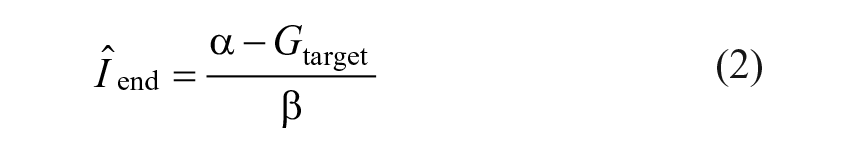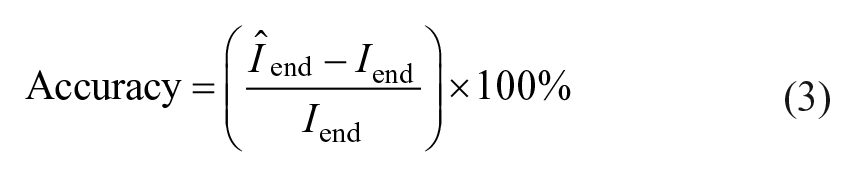Abstract
Background:
Treatment inertia and prescription complexity are among reasons that people with type 2 diabetes (T2D) do not reach glycemic targets. This study investigated feasibility of a new approach to basal insulin initiation, where the dose needed to reach a glycemic target is estimated from two weeks of insulin and continuous glucose monitoring (CGM) data.
Methods:
This was an exploratory single arm study with a maximum length of 84 days. Eight insulin naïve people with T2D, planning to initiate basal insulin, wore a CGM throughout the study period. A predetermined regime was followed for the first two weeks after which the end dose was estimated. The clinician decided whether to follow this advice and continued the titration until target was reached using a twice weekly stepwise titration algorithm. The primary outcome was the comparison between the estimated and the actual end doses.
Results:
Median age of participants was 57 years (range: 50-77 years), duration of diabetes was 16 years (range: 5-29 years), and Bodi Mass Index (BMI) was 30.2 kg/m2 (range: 22.0-36.0 kg/m2). The median study end dose was 37 U (range: 20-123 U). The estimated end dose was smaller than or equal to the study end dose in all cases, with median error of 26.7% (range: 0.0%-75.8% underestimation). No self-monitoring of blood glucose values were below 70 mg/dL and no severe hypoglycemia occurred.
Conclusion:
While accuracy may be improved, it was found safe to predict the study end dose of insulin degludec from two weeks of data.
Keywords
Background
Approximately one-third of people with type 2 diabetes (T2D) are prescribed insulin. 1 Insulin effectiveness in terms of lowering HbA1c in a real-world setting is low compared to clinical trial outcomes. The main reason is poor adherence to treatment mainly caused by perceived need for medication and fear of hypoglycemia.2,3 Other reasons include lack of patient confidence in managing insulin therapy and uncertainties due to vague prescribing information from professional societies. 4 The lack of adherence negatively impacts metabolic control 5 and results in over 60% of people not reaching recommended treatment targets. 6 Therefore, new dose guidance tools are needed to support clinicians and promote adherence and motivation of people with T2D. 7
Research groups have previously developed automated guidance systems to substitute paper-based algorithms to simplify and increase adherence to treatment, such as the Intelligent Dosing System and the Diabetes Insulin Guidance System.8,9 Both systems iteratively adjust doses based on prebreakfast self-monitoring of blood glucose (SMBG) to reach a fasting glucose target.
Our research group envisions that decision support tools leveraging brief use of continuous glucose monitoring (CGM), in particular in the primary care setting, will support future treatment decisions. In this context, this study evaluates an approach to rapidly identify the right insulin dose for people with T2D. The approach uses initial CGM and injection data to generate an individual dose response model. This model is then used to estimate the dose needed to reach a fasting glucose target. The expected implications of such an estimation early in the titration would be to improve perception of need for medication, prescription support, and reduced fear of hypoglycemia. The specific aim of the study was to evaluate the feasibility of this treatment approach.
