Abstract
Ascertaining the utility of continuous glucose monitoring (CGM) in pregnancy complicated by diabetes is a rapidly evolving area, as the prevalence of type 1 diabetes (T1D), type 2 diabetes (T2D), and gestational diabetes mellitus (GDM) escalates. The seminal randomized controlled trial (RCT) evaluating CGM use added to standard care in pregnancy in T1D demonstrated significant improvements in maternal glycemia and neonatal health outcomes. Current clinical guidance recommends targets for percentage time in range (TIR), time above range (TAR), and time below range (TBR) during pregnancy complicated by T1D that are widely used in clinical practice. However, the superiority of CGM over blood glucose monitoring (BGM) is still questioned in both T2D and GDM, and whether glucose targets should be different than in T1D is unknown. Questions requiring additional research include which CGM metrics are superior in predicting clinical outcomes, how should pregnancy-specific CGM targets be defined, whether CGM targets should differ according to gestational age, and if CGM metrics during pregnancy should be similar across all types of diabetes. Limiting the potential for CGM to improve pregnancy outcomes may be our inability to maintain TIR > 70% throughout gestation, a goal achieved in the minority of patients studied. Adverse pregnancy outcomes remain high in women with T1D and T2D in pregnancy despite CGM technology, and this review explores the potential reasons and questions yet to be investigated.
Introduction
Despite major advances in diabetes technology use in pregnancy, serious maternal and fetal morbidities are common, including a greater than 50% risk of large for gestational age (LGA) infants, a powerful risk factor for offspring obesity and diabetes. Glycemic metrics derived from continuous glucose monitoring (CGM), including percentage of time spent within the recommended glucose target range (time in range [TIR]) during pregnancy, have been associated with adverse pregnancy outcomes, including maternal complications, fetal overgrowth, and neonatal intensive care unit (NICU) admissions.
Early assumptions about the ability of CGM to improve both maternal and fetal outcomes underestimated the complex and dynamic physiology of pregnancy. Marked alterations in maternal glucose metabolism driven by rapidly changing fetal-placental glucose demands alter diurnal, postprandial, and gestational-specific glucose “norms.” Furthermore, these timed glucose exposures to the fetus do not contribute equally to the risk of fetal malformations, fetal overgrowth, or to multiple fetal and neonatal morbidities. Given the dramatic changes in insulin resistance and varying fetal developmental stages across gestation, optimal targets for percent TIR, time above range (TAR), time below range (TBR), and mean glucose may change in different trimesters. Nocturnal glucose levels may be a more powerful driver of LGA than daytime or postprandial glucose in some populations. 1 Yet, distinct targets for daytime versus nocturnal TIR or mean glucose are not currently established. There are compelling data from studies of CGM use among pregnant women with type 1 diabetes (T1D) that even 4% to 7% TIR improvements can result in an approximate 50% decrease in LGA and NICU admissions, 2 resulting in an international recommendation for CGM use in T1D.3,4 Some experts also recommend CGM for pregnant women with type 2 diabetes (T2D) or gestational diabetes mellitus (GDM) treated with intensive insulin therapy, but data are much more limited and conflicting. 5 Given that fetal hyperinsulinemia may begin by 14 weeks of gestation and drive fetal overgrowth early, optimizing glycemia in the first half of pregnancy may be as critical as in late pregnancy. Furthermore, glycemic variability (GV), which is not adequately reflected by TIR or mean glucose, may be an additional determinant of pregnancy outcomes. Finally, optimal targets for CGM metrics during pregnancy may vary in pregnancies complicated by T1D compared with T2D or GDM.
We must reconceptualize CGM use and how to apply it clinically during pregnancy. Thus, we set out to critically appraise the current evidence for CGM use during pregnancy, the targets for CGM metrics in clinical practice, as well as the most pressing areas for future investigations.
Methods
We conducted a PubMed review to identify major trials and analyses evaluating associations between CGM use and metrics with adverse pregnancy outcomes. Based on the above literature review, we critically appraise the current evidence for CGM use during pregnancy and targets for CGM metrics in clinical practice, and we describe the major benefits and challenges of CGM use during pregnancy. In this perspective piece, we describe: (1) the associations between CGM metrics and adverse pregnancy outcomes in T1D, (2) challenges in attainment of the recommended CGM targets during T1D pregnancy, and (3) controversies surrounding CGM use in pregnancy, including a discussion of whether CGM metrics or conventional blood glucose monitoring (BGM) targets should be prioritized, whether differential nocturnal versus daytime CGM target ranges should be utilized, challenges in interpretation of the ambulatory glucose profile (AGP) during pregnancy, controversies regarding CGM use in the less-studied populations of T2D and GDM, and controversies in establishing optimal CGM targets during pregnancy.
