Abstract
On July 21, 2021, Diabetes Technology Society convened the virtual Green Diabetes Summit. The event consisted of 23 representatives from key stakeholder groups based in both the United States and Europe. The purposes of the summit were to (1) provide background on the complexity of addressing sustainability-related issues, including waste management, of diabetes devices from many different perspectives along the products’ life cycle stages, and (2) determine the feasibility and role of a coalition of stakeholders to find solutions, particularly in the design, use, and proper disposal of diabetes devices used in home care that no one stakeholder can resolve on their own.
Introduction
Diabetes Technology Society (DTS) launched the first ever Green Diabetes Initiative in 2020. 1 The Green Diabetes Summit (GDS) was the culmination of a two-year planning process to assemble representatives from the patient community, clinicians, government officials, waste handling service providers, and industry representatives to discuss what can be done to improve sustainability and waste management associated with the use of medical products by people with diabetes (PwD) for their therapy. The number of PwD is increasing and the types of wearable devices and wireless sensors for diabetes therapy are also increasing. Many of the devices contain needles, electronics, batteries, and so on. Although workplace safety laws are in place for sharps, there are no such laws to protect home health workers who might be exposed to dangerous sharps in the home workplace. When sharps are placed in recycling bins or discarded with household trash, there is a high risk of injury for home health workers and solid waste workers.
While DTS has published a number of articles in the past regarding issues about diabetes-related waste and the environment,2-6 the GDS was intended to bring together key stakeholders to explore how to design, manufacture, store, and transport diabetes devices with better sustainability and how to best handle them at the end of life with sound waste management policies, also enabling recycling. The discussions during the patient/health care professional, industry, and coalition panels were conversational and are presented as such in the meeting report. The discussion during the government panel followed a presentation approach and was also presented accordingly.
Sustainability
Introduction to Sustainability
Donna A. Seid Consulting, The Hague, the Netherlands
Bullet Points
Catastrophic global challenges such as dwindling natural resources, climate change, and pollution are some of the largest drivers for companies to incorporate sustainability initiatives, targets, and goals into their business strategies.
Aside from looking at environmental impacts, social, ethical, and regulatory impacts that affect a product at each stage of its life cycle should also be considered.
Medical device manufacturers can contribute to the environment and society through better practices in sustainability and waste management at each stage of a device’s life cycle.
Important Drivers for More Sustainable Practices and Products
As we face increasingly catastrophic environmental challenges around the globe, the push for addressing sustainability in the medical device industry becomes ever more important. Climate change remains a priority, as evidenced by recent events: wildfires in California, the heat dome over the northwestern United States, and the flooding in parts of Europe and China. Pollution continues to intensify, as observed by plastics in the oceans and increasing generation of electronic waste. At the same time, resources and raw materials are being consumed more rapidly than they can be naturally replenished. 7 Companies are competing for dwindling natural resources. As the global population continues to grow—expected to reach 9.9 billion people by 2050 8 —use of products, with their associated raw material consumption, and generation of waste are expected to increase, further exacerbating current dilemmas.
Although environmental impacts continue to be the major driver for sustainability, consideration of the social, ethical, and regulatory impacts throughout a product’s supply chain are inclusive within the scope of sustainability. Issues around social injustice and human rights such as diversity, equity, and inclusion, and assurances against human trafficking, slavery, and child labor should also be addressed. This can be addressed through obligatory commitments to “Code of Conducts,” third-party partnerships, and both internal and external audits. Ethical performance should also be scrutinized in such areas of anti-corruption, anti-bribery, and fair market practices, including consideration of fair-trade pricing when sourcing raw materials from third world countries. There exist many global regulations and conventions to which companies comply in the areas of environmental preservation, health and safety, and protection of endangered species and biodiversity. There are also emerging regulations regarding other company-related actions with regard to sustainability, product stewardship, and extended producer responsibility (EPR). The European Union’s Green Deal is a set of policy initiatives aimed to meet the region’s goal to be climate neutral by 2050. 9 The initiatives range from decreasing greenhouse gas emissions, to investing in companies that take into account natural and social capital.
Companies are signing up to voluntary initiatives. They are helping their local communities through donations and volunteerism, sponsoring foundations whose efforts and goals align with their business interests. Company CEOs (chief executive officers) have joined the World Business Council for Sustainable Development to work toward their shared aim to provide maximum positive impact for shareholders, the environment, and society. 10 Companies have also signed on to voluntary initiatives with particular environmental focuses, such as the Ellen MacArthur Foundation’s Plastics Pact Network whose purpose is tackling plastic waste pollution by bringing together stakeholders to implement solutions toward a circular economy for plastic. 11 Many companies have also committed to the 2030 Agenda for Sustainable Development, which includes 17 sustainable development goals.12,13 The 17 goals, presented in Table 1, are aimed to end poverty, reduce inequality, ensure prosperity, and protect the planet.
Product Life Cycle Impacts
Throughout a product’s life cycle, many different considerations should be made to improve a product’s sustainability. A systemic review of a product’s life cycle presents a number of sustainability risks and opportunities. The typical stages of a product life cycle are shown in Figure 1: innovation, sourcing, manufacturing, distribution, use, and end-of-life for products and their associated packaging. Hypothetical examples of sustainability-related considerations at each product life cycle stage are presented in Table 2.
