Abstract
As closed-loop insulin therapies emerge into clinical practice and evolve in medical research for type 1 diabetes (T1D) treatment, the limitations in these therapies become more evident. These gaps include unachieved target levels of glycated hemoglobin in some patients, postprandial hyperglycemia, the ongoing need for carbohydrate counting, and the lack of non-glycemic benefits (such as prevention of metabolic syndrome and complications). Multiple adjunct therapies have been examined to improve closed-loop systems, yet none have become a staple. Sodium-glucose-linked cotransporter inhibitors (SGLTi’s) have been extensively researched in T1D, with average reductions in placebo-adjusted HbA1c by 0.39%, and total daily dose by approximately 10%. Unfortunately, many trials revealed an increased risk of diabetic ketoacidosis, as high as 5 times the relative risk compared to placebo. This narrative review discusses the proven benefits and risks of SGLTi in patients with T1D with routine therapy, what has been studied thus far in closed-loop therapy in combination with SGLTi, the potential benefits of SGLTi use to closed-loop systems, and what is required going forward to improve the benefit to risk ratio in these insulin systems.
Introduction
Type 1 diabetes (T1D), which accounts for 5%-10% of cases of diabetes, 1 is marked by the lifelong need for intensive insulin therapy at diagnosis, which often occurs before adulthood. The goal of intensive insulin therapy is to achieve glycated hemoglobin (HbA1c) levels of ≤7.0%, as reflected in multiple guidelines, to reduce complications.2-5 Optimal control is only achieved by a few with T1D worldwide, specifically 21% in the United States.6-10
Closed-loop insulin systems are the most advanced device-based treatment in T1D care,11,12 and with systems such as the MiniMed 670G®, Tandem’s Control-IQ®, CamDiab®, and DiabeLoop®, they are now part of clinical practice.13,14 Hybrid closed-loop therapy (HCL), compared to routine therapy, increase time-in-range (TIR), reduce time in hypoglycemia, improve HbA1c, and increase patient satisfaction.15-21 Unfortunately, HCL has not been able to perfect T1D care; in the iDCL trial using Control-IQ®, there remained almost half of the participants who had HbA1c levels above 7%. 20
A possible way to rectify this problem is the use of adjuvant pharmacotherapy, which has been previously assessed as adjunct to standard of care and HCL.17,19,22-28 Metformin has some glycemic improvement, but has not been tested with HCL. 29 Glucagon-like Peptitde-1 (GLP1)-receptor agonists have demonstrated conflicting outcomes in benefits as adjunct to T1D care, both with and without HCL.24-27,30-33 The effect of dipeptitdyl peptidase-4 inhibitors is not as significant compared to other adjunctive agents in T1D, 34 with one inpatient study demonstrating reduced post-prandial glycemia with sitagliptin using HCL. 27 While pramlintide has been studied as an injectable pharmacotherapy adjunct to routine insulin therapy or integrated into HCL,19,24,25 and has shown benefits,22,24 its clinical application is limited by the lack of an existing co-formulation with insulin and the need for separate injections or infusion systems.
A separate group of medications is the sodium-glucose-linked cotransporter inhibitors (SGLTi’s). Sodium-glucose-linked cotransporters (SGLTs) are solute symporters located in the small intestine (SGLT1) and kidneys (SGLT2) which exchange sodium and glucose.35,36 The inhibition of SGLT2 in the renal proximal tubule (ie, SGLT2 inhibitors, such as empagliflozin, canagliflozin, dapagliflozin) reduces urinary glucose reuptake, and in doing so, reduces serum glucose levels. 35 These medications have revolutionized pharmacology for type 2 diabetes (T2D), given additional non-glycemic benefits such as cardiac and renal protection. This narrative review seeks to delve into their data in T1D and their potential in HCL.