Associations Between Improved TIR and Neonatal Outcomes in T1D
In this review, we will report glycemic targets as recommended percentage times spent in each range, as is recommended by the American Diabetes Association Standards of Care in Diabetes 6 and the International Consensus on TIR. 4 The pregnancy-specific target glucose range is defined by convention as 63 to 140 mg/dL, though it should be noted that the lower threshold has been questioned. 6 Recommendations per current expert clinical guidance3,4 for percentage of time spent within the pregnancy-specific TIR, TAR, and TBR are shown in Table 1 and are widely used in clinical practice. Most studies assessing the impact of CGM use on neonatal and maternal outcomes as well as the relationships between CGM metrics and pregnancy outcomes included women with T1D, alone or less commonly in combination with T2D and/or insulin-treated GDM (Table 2).2,8-23,25 Notably, the major trials evaluating impact of CGM use on pregnancy outcomes evaluated CGM as adjunctive therapy to BGM, and these trials did not compare use of CGM alone to standard care (BGM). Given the need for extremely tight glucose control in pregnancy, it may still be beneficial to confirm CGM measurements with BGM measurements for actionable events (eg, need to bolus insulin or treat hypoglycemia). More studies are needed to determine if non-adjunctive use of CGM without corroborating BGM is safe in pregnancy. Providers must also be aware of potential medication interferents that may impact CGM accuracy, as well as potential adverse reactions, such as allergic or irritant skin reactions that may result from CGM use, both during and outside of pregnancy. 26
CGM Metric Targets for Pregnant Individuals With T1D.
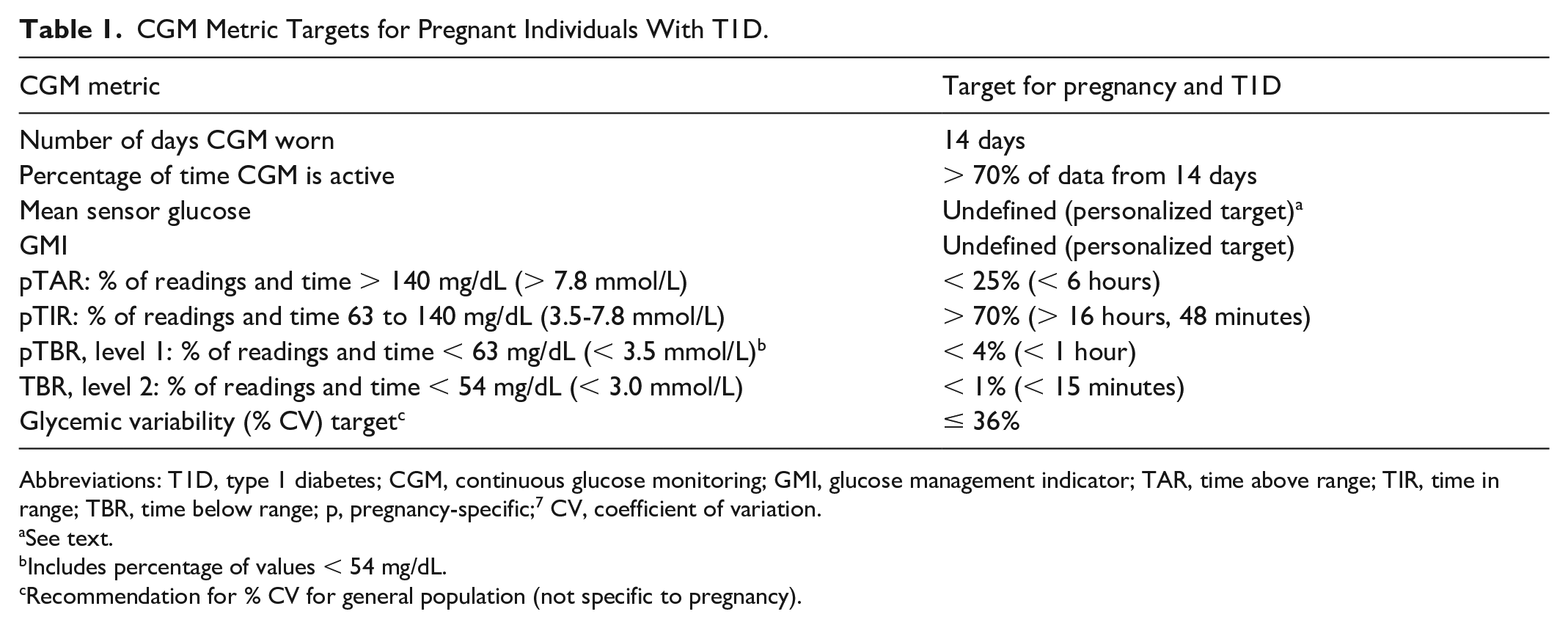
Abbreviations: T1D, type 1 diabetes; CGM, continuous glucose monitoring; GMI, glucose management indicator; TAR, time above range; TIR, time in range; TBR, time below range; p, pregnancy-specific; 7 CV, coefficient of variation.
See text.
Includes percentage of values < 54 mg/dL.
Recommendation for % CV for general population (not specific to pregnancy).
Associations of CGM Metrics With Neonatal and Maternal Outcomes in T1D.