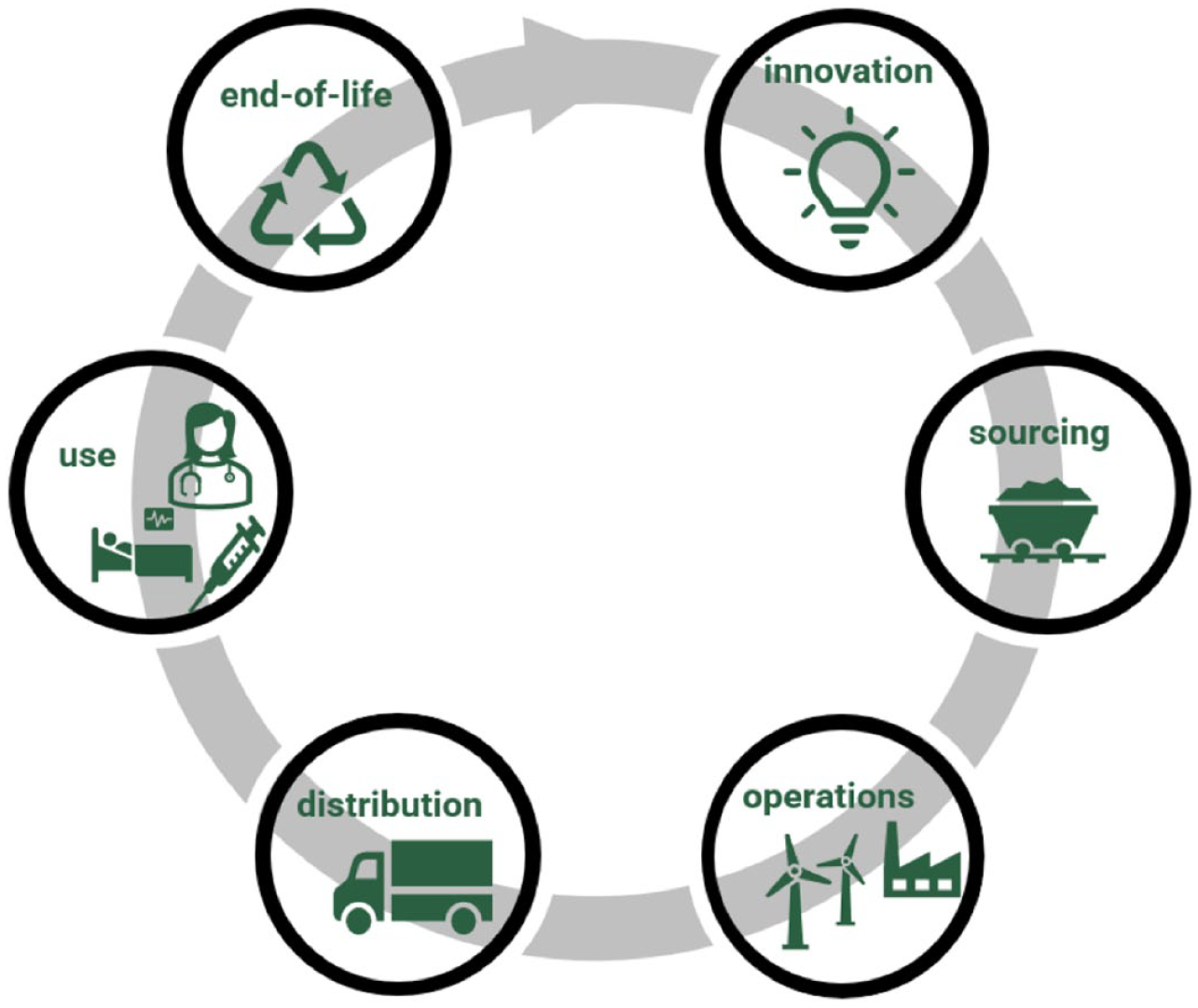
Typical product life cycle.
Hypothetical Examples of Sustainability-Related Considerations at Various Stages of a Product’s Life Cycle.

Diabetes Sustainability and Waste Management
Science Consulting in Diabetes GmbH, Kaarst, Germany
Diabetes mellitus is one of the most common chronic medical conditions globally. According to estimates from the International Diabetes Federation, 463 million adults worldwide were living with diabetes in 2019. 14 By 2045, the number of adults living with diabetes is estimated to reach 700 million. 14 As technology to treat diabetes continues to develop, PwD are increasingly utilizing medical products in their at-home care. In addition, these products are also used in hospitals and practices. Although reasons for use vary, including improved safety and convenience of treatments, one thing is certain. A vast amount of waste, especially plastic waste, is and will be generated by used diabetes devices and their packaging. In addition, the disposal and recycling of medical devices represent a more particular challenge. A single product may contain many different components, each constructed from a different material requiring thought and effort to disassemble and sort into the appropriate bins to enable recycling of these materials. As a result, diabetes devices are normally left assembled and thrown away with general household waste, leaving the issue of disposal to local waste handling facilities and putting their employees at risk of injury or infection in case the waste also contains sharps. Patients are well aware of this and will start to complain about this.
A better balance between the performance of the products and their environmental impact has to be found; however, this is a complex story with many different aspects. At least in Europe, the regulatory environment toward plastic waste is changing rapidly in general and stricter guidelines are already being approved. One logical next step is that new devices should be better designed, use more renewable or recyclable materials, require less packaging, and rely on more environmentally-friendly manufacturing processes and distribution options. Ideally, only reusable devices would be manufactured and used to reduce the disposal of such products to a large extent.
Recycling of medical devices can be costly and there is not a good market for plastic waste. However, there are interesting differences toward this topic between countries/continents. The medical device industry and also the packing industry are aware of such requirements (at least the statements on the homepages of the different companies indicate this); however, the changes associated are time-consuming and costly, which represents an issue in a highly competitive area. As industry also has to stick to regulatory requirements and legal aspects, there is a need for willingness for change also at the political level.
The complexity of the topic and the costs associated are a hurdle to changing the situation; however, there will be political pressure and additional pressure from users. Patients might have environmental aspects more in mind in the future when—for example—systems for continuous glucose monitoring (CGM) with different ecological footprints are available. Companies that are willing to invest in our future should not only be applauded, but they should also have a clear advantage that is paying back to them. Only a combined reaction of all parties involved can make a change happen, and changes have to be made at all levels: patients, hospitals, practices, storage facilities, and so on. We will have to move out of our comfort zone.