A Review of the Unmet Needs in Closed-Loop Therapy
Post-Prandial Hyperglycemia
While closed-loop therapy is excellent at reducing hypoglycemia and increasing the safety of intensive insulin therapy, it does not eliminate hyperglycemia. On average, those on HCL spend 5-8 hours per day in hyperglycemia.15,16,37 This is influenced by the carbohydrate load, meal composition, the timing of insulin administration, insulin pharmacokinetics, and gastric emptying. Figure 1 demonstrates insulin-dependent and insulin-independent means to potentially ameliorate hyperglycemia in closed-loop therapy; note SGLTi’s address 2 of them.
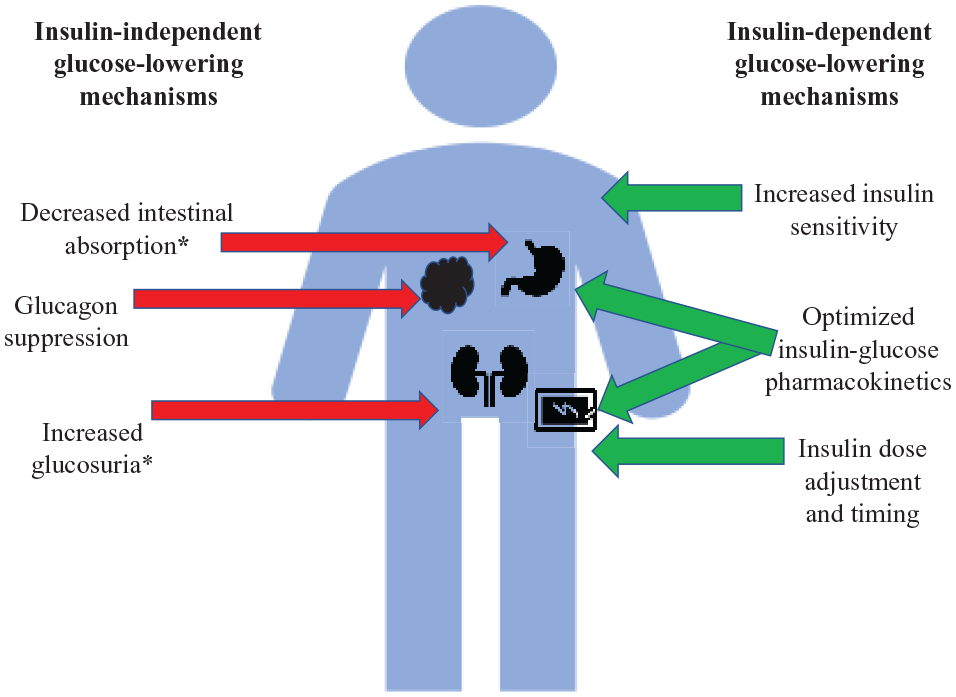
Methods of glucose normalization in type 1 diabetes while on closed-loop therapy.
Carbohydrate Counting
A major barrier to glycemic control in T1D is accurate carbohydrate counting, an essential aspect of insulin dosing.38,39 Prior studies have depicted high rates of counting inaccuracies40,41 and mental health impacts due to disease burden. 42 Carbohydrate counting alleviation could be potentially aided through adjunctive pharmacotherapy, particularly if done through an insulin-independent elimination of glucose.
The Lack of Non-Glycemic Benefits
T1D care includes the reduction of vascular complications and optimization of metabolic health. There is an increasing prevalence of overweight and obese individuals with T1D, with up to 31% higher BMI than the general population.43-45 This results in worsened clinical outcomes; not only is increased BMI in T1D linked with poor glycemic control, 46 but it is further linked with increased cardiac risk factors and complications.46-49
Cardiovascular and kidney disease are significant causes of mortality for T1D.50,51 Though glycemic control improves cardiac and renal outcomes,2,3,52 mortality is still increased in those with well-controlled T1D compared to those without T1D. 53 It is estimated that those with T1D have a life expectancy 11-13 years shorter than those without diabetes of the same age. 54 The treatment of nephropathy in T1D has not seen a breakthrough in over a decade, which includes glycemic and blood pressure control as well as specific pharmacotherapy.2,55-57 Unfortunately, there are not as much data in cardiovascular protection in T1D as there are for renal protection, as T1D is often under-represented in cardiovascular trials. Glycemic control was shown to improve cardiovascular outcomes 17 years later after DCCT, but appears to not be sufficient to completely normalize cardiovascular risk.2,3,58 Adjunct pharmacotherapy may be a way to allow for further metabolic benefits.
