Abstract
Background:
Recent advances in diabetes care and technology, such as real-time continuous glucose monitoring, can help people live more freely, with more flexibility and fewer constraints, thereby enhancing quality of life (QOL). To date, there has been no validated means for measuring this key psychological dimension. We developed the Diabetes Constraints Scale (DCS) to assess perceived constraints pertaining to diabetes self-management.
Methods:
Six items were developed from qualitative interviews (20 adults with type 2 diabetes [T2D], 8 adults with type 1 diabetes [T1D]). Items were included in one study with T2D adults (N = 458) and one with T1D adults (N = 574). Scale reliability was analyzed for each study using exploratory factor analyses. Associations between DCS and key psychosocial and glycemic variables were assessed.
Results:
In both studies, factor analyses revealed a single factor, with adequate internal reliability (Cronbach’s alpha >.80). Both studies demonstrated significant associations in the expected direction between DCS and overall well-being, diabetes-specific QOL, and diabetes distress (all P < .001). In both studies, DCS was positively linked with the number of missed insulin boluses and the frequency of severe hypoglycemic episodes (T1D both P < .001; T2D both P < .005) and—in the T1D group only—with HbA1c (P < .001).
Conclusions:
The DCS is a reliable and valid method to determine the degree to which adults with diabetes feel constrained or limited by the disease. It may serve as a useful tool for assessing how new interventions can help individuals feel freer in the face of the demands of diabetes.
Introduction
Decades of research have documented how living with diabetes can negatively impact quality of life (QOL), impairing one’s physical and emotional health as well as one’s work and social life.1,2 Quantitative and qualitative data suggest that one major contributor to diminished diabetes-related QOL (dQOL) is the individual’s sense of feeling constrained, limited, and/or controlled by the demands of diabetes.2,3 When a person comes to believe that their personal freedom—their sense of spontaneity and flexibility in life—must be compromised to manage diabetes effectively, dQOL may understandably suffer.
Recent evidence suggests that new diabetes medications and devices may ease at least some of these dQOL constraints, allowing individuals with type 1 diabetes (T1D) or type 2 diabetes (T2D) to experience a reduced sense of cognitive and behavioral strain and responsibility.4 -6 As the attention and effort required to manage diabetes effectively lessens, there is an expanded sense of freedom and adaptability to live one’s life with fewer restraints. For example, reductions in perceived diabetes-related constraints have been documented in qualitative studies assessing the effect of new insulins for T2D adults4,5 and hybrid closed-loop systems for T1D adults. 6 These and other relatively recent innovations in diabetes care (eg, real-time continuous glucose monitoring [RT-CGM], glucagon-like peptide 1 [GLP-1] therapy, inhaled insulin) may help people live more freely, with more flexibility and fewer constraints. Indeed, these may be the key means by which dQOL can improve.
Until now, however, there has been no direct way to measure this critical dimension. Several scales include single or multiple items that touch on this construct—such as the “your freedom to eat as you wish” item on the DAWN2 Impact of Diabetes Profile (DIDP) 7 and the “it bothers me that diabetes seems to control my life” item on the Type 1 and Type 2 Diabetes Distress Assessment Systems (T1-DDAS and T2-DDAS),8,9 but no comprehensive scale has yet been developed that targets this dimension specifically. The development and validation of a brief tool to measure the perceived constraints pertaining to diabetes self-management could contribute to a more comprehensive assessment of individual responses to diabetes treatments, including new medications and devices, which can influence dQOL positively or negatively.
To address this need, we developed the six-item Diabetes Constraints Scale (DCS) and included it in two different studies—one with T2D adults 10 and one with T1D adults. 9 This report documents the construction, evaluation, and validation of the DCS: relationships between the DCS and key psychosocial and glycemic variables and associations between diabetes-related perceived constraints and patient characteristics.
Study 1 (Type 2 Diabetes)
Research Design and Methods
Objectives, participants, and procedures
Polonsky and Soriano 10 conducted a six-month, prospective, quasi-experimental study examining psychosocial, glycemic, and behavioral changes among T2D adults on multiple daily injections (MDIs) who were interested in starting a continuous subcutaneous insulin infusion (CSII) device (Omnipod DASH). The study compared those who then chose to start on DASH vs those who decided to remain on MDI. The current report is based solely on the study’s baseline data, which were collected prior to any decisions regarding the initiation of DASH.