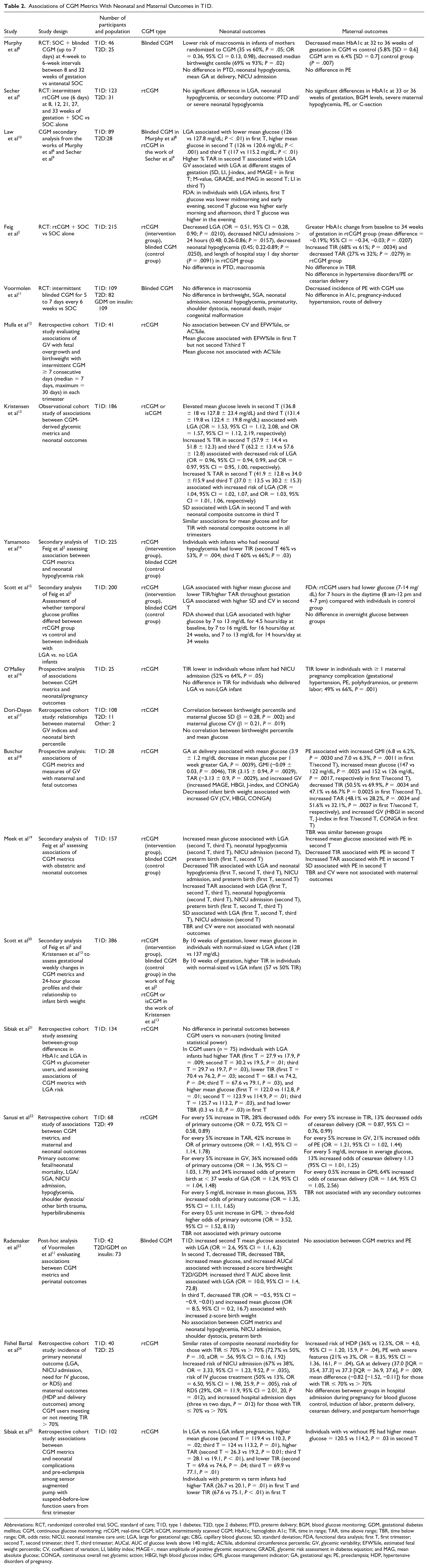
Abbreviations: RCT, randomized controlled trial; SOC, standard of care; T1D, type 1 diabetes; T2D, type 2 diabetes; PTD, preterm delivery; BGM, blood glucose monitoring; GDM, gestational diabetes mellitus; CGM, continuous glucose monitoring; rtCGM, real-time CGM; isCGM, intermittently scanned CGM; HbA1c, hemoglobin A1c; TIR, time in range; TAR, time above range; TBR, time below range; OR, odds ratio; NICU, neonatal intensive care unit; LGA, large for gestational age; CBG, capillary blood glucose; SD, standard deviation; FDA, functional data analysis; first T, first trimester; second T, second trimester; third T, third trimester; AUCal, AUC of glucose levels above 140 mg/dL; AC%ile, abdominal circumference percentile; GV, glycemic variability; EFW%ile, estimated fetal weight percentile; CV, coefficient of variation; LI, lability index; MAGE+, mean amplitude of positive glycemic excursions; GRADE, glycemic risk assessment in diabetes equation; and MAG, mean absolute glucose; CONGA, continuous overall net glycemic action; HBGI, high blood glucose index; GMI, glucose management indicator; GA, gestational age; PE, preeclampsia; HDP, hypertensive disorders of pregnancy.
Early trials of intermittent real-time (rt) 9 or blinded8,11 CGM showed mixed results (Table 2), with CGM use leading to decreased risk of macrosomia and decreased median birthweight centile in one trial, 8 no differences in any neonatal or maternal outcomes in another trial, 9 and an isolated decrease in incidence of preeclampsia without impact on any neonatal outcomes in another. 11 Notably, blinded CGM does not allow for therapeutic adjustments in real time in response to hypoglycemia or hyperglycemia. In addition, intermittent, as opposed to uninterrupted, rtCGM use may not afford CGM users sufficient exposure to CGM data to recognize glycemic patterns and derive downstream clinical benefits. In the above trial evaluating intermittent rtCGM use, 9 only 64% participants used rtCGM per the specified protocol, which may have diluted any beneficial impact of CGM use on outcomes.
In the Continuous Glucose Monitoring in Women with Type 1 Diabetes in Pregnancy Trial (CONCEPTT), the seminal multicenter open-label RCT evaluating the impact on maternal glycemia and neonatal health outcomes of rtCGM added to standard BGM compared with BGM alone in T1D, CGM use led to increased TIR, decreased TAR, no decrease in hypoglycemia, and a slight decrease in hemoglobin A1c (HbA1c) (Table 2). 2 Neonatal health outcomes were markedly improved with CGM use, including a decrease in LGA, fewer NICU admissions lasting more than 24 hours, and fewer cases of severe neonatal hypoglycemia (with odds ratios [OR] of ~ 0.5), which was thought to be due to the modestly higher TIR (~6%-7%) and fewer postprandial glucose excursions rather than the slight decrease in HbA1c (0.2%). 27 The numbers needed to treat (NNT) with CGM use to avoid one adverse event were low despite the modest glycemic benefits noted: six for NICU admission and LGA, and eight for neonatal hypoglycemia. Thus, this pivotal trial showed that even modest glycemic improvements in HbA1c and CGM metrics associated with CGM use led to significant improvements in neonatal health outcomes.
A subsequent retrospective analysis of rtCGM data of women with T1D or T2D similarly found that even modest increments in multiple standard CGM metrics were associated with meaningful reductions in risk of the composite neonatal outcome, and selected maternal outcomes (Table 2). 22 Every 5% increase in TIR translated into a 28% decrease in the risk of the neonatal composite outcome.
Associations Between Improved TIR and Maternal Outcomes in T1D
The development of hypertensive disorders of pregnancy (HDP) is a major cause of both maternal and fetal morbidity and mortality, 28 and suboptimal glycemic control is a contributing factor to the development of HDP. 29 Individuals with T1D are at 3.5 times higher risk of developing HDP, potentially leading to preterm delivery (PTD; < 37 weeks) or complications, such as pulmonary edema and stroke. 30
In pregnant individuals with T1D and T2D, those who spent < 70% TIR had a four-fold increased risk for developing HDP compared with those achieving ≥ 70% TIR (Table 2). 24 Among those who developed HDP, individuals with < 70% TIR were eight times more likely to develop severe features of preeclampsia.