Method
Design
The study was an exploratory single arm study with variable duration, with a maximum length of 84 days. People with T2D were recruited from the outpatient clinics at Hvidovre University Hospital and Steno Diabetes Center Copenhagen, Denmark. After screening, the clinician and the participants met three times in the clinic. Phone calls were scheduled every three or four days between visits (Figure 1). The procedure was as follows:
Visit 1 (day 1): The clinician introduced insulin degludec (IDeg) therapy, the CGM, and a smart phone for transferring data, and demonstrated logging of insulin doses and SMBG measurements in logbooks. Participants were asked to perform three daily SMBG measurements, one before breakfast and two evenly distributed throughout the rest of the day. The SMBG measurements were used for CGM calibration.
Insulin initiation (days 1-14): The participants did not inject insulin during the first four days. On day 5, insulin treatment was initiated with 10 U IDeg in the morning. This dose was kept unchanged for the next five days. On day 10, the clinician evaluated whether 10 U were sufficient for reaching the fasting glucose target of 72 to 108 mg/dL. If more insulin was needed, the daily basal insulin dose was increased to 0.2 U/kg body weight according to local standard practice guidelines.
Visit 2 (day 15): Continuous glucose monitoring data from the insulin initiation period were downloaded to the clinician’s computer. A dose estimation algorithm estimated the study end dose and suggested 75% of this dose to the clinician. If the suggested dose was assessed safe, the participant was prescribed this dose for the next four days. If the suggested dose was lower than the dose prescribed prior to day 15 and no events of hypoglycemia had occurred, the dose was maintained during the next four days and stepwise titration should continue from the dose already reached.
Titration to target (days 20-84): Titration continued twice weekly according to a stepwise titration algorithm (dose change increments of −4 to +8 U) with a fasting glucose target of 72 to 108 mg/dL, until the study end dose was achieved or until day 84. The study end dose was defined as the dose when the sum of three consecutive dose changes was within or equal to ±2 U. The clinician could deviate from the titration algorithm if assessed relevant, eg, the CGM data indicated a risk of hypoglycemia at any time of the day. The clinician determined and communicated dose changes during phone consultations.
Visit 3 (day 84 or end of study): When the study end dose was identified, the participant was called in for a last visit.
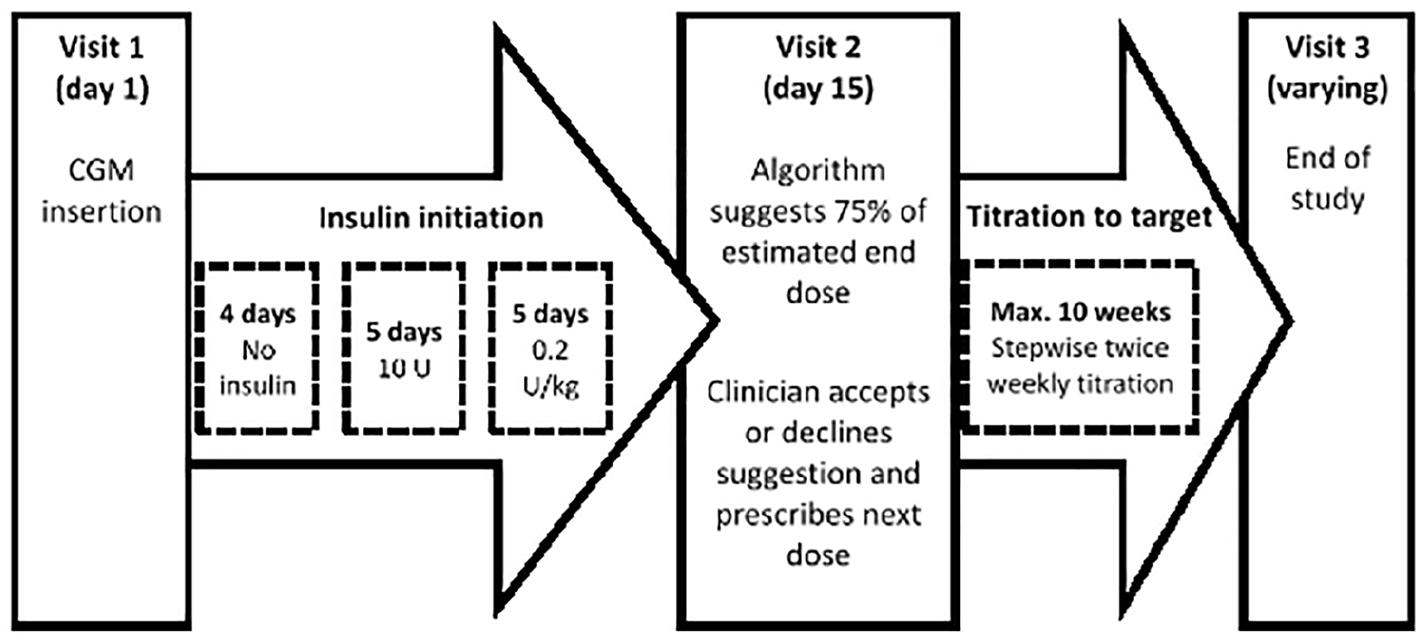
Overview of the study procedure. A predetermined regime was followed for the first two weeks. The dose prediction software suggested 75% of the estimated end dose. The clinician determined whether to follow this advice and continued the titration until target was reached. Titration continued twice weekly using a stepwise titration algorithm (−4 to +8 U steps) with target range of 72-108 mg/dL until the study end dose was achieved or until day 84.
