Abstract

Introduction
The past decade has seen an exponential growth in concerns related to threats to the environment and the sustainability of natural resources for future generations. From the perspective of the environment and health, the focus has been on the impact of the environment on diabetes risk and management.1-3 In parallel with the recognition of a direct link to environmental changes and human health, there has also been a great increase in the use of technology to support diabetes self-management 4 over the past ten years. Although these technologies have delivered measurable beneficial clinical results, there is an increasing recognition that the use of such technologies may also have a negative environmental impact. During the manufacturing process of devices for monitoring, diagnosing, and treating diabetes, as well as ones that use smartphone-based controllers, the use of “disposable” components during the manufacturing processes has become ubiquitous to create single-use devices. The reasons for this are easily understood in terms of ease of use and avoidance of biological hazards. The challenge for manufacturers of technologies to support diabetes care is to introduce sustainable change to reduce environmental costs and waste and at the same time, to increase the use of recyclable materials, all without adversely affecting either device performance, user experience, or personal financial cost as well as the personal time burden associated with the use of the technology for people living with diabetes.
Diabetes Technology Society (DTS) is committed to conserving natural resources and waste management processes to promote environmental sustainability. The used disposable diabetes devices intended for one-time use in the home create a large amount of waste. These devices include injection needles, syringes, lancets, blood glucose monitoring strips, blood glucose monitors, continuous glucose sensors, insulin bottles, pens, infusion tubing, and disposable pumps, as well as device batteries and packaging, which all create large amounts of waste. Diabetes Technology Society hereby announces that we are prioritizing a mission to advocate for the development and deployment of “green” diabetes technology. Our goal is to reduce the mass of medical waste associated with diabetes care.
The Five “R” Strategy for Waste Management
At DTS, we believe that it is now time to develop policies for minimization, collection, separation, treatment, and disposal of diabetes device waste, as well as to address waste management policy and education. Our belief is that prudent waste management strategies for diabetes devices should emphasize the waste hierarchy five “R’s.” We call for (1) reducing, (2) reusing, (3) recycling, (4) redesigning, and (5) re-educating in the area of diabetes device waste. These five “R” strategies are intended to support extraction of the maximum practical benefits from disposable diabetes devices while generating the minimum amount of waste.
Manufacturers are increasingly interested in lifecycle analysis of devices to avoid unnecessary generation of waste and to optimize the use of often limited resources. Many clinicians and patients who prescribe and use these devices are interested in knowing how to safely dispose of diabetes devices. They need more information and better tools to avoid disposing of these products in an unsafe way. 5 Safe disposal means avoiding putting waste handlers at risk of being stuck by a needle or exposed to blood or other body fluids. 6
Regulation
Regulatory agencies at the local and federal level have been tightening up regulations for medical waste disposal in many ways; however, household wastes that include much of the diabetes device waste management problems have been traditionally excluded from most government regulations. The US Department of Transportation has defined medical waste, sharps, and a used healthcare product 7 (see Table 1).
Definitions of Medical Waste, Sharps, and a Used Healthcare Product According to the U.S. Department of Transportation. 7

Per the US Environmental Protection Agency (EPA) regulations, “household wastes” are excluded from the definition of “hazardous waste” subject to regulation under the Resource Conservation and Recovery Act. 7 The Medical Waste Tracking Act was in effect from 1989 to 1991. This US federal law assigned to the EPA the regulation of medical waste management including disposal of body tissues, blood wastes, and other contaminated biological materials. Since 1991, medical waste has been primarily regulated by state environmental and health departments, whose rules for disposal vary from state to state. 8 The EPA recently published its highly anticipated final rule on the Management Standards for Hazardous Waste Pharmaceuticals. Medical devices were not addressed in this standard. 9 Workers who handle medical waste are protected by standards and research from Occupational Safety and Health Administration (OSHA) and National Institute of Occupational Safety and Health (NIOSH). OSHA is an agency of the U.S. Department of Labor that sets and enforces regulations for worker safety and health. 10 NIOSH is a research agency of U.S. Centers for Disease Control and Prevention that studies how to create safe and healthy workplaces. 11
Household waste means any solid waste (including garbage, trash, and sanitary waste from septic tanks) derived from households. 12 This term generally refers to wastes that are (1) generated by individuals on the premise of a temporary or permanent residence and (2) composed primarily of materials found in wastes generated by consumers in their homes. Much waste from disposable diabetes devices intended for one-time use is either household waste or hospital waste. In these two settings, we see a need for two paradigm shifts. First, better educational tools are needed to educate patients and healthcare workers to practice safe diabetes device waste management of household wastes.13,14 This need has been increasing in recent decades as disposable diabetes devices are more frequently used in hospitals and significantly more frequently used in the home. Second, standardized sorting of medical waste streams is needed to increase the efficiency of efficient waste management at healthcare facilities. 15 The color of receptacles used for various waste types varies from country to country, with some using the source of the waste as a basis for sorting and others using an estimation of the risk of infection of an object to determine its disposal waste stream. 16 Other specialties in hospital healthcare have also been addressing their environmental footprint, such as the operating room, radiology, and interventional cardiology, by collecting and repurposing devices. The range of diabetes care products needs a similar assessment.