The MDI-using adults with T2D who contacted Insulet Corporation about starting on Omnipod DASH were invited to participate in the Behavioral Diabetes Institute’s independent online survey study. Inclusion criteria were as follows: T2D duration ≥5 years, age ≥25 years, MDI use ≥1 year, no prior CSII experience, and actively planning or hoping to start on CSII in the future. A total of 1426 individuals indicated an initial interest in participating and provided their contact information, which was then forwarded by Insulet staff to the research team.
Interested subjects received an email invitation explaining that there would be two online survey batteries to complete—–one at baseline (prior to starting CSII) and one at six months later. Because only baseline data were used for the current report, the results do not reflect participants’ eventual decision about starting DASH. Participants who met screening criteria and completed the baseline survey received a $25 electronic gift card for completing the baseline survey. The research protocol was approved by Ethical and Independent Review Services, a community-based Institutional Review Board.
Scale development
The original idea for the DCS emerged from a small qualitative study that identified a set of individuals with T2D who reported “feeling better” after switching to a new insulin that could be used in a more time-flexible manner than their previous insulin. They were then interviewed in an effort to clarify what exactly “feeling better” meant to them. In total, 20 participants were recruited from two clinical sites in the United States and one in Switzerland. Of the four factors that were found to contribute to “feeling better,” we were particularly intrigued by two: (a) a reduced sense of diabetes as burdensome and requiring excessive attention and (b) an enhanced sense of adaptability, spontaneity, and freedom. 5 As we considered these two factors, we realized that this sense of being constrained or limited by diabetes had never been captured in a coherent manner by any currently available self-report instrument. And so, with these qualitative results, together with comments from our own T2D clinic patients, we constructed a set of items to characterize the constraints underlying these reported benefits. Following the scale construction approach developed by Clark and Watson, 11 we reviewed these items with a second group of T2D adults, leading to further refinement and final selection of the six items as follows: “I spend more time thinking about my diabetes than I’d like to,” “I feel pressure to eat snacks to avoid low blood sugar problems,” “I feel restricted about if and/or when I can exercise,” “I feel limited about what I should and shouldn’t eat,” “I can’t be as spontaneous in my life as I’d like to be,” and “I don’t feel as free to live my life the way I want.” Respondents were asked to rate each item on a Likert-type scale (1 = strongly disagree, 2 = agree, 3 = neutral, 4 = agree, and 5 = strongly agree).
Measures
In addition to the DCS, the survey battery included demographic information (eg, age, gender, race/ethnicity, education, annual income), years since diabetes diagnosis, diabetes history, the most recent self-reported HbA1c value (within the past three months only), the number of missed insulin doses in the past week, and the number of severe hypoglycemic episodes (ie, requiring the assistance of another person) in the past month. Also assessed were relevant psychosocial dimensions, including the DIDP, 7 which assesses the perceived impact of diabetes on QOL; the World Health Organization Well-Being Index (WHO-5), 12 which assesses overall QOL and well-being; the Core scale of the Type 2 Diabetes Distress Assessment System (T2-DDAS), 8 which measures overall diabetes distress; and the Hypoglycemia Attitudes and Behavior Scale (HABS), 13 which evaluates hypoglycemia-related Anxiety, Avoidance, and Confidence.
Data analysis
Analyses were conducted with SPSS software (IBM Corp, 2023, Version 29.0.2.0). To determine the underlying factor structure of the DCS, an exploratory factor analysis was performed on its six items using the Kaiser-Guttman rule (retain factors with eigenvalues >1.0; Kaiser, 1960). After evaluating the factor structure of the DCS, bivariate correlations were calculated between the DCS total score and other measures to evaluate construct validity. Correlations were computed between DCS and the DIDP, WHO-5, T2-DDAS, and HABS, as well as other demographic and clinical metrics, including age, diabetes duration, self-reported HbA1c, missed insulin doses, and number of severe hypoglycemic episodes (recoded dichotomously as zero vs at least one episode in the prior month). Pearson Product-Moment Correlations were computed for continuous variables, and the Point-Biserial Correlation was calculated for the dichotomous variable.