How Patients Dispose of Medical Waste and Sharps
Moderator
WNWN International, Burlington, Connecticut, USA
Panelists
Type1EU, Brussels, Belgium
Mayo Clinic Arizona, Scottsdale, Arizona, USA
American Association of Diabetes Care and Education Specialists, Montross, Virginia, USA
Pfützner Science and Health Institute, Mainz, Germany
Diabetes Technology Society, Burlingame, California, USA
Bullet Points
Improper sharps disposal into the environment is a global concern.
A lack of standardization in waste disposal options contributes to unsafe disposal of medical waste and sharps.
More research into how people are currently disposing of their medical waste and sharps is needed to define the current barriers that exist for the proper disposal of waste before a solution can be developed.
Proper education is an imperative first step for encouraging safe waste disposal habits. Health care professional advocates should take steps to identify key journals and sites where information is available to share with patients and to publish articles about safe medical waste and sharps disposal.
Consumer convenience for safe disposal options also needs to be considered because if disposal is too inconvenient, then the user may just dispose of waste improperly.
It is necessary to promote and produce more sustainable designs of diabetes devices, ideally with patients included early on in the design process.
The entities that will be trying to disassemble these devices to promote recycling should also be included in the design process.
Patient Community Concerns
There are many factors that influence a person’s decisions on what to do with the medical waste generated from diabetes management. Unfortunately, misunderstanding and misinformation from different sources, such as hospitals versus pharmacies, and inconvenience in finding sharps disposal locations and other resources can cause many PwD to dispose of their trash into normal trash bins, which puts themselves and others at risk for injury or infection. Individuals may not be aware of the dangers of inappropriate disposal of medical waste and sharps, or there may be minimal or no resources or programs available for PwD to safely dispose of diabetes waste. Furthermore, not every country has diabetes associations engaged in the topic of appropriate waste handling. There are often available programs and resources, but they are typically not person-centered, often designed to cater to manufacturers, and not well publicized. Managing diabetes waste can also be expensive because separate containers for medical waste and sharps disposal are not provided to PwD. People who wish to dispose of their waste safely may also need to travel to waste facilities or mail it to waste handling companies. All of these factors play a role in discouraging PwD from practicing proper disposal habits. Internationally, other countries may not have started pursuing appropriate waste management initiatives despite the fact that this is a global concern. A product that has many different parts with various disposal requirements for each component may also discourage proper waste disposal habits. Although sterile packaging is certainly necessary to protect the contents from contamination, an opportunity to reduce excessive waste exists in the materials used to create packaging.
Even so, more and more PwD, endocrinologists, medical device manufacturers, and diabetes care and education specialists are making efforts to prioritize managing diabetes-associated waste. In the early 2000s, the Safe Needle Disposal Coalition was formed and there was legislation passed in individual states in the United States to identify where PwD could safely dispose of their sharps waste and to educate PwD on safe waste disposal practices. Web sites with specific information for sharps disposal in certain regions were established and readily available for people who were curious about what to do with their sharps and medical waste. Diabetes care and education specialists often include safe sharps disposal as part of their curriculum, especially in patient-intensive diabetes education classes. Information booths for sharps disposal are also available and consistently popular at annual meetings for diabetes care and education specialists because PwD want information and resources. Initial individual steps have also been taken by organizations to tackle the problem of waste disposal. In the realm of industry, medical device manufacturers often offer mail back services for collecting the waste generated by their consumers. However, the supplies for collecting the sharps waste are not always readily available to the consumer, which provides another barrier to managing waste properly.
Studies have been conducted to examine and characterize current issues regarding waste disposal practices. In a study reported by Montoya et al in 2021 at the Mayo Clinic in Arizona, USA, of 150 respondents with diabetes, 64% indicated that they did not receive proper education on how to dispose of their waste, and around 40% indicated that they disposed their lancets in common household trash. 15 Evidence indicates that this is a global problem. DTS also conducted a study to describe the current landscape of sharps disposal options in commercial airports in California. This study found that even out of the airports that do offer sharps disposal options (11 of 18 respondents indicated that they did offer sharps disposal options), the implementation of sharps disposal options varies greatly. 16 Some airports have sharps disposal bins in all of their restrooms while other airports have bins in less than half. Different types of sharps disposal bins are used and it is uncertain what kind of publicizing is being done to let people know where to dispose of sharps. There is a lack of standardization with what options are being offered to people who need to dispose sharps and this likely contributes to the mishandling and unsafe disposal of medical waste and sharps.
Although it is clear that waste disposal is a problem, efficient and feasible solutions are not well explored or delineated. More research is needed to identify key issues that are leading to unsafe waste management to pave the way for effective solutions. It is known that sharps waste is handled differently from country to country and from state to state. There are no standard medical waste or sharps containers for disposal at home and there are no standard bins to identify for disposal in public spaces. It is clear from waste management habits that there is a need for standardized and available education and resources around safe medical waste and sharps disposal. There is not enough conversation about waste—how it is generated, how to handle it safely, and where to dispose of it. Managing waste needs to be made more sustainable, accessible, and standardized. Forming a coalition and publishing articles on the necessity of change could be a good first step toward inspiring federal legislation. While PwD make up a large percentage of people who need to dispose of medical waste and sharps, it may be fruitful to collaborate with representatives from populations without diabetes that may need to dispose of medical waste and sharps to create solutions for waste management. Eventually, a global public health movement and national policies would be necessary to prevent the waste management situation from getting worse, especially as the prevalence of diabetes continues to increase each year. Overall, diabetes waste management is undeniably a serious public health problem that could benefit from a solution on a global level.
The Role of Government
Moderator
National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda, Maryland, USA
The Issue: Lack of Safety Regarding Disposal of Diabetes Medical Waste and Sharps
South Bayside Waste Management Authority, San Carlos, California, USA
Bullet Points
Improper medical waste and sharps disposal poses a significant safety concern.
