Abstract
Increasing numbers of people, particularly with type 1 diabetes (T1D), are using wearable technologies. That is, continuous subcutaneous insulin infusion (CSII) pumps, continuous glucose monitoring (CGM) systems, and hybrid closed-loop systems, which combine both these elements. Given over a quarter of all people admitted to hospital have diabetes, there is a need for clinical guidelines for when people using them are admitted to hospital. The Joint British Diabetes Societies for Inpatient Care (JBDS-IP) provide a scoping review and summary of guidelines on the use of diabetes technology in people with diabetes admitted to hospital.
JBDS-IP advocates enabling people who can self-manage and use their own diabetes technology to continue doing so as they would do out of hospital. Whilst people with diabetes are recommended to achieve a target of 70% time within range (3.9-10.0 mmol/L [70-180 mg/dL]), this can be very difficult to achieve whilst unwell. We therefore recommend targeting hypoglycemia prevention as a priority, keeping time below 3.9 mmol/L (70 mg/dL) at < 1%, being aware of looming hypoglycemia if glucose is between 4.0 and 5.9 mmol/L (72-106 mg/dL), and consider intervening, particularly if there is a downward CGM trend arrow.
Health care organizations need clear local policies and guidance to support individuals using diabetes technologies, and ensure the relevant workforce is capable and skilled enough to ensure their safe use within the hospital setting. The current set of guidelines is divided into two parts. Part 1, which follows below, outlines the guidance for use of CGM in hospital. The second part outlines guidance for use of CSII and hybrid closed-loop in hospital.
Keywords
Introduction
People with diabetes are at higher risk for hospitalization and complications due to the added problems of hyperglycemia or hypoglycemia and metabolic co-morbidities.1,2 Improving glycemia, whilst minimizing the rate of hypoglycemia, is of major importance in the hospital setting, as both hyperglycemia and hypoglycemia have been shown to be associated with worse clinical outcomes and mortality.
Over the last two decades, wearable diabetes technology has evolved rapidly. 3 Outpatient use of continuous glucose monitoring (CGM) systems improve glycemia,4 -6 increase satisfaction, reduce fear of hypoglycemia,7,8 improve quality of life, 8 and reduce admissions with diabetes emergencies.9,10 Thus, it may be reasonable to extrapolate that most individuals admitted to hospital would choose to continue self-management in hospital. 11 Furthermore, knowledge of predicted hypoglycemia (eg, where the CGM predicts hypoglycemia within a given time frame, and/or when the glucose is between 4.0 and 5.9 mmol/L (72-106 mg/dL) and trending downward) can aid hospital staff in preventing hypoglycemic events and potentially reduce risk for the person with diabetes.
Currently, CGM is predominantly a tool for self-management, but if integrated with electronic health records could provide alerts to the diabetes team to out-of-range results and support better glycemic control in hospital.
As described in the accompanying article by Misra et al (accompanying Misra et al paper available at https://doi.org/10.1177/19322968221137360), currently, the gold standard for glucose monitoring and diagnostic purposes remains laboratory blood glucose. For monitoring purposes, capillary blood glucose (CBG) via point-of-care (POC) testing remains the mainstay of glucose monitoring in the hospital. Whilst CGM may be potentially used, we need further data on their accuracy and use in hospital before they become widely used as replacements for capillary glucose testing.
Here, we present a scoping review and summary of the Joint British Diabetes Societies for Inpatient Care (JBDS-IP) guidelines on the use of CGM in people with diabetes admitted to hospital. These guidelines are the first of a two-part series, with Part 1 outlining use of CGM in hospital and Part 2 providing guidance on use of CSII and hybrid closed-loop systems in hospital.
In-Hospital Use of CGM Systems
Recent guidance from the American Endocrine Society recommends the use of CGM in adults with insulin-treated diabetes hospitalized for non-critical illness, who are at high risk of hypoglycemia, in addition to confirmatory bedside CBG monitoring. 12 It is likely that CGM will become a standard of care for glucose monitoring in hospitals in the future, however, at present, we acknowledge that there are several challenges in implementing this safely and equitably across hospitals nationally in the United Kingdom, and therefore, our guidance is restricted to people with diabetes coming into hospital already using CGM.
Traditional CBG monitoring measures glucose at a few isolated time points in the day and may miss much of the hyperglycemia and hypoglycemia seen during an admission, much of which is aysmptomatic. 13 Meanwhile, CGM devices measure interstitial glucose, which could potentially be affected by temperature, hydration status, rapid changes in blood glucose, and tissue perfusion. However, they allow for more complete 24-hour retrospective data review, with prospective alerts and alarms for hypoglycemia and hyperglycemia detection, as well as real-time trends in glucose change over time. Hence, use of CGM in the hospital setting has the potential to lead to improved glycemia, and thus possibly mitigate increased length of stay and adverse outcomes related to suboptimal glucose control. 14
An advantage of CGM over self-monitoring blood glucose alone is the presence of trend arrows. These arrows are based on interstitial glucose variation over the previous 15 minutes and allow estimation of the rise or decline in the rate of glucose over the next 30 to 60 minutes. 15 Table 1 summarizes the definitions of trend arrows according to the CGM device (Table 1).
Continuous Glucose Monitoring Arrows and Trends.