Subjects
People with T2D who planned with their physician to initiate basal insulin treatment were included. The inclusion criteria were 18 to 80 years of age, insulin-naive people with HbA1c 53 to 100 mmol/mol (7.0%-11.3%), Body Mass Index (BMI) 20 to 40 kg/m2, willing to use CGM during the study and send/receive data and dose advice to/from clinician via phone. Exclusion criteria were pregnancy, breast-feeding or intention to become pregnant, active proliferative retinopathy or mild background retinopathy with HbA1c > 86 mmol/mol (10.0%), mean blood glucose (BG) > 270 mg/dL the week prior to screening, BG > 360 mg/dL and nonfasting ketones >0.5 mmol/L on the screening day, conditions which made tight diabetes control undesirable, and other concomitant conditions that made participation unsuitable. No use of sulfonylurea within two weeks prior to or during the study or corticosteroids within 30 days prior to or during the study was allowed. Change in other antidiabetic medicine than basal insulin and marked change in lifestyle within 30 days prior to or during the study were not allowed.
Devices
All participants were provided with an insulin pen (Tresiba FlexTouch), a smart phone (iPhone 5S), a CGM (Dexcom G5), and logbooks for SMBG and insulin data. The purpose of the CGM was (1) intensive data capture during the first period for dose estimation and (2) the glucose safety alarm (alarm level set at 70 mg/dL) during the full study period. The purpose of the smart phone was to (1) receive and transfer CGM data to Diasend and (2) telephone consultations every three or four days. Data from the CGM and logbook entries were transferred to the study computer during visits. The clinicians had a laptop with the dose estimation algorithm.
Algorithm
For IDeg, the total glucose-lowering effect increases linearly with increasing doses in the range 0.4 to 0.8 U/kg. 10 Therefore, a linear dose response model was used to estimate the study end dose based on two weeks of CGM and insulin data. The algorithm assumes that the fasting blood glucose response to long acting insulin is linear:
where
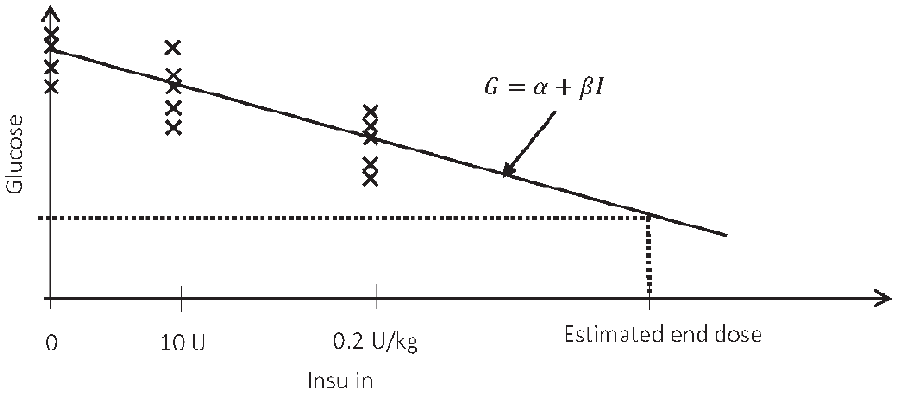
A concept illustration of the linear dose response model. The model assumed linear response of fasting glucose (x markers) to basal insulin. This model was used to estimate the end dose.