Standardization and further action must be taken toward a safer disposal process.
Safety Jeopardization—Puncture-Resistant, Not Puncture-Proof
Artificial intelligence is used in waste sorting at solid waste management facilities, although machines are unable to sort everything. Materials Recovery Facility (MRF) handlers and operators manually sort through waste, where needle sticks are common, while wearing $30 per pair Kevlar puncture-resistant, but not puncture-proof gloves. Sharps sticks are an issue not only for the solid waste industry but also for the wastewater industry because sharps often get flushed, resulting in the wastewater industry adjusting methodology for sewer system maintenance to avoid further sharps stick incidents. When a needle is found, the manual sorting process in an MRF must be completely halted, thereby resulting in increased time to sort through the recyclable materials and higher community curbside costs. Continual and frequent needle sticks to MRF handlers requires increased safety procedures regarding the sharps disposal process.
Safety Proposal 1: Coalition Formation and Standardization of Sharps Containers
Coalition formation between drug and equipment manufacturers can increase safety regarding sharps disposal. The coalition could pay for the production of standardized uniform containers and also for solid waste management districts to collect and properly dispatch the containers per governmental regulations. There is also a need for the standardization of sharps waste containers and for separate sharps waste collection, just as California Law requires separate battery disposal and collection. One way to address these safety concerns around sharps disposal is to treat it as a separate waste stream with a specific set of safety rules and have a final uniform destination funded by the industry producing the product itself. It is the work of the government to enact regulations and enforce a standardized method of sharps disposal and processing.
Safety Proposal 2: Standardization, Increased Governmental Regulation, and Public Education
With the current gap in information between sharps disposal process and the public, increased governmental intervention and government offered grants toward public education may narrow the gap in information. Patients need to be made aware of waste sorting and disposal processes from when sharps waste leaves the home, which is currently largely ungoverned.
Conclusions
Sharps waste is hazardous to waste management facility workers and is a source of major safety concerns.
Standardization of sharps waste collection and increased governmental intervention and regulation are important to increase sharps waste safety.
Public user education is vital, from the time of acquisition to the time of disposal.
Current Program Actions Promoting Safe Management of Medical Waste and Sharps
NYSDOH Opioid Overdose Prevention Initiative & Expanded Syringe Access Program, Albany, New York, USA
Bullet Points
Sharps litter is prevalent throughout communities and specific programs are available to aid in increased safety and proper sharps disposal.
A collaborative effort across several different entities, including individual representatives of various programs and local governments, will be required to bring about increased efforts regarding a standardized method for sharps disposal.
The Expanded Syringe Access Program
In May 2000, the New York State Legislative enacted Chapter Fifty-Six the Expanded Syringe Access Program (ESAP). 17 ESAP is a New York–funded Public and Environmental Health Initiative that (1) supports the provision of sharps and (2) promotes and supports the proper disposal of used sharps aiming to attain a statewide goal of reducing and preventing transmission of blood-borne diseases such as HIV, Hepatitis B, and Hepatitis C in the community. ESAP works closely with funded programs and local regional partners to secure various disposal sites throughout New York State. ESAP emphasizes the importance of educating and supporting the New York State community by offering free educational materials, including simple pamphlets and posters specific to the diabetes population, and working with partners and community-based organizations by purchasing and offering syringe disposal kiosks. ESAP also works with local programs, mayors, and local governments to identify various locations with sharps and medical waste litter in efforts to secure and deliver syringe disposal kiosks to those locations within New York State. Once these kiosks are delivered, the regional site determines the specific method of appropriate disposal of kiosk-collected waste, which includes a contract or memorandum agreement with a waste disposal company or programs at local hospitals and nursing homes.
ESAP in the Community
As of 2019, ESAP successfully installed more than 300 syringe disposal sites throughout New York State in 63 counties. Ninety collection sites reported a collection total of nearly 15,000 pounds of syringe waste. The amount of collected syringe waste is exponentially increasing. As of 2020, ESAP-supported syringe collection sites reported and surpassed 20,000 pounds of syringe waste collected in the second quarter of 2020 alone.
Conclusions
ESAP’s ultimate goal is to remove syringes from community circulation and safely dispose of used syringes.
ESAP strives to achieve the aforementioned goal by involving multiple entities within a community to educate needle users and agree upon a plan to ensure proper syringe waste disposal through installation of ESAP-supported syringe waste disposal kiosks.
The amount of waste collected by ESAP-supported sharps kiosks is increasing with time.
How the Government Promotes Proper Waste Management of Household Waste Generated by Disposal of Diabetes Devices and Home-Use Sharps
California Product Stewardship Council, Sacramento, California, USA
Bullet Points
Manufacturers must be responsible for managing the safe and proper return of their materials and products to ensure public safety.
Governmental regulation and policy can help enforce the safe return and disposal of sharps.
The sharps industry is far from attaining environmental sustainability; however, several adjustments may be steps toward attaining higher environmental sustainability. These include assessing the reliability and recyclability of specific components within devices.
About the California Product Stewardship Council
California Product Stewardship Council (CPSC) is a nonprofit organization that represents cities and counties throughout California and urges manufacturers to take responsibility for their product’s end-of-life and for designing greener products in efforts to achieve greater environmental sustainability.
The CPSC-Sponsored SB 212 Bill
In 2018, CPSC sponsored a bill that addresses sharps waste and pharmaceutical waste. Senate Bill No. 212 (SB 212), authored by Senator Hannah-Beth Jackson and co-authored by Assembly Members Ting and Grey, requires sharps and pharmaceutical manufacturers to manage the materials at the end of their product life. 18 Upon direct negotiation between CPSC and sharps manufacturers, it was agreed that sharps manufacturers are required to provide a free, postage paid, safe return container to the customer at the point of sale at a sufficient size to handle the number of sharps sold to the user. It is then the user’s responsibility to seal and mail back the sharps disposal container to the sharps manufacturer where it will undergo a safe and standardized disposal process. There are two main options regarding customer sharps return, including (1) customer mail back and (2) customer return to physical sharps disposal kiosks. Because of the high cost required to operate return kiosks, sharps manufacturers chose the customer mail back method. Sharps manufacturers must fully implement SB 212 provisions by July 7, 2022. The program can be replicated across the United States upon full implementation of the program in California.