SGLTi’s in T1D: A Review of the Randomized Controlled Trials (RCTs) and Real-World Use
A natural interest in SGLTi use sparked in T1D given their novel action and benefit in T2D. Table 1 depicts RCTs assessing SGLTi use in T1D. Canagliflozin was the first drug studied in a major RCT, revealing significant benefits in glycemic control and body weight, while reducing insulin needs without increasing hypoglycemia.59,60 Unfortunately, the rates of diabetic ketoacidosis (DKA) were markedly elevated at 4.3% and 6% with 100 and 300 mg respectively versus none in the placebo group. Following this trial, the DEPICT trials (dapagliflozin), the inTandem trials (using sotagliflozin, an SGLT1 and 2 inhibitor), and the EASE trials (empagliflozin) assessed these agents as adjunct to routine care to T1D management.59,61-69 Various meta-analyses have assessed the collective data on these medications in T1D. The summated reductions are in HbA1c by 0.39%, weight by 3.47%, mean glucose by 1.07 mmol/L, TDD by 10.4%, and systolic blood pressure 3.37 mmHg.70-73 CGM data revealed improvements in TIR without increased hypoglycemia.59,62,64,66,74 Unfortunately, the relative risks of ketoacidosis in those treated with SGLTi compared to placebo were 3-5.70-73
A Summary of Trials in the Use of SGLTi as Adjunct to Insulin Therapy in T1D.
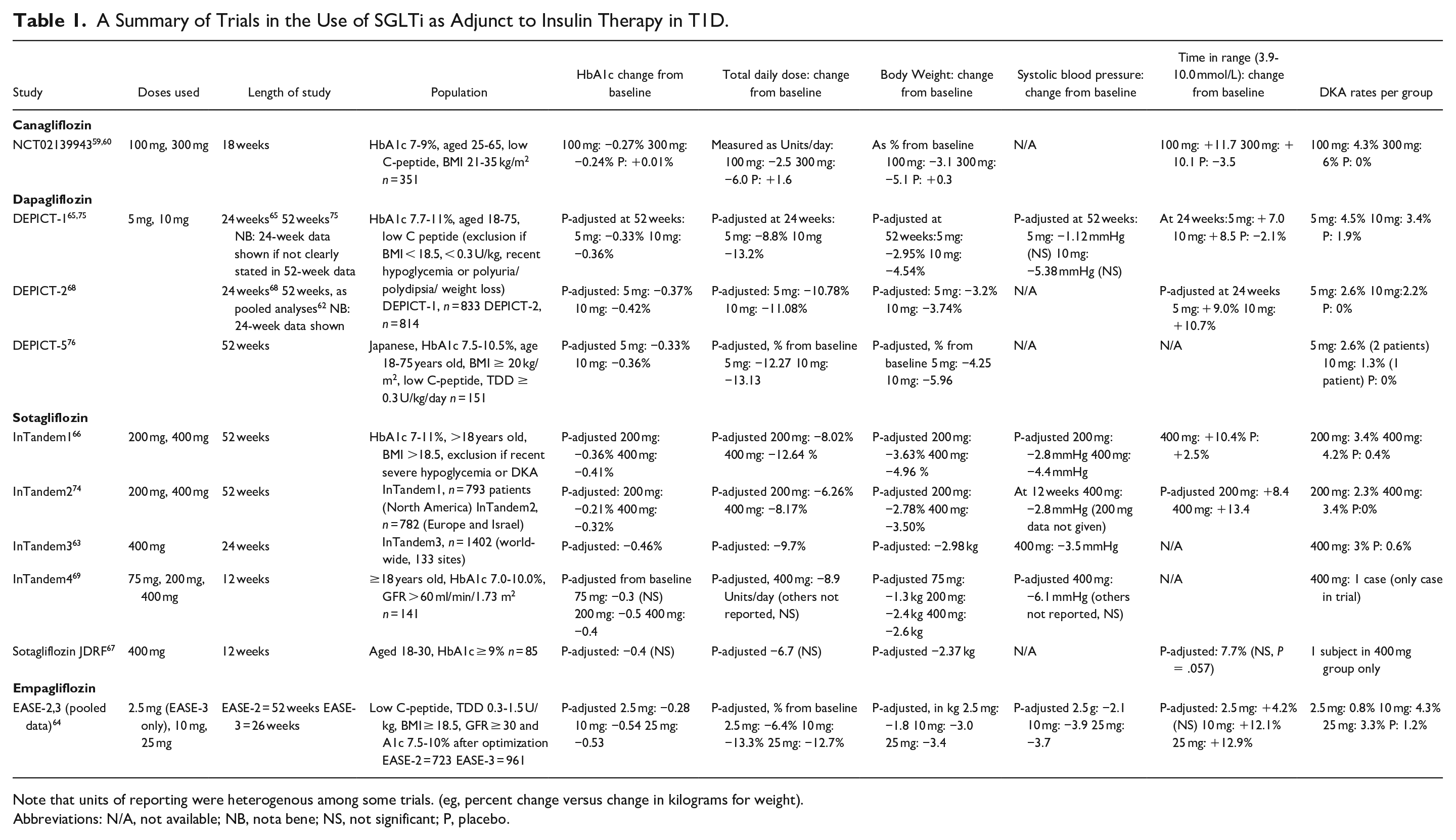
Note that units of reporting were heterogenous among some trials. (eg, percent change versus change in kilograms for weight).
Abbreviations: N/A, not available; NB, nota bene; NS, not significant; P, placebo.