Results
Sample characteristics
Of the 854 who responded to the invitation, 458 (53.6%) were eligible and completed the baseline survey. Most were female (60.7%) and non-Hispanic white (70.3%), and 50.0% reported an annual household income <$50,000 (see Table 1). Mean age was 52.1 (±14.6) years, mean body mass index was 35.9 (±8.9) kg/m2, and mean HbA1c was 8.8% (±2.0). Most participants (68%) had T2D for more than ten years.
Sample Characteristics.
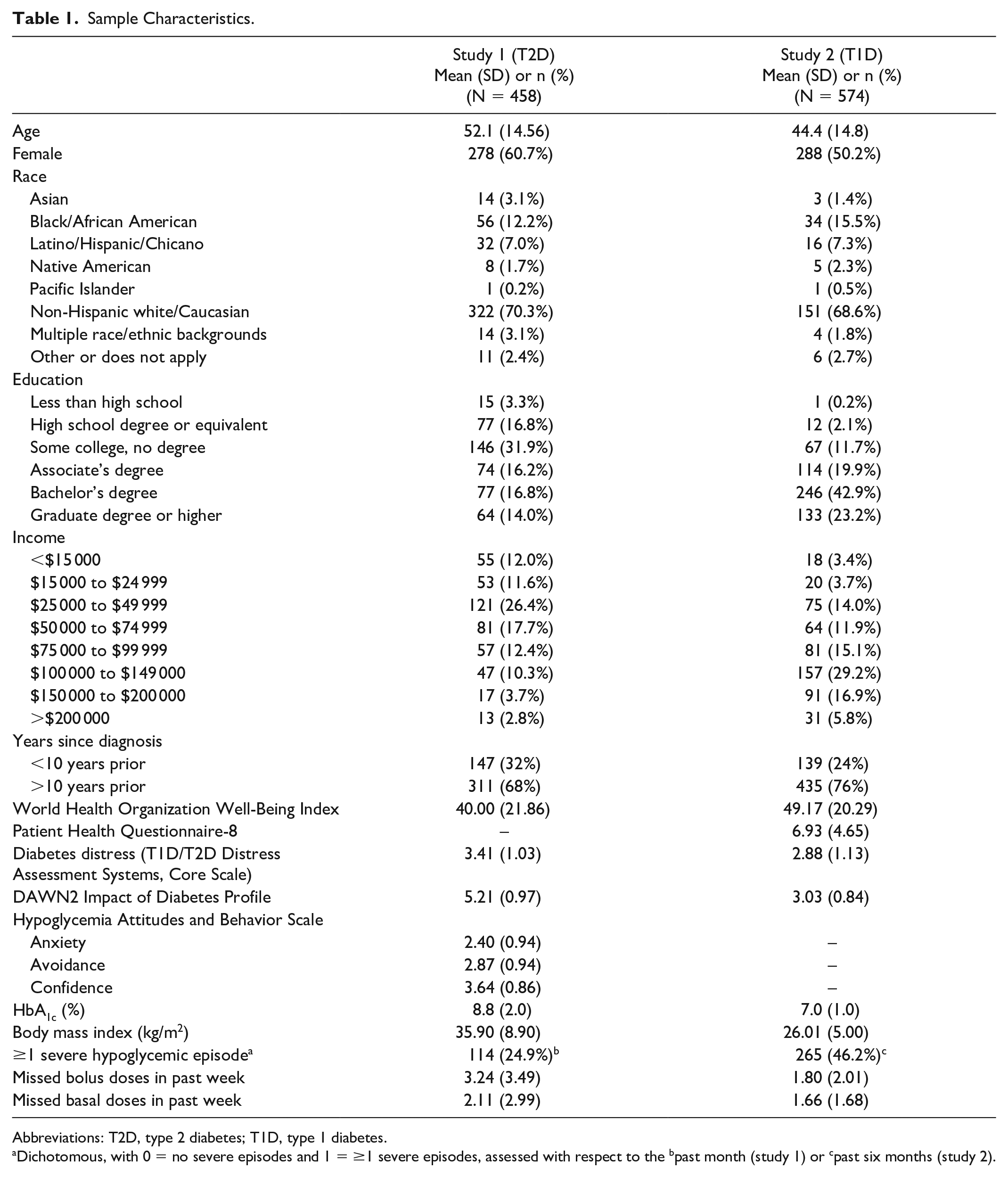
Abbreviations: T2D, type 2 diabetes; T1D, type 1 diabetes.
