Abstract
Background:
Although real-time continuous glucose monitoring (rtCGM) has been shown to improve glycemic control in patients with type 1 diabetes mellitus and type 2 diabetes mellitus treated with insulin, rates of adoption have been low. A novel approach, with the use of a long-term implantable continuous glucose monitoring (LTI CGM) has the potential to overcome barriers to rtCGM. The purpose of this review is to provide a background on the first LTI CGM technology to be approved, along with a review of contraindications, interference, safety, accuracy, and efficacy. Considerations for patient selection are discussed based on the available evidence.
Methods:
PubMed, EMBASE, and Cochrane Library were searched for keywords and subject headings to identify studies assessing LTI CGM.
Results:
Seven studies were identified which assessed LTI CGM. Mean absolute relative difference is similar to available CGM devices. Rates of adverse events were low. Change in hemoglobin A1c with LTI CGM may be comparable to rtCGM.
Conclusions:
Based on the available evidence, LTI CGM appears to be safe and accurate. Additional clinical trial investigation is warranted to evaluate the glycemic efficacy of LTI CGM.
Background
Continuous glucose monitoring (CGM) is an alternative to self-monitoring of blood glucose (SMBG) and allows patients with diabetes to react to glycemic patterns, which can inform meal intake, physical activity, carbohydrate counting, and medication adjustments. 1 As compared with SMBG, real-time CGM (rtCGM) and intermittent-scanned CGM (isCGM) technology have been shown to improve glycemic control in patients with type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM).2-5 Although there are clinical benefits to rtCGM and isCGM, a common barrier is the need to frequently replace sensors every 6 to 10 (depending on the device) or 14 days, respectively. Moreover, up to 41% of patients in the T1D registry discontinued rtCGM use in the first year from discomfort, trouble inserting the sensor, poor sensor adherence to skin, cutaneous reactions, and interference with sports and physical activity. 6
Eversense Long-term Implantable CGM
A novel long-term implantable (LTI) subcutaneous CGM device (Eversense CGM System; Senseonics, Inc., Germantown, MD, United States) was approved by the U.S. Food & Drug Administration (FDA) on June 6, 2019, and has been available in the European Union and European Economic Area since May 2016. 7 The device is indicated in the United States for adults aged 18 years or older with diabetes. Historical data from the system can be used nonadjunctively to replace SMBG for making diabetes treatment decisions, although it requires two fingersticks daily with SMBG for calibration. The Eversense LTI CGM system contains an implantable fluorescence-based sensor, transmitter, and a mobile application (downloadable by smartphone or tablet).
Technology
Sensor
The sensor (3.5 × 18.3 mm) contains core electronics and optics, and is sealed in epoxy within a polymethyl methacrylate (PMMA) encasement. A 100-µm-thick copolymer matrix is grafted to the outside of the PMMA encasement. This polymer is fluorescent and uses selective, fully reversible binding between glucose and a covalently attached molecular complex to sense changes in interstitial glucose. When glucose binds, there is an increase in fluorescent intensity, which is measured by the sensor’s optical system. The sensor sends fluorescent measurements to the externally worn transmitter for calculation and storage of glucose values every five minutes via Bluetooth transmission.
Within the sensor is a silicone collar component containing 1.75 mg of dexamethasone, which is released at 3 µg per day over the sensors life to reduce tissue inflammation around the sensor. The sensor must be inserted by a healthcare provider in-office and needs to be replaced every 90 days (Eversense; currently available in the United States) or up to 180 days (Eversense XL; not available in the United States). The sensor is also designed to provide an alert to the user when it needs to be replaced.
Transmitter
The transmitter (1.5 × 1.7 inches; 0.6 inches thick) is worn externally over the inserted sensor and uses Bluetooth low energy technology to wirelessly send interstitial glucose information to the mobile medical application (MMA) for display on a handheld device. The transmitter uses a rechargeable battery and can be charged with a supplied universal serial bus powered cradle. The transmitter is also capable of providing vibration signals to the user for the detection and prediction of episodes of hypo- or hyperglycemia, even if the MMA is not close by the user.
Mobile Medical Application
The MMA receives and displays calculated glucose information from the transmitter. Glycemic information available to display includes real-time glucose readings, glucose trend information, and glucose alerts.