SGLTi use in T1D has had variable uptake worldwide. In 2019, Europe approved both sotagliflozin and dapagliflozin for T1D.77,78 The National Institute for Health and Care Excellence (NICE) approved the use of dapagliflozin as adjunct to insulin therapy for those with T1D with inadequate control, BMI ≥ 27 kg/m2 and insulin requirements of ≥0.5 Units/kg.77,79 Japan has also approved dapagliflozin and ipragliflozin, a selective SGLT2 inhibitor, for T1D.80,81 In the United States, empagliflozin and sotagliflozin were refused approval by the FDA,82,83 but American off-label use in T1D is still present. 84
SGLT Inhibitor Use and Closed-Loop Therapy: Research thus Far
Though the trials assessing SGLTi’s as adjunct to routine T1D have been extensive, there are only small pilot studies published thus far assessing their potential use in closed-loop therapy (see Table 2). Two studies have used dapagliflozin and empagliflozin as adjunct to closed-loop therapy.85,86 In the study described in Biester et al. 85 dapagliflozin 10 mg BID (2 doses) was used as adjunct to fully closed-loop; participants were given 2 mixed meal tests 6 hours apart without meal announcement. The TIR with dapagliflozin was 68% compared to 50% in the placebo arm (P < .001), and required 22% less insulin.
Prior Studies Assessing the Use of SGLT2i’s as Adjunct to Closed-Loop Therapy.

In Haidar et al. 86 empagliflozin 25 mg daily as adjunct to closed-loop therapy was assessed for alleviation of carbohydrate counting via 3 arms: full carbohydrate counting, simple meal announcement, or no meal announcement. The simple meal announcement with empagliflozin resulted in a daytime mean glucose of 8.5 mmol/L and daytime TIR of 68%, which was non-inferior to full carbohydrate counting alone (TIR 70% with mean glucose 8.5 mmol/L, non-inferiority P = .007). Empagliflozin with full carbohydrate counting resulted in the highest daytime TIR (84%) and lowest mean glucose (7.4 mmol/L), which was significantly lower compared to full carbohydrate counting alone with no empagliflozin (P = .004).