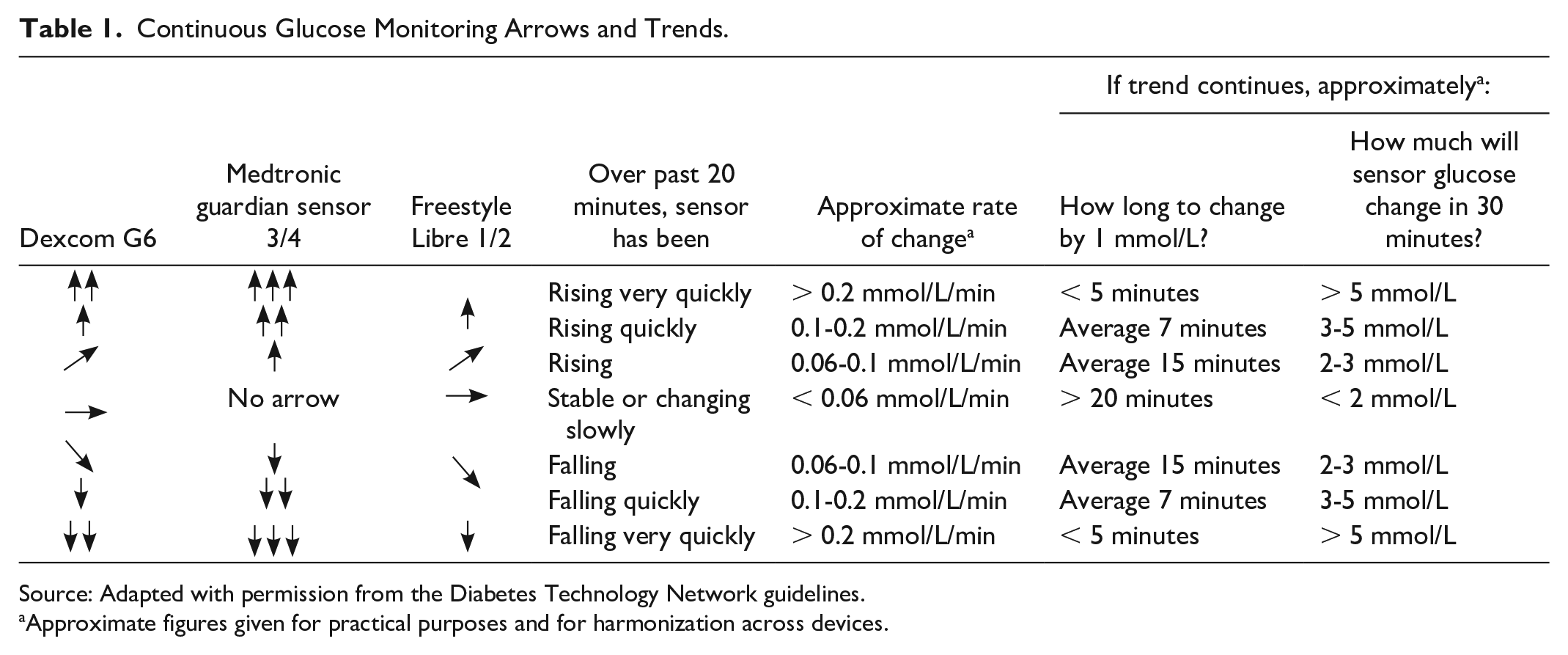
Source: Adapted with permission from the Diabetes Technology Network guidelines.
Approximate figures given for practical purposes and for harmonization across devices.
At present, CGM data do not directly link in with electronic health records, therefore on admission and during the hospital stay, glycemia can be downloaded, shared through the mobile app, or reviewed using the “history” function on the device. This allows the diabetes team and clinician to review and monitor the individual’s glucose levels. The individual will need to give permission for their data to be shared with the hospital team (if not already doing so).
Glycemic Targets in Hospital
The International Consensus on Time in Range consensus guidelines recommend a target percentage time in range (3.9-10 mmol/L, [70-180 mg/dL]) in the outpatient setting is 70%, but this is reduced to 50% for those at risk of hypoglycemia. 16 There are no guidelines on percentage times in range that should be maintained in hospital. Percentage time in range should ideally be calculated using > 70% data for at least 14 days, 16 and the overall calculation from short hospital admissions whilst people are unwell may be inappropriate. Furthermore, glycemia is likely to change hour by hour, and day by day, whilst people are unwell and it is difficult to meaningfully use the concept of “time in range” in that situation. Overall, published data relating to glucose control, time in range in hospital and clinical outcomes are unclear. 17 The consensus view is that avoiding hypoglycemia is the priority, with the secondary aim of avoiding significant hyperglycemia.
JBDS-IP propose ensuring hypoglycemia avoidance (percentage time below range < 1%) and to minimize hyperglycemia. The usual JBDS-IP glycemic targets in hospital are 6 to 10 mmol/L (108-180 mg/dL) 18 for the acutely unwell person, but we recognize for some individuals who are well in hospital, then the outpatient target of 3.9 to 10 mmol/L (70-180 mg/dL) may be acceptable (eg, in otherwise fit, well adults awaiting elective surgical procedures). Targets should therefore be individualized and the reasons why targets in hospital differ from the outpatient setting should be discussed with the individual. Further studies are required to establish appropriate CGM metrics for people in hospital.