Dichotomous, with 0 = no severe episodes and 1 = ≥1 severe episodes, assessed with respect to the bpast month (study 1) or cpast six months (study 2).
Factor structure
Exploratory factor analysis revealed that only a single factor met the Kaiser-Guttman eigenvalue >1.0 criterion (eigenvalue = 3.03), explaining 50.6% of the total variance. Factor loadings are shown in Table 2; all were significant (P < .001). Cronbach’s alpha for the scale = .80, indicating good internal reliability. The six items of the DCS, including descriptive statistics, are listed in Table 3. Higher DCS scores indicate greater perceived constraints.
Diabetes Constraints Scale Item Loadings From Exploratory Factor Analysis.
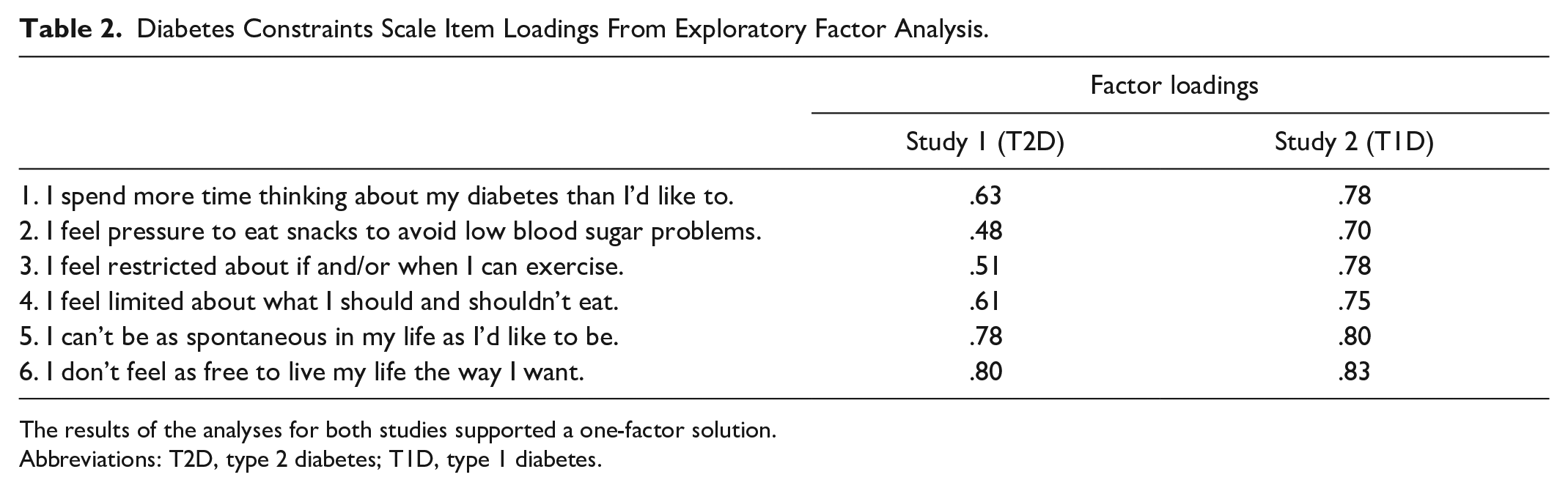
The results of the analyses for both studies supported a one-factor solution.
Abbreviations: T2D, type 2 diabetes; T1D, type 1 diabetes.
Descriptive Statistics for Diabetes Constraints Scale Items and Total Score.