The Benefits of SGLT Inhibitor Use in Closed-Loop Therapy
Glycemic Outcomes and Insulin Dose Optimization
The trials demonstrated in Table 1 revealed decrease in mean fasting glucose, mean glucose throughout the day, and glycemic variability with SGLTi use.70,72,73,87 While improved overnight glucose control is novel in SGLTi use with routine insulin therapy, HCL has already shown superior nocturnal glycemia.18,20,88 Given daytime hyperglycemia is the main obstacle, SGLTi-induced reductions in glucose variability may blunt these rises. The pilot study for sotagliflozin was one of the few studies looking at postprandial effects of SGLTi, where post-breakfast 3-hour CGM data had lower glucose levels compared to placebo. 89 Though postprandial glucose levels were not reported for Haidar et al. (see Table 2), the daytime time-in-range and standard deviation were significantly reduced in the empagliflozin 25 mg group on HCL compared to placebo on HCL. 86 In Biester et al. 85 the areas under the curve for both meals were reduced in the dapagliflozin group compared to placebo, as was standard deviation of glucose.
SGLTi use also reduces the amount of insulin needed to accomplish these glycemic changes. Basal and bolus insulin doses were decreased in the SGLTi trials.72,73 The comparison of basal vs bolus reduction was usually not assessed except for the EASE trials, which found the ratio unchanged. 64 In the small closed-loop studies using SGLT2i’s, the findings were different. In Biester et al. 85 bolus but not basal insulin was significantly reduced in dapagliflozin compared to placebo. In Haidar et al. 86 empagliflozin combined with simple meal announcement on closed-loop had reduced bolus requirements compared to HCL on placebo, yet with non-inferior changes to glycemic control or basal dose. 86 In that same study, empagliflozin with carbohydrate counting resulted in no statistically significant change in mean basal or bolus dose compared to placebo with carbohydrate counting, but TDD was significantly reduced. Though these are small studies, this may suggest insulin utilization is different when SGLTi is combined with closed-loop therapy, and urinary excretion of glucose could bypass the difficulties in insulin pharmacokinetics in HCL.
Alleviation of Carbohydrate Counting
Though commercial HCL may remove a certain amount of nutritional distress, 90 simplified meal announcements and fully closed-loop are the next frontiers in closed-loop insulin delivery systems research.91-95 The main objectives of the studies described in Table 2 were to alleviate carbohydrate counting with the use of closed-loop therapy. While Biester et al demonstrated time-in-range was higher with SGLT2i compared to placebo without meal announcement, Haidar et al. 86 revealed this is still inferior to carbohydrate counting. Adding SGLTi pharmacotherapy to reduce glucose levels while simultaneously optimizing automated insulin delivery through bolus simplification may relieve some of the burden of carbohydrate measurement.
Improved Metabolic Outcomes and Potentially Reductions in Complications
HCL has increased the ability to achieve target glycemic control, which in itself has shown improvements in micro- and macrovascular complications,2,3 but glucose control alone cannot eliminate the increased mortality and vascular complications seen in T1D.53,58 The latest advances in T2D pharmacotherapy, alternatively, have shown advantages in weight management, and renal and cardiovascular protection independent of glucose-lowering properties.96-98
Intensive insulin therapy is known to cause difficulties in weight loss. 99 A comparison of pharmacotherapies in T1D demonstrated SGLTi’s as one of the most effective pharmacotherapies for weight loss.59,63-66,68,100,101 Though SGLTi’s have not yet had an RCT in T1D dedicated to renal outcomes, post-hoc analyses from DEPICT revealed reduction in albuminuria with dapagliflozin; 102 a meta-analysis also revealed benefits with sotagliflozin. 103 A study using empagliflozin 25 mg for 8 weeks in those with T1D demonstrated that in clamped euglycemia, renal hyperfiltration (an early sign of diabetic nephropathy) was attenuated by the use of empagliflozin. 104 In the trial DAPA-CKD where dapagliflozin was administered to participants with chronic kidney disease, of whom 32% did not have diabetes, reductions in progression of chronic kidney disease were seen in those taking dapagliflozin, independent of diabetes diagnosis. 105 Because benefits were seen without diabetes, their mechanism of action may then be also protective in those with T1D.