In a small prospective cohort of pregnant individuals with T1D, multiple CGM metrics in the first and second trimesters were associated with the development of preeclampsia. 18 Those who developed preeclampsia had higher mean glucose (~25 mg/dL) in the first and second trimesters compared with the normotensive group (Table 2). Time in range was consistently lower in early and mid-pregnancy in the HDP group, and TAR was higher compared with those who remained normotensive. A higher mean glucose and TAR were also significantly associated with an earlier gestational age at delivery but not with PTD.
Finally, a sub-analysis of CONCEPTT found that increased TIR was associated with a 50% reduction in both preeclampsia and PTD. 19 These findings were primarily seen in the second trimester. Given that preeclampsia is associated with early abnormal placentation and angiogenesis as well as preexisting chronic hypertension, the extent to which these or other CGM metrics contribute to development of preeclampsia or other adverse maternal outcomes at varying gestational ages should be further explored.
Importance of timing – early vs. late exposure to hyperglycemia in pregnancy
The increased risks of congenital malformations, 31 preeclampsia, 32 and PTD 33 associated with hyperglycemia may depend on the timing of exposure to hyperglycemia.34-37 Data derived from CGM worn throughout gestation allows for granular depiction of associations between CGM metrics and pregnancy outcomes. Scott et al 20 used data from two studies to demonstrate that mean glucose and TIR diverged at 10 weeks for average for gestational age infants (AGA) versus LGA (Table 2). Time in range was stable from 10 to 20 weeks of gestation and increased from 20 to 30 weeks, when it was 8% to 0% lower in the LGA group until ~34 weeks of gestation. Time below range peaked at 10% at 10 weeks in both groups but decreased to a nadir of 6% in the LGA group and remained 8% to 10% throughout pregnancy in the AGA group. It may be difficult to disentangle the impact of higher TBR compared with TIR in reducing LGA. Of note, hybrid closed-loop (HCL) insulin pumps, which may be effective in lowering TBR, were not available to the participants of these studies.
Beyond TIR: Utility of Additional CGM Metrics in Pregnancies Complicated by T1D
Mean glucose has emerged as a significant predictor of neonatal outcomes in multiple studies (Table 2).10,13,15,19-23 Higher CGM mean glucose in the second and third trimesters is associated with LGA in T1D and GDM,1,10,13,15 and may be the glycemic marker with the strongest association with LGA. 13 Surprisingly, first trimester mean glucose was found to be significantly lower in the first trimester but higher in the second and third trimesters in individuals who delivered LGA infants. This is in contrast to other studies linking early pregnancy hyperglycemia to LGA in GDM38,39; this may be related to increasing insulin sensitivity in some individuals with T1D in the first trimester or early normoglycemia allowing healthier trophoblastic invasion and development, which later increases LGA potential. 10 Targeting mean glucose of 115 to 120 mg/dL has been proposed based on observed levels among individuals who did not deliver LGA infants in two CGM RCTs,10,40 but this target requires further validation.
Glycemic variability has been evaluated as an independent predictor of pregnancy outcomes. Some measures of GV have been associated with pregnancy outcomes at varying stages of gestation,10,12,13,17,18,22 but the GV metrics have not been consistent across studies (Table 2). The clinical impact of targeting GV goals is unknown. In the seminal CONCEPTT RCT, CGM use led to a non-significantly reduced glucose coefficient of variation (32% [28-37] vs 34% [29-39],
Use of mean glucose in conjunction with TIR may better capture situations in which fasting glucose deviates from the optimal target range. For example, if mean overnight glucose is approximately 120 mg/dL (above the recommended pregnancy fasting glucose target of < 95 mg/dL 3 ), with minimal postprandial rise in glucose over the day, TIR could meet or even exceed the goal of > 70% despite fasting glucose levels markedly above the pregnancy target. In contrast, isolated use of mean glucose targets would not identify situations with significant glycemic variation (with frequent hypoglycemia and hyperglycemia). Thus, combining use of mean glucose with TIR and/or indicators of GV may provide a more comprehensive glycemic picture; more research is needed to determine if this is the case.
The glucose management indicator (GMI) is useful for converting CGM data into a metric similar to HbA1c that providers and patients readily understand, and allows focus on a 14-day rather than three-month period. This could be especially valuable and potentially superior to HbA1c in estimating the risk for major malformations during organogenesis during the limited window of five to ten weeks of gestation. Since pregnancy alters the relationship between HbA1c and average glucose, use of mean glucose may be more accurate than use of estimated HbA1c calculations. 41 In one small study, GMI correlated better with TIR in the first trimester than HbA1c, 42 likely because first trimester HbA1c may better reflect preconception glycemia than first trimester glycemia.
Studies have not demonstrated significant associations of TBR with adverse neonatal and maternal outcomes,2,19,22 though several have shown that CGM detects subclinical hypoglycemia unrecognized by BGM alone.43-45 Avoidance of severe maternal hypoglycemia is especially important in T1D with hypoglycemic unawareness and remains a cornerstone of diabetes management due to the potential harms incurred by severe hypoglycemia and its attendant consequences. 46 Furthermore, the fetus does not have gluconeogenic enzymes until close to term, 47 so that, protracted severe maternal hypoglycemia may pose fetal risk. To date, studies have not shown that CGM use reduces incidence of severe hypoglycemic events in pregnancy, but more research is needed on this.