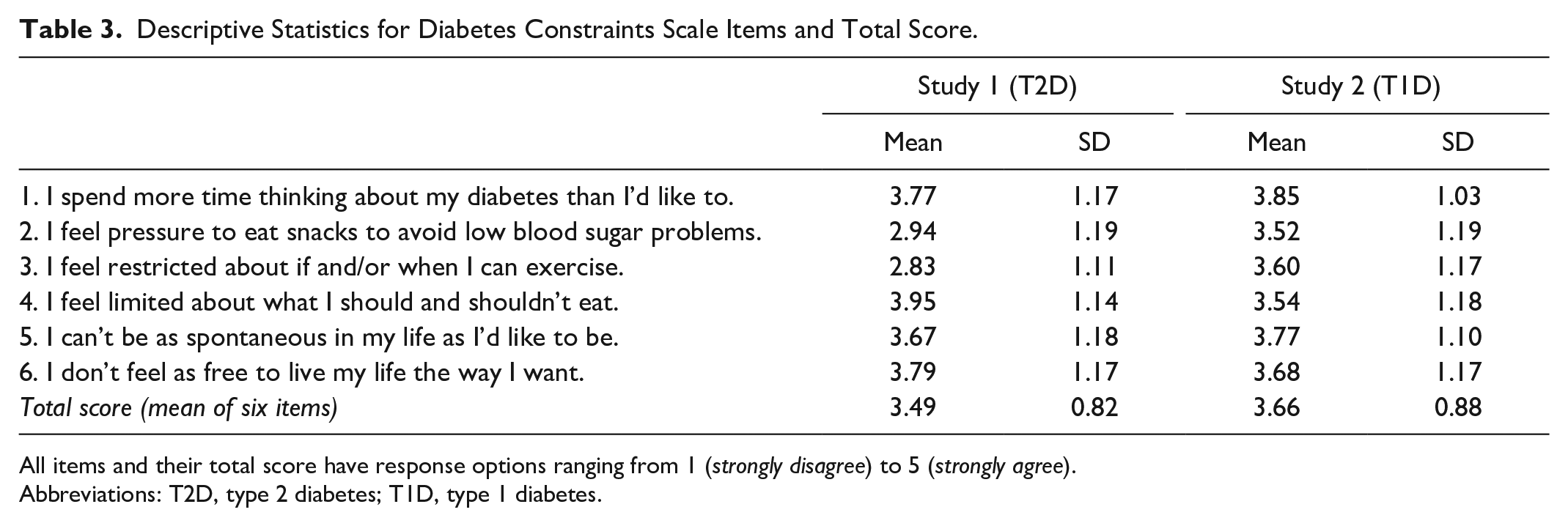
All items and their total score have response options ranging from 1 (strongly disagree) to 5 (strongly agree).
Abbreviations: T2D, type 2 diabetes; T1D, type 1 diabetes.
Construct validity
Bivariate correlations between the DCS, other baseline self-report measures, and demographic and clinical metrics are shown in Table 4. The DCS scores were negatively associated with overall well-being (WHO-5 r = − .37), dQOL (DIDP r = −.29), and hypoglycemic confidence (HABS Confidence r = −.28), and were positively associated with diabetes distress (T2-DDAS Core scale r = .51), hypoglycemic worries (HABS Anxiety r = .43), and hypoglycemia-related avoidance behavior (HABS Avoidance r = .42) (all P < .001). The DCS scores were also positively associated with missed insulin doses, including bolus (r = .15, P = .002) and basal (r = .16, P = .001) doses, and the dichotomized number of hypoglycemic episodes in the prior month (r = .13, P = .005). Correlations between the DCS and gender, age, diabetes duration, and self-reported HbA1c were not statistically significant.
Bivariate Correlations Between Diabetes Constraints Scale and Other Measures.