For the acutely unwell person in hospital, if glucose is between 4.0 and 5.9 mmol/L (72-106 mg/dL) and CGM arrow(s) are trending down, this is indicative of looming hypoglycemia 19 and should be reviewed immediately by the individual and discussed with the clinical team. A small carbohydrate snack (4-8 grams) can arrest the fall to hypoglycemia (particularly if trend arrow downward). Other interventions may include adding glucose to an intravenous infusion or simply rechecking blood glucose levels sooner than planned. 20 For admission for elective procedures and/or the peripartum period, standard targets may apply.21,22
Alarms and Alerts in Hospital
People using CGM may have individualized alarms, and if the person is able and willing to self-manage, alarm settings may be kept as in the outpatient setting. Although the usual JBDS-IP recommendations for target glucose levels in hospital are 6 to 10 mmol/L (72-180 mg/dL) (in elderly frail, aim for 6-12 mmol/L [72-216 mg/dL]), 18 we do not recommend having alarms set at these targets, due to the burden of high alarm frequency. We suggest alarms to be used as safety nets, to require action (Figure 1). It is important to note that these thresholds are set at which intervention would be necessary on safety grounds, rather than efficacy. As clinical teams gain more experience, these systems may be linked with hospital electronic health records, and we expect their use for efficacy to increase. At present, the alarms could be used to trigger a CBG reading and an intervention by the ward nursing staff according to clinical advice.
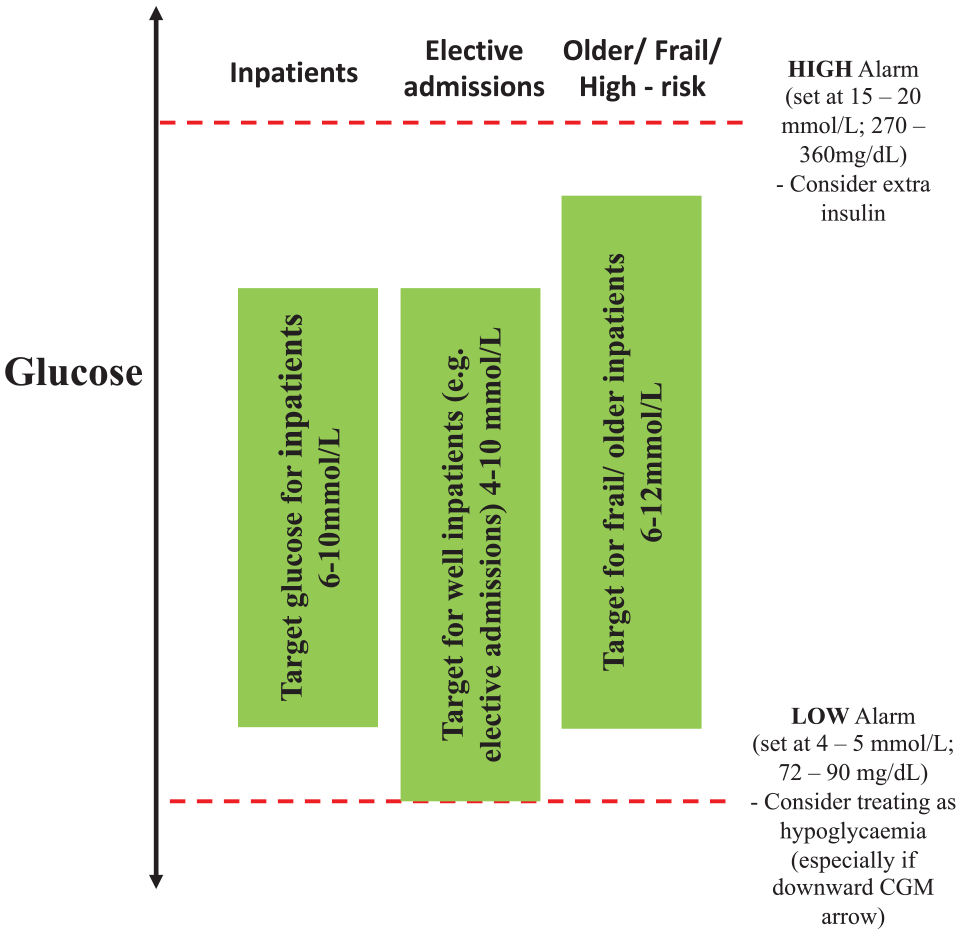
Alert/alarm settings for CGM in hospital. Abbreviation: CGM, continuous glucose monitoring.
If CGM is being used to alert the medical team, the settings can be programmed as:
HIGH ALERT: set at 15 to 18 mmol/L (270-324 mg/dL)—consider extra insulin.
LOW ALERT: set at 4 to 5 mmol/L (72-90 mg/dL)—consider treating as hypoglycemia (especially if downward arrow on CGM).
Some users may have the alarms switched off, therefore, if the system is used to alert the medical team, it must be ensured the alarms are switched on.
Capillary Glucose Monitoring Whilst the Individual Wears CGM
Due to the risk of inaccuracy during acute illness, we recommend CBG should be checked at least twice daily for people using CGM in hospital, irrespective of whether the device needs calibration. On admission to hospital, it should be explained to the person with diabetes that regular CBG monitoring is necessary for safety reasons, and for alerting staff to out of range results. Nursing staff should also be aware to perform additional CBG testing in case of any concerns of discrepancy with symptoms.
Discrepancies Between CGM and CBG Glucose
There may be a discrepancy observed between CGM and CBG glucose. We define an acceptable difference as being within ± 20% of the absolute difference between CGM and CBG glucose levels > 5.6 mmol/L (> 100 mg/dL) or within ± 1.1 mmol/L (±20 mg/dL) of the absolute difference between CGM and CBG glucose if CBG is ≤ 5.6 mmol/L (≤ 100 mg/dL). This is based on the definition for the reference standard for integrated CGM (iCGM) devices, 23 and can be referred to as %20/20.
If the discrepancy is significant, we advise more frequent CBG monitoring for the next few hours (depending on clinical need). For CGM devices that can be calibrated (eg, Dexcom G6) consider calibration with POC CBG and use if accurate (ie, within %20/20). If the discrepancy persists, remove the sensor, and replace.