Sustainability and the Circular Economy
Sustainable development means policies for growth that meet the needs of the present without depleting natural resources or compromising the ability of future generations to meet their own needs. 17 The circular economy is a subset of sustainability and applies to processes of human conversion of raw materials that is designed to minimize (1) the use of resources and (2) the creation of waste, pollution, and carbon emissions. The circular economy is a closed loop system for resource use that keeps materials and products in use. In a circular economy, raw materials that become products are eventually recycled and reused, unlike in a traditional linear economy in which raw materials become products and are then eventually converted into waste18,19 (see Figure 1). DTS advocates the principles of the circular economy, which address the preservation of limited resources through continuous recirculation, 20 and calls for maintaining the value of products and the materials from which they are made by lengthening their useful lifetime or by reusing them. 21 By maximum application of these principles, the concept of waste would be eliminated because products and materials would be endlessly reused and recycled. 22
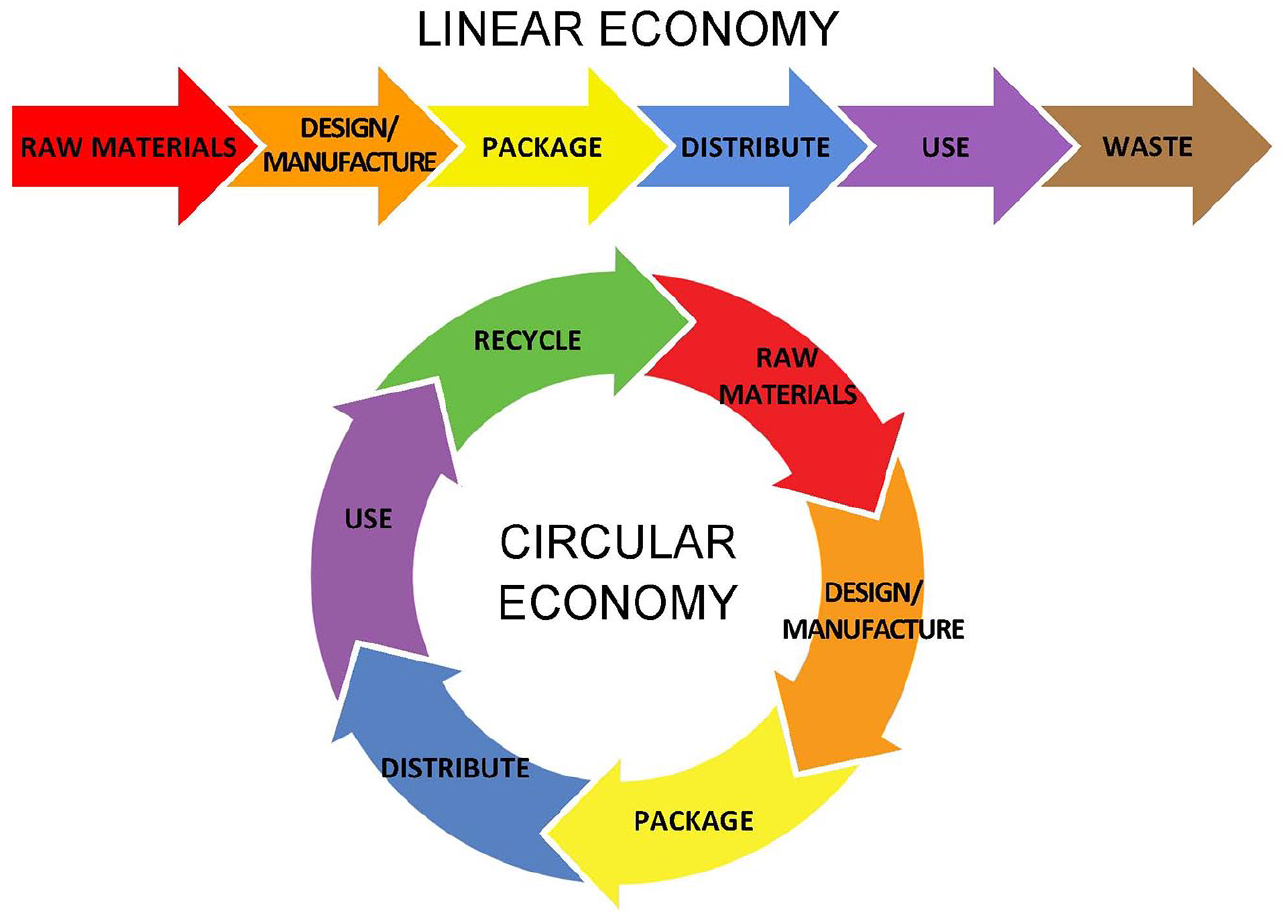
Features of a linear economy compared to features of a circular economy.