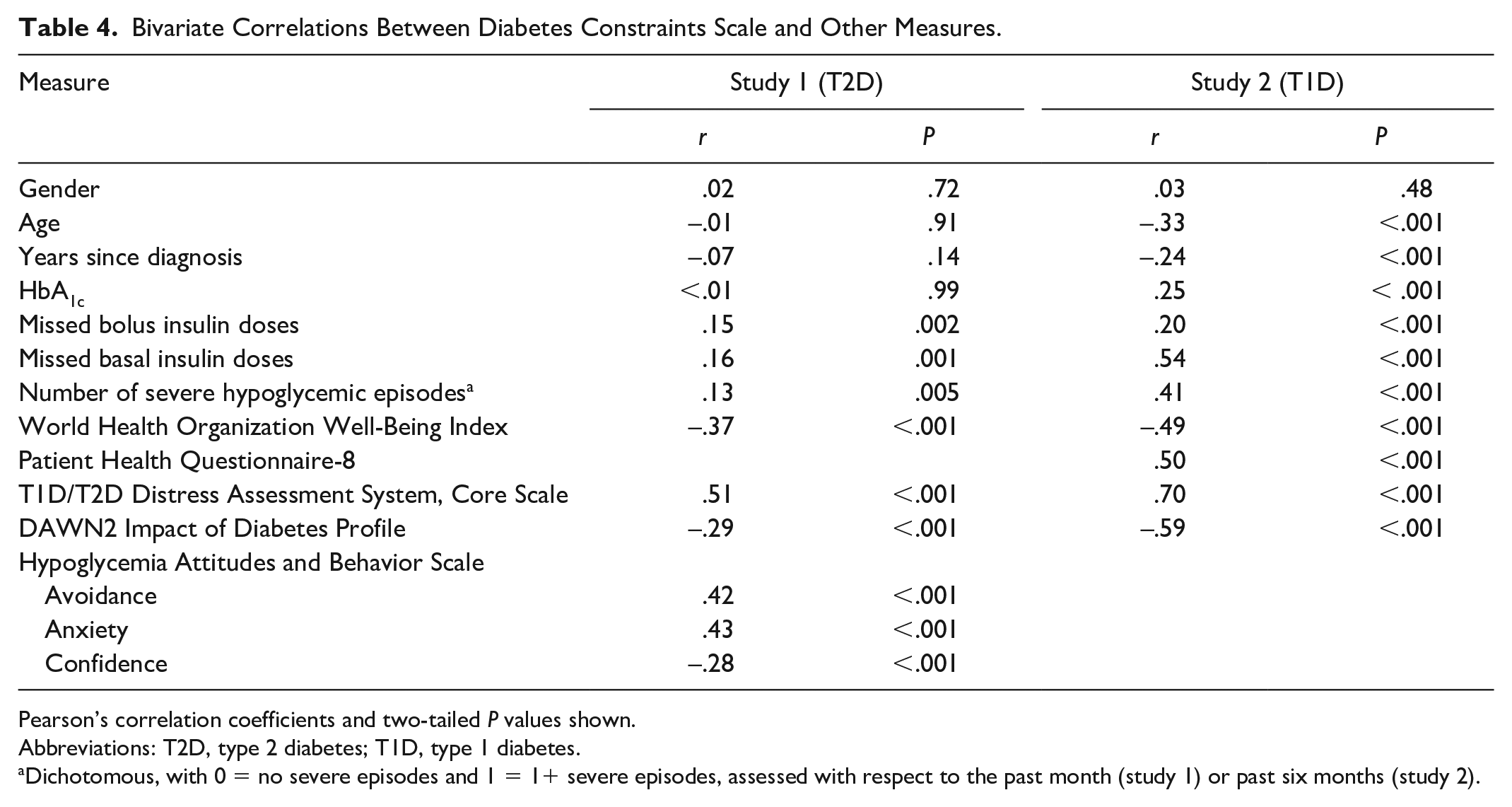
Pearson’s correlation coefficients and two-tailed P values shown.
Abbreviations: T2D, type 2 diabetes; T1D, type 1 diabetes.
Dichotomous, with 0 = no severe episodes and 1 = 1+ severe episodes, assessed with respect to the past month (study 1) or past six months (study 2).
Summary
Results show that the DCS is a coherent, reliable, and valid measure for assessing the degree to which MDI-using adults with T2D feel constrained by diabetes. The DCS demonstrated good internal consistency and was significantly associated with overall well-being, diabetes distress, and diabetes QOL, in the expected directions, providing evidence of the scale’s reliability and validity.
Study 2 (Type 1 Diabetes)
Research Design and Methods
Objectives, participants, and procedures
To develop and validate a new measure of diabetes distress for adults with T1D (the T1 Diabetes Distress Assessment System or T1-DDAS), Fisher and colleagues 9 asked participants to complete a multipart assessment battery that included demographic and disease variables, key psychosocial factors, and a draft version of the new DCS (as described in study 1). Participants completed the same assessment battery six months later. The DCS was included only in the six-month survey; these data are the basis for this report.