Table 2 outlines some situations in which CGM users in hospital should be advised to check CBG before treatment decisions 12 (rather than using sensor glucose). Reasons for ensuring capillary glucose checks are performed in hospital, include quality assurance, and for hospitals with integrated electronic records, point of care glucose may be linked to the diabetes team. As per manufacturer guidance, it is important to ensure CGM devices are not inserted into an area of generalized edema or cellulitis. If the patient is due for a procedure or operation, ensure the sensor is on a different area of the body (contralateral side) so that it is not affected.
Situations to Always Check Fingerstick CBG.

Abbreviations: CBG, capillary blood glucose; CGM, continuous glucose monitoring.
Documentation of Glucose Readings in Hospital
Currently most CGM data are not integrated with hospital electronic health records, therefore, health care professionals need to be mindful of how data are documented. Due to the influx of glucose readings available from CGM devices, we recommend a minimum standard of documentation in hospital records of fasting, pre-meals, and before bed readings. Episodes of hypoglycemia and hyperglycemia, triggered through alarms, should also be documented and the method through which glucose values have been obtained.
Diabetes teams receiving automated alerts from CBG monitoring should be mindful that until CGM systems are integrated with electronic health records, they will not be directly alerted like POC glucose systems.
In the future, we anticipate formal linkage of CGM data into the electronic health records. The integration of Continuous Glucose Monitoring Data into the Electronic Health Record (iCoDE) Project aims to facilitate integration of CGM data and provides standards towards supporting this.24,25
CGM use in Hospital
CGM management in the well person
Unless incapacitated or acutely unwell, most people using CGM in a medical or surgical ward are safe to continue to use their CGM if admitted to hospital. 26 A large multicenter pooled analysis on accuracy of CGM data using Dexcom G6 from 218 non-critically ill people with diabetes on medical and surgical wards (n = 4067 matched glucose pairs) revealed a mean absolute relative difference (MARD) of 12.8% and median absolute relative difference of 10.1%. 27 Table 3 summarizes some of the key studies assessing accuracy and efficacy of CGM use in hospital.
Summary of Key Studies Assessing Current CGM Systems Used on General Wards, the Critical Care Setting, Perioperatively, and During Diagnostic Imaging.
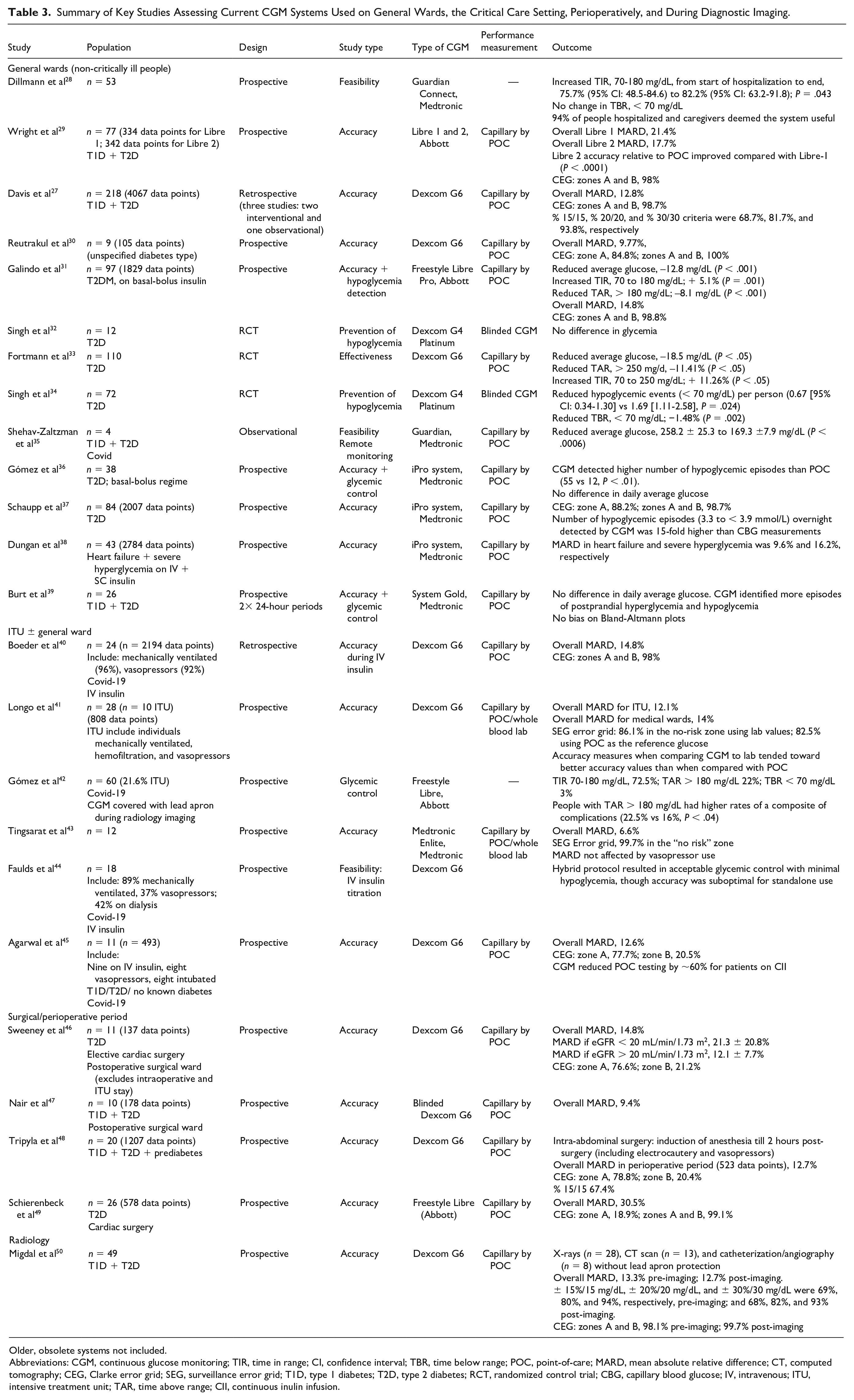
Older, obsolete systems not included.