Contraindications and Interference
The device should not be used if the patient is going to undergo magnetic resonance imaging (MRI) procedures, unless the MRI meets criteria including a static magnetic field of 1.5 or 3.0 T, the maximum spatial field gradient is 2000 G/cm, and the maximum whole body specific absorption rate is 4 W/kg. 8 Additional contraindications include lithotripsy, diathermy, or electrocautery. Forty-one potentially interfering substances were evaluated in an in vitro study. 9 Tests were performed at two glucose concentrations (77 and 321 mg/dL) except for sugar alcohols, which were tested at 54, 108, and 271 mg/dL. 9 The results indicated that only two substances, mannitol, and tetracyclines, may interfere with glucose measurement with the LTI CGM system. 9 Notably, neither acetaminophen nor ascorbic acid produced a sensor bias that exceeded International Standardization Organization (ISO) limits. 9
Safety and Accuracy
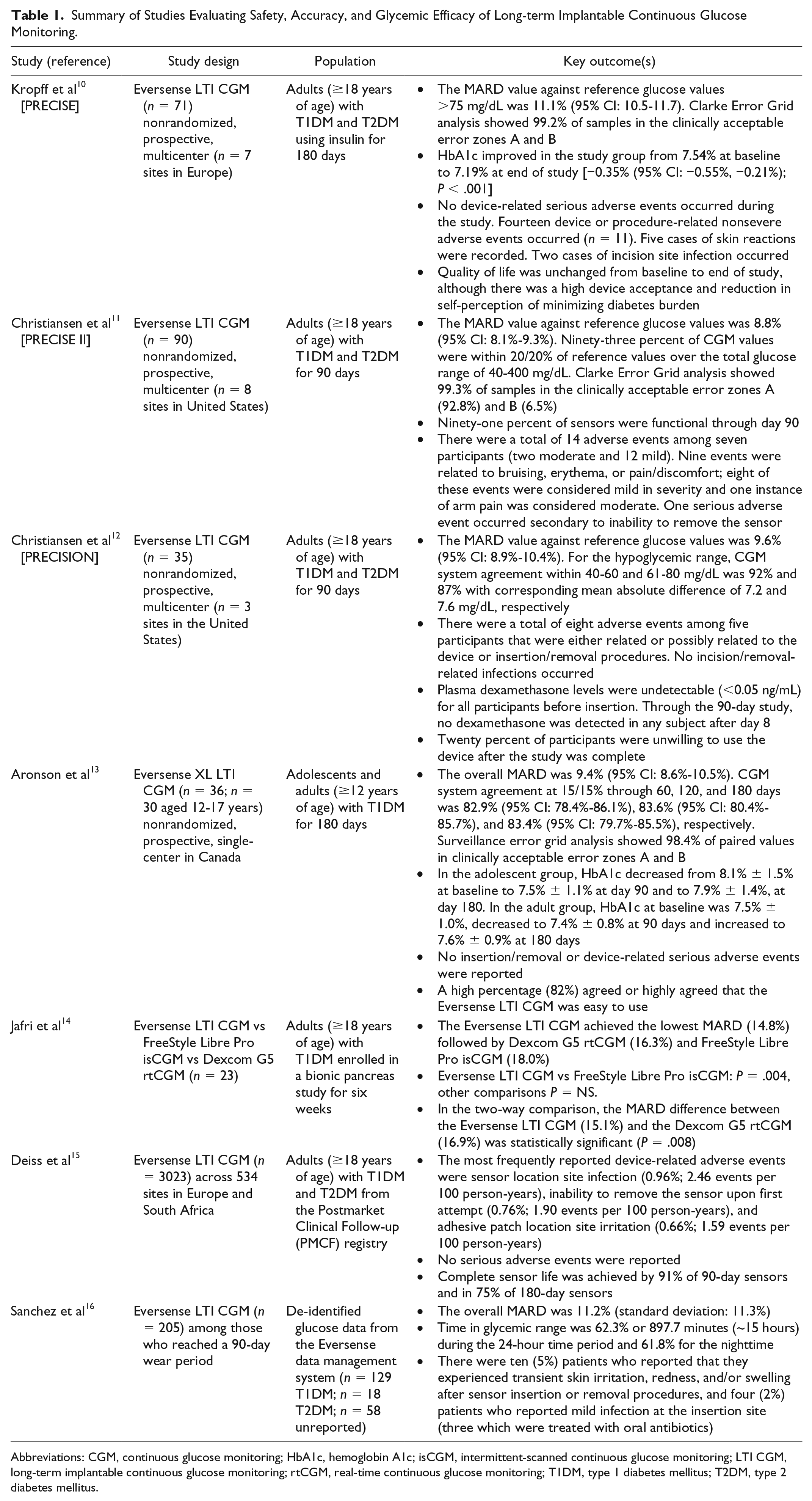
The PRECISE trial evaluated the accuracy, longevity, and impact of the Eversense LTI CGM device for up to 180 days. 10 This study was a prospective, multicenter trial in adults (≥18 years of age) with T1DM and T2DM conducted in Europe. 10 Results demonstrated that the overall mean absolute relative difference (MARD) was 11.1% (95% CI: 10.5%-11.7%) for samples >75 mg/dL, and 84% of CGM values were within 15 mg/dL or 20% of the Yellow Springs Instruments (YSI) reference glucose values. 10 Clarke Error Grid analysis showed 99.2% of samples to be within the clinically acceptable error zones A and B. 10 While no serious device-related adverse events occurred, 14 device- or procedure-related nonsevere adverse events occurred (n = 11). 10 Five cases of skin reactions were recorded. 10 Two cases of incision skin infection occurred of which one required antibiotics. 10 Although quality of life was unchanged at study conclusion, 84% of subjects stated that they would want to use the Eversense LTI CGM in the future and 90% felt that the system helped them minimize diabetes burden. 10
The PRECISE II trial sought to assess the accuracy and safety of the Eversense LTI CGM device in adults (≥18 years of age) with T1DM and T2DM for up to 90 days. 11 This was a nonrandomized prospective blinded, single-arm multicenter study with 90 subjects conducted in the United States. 11 The overall MARD was 8.8% (95% CI: 8.1%-9.3%). Ninety-three percent of Eversense LTI CGM values were within ± 20 mg/dL or 20% of YSI reference glucose values. 