Current Disposal of Diabetes Waste
Disposal of loose sharps into the municipal solid waste system is unacceptable. 23 A recent survey study of 150 respondents on insulin therapy revealed that approximately one-third of respondents disposed of their lancets and insulin needles in an unsafe manner noncompliant with the current recommendations. 24 The findings from this study are similar to the limited data from the United States and other countries that indicate many patients with diabetes mellitus (DM) unsafely dispose of their sharps.25-31
Sharps disposed of improperly pose a risk to waste industry workers. A recent report estimated that up to 1484 needle stick injuries occur annually at material recovery facilities in the United States and Canada, injuries that result in $2.25 million dollars in cost for treatment, prophylaxis, and monitoring. The number of used needles disposed of in household garbage nearly tripled from 2001 through 2011, a number likely to increase with the DM epidemic. 23 Although diabetes is not the only disease that generates disposable one-time use devices in the home that become medical waste, it is the disease that contributes the greatest burden of such waste because of the large number of patients with diabetes who need to use these products and the high frequency of use of these products, which can be generated at a rate of over ten disposable devices per day by some patients. Recommendations for safe disposal of sharps and diabetes-related medical waste exist. The US Food and Drug Administration (FDA) recommends placing used needles and other sharps into a sharps-disposal container to reduce the risk of needlestick injuries. If an FDA-cleared container is not available, then a heavy-duty plastic household container may be an alternative, although, depending on the thickness of the container, it may not be entirely safe.32,33 Additional guidelines are available from the EPA34,35 and may vary by state. 36 Despite the fact that clear recommendations for proper sharps disposal exist and are mandated by FDA to be included in package labels of sharps, the aforementioned survey study of 150 respondents using insulin therapy indicated that the majority (64%) stated they had never received instruction about safe sharps disposal as part of their diabetes education. In addition, the majority (87%) indicated they had never visited their municipal website for medical waste disposal. 24 As the incidence of diabetes increases, it becomes even more important to provide patients with education on safe sharps disposal.
A History of Leadership
Diabetes Technology Society has a history of leadership in safe waste management and sustainability. In 2011 our organization’s official journal, which is Journal of Diabetes Science and Technology, published the first special section in any medical journal ever devoted to diabetes and the environment, which was edited by one of us (DK). At the 2019 DTS’s annual meeting in Bethesda, Maryland, we presented a well-received session on Disposal of Waste from Diabetes Products. Two of us (LH and EK) were among the speakers at this session.