Abbreviations: CGM, continuous glucose monitoring; TIR, time in range; CI, confidence interval; TBR, time below range; POC, point-of-care; MARD, mean absolute relative difference; CT, computed tomography; CEG, Clarke error grid; SEG, surveillance error grid; T1D, type 1 diabetes; T2D, type 2 diabetes; RCT, randomized control trial; CBG, capillary blood glucose; IV, intravenous; ITU, intensive treatment unit; TAR, time above range; CII, continuous inulin infusion.
As per manufacturer guidance, it is important to ensure that CGM devices are not inserted into an area of generalized edema or cellulitis. If the patient is due for a procedure or operation, ensure the sensor is on a different area of the body (contralateral side), so that, it is not affected.
Figure 2 summarizes the CGM management for acute admissions presenting to hospital.
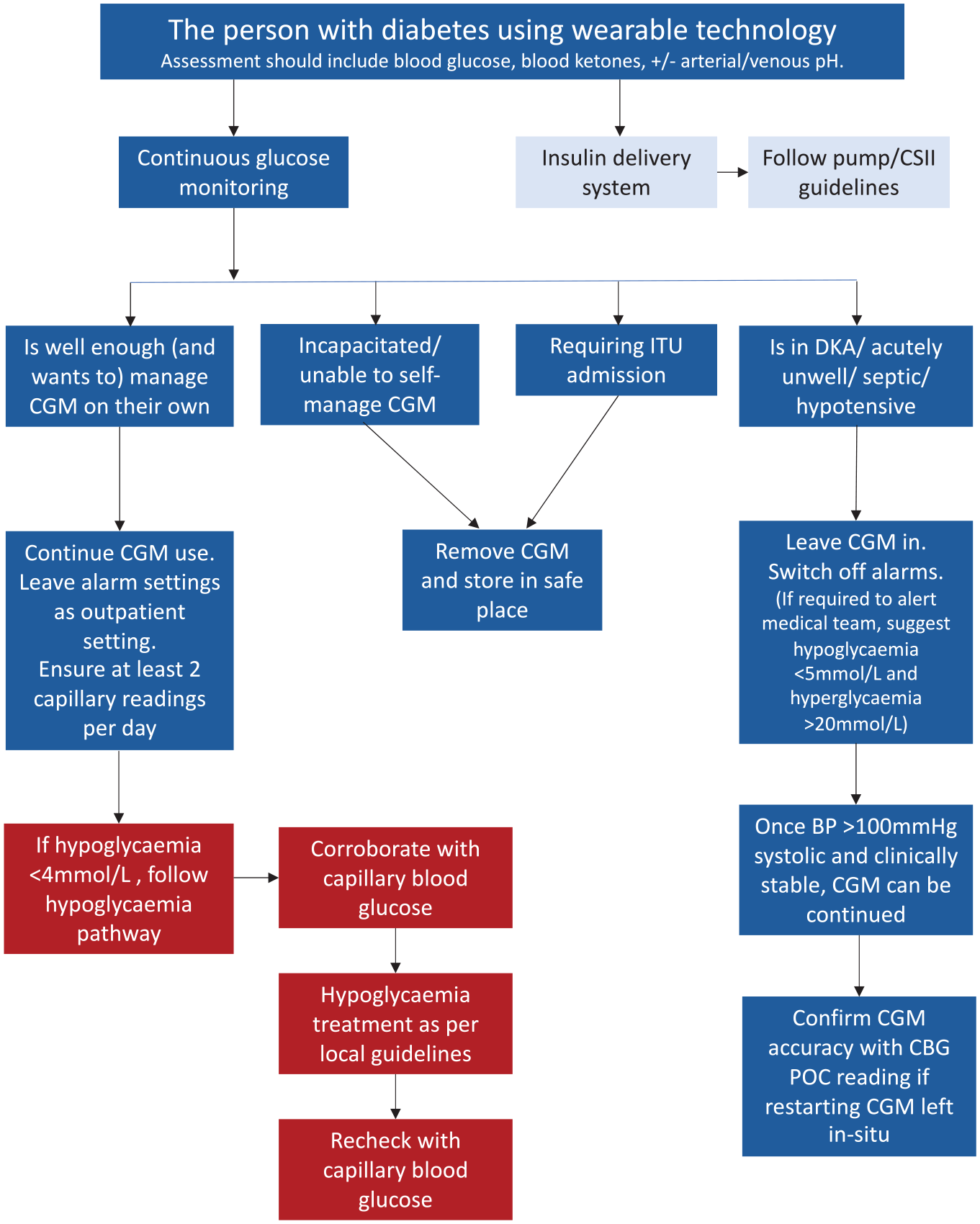
Emergency admissions and CGM management. Abbreviations: CGM, continuous glucose monitoring; CSII, continuous subcutaneous inulin infusion; ITU, intensive treatment unit; DKA, diabetic ketoacidosis; BP, blood pressure; CBG, capillary blood glucose; POC, point-of-care.