Adults were recruited nationally from multiple sources, including the TCOYD Research Registry and the T1D Exchange. Main inclusion criteria were T1D duration ≥1 year and age ≥21 years. A total of 650 people completed the baseline survey, of whom 574 (88.3%) completed the six-month survey. Participants received a $50 electronic gift card for completing each survey. The research protocol was approved by Ethical and Independent Review Services, a community-based Institutional Review Board.
Measures
The DCS items, originally developed for adults with T2D in study 1, were reviewed informally by eight T1D adults to determine if the six items were relevant to this population and to indicate whether any changes or additional items would be necessary. There was general agreement that the items adequately covered the experience of diabetes-related constraints for T1D adults. In addition to the DCS, the survey included demographic information (eg, age, gender, race/ethnicity, education), years since diabetes diagnosis, most recent self-reported clinical HbA1c value (within the past three months only), the number of missed insulin boluses in the past week, and the number of severe hypoglycemic episodes (requiring the assistance of another) in the past six months. The number of episodes was recoded dichotomously as zero vs at least one episode in the past six months. Also assessed were relevant psychosocial dimensions, including perceived impact of diabetes on QOL (DIDP), overall well-being (WHO-5), diabetes distress (Core scale of the T1-DDAS), and depressive symptoms (PHQ-8). 14 Note that the T1-DDAS Core Scale (used in study 1) and the T2-DDAS Core scale (used in study 2) are identical.
Data analysis
The same exploratory factor analysis outlined for study 1 was repeated in study 2. Likewise, bivariate correlations were estimated between the DCS and other measures to establish construct validity and relations with demographic variables.
Results
Sample characteristics
Of the 574 respondents who completed the survey, the majority were female (50.2%) and non-Hispanic white (66.7%), and 21.1% reported an annual household income <$50,000 (see Table 1). Mean age was 44.4 (±14.8) years, mean HbA1c was 7.0% (±1.0), and mean diabetes duration was 22.7 (±15.6) years.
Factor structure
One factor met the Kaiser-Guttman eigenvalue >1.0 criterion (eigenvalue = 3.60), explaining 59.9% of the total variance. Standardized factor loadings are shown in Table 2; all were significant (P < .001). Cronbach’s alpha for the six items = .87, indicating good internal reliability. Descriptive statistics of the six items are presented in Table 3.
Construct validity
The DCS scores were negatively associated with overall well-being (WHO-5 r = −.49) and dQOL (DIDP r = −.59) and were positively associated with diabetes distress (T1-DDAS Core scale r = .70) and depressive symptoms (PHQ-8 r = .50). The DCS scores were also positively associated with the number of severe hypoglycemic events (dichotomized) over the past six months (r = .41), frequency of missed bolus (r = .20) and basal (r = .54) insulin doses, and self-reported HbA1c (r = .25) (all P < .001) (see Table 4).
Summary
Similar to study 1 with T2D adults, the results of study 2 with T1D adults indicated that the DCS is a coherent, valid, and reliable measure for assessing how strongly adults feel limited by diabetes. The DCS demonstrated adequate internal reliability and was significantly associated with other relevant variables, such as overall well-being, diabetes distress, dQOL, depressive symptoms, and HbA1c, all in expected directions.
Discussion
The DCS is a brief self-report measure developed to assess how people with diabetes perceive the limitations and constraints associated with diabetes and its management. To the best of our knowledge, this is the first measure to directly target this important dimension of the diabetes experience. We found that the internal consistency of the DCS was adequate in both studies, and its construct validity was supported in both studies by significant negative associations between the DCS and overall well-being and dQOL, and significant positive associations with diabetes distress and the number of missed insulin boluses. Recent frequency of severe hypoglycemia was also linked with higher DCS scores in both groups. Finally, DCS was positively associated with HbA1c, but only in the T1D group.
In further support of DCS validity, the T1D study also examined depressive symptoms, which were positively associated with DCS scores. The T2D study examined hypoglycemic attitudes, where the DCS was significantly linked to greater hypoglycemic anxiety and avoidance behaviors and lower levels of hypoglycemic confidence.