In the EMPEROR and DAPA-HF trials, cardiac outcomes were assessed in those with heart failure, with or without T2D, after using empagliflozin and dapagliflozin respectively, which showed cardio-protective benefits regardless of diabetes diagnosis.106,107 Unfortunately, EMPEROR excluded those with any history of ketoacidosis, while DAPA-HF excluded those with T1D altogether. Heart failure, whether it be due to preserved or reduced ejection fraction, is often underdiagnosed with individuals with T1D. 108 SGLT2i's may at least improve the cardiovascular risk of hypertension; many of the studies of SGLTi as adjunct in T1D showed blood pressure reductions (Table 1). Though SGLTi use may slightly increase both LDL and HDL cholesterols, this was not linked with increased cardiac risk.96,109
Potential Risks of SGLT Inhibitor Use in Closed-Loop Therapy
The main barrier to implementation of SGLTi use in T1D care is the fear of DKA. It is a common concern in T1D; in the T1D Exchange, 2%-7% of adults with T1D had an episode of DKA in 2016. 110 This is particularly concerning because RCT data often underestimates the complications eventually seen in real-world data, as what was seen with DKA with SGLT2i use in T2D.111-114
The studies assessing SGLTi use in closed-loop therapy were too small to assess DKA risk but demonstrate slight but significant elevations in ketone levels.85,86 A case report of DKA with SGLTi while on HCL has previously been published, 115 which highlights the risks of ketoacidosis during reduced insulin delivery and decreased patient oversight when a pump’s algorithm is fully trusted. Though these risks differ from non-automated pump therapy, the known risks on routine pump therapy, such as catheter malfunctions, are still present. Many of the alarms on commercial systems to warn of ketone risk are geared towards hyperglycemia, which do not acknowledge euglycemic ketoacidosis with SGLTi use.
So why the increased risk? Increased urinary glucose excretion reduces insulin requirements, increasing lipolysis and fatty acid delivery to the liver, as well as increasing glucagon secretion, thus switching energy use to ketosis.116-118 In a study where markers of lipolysis were measured during insulin withdrawal in those with T1D on dapagliflozin vs placebo, the increase in lipolysis markers were higher in those taking dapagliflozin compared to the expected rise in placebo. 119
Further subanalyses pertaining to DKA were performed within the large studies assessing SGLTi use in T1D. As the first large trial, the canagliflozin study originally lacked strategic DKA prevention strategies. 59 All cases were linked to known precipitants of DKA such as acute illness or catheter malfunctions without differences in baseline characteristics from the study population. 120 For InTandem-1,2, the exposure-adjusted incidence rates (EAIRs) for DKA were 3.1 events per 100 person-years for sotagliflozin 200 mg and 4.2 per 100 person-years for sotagliflozin 400 mg, in comparison to 0.2 for placebo. Larger reductions in TDD were associated with increased risk of DKA. Those who developed DKA on sotagliflozin were (for sotagliflozin 400 mg): more likely to be female (EAIR 6.2), on pump therapy (EAIR 6.0), with TDD < 0.7 IU/kg (EAIR 4.9), and body mass index (BMI) < 27 kg/m2 (EAIR 4.4). Higher BMI may be protective due to lower rates of lipolysis compared to lean individuals.121,122 A detailed risk mitigation plan initiated later on in InTandem-1 and -2 allowed for reductions in DKA incidence rates. 123
Risk factors similar to InTandem were seen in the EASE trials, where of the 72 cases of DKA, 53 were in women, and 48 were with pump therapy. 64 The increased risk of DKA in women is seen independent of SGLT2i use; in the T1D exchange, DKA rates were higher in women. 110 This could be related to an increased rate of lipolysis in women. 124
Going Forward: Research and Clinical Strategies to Optimize SGLTi Use in HCL
There are potential benefits and risks by adding SGLTi pharmacotherapy to HCL. Given the small studies, more research must be done. Though empagliflozin 2.5 mg’s effect on CGM did not reach significance in EASE-3, 64 its potential may be synergistic with HCL and enough to reduce the post-prandial excursions seen in HCL. Further studies are underway to assess empagliflozin’s utility in HCL (NCT04450563, NCT03979352, NCT04201496). Studies of larger sample sizes and longer duration will aid in assessing therapeutic effects, specifically (1) degree of glycemic improvement, particularly in those who do not achieve goals on HCL, (2) what aspect of HCL is modified (ie, bolus vs basal changes, prandial vs fasting glucose), and (3) the risks associated with this regimen. Further observational studies in commercial HCL systems will also provide guidance.