CGM in the unconscious or incapacitated individual
If the person is unable to self-manage the CGM, and nursing staff are unable to manage/use to device, JBDS-IP advise that staff should revert to standard POC CBG monitoring. During this time, the CGM sensor device can be left in situ (unless the duration of illness is longer than the manufacturer’s guidance on sensor wear).
If the individual is unlikely to be able to self-manage for a significant period of time (eg, due to intubation, traumatic brain injury), the sensor should be removed.
We suggest avoiding placing CGM sensors on the abdomen in the prone individual, as increased pressure may reduce sensor accuracy. Studies involving sleep51,52 and belt compression 53 have suggested sensor performance may be affected by the effect of local compression pressure on reducing blood flow in the area of the sensor. This is particularly relevant to individuals in the high dependency or intensive care setting, where proning is recommended for Covid.
CGM in the septic, unwell individual
In the acutely unwell person (ie, hypotensive systolic blood pressure [BP] < 100 mm Hg and/or hypothermic/hyperthermic) and who may be volume deplete, it has not yet been established whether CGM systems intended for the outpatient setting can deliver adequately accurate results. Therefore, during period of acute illness, and in particular hypotension, JBDS-IP do not recommend CGM use during this time and recommend that staff revert to standard POC CBG monitoring. The CGM devices may be left in situ and once individuals are stabilized with systolic BP > 100 mm Hg, CGM use may be continued on the ward. We recommend reassessing and confirming CGM accuracy with POC CBG following the individual being acutely unwell to ensure the CGM sensor is functioning properly.
CGM during hyperglycemic emergencies
Due to rapid changes in blood glucose during a hyperglycemic emergency and on a fixed or variable rate intravenous insulin infusion (FRIII/VRIII), we do not recommend CGM use during this time. POC CBG monitoring should be performed to adjust insulin infusions (due to lack of evidence in this area).
The Freestyle Libre system has been assessed for adjustment of insulin infusion rates in 10 people with DKA (167 paired values). In comparison with CBG, the mean CGM glucose was lower than CBG (CGM glucose: 9.2 ± 3.2 mmol/L vs mean CBG 11.1 ± 3.2 mmol/L [166 ± 57 vs 200 ± 57 mg/dL]), though both were highly correlated (r = 0.84, P < .001). 54
Due to the paucity of data, we do not yet recommend CGM use for insulin management during hyperglycemic emergencies. Continuous glucose monitoring devices may be left in situ and once individuals are stabilized back on their usual insulin regime and systolic BP > 100 mm Hg, CGM may be used to guide care on the ward. We suggest reassessing and confirming CGM accuracy with POC CBG following the individual being acutely unwell to ensure the CGM sensor is functioning properly.
CGM in the intensive therapy unit
Continuous glucose monitoring can be an important tool in the critical care setting, but there is as yet insufficient evidence to support its immediate introduction into the intensive therapy unit (ITU; also known as intensive care unit “ICU”) setting. 55 Concerns include lack of accuracy in critically ill people with impaired tissue perfusion, 56 hypotension, hypothermia, hypoxia, vasopressor use, and potential substance interference (acetaminophen [ie, paracetamol], maltose, ascorbic acid, dopamine, mannitol, heparin, uric acid, hydroxyurea, and salicylic acid). 57 In addition, in the ITU setting, there are already systems in place for much more frequent blood glucose monitoring to be a routine than occurs outside the ITU setting.
Although there are increasing studies on the use of CGM in ITU, the major barrier for incorporating CGM systems within the critical care setting at present is its implementation. Many institutions have needed to rely heavily on the diabetes team for nursing/clinician training, person selection, device placement/set-up, and ongoing support. 58 The lack of a 24/7 diabetes team and infrastructure to support in majority of the UK hospitals (manuscript submitted for publication) remains a challenge to effective implementation within critical care.
At present, CGM remains contraindicated for use in critically ill people, 59 and therefore should not be used in the ITU setting. It is likely with increasing emerging evidence of CGM in the ITU setting, 45 this guidance may be updated in due course.
CGM and radiology investigations
Currently, no CGM device is labeled to allow for exposure to X-rays, computed tomographic (CT) scans, magnetic resonance imaging (MRI), diathermy, radiation therapy, or other types of radiation. However, CGM accuracy with Dexcom G6 has been assessed during certain radiologic procedures (X-rays, CT scan, and angiography), reporting an overall MARD of 13.3% pre-imaging and 12.7% post-imaging. 50 In non-clinical bench studies, Abbott Freestyle Libre 1, 2, and 3 systems have been tested in simulated diagnostic radiologic procedures, and the sensors maintained functionality with both X-rays, CT, and specified MRI conditions. 60
Diabetes teams that have covered their CGMs with a lead shield have not reported any adverse events for CT, radiotherapy or when electrocautery is used during surgical procedures. 61 Therefore, guidance may be individualized by each center, with the device either being removed or covered with a lead shield during these procedures.
For MRI procedures, although the transmitter data storage and communications function is suggested to remain intact, there is a small displacement force, with minimal heating and current in sensor/ transmitter pairs. 62 We therefore recommend all CGM devices should be removed during MRI procedures. The only exception is the implantable long-term real-time continuous glucose monitoring (rtCGM) sensor, Eversense (Senseonics, Maryland), which can be used during MRI in prespecified conditions as per manufacturer guidance.63,64 However, the external Eversense Smart Transmitter must still be removed during scanning, and once scanning is completed, the transmitter can be easily repositioned on the skin. 64
CGM during in the perioperative surgical period
Glycemic data obtained from CGM devices intraoperatively would be ideal for an anesthetist. Concern has been previously raised that CGM technology has not been consistently reliable intraoperatively, with associated signal loss and negative bias (during 60% of patients in one study) 65 . However, there are case reports to suggest that rtCGM can be safely used. 48 There are no studies of intermittently scanned continuous glucose monitoring (isCGM) (Freestyle Libre) use during surgery and the frequency of scanning.
