Afrezza was used at a low and high dose (rounded down vs. up to the closest Afrezza dose cartridge) to compare it with aspart premeal bolus during closed-loop treatment and high dose Afrezza was shown to decrease early glycemic excursion. 66 Overall, studies of newer insulin analogs are promising and more studies are needed to better understand their role in CL systems.
Conclusions
CL systems are increasingly being used and have changed the landscape of T1D management. While CL systems have evolved through the years and showed superior glycemic control compared to conventional diabetes treatment, data from clinical trials clearly demonstrate significant barriers in meeting glycemic targets for people with T1D indicating the need for advanced systems. Also, with increasing rates of overweight and obesity in T1D there is a need for therapies that promote weight loss and help prevent obesity related complications. Physiologic blood glucose regulation is a highly dynamic and complex system that is influenced by numerous factors. First generation CL systems cannot perfectly mimic the intricacy of the human glucoregulatory system and its response to different macronutrients during various conditions. The postprandial hyperglycemia challenge is more pronounced after unannounced meals and remains an important barrier for the implementation of fully automated CL systems. Moreover, insulin treatment alone might not be sufficient to address additional treatment challenges such as rising obesity and insulin resistance in people with T1D. To further optimize glycemic control, several adjunct drugs have been evaluated with CL systems with the most data existing for the use of glucagon, pramlintide, GLP-1 analogs, SGLT inhibitors and new insulin analogs. While each of the above therapeutic modalities offer varying degrees of benefit, they also have side effects that have to be factored into therapeutic considerations. Additionally, managing T1D is a daily burden, and complicated regimens may increase burnout.
Adjunctive therapies have the potential to help people with T1D reach glycemic goals by mitigating postprandial hyperglycemia, promoting weight loss and reducing daily insulin dose. Overall, studies of adjuvant therapies for CL systems in T1D are in their infancy with promising results, and further studies are needed to better evaluate these therapies. In the meantime, with careful consideration, select drugs may be beneficial for select individuals after weighing the risks and benefits.
Footnotes
Acknowledgements
None
Abbreviations
AHCL, advanced hybrid closed-loop; AUC, area under the curve; CE, Conformite’ Europeenne; CL, closed-loop; CGM, continuous glucose monitor; DPP-4, dipeptidyl-peptidase-4; GLP-1, glucagon-like-peptide-1; GIP, glucose-dependent insulinotropic polypeptide; MPC, model predictive control; PLGS, predictive low glucose suspend; PID, proportional–integral–derivative; SGLT, sodium-glucose co-transporter; TIR, time in range; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Laya Ekhlaspour has received consulting fee from Tandem Diabetes and Ypsomed. Eda Cengiz is an advisory board member and consultant for Novo Nordisk, Adocia, MannKind, Lexicon, Arecor. Eda Cengiz was also a speaker for Novo Nordisk.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shylaja Srinivasan is funded by NIH 1K23DK120932. Laya Ekhlaspour is funded by NIH 1K23DK121942, Eda Cengiz is funded by 2016PG-T1D006 Leona M. and Harry B. Helmsley Charitable Trust and 2 R44 DK101280-02 NIH.