The mean DCS score was >3.0 in both samples, with a response of 3 indicating “neutral.” Overall, 79.4% of T1D study participants and 71.8% of T2D study participants reported a mean DCS score >3.0, suggesting that feelings of being constrained by diabetes are common. Inspection of individual DCS item scores points to expected differences between the two samples. The largest numerical differences between the T1D and T2D samples (>0.4; see Table 3) were for items regarding hypoglycemic concerns (“feel pressure to eat snacks to avoid low blood sugar problems,” “feel restricted about if and/or when I can exercise”), which had higher mean values in the T1D group compared with the T2D group. Because hypoglycemic fear is reported to be more widespread in T1D than T2D populations, 15 it makes sense that the T1D sample perceived greater constraints due to hypoglycemia. A second notable between-group difference occurred on the item, “I feel limited about what I should and shouldn’t eat,” where the T2 sample scored markedly higher than the T1D sample. This may reflect the additional pressure that people with T2D often experience, typically due to longstanding weight management concerns and/or social stigma. In this regard, the DCS appears sufficiently sensitive to detect differences between T1D and T2D respondents.
Despite the rapid spread and uptake of new technologies, such as RT-CGM, where patients in the clinical setting typically report significant glycemic and dQOL benefits, it is striking that studies to date have uncovered relatively small, or absent, quality of life benefits.16,17 We suspect that one reason for this discrepancy is that feeling less constrained by diabetes and freer to live one’s life (a likely chief benefit of these innovations) has not been directly and adequately assessed until now. Indeed, the sense of feeling constrained fits well within the broader context of motivational theory, where the role of perceived autonomy is highlighted as a critical contributor to intrinsic motivation. 18 When self-management actions are feeling less constraining, the individual may perceive them as more positive, autonomous choices, thereby enhancing motivation. Future studies to assess the ability of the DCS to predict and detect clinically relevant change over time in response to the introduction of new diabetes technologies should help to determine the further utility of the DCS.
This study has several limitations. First, both samples are comprised of adults with elevated levels of diabetes distress and may not represent the broader T1D and T2D populations in the United States. Second, more than two thirds of both groups were non-Hispanic white, and all participants were English speakers. Third, the study did not document change in the DCS over time; future research should assess the ability of the DCS to detect how feelings of being limited by diabetes respond to the use of new diabetes technologies. Last, because all measures, including HbA1c, were self-reported, results should be viewed with caution. However, prior studies have reported close agreement between self-reported and laboratory-assessed HbA1c (r = .84). 19
Conclusions
These results suggest that the DCS is a reliable and valid method to quantify the degree to which adults with either T1D or T2D feel constrained by their disease. It may prove to be a useful tool to document how new interventions can help people faced with the demands of diabetes management to feel freer.
Footnotes
Abbreviations
QOL, quality of life; DCS, Diabetes Constraints Scale; T1D, type 1 diabetes; T2D, type 2 diabetes; HbA1c, glycosylated hemoglobin; dQOL, diabetes-related QOL; RT-CGM, real-time continuous glucose monitoring; GLP-1, glucagon-like peptide; DIDP, DAWN2 Impact of Diabetes Profile; T1-DDAS, Type 1 Diabetes Distress Assessment System; T2-DDAS, Type 2 Diabetes Distress Assessment System; CSII, continuous subcutaneous insulin infusion; MDI, multiple daily injections; HABS, Hypoglycemic Attitudes and Behavior Scale; WHO-5, World Health Organization-5; TCOYD, Taking Control of Your Diabetes; PHQ-8, Patient Health Questionnaire-8.
Author Contributions
WHP contributed to study conception. ES and LS contributed to statistical analysis. WHP, LF, ECS, and LAS contributed to interpretation of data. WHP, LF, ECS, and LAS contributed to manuscript development. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Behavioral Diabetes Institute licenses the DCS to pharmaceutical companies and other for-profit organizations; otherwise, the DCS is available free of charge for use in clinical care and research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: While the current investigator-initiated study received no external funding, the original studies from which these data were derived did receive support from Insulet (study 1) and the Leona M. and Harry B. Helmsley Charitable Trust (study 2).