The Balance Between Disposal and Environmental Sustainability
Disposal and environmental sustainability are not always compatible, as not every aspect of a diabetes device is recyclable. However, efforts toward increasing devices’ reusability, durability, repairability, longevity, and recyclability are all examples of activities intended to attain higher environmental sustainability. CPSC presently works on campaigns for all different product categories; however, this is not a simple solution because not every device component is recyclable. CPSC identifies products individually and/or by category and then decides which specific individual parts of the device can be reduced, reused, or recycled.
Conclusions
SB 212 was passed in 2018 in efforts to coordinate a standardized sharps disposal system with sharps manufacturers in California.
Full implementation of SB 212 is required by July 7, 2022, and might then be replicated across the United States.
The Pitfalls and Promotion of Sharps Waste Programs
Bureau of Environmental Health, Florida Department of Health, Tallahassee, Florida, USA
Bullet Points
Sharps disposal instructions are often minimal, vague, and convoluted.
The Bureau of Environmental Health (BEH) regulates sharps businesses with minimal regulation on user sharps disposal.
The BEH voluntary sharps disposal programs are present throughout Florida, mainly in urban areas.
Responsibilities of the Bureau of Environmental Health
The BEH has a biomedical research program in Florida primarily to regulate generators, transporters, storage, and treatment facilities, and the businesses that generate biomedical waste, from generation to disposal. However, home users are only lightly regulated by the Department of Health. Current rules require the health care professional to educate the home user on how to properly dispose of home-generated sharps waste. Rules also require the home user to package the waste to prevent public exposure to used sharps. As of 2021, the BEH has developed little regulation on home user sharps disposal.
Method to Collect Sharps Waste From Home Users
In Florida, the BEH operates a nonfunded, voluntary community service sharps collection program currently operating in 37 of the 67 counties in Florida, mainly in urban communities. The collection site type varies among counties; collection sites include hospitals, local pharmacies, health department sites, fire stations, and household hazardous waste sites. The voluntary sharps disposal collection program reveals several challenges, including (1) the difficulty in acquiring a new site to agree to being a collection site, (2) liability concerns, and (3) fear of staff exposure to collected sharps. However, with experience in the Tampa Bay Area, when one collection site joins, such as a hospital, then a chain reaction may occur among other hospitals which then agree to be a collection site out of competition and desire to promote community service.
Challenges With Rural Counties
Most of the counties actively involved in the BEH’s sharps collection program are within urban communities. The majority of the 30 counties lacking an official sharps disposal program are smaller rural communities. The BEH continues to face several challenges regarding sharps disposal in rural communities in Florida:
Sharps in rural areas are often disposed of in landfills, due in part to the county’s lack of local ordinance prohibiting the disposal of sharps in landfills.
There is often a great distance from rural areas to sharp disposal sites located in urban areas. The rural home sharps user may not have access to transportation and/or may not want to travel the distance to a sharps disposal site in urban areas.
If some programs require the home user to purchase a sharps container, then they may be sold at a nominal fee. However, if the home user possesses limited income, then they might opt out of the program because of a lack of financial means to pay for the sharps container.
Potential lack of disposal information from the health care professional (eg, the professional tells the patient to check their blood sugar every day, but does not explain the importance of the disposal process for the single-use finger stick device). Thus, patients may be disposing of loose used sharps in trash bins out of ignorance or lack of awareness regarding nearby sharps disposal collection sites.
Some pharmaceutical companies provide sharps containers to patients, although the packaging lacks further instructions regarding how to dispose of the sharps containers themselves, so sharps containers may be disposed of in recycling bins, sharps container lids may open, and sharps are spilled, thereby posing a public and community safety hazard.
Spreading the Word and Increasing Participation in Sharps Disposal Programs
To promote participation in sharps disposal programs, three actions may be taken:
Education of residents through simple brochures and fliers available in counties for every pharmaceutical prescription injectable picked up.
Encouragement of pharmaceutical manufacturers to provide more instruction on sharps disposal with more detail than the minimal “follow local, state and federal government disposal requirements.”
Promotion of individual sharps disposal programs on social media, targeting audiences composed of home sharps users.
Conclusions
It is the health care professional’s responsibility to educate patients about safe sharps disposal methods.
Access to sharps disposal programs in rural counties reveal challenges because of insufficient finances, transportation, and understanding of local ordinances, regarding sharps disposal.
An emphasis on education and promotion regarding local sharps disposal programs must be circulated to increase participation in such programs.
Occupational Perspective on Safe Disposal of Sharps Waste
CDC National Institute for Occupational Safety and Health, Morgantown, West Virginia, USA
Bullet Points
The National Institute for Occupational Safety and Health (NIOSH) makes research-based recommendations to protect workers, including for safe disposal of sharps in hospital and home health care settings.
There are current regulations from the Occupational Safety and Health Administration (OSHA) that are in place to protect workers from at-work sharps injuries.
NIOSH has developed educational resources for sharps safety, including safe sharp disposal procedures.
NIOSH’s Role in Improving Sharps Disposal Safety
NIOSH resides within the Department of Health and Human Services in the Centers for Disease Control and Prevention (CDC) and conducts research, disseminates research findings, and moves research into practice by making recommendations based on research conclusions. Sharps injuries are a major issue. In hospitals alone, there are about 385,000 sharps injuries per year. Sharps injuries also occur in other settings, including home health care settings, where one of the largest risk factors for sharps injuries is the inappropriate disposal of sharps, that is, sharps laying around in unexpected places such as bedding or patient clothing, posing a significant safety concern when a health care professional changes a bed or a patient and risks getting stuck. Thus, addressing improper sharps disposal is very important.