Launch of the Green Diabetes Initiative
In response to both a growing awareness that we must conserve the environment as well as a need to better manage the sustainability of disposable diabetes devices for home use, DTS is launching the Green Diabetes Initiative in 2020. Our new green colored logo which we will use in 2020 indicates the seriousness of our interest in this topic (see Figure 2). Diabetes Technology Society will present the first ever Diabetes Environment Summit: (Sustainability, the Circular Economy, and Waste Management) in the second half of 2020. The meeting will cover topics which are part of product stewardship. This concept is defined as an ethic that embodies the responsible planning and management of resources by manufacturers, retailers, users, and disposers to make products safer for people and reduce negative impacts to the economy, environment, public health, and worker safety.37,38 The focus of this first-ever meeting is intended to bring together key opinion leaders in diabetes device stewardship to find ways to massively reduce waste production in order to minimize environmental effects and health risks. This event will cover advances in (1) sustainability and (2) the circular economy. The summit will address (1) waste management of disposable diabetes devices and paraphernalia for home use at the end of a product’s lifecycle; and (2) design, manufacturing, packaging, and distribution processes of medical products throughout a product’s lifecycle. Participants will represent the industry, government, clinical, and patient communities. We will discuss new technologies and policies to reduce waste through redesign, reuse, and recycling of products as well as education of device prescribers and users. We will also identify necessary resources to achieve a safe and sustainable waste management system for disposable diabetes devices according to stakeholder sectors (see Table 2). 39

The new green logo for Diabetes Technology Society for 2020.
Necessary Resources to Achieve Safe and Sustainable Waste Management Systems for Disposable Diabetes Devices According to Stakeholder Sectors. 39

The Diabetes Environment Summit: (Sustainability, the Circular Economy, and Waste Management) will have three purposes. First, the meeting will deliver information about processes that can enhance the sustainability of disposable diabetes devices used in the home. Second, the meeting will provide a forum for networking among sustainability professionals from various backgrounds that can lead to future collaborations. Third, the meeting will allow DTS to determine whether there is interest among manufacturers, regulators, prescribers, and users of diabetes devices to work together to create guidelines for regulators, conduct research for cleaner product development, and develop waste management tools for patients.
Two examples of the types of topics we will cover at this Summit will be (1) “Patterns of sharps handling and disposal among insulin-using patients with diabetes mellitus,” which is the title of an eye-opening article by Bithika Thompson and Curtiss Cook et al that we published in Journal of Diabetes Science and Technology (JDST) six weeks ago 24 ; and (2) “Diabetes technology and waste: a complex problem piling up,” which is the title of a thoughtful article by Lutz Heinemann and Ed Krisiunas that was published in JDST last year. 40 We will also discuss how to move waste management processes from less preferable to more preferable according to a seven-level hierarchy, as described by the United Nations Global Healthcare Waste Project 41 (see Figure 3). 42 Additional parts of our Green Diabetes Initiative will include research on best practices for diabetes device disposal, a special section in JDST on topics related to this initiative (to be edited in 2021 by Lutz Heinemann, Edward Krisiunas, Curtiss Cook, and Tobias Stumpe), and the publication of a summary of the summit in JDST. We will announce specifics of the date and location of this meeting soon.

The waste management hierarchy. 42
Conclusion
Sound medical device stewardship improves environmental quality, public health, and worker safety. The triple bottom line, which is used for assessing the sustainability of a business or organization, is an accounting framework that incorporates financial performance as well as social and environmental benefits. 43 The goals of the DTS’s Green Diabetes Initiative will enhance all three elements of this triple bottom line paradigm for manufacturers of disposable diabetes devices intended for home use. This stewardship initiative will encourage the diabetes technology industry to practice sound material and waste management, protect the environment, aim for sustainability, and meet the triple bottom line of planet, people, and profit.
Footnotes
Acknowledgements
The authors thank Annamarie Sucher for her expert editorial assistance.
Abbreviations
DM, diabetes mellitus; DTS, Diabetes Technology Society; EPA, Environmental Protection Agency; FDA, US Food and Drug Administration; JDST, Journal of Diabetes Science and Technology; MSW, municipal solid waste, NIOSH, Occupational Safety and Health Administration; OSHA, National Institute of Occupational Safety and Health.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant for Abbott, Ascensia, EOFlow, Fractyl, Know, Lifecare, Novo Nordisk, Roche, and Thirdwayv. LH is a consultant for Becton Dickinson, Lifecare, Roche Diabetes Care, and Zucara. He is a shareholder of Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany and Prosciento, San Diego, United States. CBC has nothing to disclose. BMT has nothing to disclose. DK is a medical advisor for Glooko and has received consultancy fees from NovoNordisk, Abbott Diabetes Care, Sanofi and research support from Lilly. JH has nothing to disclose. EPK is a consultant to CDC, Future Healthcare Systems, Stericycle, Sterigerms, Technopath, Tesalys/Spire, and USDHS.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