We suggest that CGM may be used to guide the use of capillary or blood gas glucose monitoring, but treatment decisions should be based on POC CBG or blood monitoring particularly for major surgical procedures.
For minor procedures without sedation (eg, esophago-gastro-duodenoscopy and colonoscopy), the CGM can be continued as for any person with diabetes. The CGM sensor should be situated away from the operative site and any diathermy pad(s).
In terms of diathermy, manufacturers advise removal of CGM devices prior to diathermy treatment as there are limited data on safety and impact on sensor performance. However, rtCGM is increasingly used during cesarean section with both unipolar and bipolar diathermies. 23 The guidance may therefore be individualized for each center. We recommend checking POC CBG after procedures to ensure the individual’s CGM is functioning properly.
Glucose levels should be within an acceptable range pre-procedure (ie, 6-12 mmol/L; 108-216 mg/dL), with an aim to keep glucose in the range 6 to 10 mmol/L (108-180 mg/dL) ideally. Glucose between 4 and 6 mmol/L (72-108 mg/dL) indicates looming hypoglycemia. 21 Continuous glucose monitoring trends at this stage can be particularly helpful, with a downward arrow being more suggestive of looming hypoglycemia than an upward arrow. Consider intervening at glucose < 6.0 mmol/L (108 mg/dL) to prevent hypoglycemia with oral or intravenous carbohydrate. 22 However, for many optimally managed people with diabetes, a range of 4 to 6 mmol/L (72-108 mg/dL) may be their normal when there are not eating. In these cases, it is important to have a discussion with the person with diabetes about the need to avoid severe hypoglycemia.
Continuous glucose monitoring should not be used for glucose monitoring in the event of intraoperative hypotension or hemorrhage.
CGM management during labor
Continuous glucose monitoring systems can be safely used during pregnancy and labor delivery. 66 Glycemia during labor should be maintained in either the target range advocated in the UK guidelines from the National Institute of Health and Care Excellence (4.0-7.0 mmol/L; 72-84 mg/dL) or in the more liberal range of 5.0 to 8.0 mmol/L (60-96 mg/dL) due to lack of randomized control trial (RCT) evidence for either target. 23
Women should usually continue their recommended glucose monitoring until they are in established labor or fasting prior to cesarean section. The peripartum diabetes management plan includes hourly glucose monitoring using CGM or POC CBG during established labor or after artificial rupture of membrane or on admission for cesarean section. 21
Women should be made aware to move the sensor to the arm prior to cesarean section, so that, they do not interfere with the operative field.
Women continuing CSII or multiple daily insulin injections during labor or leading up to a cesarean birth, who are using either rtCGM or isCGM can continue to use this during delivery ensuring that a glucose level is recorded at least hourly.
Stopping and restarting CGM
The person with diabetes is ideally best placed to remove the sensor, with the phone app or receiver to be informed of sensor session ending. If this is not possible, the sensor may be removed and safely stored.
As some sensors have the transmitter attached (ie, Dexcom One/G6, Medtrum A6, Glooko Rx and Medtronic Guardian 3/4), we recommend any devices removed, be labeled, and stored in a safe place and documented. This should ensure that the relatively expensive transmitter is not discarded. Even though the implantable sensor (Eversense) is not available in many parts of the world, including the United Kingdom, the transmitter can be removed and then worn after.
For health care professionals familiar with the diabetes technology, the sensors can be safely disposed of. For health care professional unfamiliar with the technology devices, due to the cost of diabetes wearable technology, we suggest that they should be safely stored until further input from the diabetes team.
For restarting CGM, the person with diabetes is ideally best placed to restart this as they will have received training and will be experienced in this process.
CGM use during cardiac arrest situations
During a cardiac arrest situation, CGM devices should be removed for external DC cardioversion. This is due to metal within the CGM sensor, which may act as a conductor, and therefore, there is a theoretical risk of energy dispersed through the sensor. We recognize that in a cardiac arrest situation, this may be difficult, and resuscitation should not be delayed, but where possible the device should be removed.
Continuous glucose monitoring derived glucose should not be used to guide treatment of hypoglycemia in cardiac arrest situations.
Limitations and Accuracy
There are no currently defined standards for CGM accuracy in the inpatient hospital setting. 58 The US Food and Drug Administration (FDA) has defined overall accuracy as 87% of values within ± 20% of the reference standard for iCGM devices. 23 Overall, lower MARD is seen as representing better sensor performance, with a MARD of < 10% representing sufficient accuracy for CGM device readings to make insulin dosing decisions. 67 As demonstrated in Table 3, majority of the studies have been compared against POC CBG, which remains a limitation of most accuracy studies. The MARD calculated from lab blood glucose as a reference was significantly lower than the MARD using POC as a reference glucose. 68 Lab blood glucose is the gold standard of reference and in-hospital POC glucose meters may have up to ± 15% deviation from the lab glucose per the International Organization for Standardization (ISO) standards.