11 Clarke Error Grid analysis showed 99.3% of samples to be within the clinically acceptable error zones A (92.8%) and B (6.5%). 11 During the study, 14 adverse events were noted and related to device insertion or removal. 11 Twelve of the events were classified as mild, and two were classified as moderate in severity. 11 There were nine documented events of bruising, erythema, or pain/discomfort. 11 One serious adverse event was documented because the sensor could not be removed on first attempt. 11
In a third study (PRECISION), accuracy and safety of the Eversense LTI CGM device in adults (≥18 years of age) with T1DM and T2DM was further confirmed up to 90 days. 12 This was a nonrandomized, prospective multicenter study with 35 subjects conducted in the United States. Overall MARD was 9.6% (95% CI: 8.9%-10.4%). 12 The overall percentage of CGM values within 15/15% of YSI reference was 85%. 12 Notably, the Eversense LTI CGM system agreement within the hypoglycemic range was 92% between 40 and 60 mg/dL and 87% between 61 and 80 mg/dL with a corresponding MARD of 7.2 and 7.6 mg/dL, respectively. 12 There were a total of eight adverse events among five subjects either related or possibly related to the device or insertion/removal during the study. 12 Twenty percent of participants reported being unwilling to use the device again after the study. 12
Although not approved in the United States, the Eversense XL LTI CGM system has a life span of 180 days as compared with 90 days with the original Eversense LTI CGM system. In a nonrandomized, nonblinded, prospective, single-center study in Canada of patients with T1DM (aged ≥12 years), the overall MARD for the Eversense XL LTI CGM was 9.4% (95% CI: 8.6%-10.5%). 13 The Eversense LTI CGM system agreement at 15/15% (n = 7,163) through 60, 120, and 180 days was 82.9% (95% CI: 78.4%-86.1%), 83.6% (95% CI: 80.4%-85.7%), and 83.4% (95% CI: 79.7%-85.5%), respectively. 13 The surveillance error grid analysis showed 98.4% of paired values to be within clinically acceptable error zones A and B. 13 There was no insertion, removal, or device-related serious adverse events that occurred with the Eversense XL LTI CGM device. 13 In this study, 82% of participants agreed or highly agreed that the Eversense XL LTI CGM was easy to use. 13 No additional studies have been published to date including children or adolescents.