NIOSH’s Free and Publicly Accessible Educational Resources
NIOSH actively publishes Web pages such as the Stop Sticks Campaign 19 and the Bloodborne Pathogens Web site, 20 which are composed of information regarding sharps safety, including safe disposal. Additionally, in response to significant concerns during the COVID-19 pandemic regarding vaccination sites running out of sharps disposal containers, NIOSH posted detailed information about the use of appropriate alternative sharps containers, 21 their characteristics, and proper labeling criteria on their publicly accessible Web site.
OSHA’s Sharps Regulations
OSHA’s Bloodborne Pathogens Standard (BPS), which is a comprehensive set of federal regulations addressing sharps injuries, requires employers to have detailed plans regarding sharps disposal, and encourages the use of engineered devices designed to reduce injury risk. Such engineered devices may include a user-activated safety component so the sharp can no longer stick, lacerate, or puncture skin post-use. This type of safety feature improves safety not only for those in the direct care environment but also for those downstream in the MRF waste sorting sites. Another component of OSHA’s BPS is the implementation and use of the regulation itself, which requires public education and training, and mandates for employee surveillance and injury reports to ensure that employees receive appropriate post-exposure prophylaxis and treatment at the employer’s expense in case a worker encounters a sharps-related injury.
Conclusions
OSHA’s BPS regulations require employers to have detailed sharps disposal plans in place, encourages engineered safety mechanisms on devices, and aims to protect employers in the case of a sharps injury at work.
Devices can be re-engineered to contain embedded user-activated safety features after sharps device utilization.
The Position of Industry
Moderator
Science Consulting in Diabetes GmbH, Kaarst, Germany
Panelists
HealthBeacon, Dublin, Ireland
Dexcom, San Diego, California, USA
Abbott Diabetes Care, Alameda, California, USA
Novo Nordisk Pharma GmbH, Mainz, Germany
Roche Diabetes Care, Indianapolis, Indiana, USA
Bullet Points
Environmental sustainability is a growing area of interest in the medical device industry.
Many companies are working to improve sustainability and waste management of their products.
Plastics comprise a large portion of waste generated by diabetes devices.
It is necessary to reduce the amount of raw material used and to recycle and reuse plastic components of these devices to reduce environmental impact.
There is considerable value in changing regulations around instructions for use (IFUs) to better suit 21st-century technology in light of the current global environmental challenges.
Current regulation requires paper manuals for medical devices. Companies are already shifting to online resources, so it could be beneficial for the environment for regulations to be updated to reflect current trends.
There continues to be a need for education efforts to spread awareness about environmental sustainability.
Industry Initiatives for Environmental Sustainability
In addition to environmentally conscious individuals, it is important for industry-wide participation to increase the sustainability of medical device production. The divisions of diabetes device companies represented in this panel include Abbott Diabetes Care, Dexcom, HealthBeacon, Novo Nordisk Pharma GmbH, and Roche Diabetes Care. All of these companies understand the concerns for environmental sustainability and have different initiatives currently in place. Abbott has a ten-year plan looking at sustainability through the overall environmental impact that they make and at the accessibility of their devices to global communities. The Dexcom Design Team is committed to interdisciplinary collaboration to tackle sustainability starting from medical device design. HealthBeacon follows the EPR Model, has developed a reusable sharps bin for use in the home market, and recently founded a nonprofit with seven other supply chain members to address sharps waste. Novo Nordisk has a program in place called “Circular for Zero,” involving collaboration with suppliers, production companies, and distribution companies among others to aim for zero environmental impact. Roche Diabetes Care focuses on developing products that meet current needs without sacrificing future generations’ ability to meet their needs and has a “Key Performance Indicator” called Eco-Balance to examine their ecological footprint.
One of the major obstacles to diabetes device sustainability is the amount of plastic material used to manufacture them. For some devices, such as blood glucose monitors, the trend for digital devices like wearable sensors decreases the need for glucose test strips and lancets, leading to less waste generated. However, for devices like CGM systems or insulin infusion sets with no alternative, it is important to focus on the raw materials used to produce them. Using minimal necessary packaging, reducing the number of separate components of these devices, and trying to maximize their life cycle are several ways to reduce the amount of plastics used. The remaining material may also be recycled or reused as part of waste management to avoid landfills as much as possible. Device companies should provide instruction to recycling centers to ensure the components of each device are properly separated and handled as they can often include biohazards. The types of products that these materials are recycled into also need to be considered. For example, recycling plastics from a medical device to make plastic bags still ultimately has a negative environmental impact. Consumers should also be educated on proper disposal of their devices after use. Other areas to consider include using environmentally clean transportation when shipping medical devices and lessening the overall plastic usage of the company through eliminating single-use plastics in the office.
In line with the shift toward digitalized medical devices, there are more and more digital products emerging for managing diabetes. However, the regulation behind the usage of these nonphysical products has not been updated as quickly to reflect this trend. Many companies now have apps that are connected to patients’ smartphones and sometimes capable of transferring information to the patients’ care teams as well. These apps will often contain information such as instructions and videos on how to use medical devices and how to dispose of their components, which is redundant with the paper instruction manuals that companies are currently required to include with their medical devices. Using a digital platform to replace the paper format would not only reduce waste generation but would also allow companies to update any user information more frequently and more quickly. Regulatory changes to allow industry to substitute paper IFUs with digital ones would decrease the environmental impact associated with diabetes devices.
All of the aforementioned issues and potential solutions are complex and require the efforts of various companies in different sectors. Thus, coalitions that can bring together different industry players can help create concrete plans and goals. One such coalition founded in Switzerland is the “Alliance to Zero” with eight companies that represent the full supply chain for an injection device that aims to comply with net-zero emissions. Another collaborative effort is the Pharmaceutical Product Stewardship Work Group (PPSWG) that coordinates industry responses to product take-back laws and provides guidance for stewardship programs. Many manufacturers are open to working together to drive a sustainability initiative for specific diabetes devices that can eventually be scaled up to relate to medical devices as a whole.