CGM Target Attainment in Pregnancies Complicated by T1D
Continuous glucose monitoring target attainment remains concerningly low. Among pregnant individuals participating in the CONCEPTT trial,
48
>70% TIR was achieved by only 7.7/10.2/35.5% of participants, < 25% TAR was achieved by only 14.5/14.2/37.2% of individuals, and TBR < 4% was achieved by only 30.3/52.8/52.9% of participants in first, second, and third trimesters, respectively. The rate of CGM target attainment increased over the course of gestation, and at 34 weeks of gestation, the proportion of participants who achieved CGM targets was greater among subjects in the rtCGM group compared with the control group. Another T1D study reported a cumulative TIR across gestation of 59% ± 14%, TAR of 38% ± 15%, and TBR of 3% ± 3%, with only 28% (7/25) achieving the recommended ≥ 70% TIR cumulatively over the course of pregnancy.
16
In an analysis of individuals participating in the T1D Exchange, rates of adverse maternal and fetal outcomes, including cesarean delivery, PTD, LGA infants, and NICU admission were disappointingly high in spite of high rates of CGM use and insulin pump therapy.
49
It is important to note that CGM target attainment was achieved to a greater extent when CGM was paired with automated insulin delivery in a recent RCT evaluating efficacy of a novel automated insulin delivery system with pregnancy-specific glucose targets used during pregnancy (mean TIR of 68.2% ± 10.5% in the intervention group vs 55.6% ± 12.5% in the standard-care group [mean adjusted difference, 10.5%, 95% confidence interval [CI] = 7.0, 14.0;
Challenges and Controversies of CGM Use in Pregnancy
CGM Targets Versus Conventional BGM Targets: Which Should Take Precedence When Both Cannot be Achieved?
A pressing question is whether CGM-derived TIR or conventional fasting and postprandial glucose targets should take precedence for both clinical and research outcomes. Individuals can interpret and act on fasting and postprandial targets more quickly in real time than on a retrospective TIR. Use of off-label HCL technology, substantially increasing in pregnancy, may increase TIR at the expense of higher fasting or nocturnal glucose. 51 The potential benefits of increased TIR and lower rates of hypoglycemia might outweigh the risks of higher average fasting glucose, but further research is needed to determine if this is the case.
Should Nocturnal Versus Daytime CGM Target Ranges be Established?
It is unresolved whether different nocturnal versus daytime TIR targets, mirroring the conventional pregnancy blood glucose targets (Table 3), should be utilized. Physiologically, both nocturnal and daytime maternal glycemia may contribute to 24-hour fetal glucose exposure and resultant fetal hyperinsulinemia, though data from the GDM population suggests that LGA may be specifically driven by nocturnal hyperglycemia. 1 The consensus CGM targets (Table 1) do not differentiate between fasting and postprandial time points. 4
ADA-Recommended Targets for Pregnant Individuals With Type 1 or Type 2 Diabetes. 3

Prior to establishing nocturnal and daytime TIR, normative CGM pregnancy metrics and functional data analysis (FDA) are needed as a reference throughout pregnancy, especially since fasting glucose falls in normal pregnancy. Among individuals with normal body mass index (BMI) and glycemia at ~34 weeks of gestation, mean fasting glucose was 71 ± 8 mg/dL, with one-hour and two-hour postprandial glucose of 109 ± 13 and 99 ± 10 mg/dL, respectively. 52 Future research is needed to determine whether differential nocturnal versus daytime TIR targets will improve prediction of adverse pregnancy outcomes and what ranges are optimal.
What Are the Challenges for Use of the AGP in Pregnancy?