To date, only one study has investigated the accuracy of the Eversense LTI CGM device as compared with the Dexcom G5 rtCGM and the FreeStyle Libre Pro isCGM devices. 14 In this study conducted under real-world conditions, the MARD compared with plasma glucose for the Eversense LTI CGM was 14.8%, Dexcom G5 rtCGM was 16.3%, and FreeStyle Libre Pro isCGM was 18% (Eversense vs FreeStyle Libre Pro, P = .004; other P = NS). 14 Of note, this study was conducted in 23 subjects with T1DM who wore all three devices concomitantly. When taken together, the overall MARD from the PRECISE, PRECISE II, and PRECISION was 11.1% (95% CI: 10.5%-11.7%), 10 8.8% (95% CI: 8.1%-9.3%), 11 and 9.6% (95% CI: 8.9%-10.4%), 12 respectively. The overall MARD is comparable to that seen with several commonly used rtCGMs including the Dexcom G6 (9%), 15 Medtronic Guardian Sensor 3 (9.1%), 16 and Medtronic Guardian Connect (9.4%). 16 MARD with the isCGM (FreeStyle Libre) has been reported to be between 11.4% and 13.2%.17,18
Postmarketing data evaluating the Eversense LTI CGM among adults (≥18 years of age) with T1DM and T2DM (n = 3023) across 534 sites in Europe and South Africa demonstrated that the most frequently reported adverse events were related to sensor location site infection (0.96%; 2.46 events per 100 person-years), inability to remove the sensor upon first attempt (0.76%; 1.90 events per 100 person-years), and adhesive patch location site irritation (0.66%; 1.59 events per 100 person-years). 19 Safety and accuracy data from the aforementioned studies are summarized in Table 1.
Summary of Studies Evaluating Safety, Accuracy, and Glycemic Efficacy of Long-term Implantable Continuous Glucose Monitoring.
Abbreviations: CGM, continuous glucose monitoring; HbA1c, hemoglobin A1c; isCGM, intermittent-scanned continuous glucose monitoring; LTI CGM, long-term implantable continuous glucose monitoring; rtCGM, real-time continuous glucose monitoring; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
Glycemic Efficacy
The PRECISE study found a −0.35% decrease (95% CI: −0.55%, −0.21%); P < .001)] in hemoglobin A1c (HbA1c) at 180 days as compared with baseline HbA1c values. 10 Aronson et al also found that HbA1c decreased at day 90 (−0.6%) and at day 180 (−0.2%) as compared with baseline HbA1c among adolescents. However, among adults in the study, HbA1c decreased (−0.1%) at day 90, but increased (+0.1%) at 180 days. 13 The changes in HbA1c with LTI CGM in these two studies are comparable to that seen with several studies of rtCGM and isCGM.2,20-22
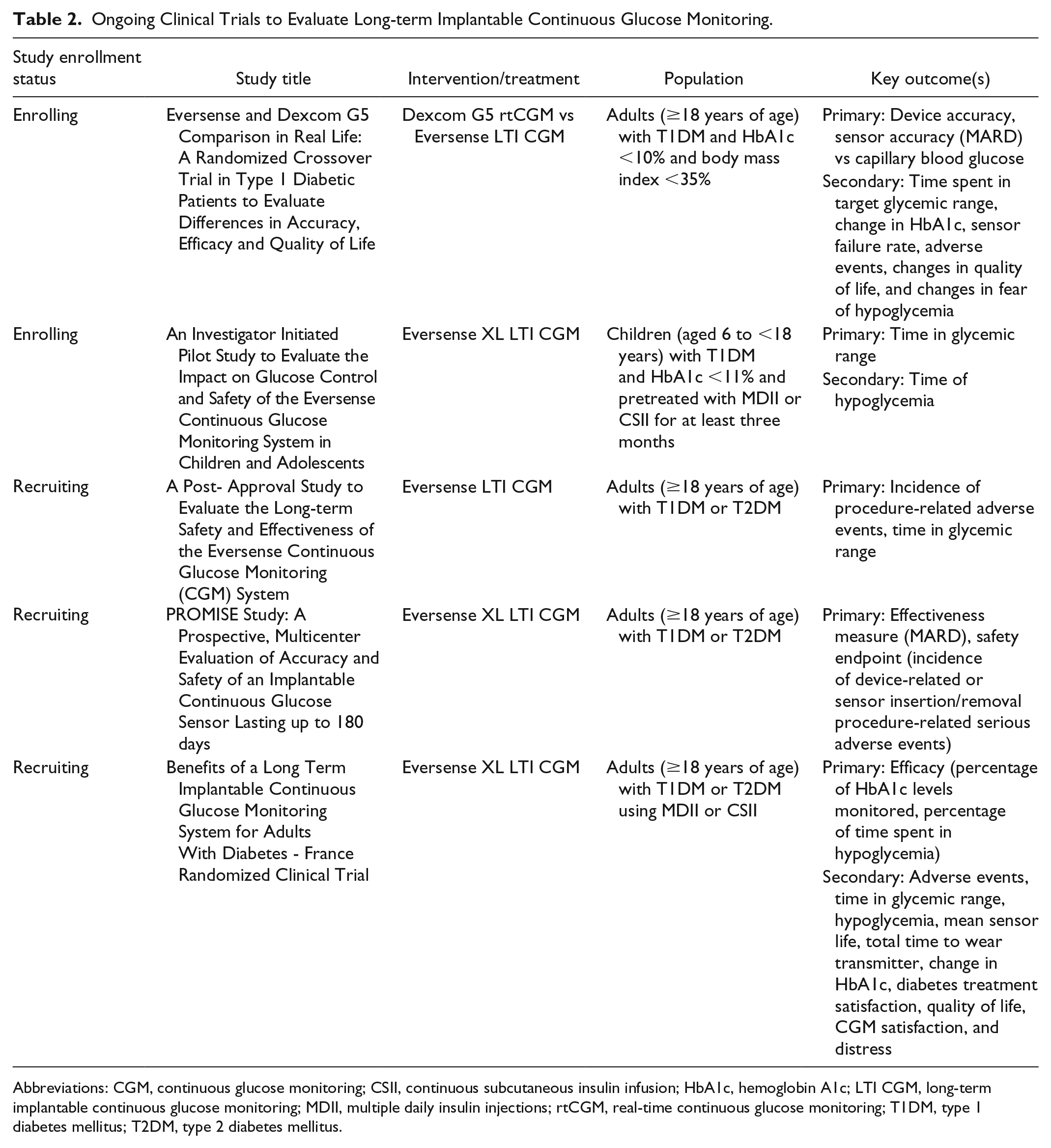