Despite the number of industry initiatives surrounding environmental sustainability, there is still a lot to be done to increase awareness of environmental issues and educate the public. In addition, the support of these efforts is often conditional on associated economic benefits. Conference presentations, meeting panel discussions, and scientific publications are also crucial for health care professionals who in turn can educate patients. Regions around the world also have differing interest in sustainability. It may be of interest to tailor efforts to spread awareness in areas that are not actively thinking about sustainability at this time.
In the future, environmental impact will be a standard consideration for diabetes device design, sourcing, manufacturing, use, transport, and end-of-life. Eventually, environmental impact will also become a standard consideration for all medical devices. Collaborative efforts between supply chain companies, manufacturers, and distributers will be needed to improve sustainability.
The Role of Coalitions
Moderator
Mills-Peninsula Medical Center, San Mateo, California, USA
Panelists
DASTRI French Health Industry Coalition for Sharps, Paris, France
Stericycle, Bannockburn, Illinois, USA
Science Consulting in Diabetes GmbH, Kaarst, Germany
WNWN International, Burlington, Connecticut, USA
ConvaTec Infusion Care, Lejre, Denmark
Bullet Points
Opportunities for partnerships with interested stakeholders may provide options that contribute to a circular economy.
Coalitions for sustainability and waste management should be built to fund research and education, align objectives, and allow stakeholders to work together and determine solutions. Success as a coalition will only be reached with the engagement of all stakeholders.
The technology to inform the public about medical waste and sharps disposal locations needs to be improved. Only a fraction of device waste streams is currently captured.
Sharps that are improperly disposed of pose a risk of infectious disease transmission and/or physical injury to those working in solid waste facilities and wastewater treatment plants.
Coalitions can leverage existing technologies and social media platforms to reach online communities of PwD, as well as communities without diabetes. Leveraging technology allows the coalition to conduct surveys to better understand consumer needs, distribute information, and communicate directly with consumers once initiatives are established.
Initial projects should be designed/selected to prioritize the environmental impact and the likelihood of success. It will then be easier to follow up with subsequent projects.
The Feasibility and Role of Coalitions in Promoting Sustainability and Waste Management
While industry-wide efforts to increase sustainability in manufacturing processes of diabetes devices will be crucial in reducing the amount of waste generated by medical devices used in home care, the issues of environmental preservation, climate change, and potential biological collapse are ultimately too substantial for any single stakeholder to address alone.
Coalitions organized for the purpose of improved waste management have been successful before. In France, “Déchets d’Activités de Soins à Risque Infectieux” (DASTRI) perforants, a coalition of 63 major companies, including Dexcom, Abbott, Roche, and Novo Nordisk, gained approval from the French government to manage the disposal of medical devices, especially sharps, used in home care. In France, there are about 2 million home care patients using a total of 1.3 billion products each year. DASTRI provides them with sharps containers, distributing approximately 3 million containers every year via a network of 22,000 pharmacies. Once sharps containers are full, patients can return them to their local pharmacy and DASTRI will collect and properly dispose of the used sharps. DASTRI has been very successful, collecting around 83% of the 1.3 billion sharps that are used every year by home care patients. Admittedly, DASTRI’s success is based on unique circumstances; EPR is required by state regulation. The French state was under pressure from local governments in response to sharps being improperly disposed with household waste and causing injuries to those working at waste sorting facilities. In addition, regulations require device manufacturers to pay for waste management programs like DASTRI. Outside of France, where there are currently 20 similar collection programs for sharps, few such programs can be found.
Although countries like the United States do not have as many regulations as France does, coalitions can still be successful in increasing public awareness and changing perspectives. Coalitions have been successful in the United States before. In 2002, the Coalition for Safe Community Needle Disposal created SafeNeedleDisposal.org as part of their campaign for safe needle disposal. Today, safe needle disposal is considered a public health and safety concern in the United States. 22 Similar to how improper sharps disposal was not considered a major safety concern until needles started washing up on beaches, not many people today understand the magnitude of sustainability and waste management for medical devices. No one group probably sees the entire picture of the product life cycle. Issues like environmental protection are simply too big to solve by any one company or organization on its own. A coalition would be important in bringing awareness to environmental issues related to improper disposal of device waste as well as the types of injuries and accidents that can occur and showing the value of safe needle devices to the public. As pressure from consumers, health care professionals, hospitals/clinics, and local waste disposal facilities continues to grow, take-back programs and other forms of EPR for medical devices are becoming the trend. Companies may be willing to volunteer to bring the initiative forward. It would be a little step in the right direction to make both people and industry more aware of sustainability and waste management.
Any coalition aiming to positively impact diabetes device sustainability and waste management must first understand the background and current landscape around device disposal. This involves identifying the current issues around the sustainability of diabetes devices from the perspective of each stakeholder group. How many consumers are improperly disposing of device waste and why? What guidance currently exists for home use of diabetes devices? What tools or information do consumers need? What are the regulations around waste disposal? What equipment is being used and who is manufacturing it? A coalition needs to update the current understanding, collect data, distribute it, and see where there are gaps in understanding. With the help of device companies, we would better understand the magnitude of the issue. Based on sales figures per year, how much waste should be expected? How many products does the typical person with diabetes use during a set time period? Coalitions should not only be concerned about the type of product, but how much is being used, how frequently it is used, and how much/what type of waste is generated by its use. Answers to these questions would be important in determining what are the most important issues that need to be prioritized.
While working to understand the scope of the issue, a coalition may be able to engage with companies to perform full life cycle assessments to uncover their largest environmental impacts during manufacturing processes. If sustainability concerns are revealed, then manufacturers may alter their production processes to minimize the amount of resources used and reuse water where possible.