The AGP report presents a standardized visual representation of CGM data (Figure 1). However, AGPs that are standardized to pregnancy-specific CGM targets are not universally and readily available. Pregnancy-specific mean glucose and GMI targets were not defined in the consensus guidance, and time spent in target ranges were not specified for GDM or T2D due to insufficient data. 4
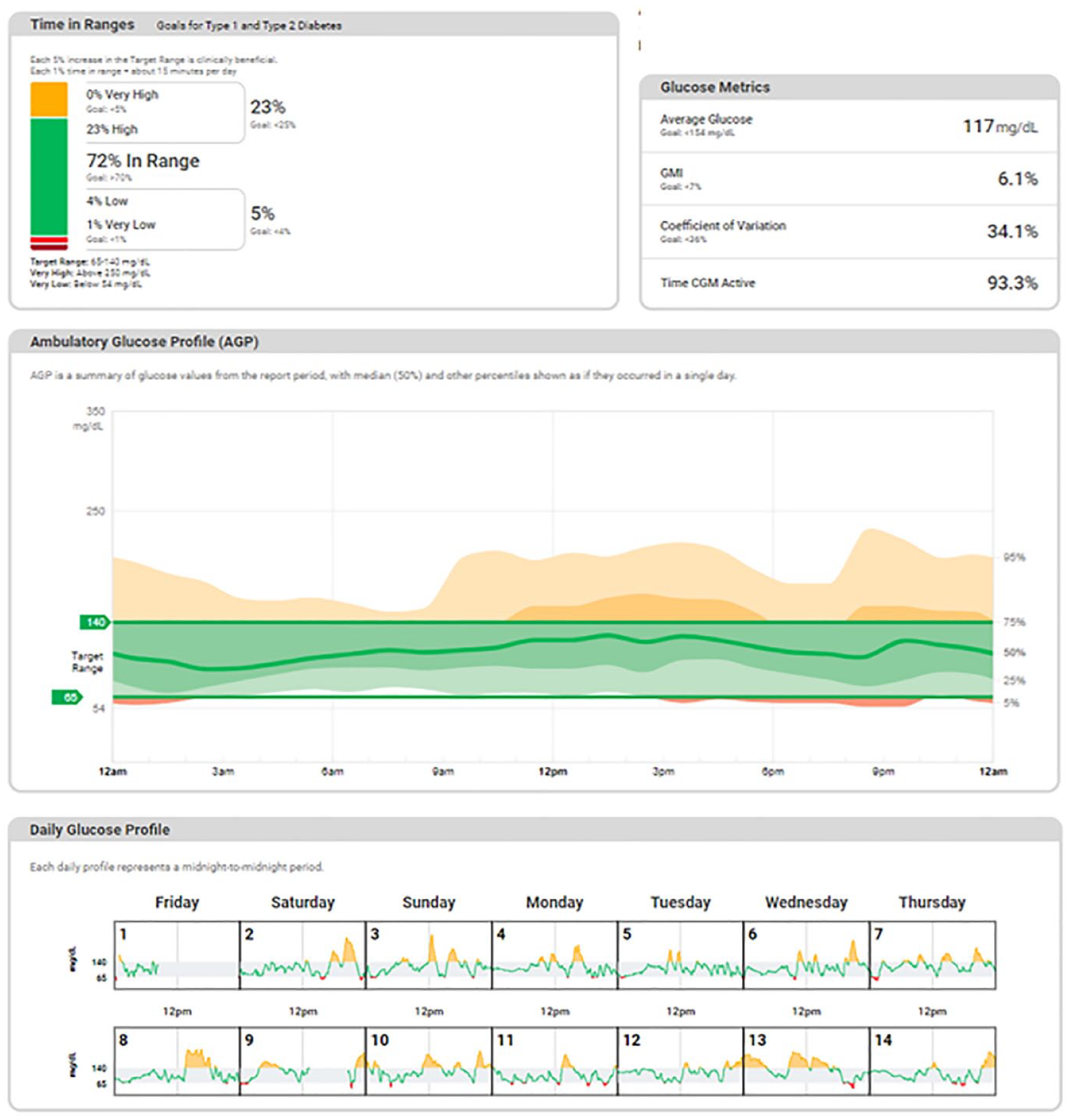
Components of the AGP report from a pregnant individual with type 1 diabetes. The lower limit of the target range is set to 65 mg/dL instead of 63 mg/dL when 5 mg/dL is the minimum increment available on the software program. The glucose metric targets displayed by default on some software programs are those recommended for non-pregnant individuals.
It is important that all CGM devices permit sharing with the treating provider and that the AGP report’s targets can be adjusted from non-pregnant glucose targets (70-180 mg/dL) to those of pregnancy (63-140 mg/dL). In the preconception period, pregnancy-specific glucose targets can be set on the CGM device and software, to monitor progress toward achievement of pregnancy glycemic targets. Yet not all CGM devices can currently be adjusted to these pregnancy-specific targets.
As described for AGP interpretation in non-pregnant populations, action is needed if TIR and TBR are not both at goal.53,54 Hypoglycemia patterns should be addressed first, both for safety and because hypoglycemia often results in overtreatment with carbohydrate and subsequent hyperglycemia. The daily glucose profiles allow identification of high or low glucose patterns that warrant adjustment of basal and/or prandial insulin doses.
Ideally, connected insulin delivery devices that communicate with CGM devices (integrated insulin pumps or smart insulin pens 55 ) should be used, so that, insulin doses and timing can be reviewed, missed or delayed boluses can be identified, and carbohydrate counting difficulties can be addressed. Behaviors that may increase TIR include pre-bolusing 15 minutes before carbohydrate consumption early in pregnancy and 30 to 40 minutes in late pregnancy. 56
What Evidence is There for Potential Use of CGM in Pregnant Populations Without T1D?
Gestational Diabetes Mellitus
A systematic review and meta-analysis of six randomized trials with overall a relatively small sample size found that GDM participants using CGM had a modestly lower HbA1c at the end of pregnancy (mean difference = 0.22%), a small decrease in gestational weight gain (1.17 kg), and slightly lower birthweight (mean difference = 116 g). 57 In a RCT not included in the meta-analysis, CGM with BGM did not lead to differences in CGM metrics, HbA1c, or adverse perinatal outcomes but was associated with a decrease in maternal gestational weight gain and lower infant birth weight. 58
In a prospective observational study of blinded CGM use for only seven days at 30 to 32 weeks of gestation, mean glucose was significantly higher in mothers with LGA infants, driven by higher mean glucose overnight. 1 A cohort study of individuals with GDM using intermittently scanned CGM (isCGM) for only 5 to 14 days at a mean of 28 weeks of gestation found that nighttime mean glucose was associated with a significant increase in birthweight percentile. 59 In another cohort study, GV as measured by mean amplitude of glycemic excursions (MAGE) was associated with increased birthweight and an adverse composite neonatal outcome, 60 while another GDM study found no association between GV and birthweight percentile or other adverse pregnancy outcomes. 61 A recent and much larger study of 1302 individuals with GDM who used CGM for 14 days at a mean of 26 weeks of gestation found that TAR, area under the curve (AUC), nighttime mean glucose, daytime mean glucose, and daily mean glucose were all associated with a composite adverse pregnancy outcome. 62 In addition, TIR, TAR, AUC, MAGE and nighttime, daytime, and daily mean glucose were positively associated with LGA, while TBR was inversely associated with risk of LGA.