Exposure to several substances may impair CGM accuracy. 58 Acetaminophen (ie, paracetamol) can reduce the accuracy of CGM measurements from certain CGM systems (Dexcom G5, Medtronic Guardian) but does not interfere with sensor glucose measurement by the Dexcom G6 CGM or Freestyle Libre (note, Dexcom G6 may be affected at higher doses). 69 Accuracy may be affected with other substances (namely maltose, ascorbic acid, dopamine, mannitol, heparin, hydroxyurea, and salicylic acid) with certain devices.11,58,70
Challenges and Future Directions
Implementation of diabetes technology in hospital presents several challenges. 61 Continuous glucose monitoring devices are currently infrequently encountered in the hospital, therefore, hospital staff are unlikely to be familiar with their operation. Upskilling of hospital staff is required for clinical and allied health care professionals, particularly in the acute setting, to understand and identify between insulin delivery systems and glucose monitoring systems. Furthermore, hospital diabetes teams are required to support implementation, and at present, there remains great variation nationally for out-of-hours and weekend specialist input (manuscript submitted for publication).
Continuous glucose monitoring accuracy studies are also required for specific hospital populations, including individuals on haemodialysis or peritoneal dialysis. There are no current consensus guidelines on target CGM metrics in hospital, and further data (including long-term outcomes) are required, including post myocardial infarction, stroke, or coronary artery bypass. In the future, integrated systems with continuous ketone monitoring could be combined with CGM technology to reduce rates of diabetic ketoacidosis, and aid in the management for people in hospital (accompanying Misra et al paper available at https://doi.org/10.1177/19322968221137360). 71
Currently, most CGM data are not integrated within the electronic health records of the user, therefore, health care professionals need to be mindful of how data are documented. There is a need for data standards and implementation policies to integrate CGM data into electronic health records, and the iCoDE Project aims to facilitate provides standards towards this integration. 72 Hospitals, CGM manufacturers, and service providers for electronic health records need to work together to integrate systems. 11 Furthermore, the data acquisition process must be compliant with regulatory privacy rules and cybersecurity policies, 11 as users would need to grant permission to transfer their CGM data to hospital health records. Future directions include the incorporation of CGM data to electronic prescribing with prompts and allowing access for inpatient diabetes teams. Additional linkage to national datasets can potentially allow comparisons between hospitals and drive improvement.
Finally, further studies are required on whether CGM systems can be effective in guiding better glucose control in hospital and as a result, reduce hospital length of stay, harms related to poor glycemic control, self-management, and/or experience in hospital. Information is also required on the impact of wearable diabetes technology use on costs for safe and effective implementation. Increased costs may arise through repeated sensor malfunctions or need for replacement in patients undergoing MRI or other procedures. These costs are likely to be negated by the benefits of safer glycemic control, reducing in hospital hypoglycemia, hyperglycemia and the potential resulting diabetic ketoacidosis or hyperosmolar hyperglycemic syndrome.
Conclusion
With an increasing number of people with diabetes using CGM and insulin pumps, it is inevitable that health care professionals working in hospitals will be increasingly exposed and need to care for people with these devices. This technology is constantly improving and is likely to play a greater role in hospital, potentially significantly reducing the burden of health care professionals and mitigating the risk people face from their diabetes when hospitalized. In the near term, the availability of accurate CGM systems combined with automated insulin dosing systems using software algorithms will likely facilitate improved glycemia, and reduction of hypoglycemia and hyperglycemia in critically and non-critically ill people with T1D and T2D. As artificial intelligence becomes more established, the dosing algorithms for insulin delivery in hospital will become individualized for closed-loop control of glycemia. 73 The challenge is likely to lie in safe implementation, integration of data and associated costs within clinical practice.
Footnotes
Acknowledgements
SM is supported by the National Institute for Health Research (NIHR) Biomedical Research Centre at Imperial College Healthcare NHS Trust.
Abbreviations
CBG, capillary blood glucose; CEG, Clarke error grid; CGM, continuous glucose monitoring; CSII, continuous subcutaneous inulin infusion; DKA, diabetic ketoacidosis; FRIII, fixed rate intravenous insulin infusion; HCL, hybrid closed-loop; iCGM, integrated continuous glucose monitoring; isCGM, intermittently scanned continuous glucose monitoring; ITU, intensive care unit; IV, intravenous; JBDS-IP, Joint British Diabetes Societies for Inpatient Care; MARD, mean absolute relative difference; MRI, magnetic resonance imaging; POC, point-of-care; RCT, randomized control trial; rtCGM, real-time continuous glucose monitoring; SC, subcutaneous; SEG, surveillance error grid; TAR, time above range; TBR, time below range; TDD, total daily dose; TIR, time in range; T1D, type 1 diabetes; T2D, type 2 diabetes; UK, the United Kingdom; VRIII, variable rate intravenous insulin infusion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AL has received payments for speaking and advisory boards from Insulet, Dexcom, Abbott Diabetes Care, Novo Nordisk, Sanofi, and Institutional Research Support from Abbott Diabetes Care, Novo Nordisk. DF is the national lead for the UK diabetes care accreditation program and has received speaker honoraria from AstraZeneca, Novo Nordisk, and Sanofi Diabetes. SM is appointed to the Board of Trustees at the Diabetes Research and Wellness Foundation and is in receipt of funds from Dexcom for an investigator-initiated research study. GR has received personal fees from Abbott Diabetes Care, Sanofi Aventis, and Eli Lilly. PC has received personal fees from Abbott Diabetes Care, Dexcom, Diasend, Eli Lilly, Insulet, Medtronic, Novo Nordisk, Roche, and Sanofi Aventis. KD is the chair of the Joint British Diabetes Societies for Inpatient Care and has received speaker fees, travel or taken part in advisory boards for AstraZeneca, Sanofi Diabetes, Boehringer Ingelheim, Lilly, and Novo Nordisk. PA has no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