Risk mitigation must be accomplished prior to widespread use of this regimen. This involves revision of clinical strategies and appropriate patient selection. While prior recommendations have described the targeted population for this therapy,125,126 Table 3 expands on these concepts to HCL and other potential benefits.
A Consideration of Patient Choice in Strategic SGLTi Use as Adjunct to Closed-Loop Therapy in Those with Type 1 Diabetes.
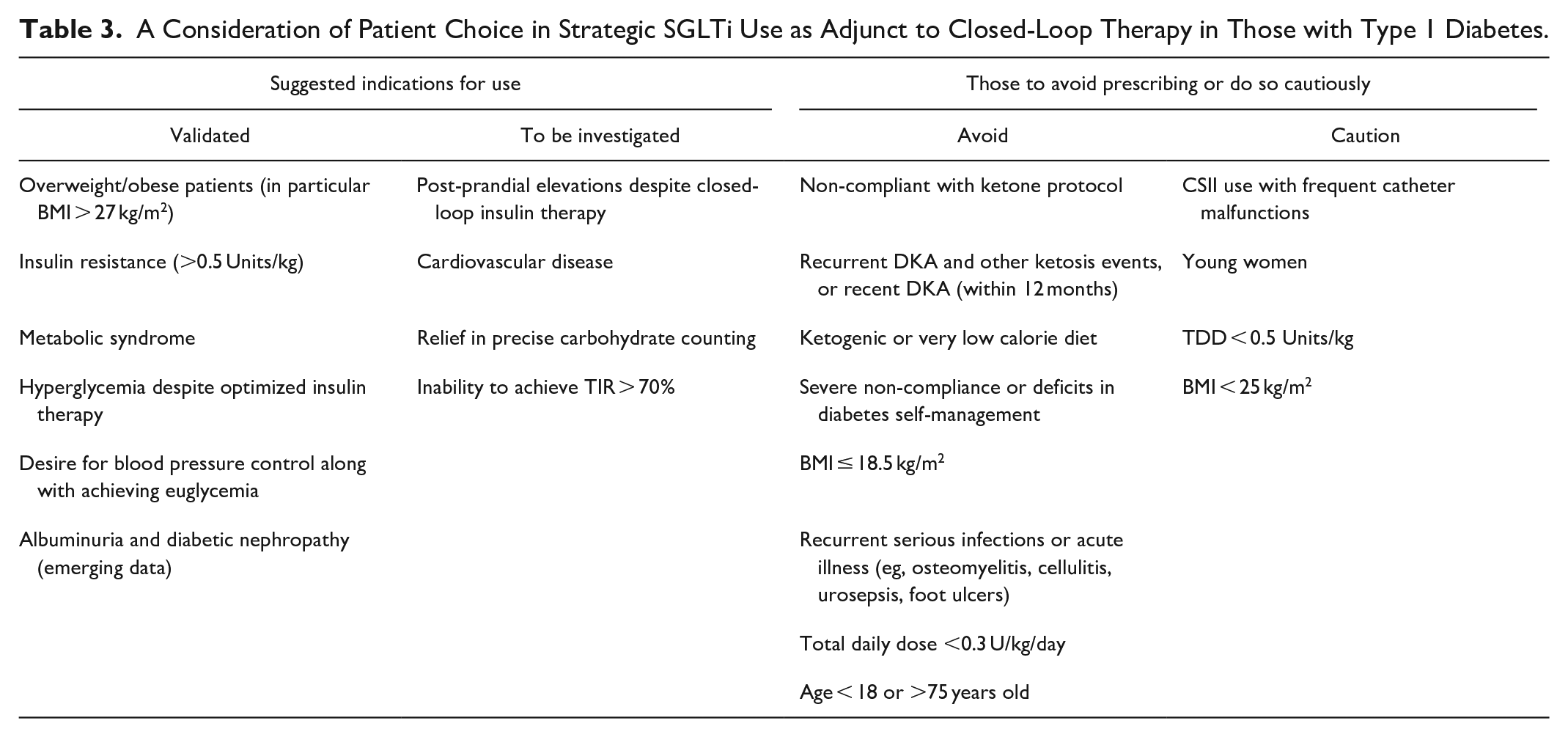
The most important method to reduce risk of DKA is ketone monitoring. Very recently a continuous ketone monitor device has been proposed. 127 Ideally, the device’s ketone data could be integrated into the insulin-dosing algorithm to increase insulin requirements in order to reduce ketone levels in an additional closed-loop fashion. Until this is feasible, clinical and technological strategies are required. The aforementioned risk factors are incorporated into various guidelines to mitigate DKA risk in SGLT2i use in T1D.126,128 Both the STICH and STOP-DKA are protocols that describe approaches to mitigating DKA risk and reducing ketone levels with SGLTi use in T1D.125,129 Unfortunately, these do not address HCL. To avoid the risk of DKA, not only from routine pump malfunction but from possible minimal or suspended insulin delivery, clear recommendations must be given. These include recommended carbohydrate intake, additional insulin bolus doses, or potentially, turning off the algorithm in order to manually increase insulin delivery.
Conclusions