Type 2 Diabetes Mellitus
Data in T2D pregnancies are very limited. A pilot feasibility study from Australia 63 enrolled 57 participants in remote rural areas, of whom 91% used insulin. Continuous glucose monitoring was well-received; most were able to use the technology and found it useful. Randomized controlled trials using CGM that included individuals with T1D and T2D are described above8,11 and in Table 2; however, there are currently no published trials in T2D alone.
Efficacy studies addressing CGM use in T2D and GDM demand immediate attention, but such widespread implementation of CGM technology would also mandate the availability of more diabetes care and education specialists to assist patients and their providers.
Controversies in Establishing the TIR Target in Pregnancy: Will Achieving Higher TIR Lead to Improved Pregnancy Outcomes?
In a study of women with GDM, receiver operating characteristic (ROC) curves were analyzed to explore the optimal thresholds for TAR and mean glucose in predicting adverse pregnancy outcomes. 62 The optimal thresholds for TAR were 1.9% for LGA, 1.2% for NICU admission, and 2.5% for any adverse pregnancy outcome. The optimal thresholds for mean glucose were 81 mg/dL for LGA and 86 mg/dL for any adverse outcome, considerably lower than the mean glucose target suggested for T1D. 40 Thus, it is possible that optimal glucose targets are lower in GDM compared with T1D pregnancies, but currently available data are not sufficient to recommend TIR targets specific to GDM or T2D, and further research is needed to define specific CGM targets for these less well-studied populations. It is important to note that hypoglycemia risk and GV are much higher in individuals with T1D compared with GDM, potentially hindering the safe achievement of these more aggressive glycemic targets in T1D pregnancies. Prospective investigation is therefore needed to determine whether CGM targets should differ in T1D compared with T2D/GDM pregnancies, and to inform evidence-based CGM targets in these different populations. In the work of Sanusi et al, 22 which included both T1D and T2D pregnant individuals, the optimal TIR to reduce an adverse composite neonatal outcome was 71% using the Youden’s index and 66% using the Liu method, thus validating the current recommendation to aim for > 70% TIR in T1D pregnancies.3,4 However, separate ROC curves could not be generated for women with T1D and T2D. The optimal mean glucose target to reduce LGA may be different in patients with T2D and vascular disease or those with hypertensive disorders at high risk for abnormal placentation and preeclampsia, given their higher risk of small for gestational age (SGA).
Conclusion
Continuous glucose monitoring use during T1D pregnancy has shown that even modest changes in TIR and other common CGM metrics are associated with meaningful differences in risk of adverse neonatal outcomes and selected maternal outcomes. If advanced technologies including HCL pumps can help achieve ≥ 70% TIR throughout pregnancy in substantially more individuals, maternal and neonatal outcomes may further improve. Some key areas of clinical practice or research requiring further work in pregnant individuals using CGM are outlined in Table 4. It is currently unclear whether TIR targets, possibly in tandem with additional CGM-derived metrics, should take precedence over traditional fasting and postprandial pregnancy glucose targets when both cannot be easily achieved. Whether targeting differential nocturnal and daytime TIR targets and other less-studied CGM metrics affords incremental clinical benefit are questions deserving research prioritization. Future research will inform whether CGM targets should vary at different gestational stages and whether achievement of higher TIR beyond what is currently recommended will incrementally improve pregnancy outcomes. In addition, future work will better define the role for CGM use and evidence-based CGM targets among pregnant individuals with T2D and GDM, who may be at lower risk for SGA (GDM) and maternal hypoglycemia.
Areas of Clinical Practice or Research Requiring Further Work in Pregnant Individuals Using CGM.