Using de-identified glucose data (Eversense LTI CGM) from the Eversense data management system, Sanchez et al found that the percentage of sensor glucose readings at <54 mg/dL was 1.2%, <70 mg/dL was 4.1%, time in normal glycemic range (70-180 mg/dL) was 62.3%, >180-250 mg/dL was 21.9%, and > 250 mg/dL was 11.6% at 90 days. 23 The median transmitter wear time in this analysis utilizing postmarketing data was 83.6%. 23 This real-world analysis suggests that the Eversense LTI CGM may provide similar benefits to isCGM and conventional rtCGM, although additional comparative efficacy analysis is lacking at this time. 23 However, there are several ongoing clinical trials designed to further evaluate the glycemic efficacy of the Eversense LTI CGM (Table 2). 24
Ongoing Clinical Trials to Evaluate Long-term Implantable Continuous Glucose Monitoring.
Abbreviations: CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; HbA1c, hemoglobin A1c; LTI CGM, long-term implantable continuous glucose monitoring; MDII, multiple daily insulin injections; rtCGM, real-time continuous glucose monitoring; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
Conclusion and Patient Selection
Based on the available evidence, the Eversense LTI CGM appears to be safe and accurate. Additional clinical trial investigation is warranted to evaluate the glycemic efficacy of the Eversense LTI CGM, especially as compared with isCGM and rtCGM devices, which are widely used in clinical practice. Based on limited efficacy data, the Eversense LTI CGM may reduce HbA1c and improve time in glycemic range.10,13,23 At this time, there is insufficient evidence to support using LTI CGM use in children <12 years of age, pregnant women, or those planning to become pregnant.
Selection considerations for conventional rtCGM still apply to the Eversense LTI CGM. These include the use of multiple daily insulin injections or continuous subcutaneous insulin infusion, high glycemic variability and frequent, problematic hypoglycemia, and high likelihood of adherence and CGM sensor wear time. Cost must also be considered, and providers must be aware of criteria for CGM use, which varies, based on payer type. Additional characteristics to identify those most likely to benefit from the Eversense LTI CGM device are as follows: (1) patients who perceive frequent sensor replacement a burden, 25 (2) patients who would benefit from on-body vibration alerts, 25 (3) those with a history of skin problems or oversensitivity to adhesives such as isobornyl acrylate,25,26 (4) those with a need for transient removal of external devices such as those with a high level of physical activity, 25 (5) those physically handicapped (visual, hearing, dexterity), 25 or (6) those with a pain or needle phobia. 25
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