As it emerges into commercial use, HCL is becoming a life-changing therapy and may eventually dominate insulin pump therapy as CGM becomes standard-of-care. As those with T1D move onto closed-loop therapy, they may want to bring their pre-existing SGLTi prescriptions with them. Those who transition to HCL may also find themselves with difficulties in reducing post-prandial hyperglycemia, or struggling with weight loss or vascular complications. The use of SGLTi could optimize HCL, and would efficiently add the benefits of weight loss, blood pressure control, and renoprotection that HCL alone cannot offer. The use of SGLTi in T1D remains a controversial topic, yet there are significant potentials for closing the gaps in routine care.
Footnotes
Acknowledgements
None
Abbreviations
BMI, body mass index; CGM, continuous glucose monitoring; DKA, diabetic ketoacidosis; EAIR, exposure adjusted incidence rates; HbA1c, glycated hemoglobin; HCL, hybrid closed-loop therapy; RCT, randomized controlled trial; SGLT(i), sodium-glucose linked transporter (inhibitors); T1D, type 1 diabetes; T2D, type 2 diabetes; TDD, total daily dose; TIR, time in target range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.H. received research support/consulting fees from Eli Lilly, Medtronic, Aga- Matrix, Adocia, and Dexcom and has pending patents in the artificial pancreas area. M.A.T. received re- search support from AgaMatrix, consulting fees from Sanofi and Adocia, and speaker honoraria from Eli Lilly, Novo Nordisk, Boehringer Ingelheim, Janssen, and AstraZeneca. No other potential conflicts of interest relevant to this article were reported.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