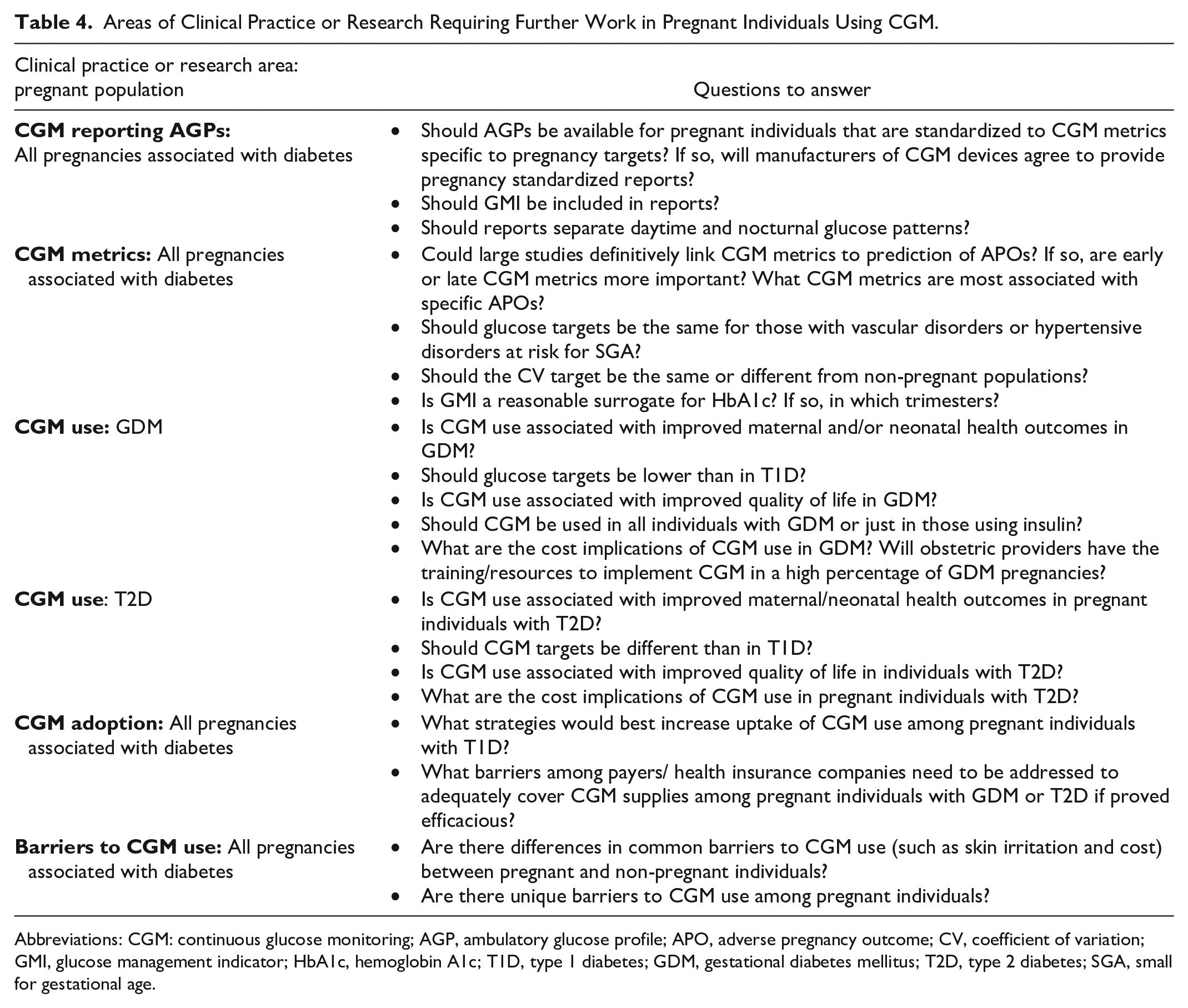
Abbreviations: CGM: continuous glucose monitoring; AGP, ambulatory glucose profile; APO, adverse pregnancy outcome; CV, coefficient of variation; GMI, glucose management indicator; HbA1c, hemoglobin A1c; T1D, type 1 diabetes; GDM, gestational diabetes mellitus; T2D, type 2 diabetes; SGA, small for gestational age.
Footnotes
Abbreviations
First T, first trimester; second T, second trimester; third T, third trimester; AC%ile, abdominal circumference percentile; AGA, average for gestational age; AGP, ambulatory glucose profile; AUCal, area under the curve of glucose levels above 140 mg/dL; BGM, blood glucose monitoring; CBG, capillary blood glucose; CGM, continuous glucose monitoring; CONCEPTT, Continuous Glucose Monitoring in Women with Type 1 Diabetes in Pregnancy Trial; CONGA, continuous overall net glycemic action; CV, coefficient of variation; EFW%ile, estimated fetal weight percentile; FDA, functional data analysis; GA, gestational age; GDM, gestational diabetes mellitus; GMI, Glucose Management Indicator; GRADE, glycemic risk assessment in diabetes equation; GV, glycemic variability; HbA1c, hemoglobin A1c; HBGI, high blood glucose index; HCL, hybrid closed-loop; HDP, hypertensive disorders of pregnancy; isCGM, intermittently scanned CGM; LGA, large for gestational age; LI, lability index; MAG, mean absolute glucose; MAGE, mean amplitude of glycemic excursions; MAGE+, mean amplitude of positive glycemic excursions; NICU, neonatal intensive care unit; OR, odds ratio; PE, preeclampsia; PTD, preterm delivery; RCT, randomized controlled trial; rt, real-time; rtCGM, real-time CGM; SD, standard deviation; SGA, small for gestational age; SOC, standard of care; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.A. has received consultant fees from Dexcom, Insulet and Medscape, and has received research support to her employer Northwestern University from Fractyl Health, Insulet, MannKind, Tandem Diabetes and Welldoc. F.M.B. received funding from Dexcom, Inc. paid to the Joslin Diabetes Center and receives royalties from UpToDate for topics on preexisting diabetes in pregnancy. C.D. has received financial support from Dexcom for advisory role for patient education and development of educational materials, receives royalties from UpToDate for topics on GDM, and has received funding from United Health Group and Helmsley Charitable Trust. D.S.F. reports grants from the Canadian Institute of Health Research and an investigator-initiated grant from Dexcom, in-kind donations from Dexcom and Tandem and speaker honoraria from Novo-Nordisk. G.O. receives research support from Dexcom, Inc, Tandem Diabetes and MannKind Corporation paid to her institution. S.P is a contributing writer for diaTribe, is on the Medical Advisory Board for Medtronic MiniMed, Inc, has received research funding from Dexcom, Inc., Eli Lilly, JDRF, Leona & Harry Helmsley Charitable Trust, NIDDK, and Sanofi US Services, and has received research support from Diasome Pharmaceuticals, Medtronic MiniMed, Inc., and Sanofi US Services.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