After understanding the complexity of the issue, a coalition can then move forward with solutions. Coalitions will determine which groups of stakeholders need to be represented. A list of stakeholders that an international coalition should potentially include is presented in Table 3. Although the answer varies depending on which aspect of the problem is prioritized, the broad range of perspectives and knowledge that can be gathered is crucial to the success of the coalition. Manufacturers and regulators may know how devices should be properly disposed of, but the consumers and local waste managers directly tasked with disposal may not. In addition, manufacturers and regulators can adapt their recommendations based on input from consumers and waste managers. Building a coalition creates an environment wherein stakeholders can most efficiently cooperate and gain the most benefit from each other’s economies of scale and knowledge about what works and what does not work. Areas where a coalition may be more impactful than individual companies, government agencies, nongovernmental organizations, medical groups, or patient groups are summarized in Table 4.
Coalitions for Sustainability and Waste Management of Diabetes Devices for Home Use Should Include, But Are Not Limited to, the Following Groups of Stakeholders.

Areas Where a Coalition May Be More Impactful Than Individual Companies, Government Agencies, NGOs, Medical Groups, or Patient Groups.

Abbreviations: NGO, nongovernmental organization; IFU, instructions for use.
Understanding the breadth of the issue also allows coalitions to share their findings with the public. Like with safeneedledisposal.org, coalitions can create a Web site to provide consumers with information about local recommendations for device disposal. Much important information about sharps disposal is currently available, but consumers may be unaware. The challenge is navigating through the current maze of noise and misinformation to raise the baseline awareness of consumers. If consumers cannot see the problem, then they will not know it needs a solution. However, while education and awareness about sustainability and waste disposal are necessary and a great starting point, coalitions have to do more to change people’s behavior and to change the system. The next step would be collaborating among stakeholders to determine which “low-hanging fruit” can be easily addressed and resolved.
A potential initiative for a coalition would be to engage with regulators and manufacturers to discuss requirements for providing printed IFUs with medical devices. In principle, printed IFUs are important resources for consumers to understand how to safely use their devices. Although IFUs should be easily accessible to consumers, most consumers purchasing medical devices may read the IFUs once, if at all, and then dispose of them with each subsequent purchase, leading to large amounts of unnecessary paper waste. In addition, the incorporation of IFUs into device packaging takes up a lot of unnecessary space, adding extra volume to shipments and consequently additional transport costs per device. Working with regulators and manufacturers, a coalition may be able to reform requirements for IFUs that are currently being provided only in printed form. Most IFUs are available in several different languages on device Web pages. Directing people who need additional information to online resources could substitute for printed IFUs included in packaging. Consumers who need to consult IFUs could easily access them by scanning QR codes included in packaging in lieu of the documents that are currently available. Manufacturers may then be able to allocate the resources saved by cutting production, packaging, and transportation costs associated with paper IFUs into voluntary initiatives for sustainability and waste management.
Coalitions often propose solutions in terms of EPR and product stewardship, but the reality of sustainability is much more complex. Producer action on its own will not do enough to solve the global environmental challenges. EPR and product stewardship are not solutions if consumers do not play their role in properly sorting waste. Consumers are the ones first and foremost dealing with waste disposal in their homes. Consumer action needs to be supplemented with the proper education and tools for patients to manage their device wastes. The objective is to have a coalition of stakeholders who want to cooperate and who are focused on sustainability and waste management.
Conclusions
The GDS assembled a variety of participants who are interested in the environment. Most of them expressed an interest in collaborating on projects related to sustainability of (disposable) diabetes devices intended for home use. The summit addressed (1) waste management of disposable diabetes devices and paraphernalia for home use at the end of a product’s lifecycle, and (2) design, manufacturing, packaging, and distribution processes of medical products throughout a product’s lifecycle. A great deal of effort will be needed to maximize sustainability in the design, production, and transport of diabetes devices, as well as in their disposal. Many diabetes devices contain sharps that must be disposed of in a way to avoid injures to waste handlers. Many projects by nongovernment organizations, by the government sector (especially in France and in California), and by the industry sector are tackling waste management. Many waste management experts believe that a coalition from multiple sectors of society can accomplish more than any single organization. In the face of an international groundswell of interest in preserving the environment these days, the GDS was a milestone event to discuss for the first time in an international forum how diabetes affects the environment and now is the time to stop environmentally damaging processes caused by the production and disposal of diabetes devices.
Footnotes
Acknowledgements
We thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
BEH, Bureau of Environmental Health; BPS, Bloodborne Pathogens Standard; CDC, Centers for Disease Control and Prevention; CEO, chief executive officer; CGM, continuous glucose monitoring; CPSC, California Product Stewardship Council; DASTRI, Déchets d’Activités de Soins à Risque Infectieux perforants; DTS, Diabetes Technology Society; EPR, extended producer responsibility; ESAP, Expanded Syringe Access Program; GDS, Green Diabetes Summit; IFUs, instructions for use; MRF, Materials Recovery Facility; NIOSH, National Institute for Occupational Safety and Health; OSHA, Occupational Safety and Health Administration; PwD, people with diabetes; SB, Senate Bill.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KTN, NYX, JYZ, TSh, and AYD have nothing relevant to disclose. LH is a consultant for a number of companies that are developing novel diagnostic and therapeutic options for diabetes treatment. He is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Kaarst, Germany. EPK is the President of WNWN International Inc, a medical waste management company in Burlington, Connecticut. He is currently a consultant for Stericycle, Daniels, HealthBeacon, and Tesalys. TSt is an Innovation Project Manager for ConvaTec Infusion Care. DAS is a sustainability consultant for multiple companies. DCK is a consultant for EOFLOW, Fractyl, Integrity Applications, Lifecare, Novo, Roche Diagnostics, and Thirdwayv.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