The only change in a glucose-lowering drug during the study is IDeg doses. The mean change in fasting glucose is therefore assumed to be caused or induced by the change in IDeg. To identify the response to an injection from the CGM data (288 data points per day), we define a titration glucose level as the lowest average one-hour window of CGM data of each day (Figure 3). The choice of a one-hour window was assessed reasonable to reflect a low glucose event while still robust to noise and outliers. The high-level steps of the algorithm are as follows:
Titration glucose level: The algorithm locates the lowest average one-hour window of CGM data of each day (Figure 3).
Linear dose response model: The algorithm estimates the parameters
Estimated end dose: The identified model is used to estimate the IDeg dose,
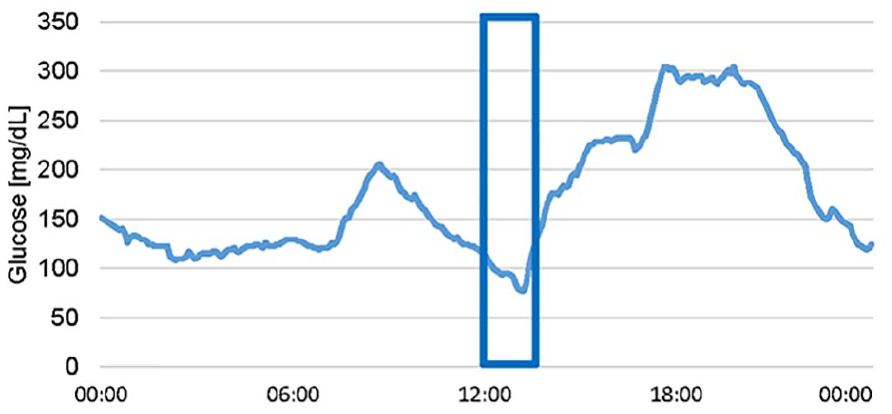
Lowest daily average one-hour continuous glucose monitoring interval. The algorithm located the lowest average one hour of continuous glucose monitoring data of each day. This glucose level was used to determine the dose response to the basal insulin.
The target,
Study Outcome
The primary outcome of this feasibility study is the percentwise deviation of the estimated end dose,
Secondary outcomes include accuracy of the estimated end dose based on SMBG data compared with the study end dose, where the dose estimate approach is the same as for the CGM-based dose estimation with titration glucose level substituted by prebreakfast SMBG values, number of participants reaching the fasting glucose target, number of titration algorithm deviations due to risk of hypoglycemia (based on the evaluation of CGM data), qualitative assessment by the clinician of the participants who do not reach the fasting glucose target within 84 days: (1) frequency of participants needing additional basal insulin and (2) frequency of participants needing additional drugs to achieve the fasting glucose target. Number of SMBG values ≤70 and <54 mg/dL, number of severe hypoglycemic events (defined as severe cognitive impairment requiring external assistance), time in hypoglycemia (<70 mg/dL), normoglycemia (70-180 mg/dL), and hyperglycemia (>180 mg/dL) were assessed by CGM during the first and last four days of the study.
Ethics and Statistical Considerations
The study is exploratory in nature and not powered for hypothesis tests, carried out in accordance with the Helsinki Declaration and good clinical practice (GCP), and monitored by the GCP unit at Copenhagen University Hospital after approval by the Regional Scientific Ethics Committee, the Danish Medicines Agency and the Danish Data Protection Agency. The study was registered at ClinicalTrials.gov, ID: NCT03365180.
Results
Demographic Data
Forty-seven participants were screened, whereof eight participants were included. Main reasons for exclusion included too high HbA1c or BMI and lack of willingness to participate. Six (75%) participants were males. Median (range) age of participants was 57 years (50-77), duration of diabetes was 16 years (5-29), HbA1c was 72 mmol/mol (64-87) or 8.8% (8.0-10.1), and BMI was 30.2 kg/m2 (22.0-36.0). All participants used other antidiabetic medicine prior to and throughout the study (2.3 on average); seven participants used metformin, five used a GLP-1 receptor agonist, four used an SGLT2-inhibitor, and two participants used DPP-4 inhibitors. Seven participants used antihypertensive drugs (2.3 on average) and seven participants used lipid-lowering drugs (1.0 on average).
Primary Objective Results
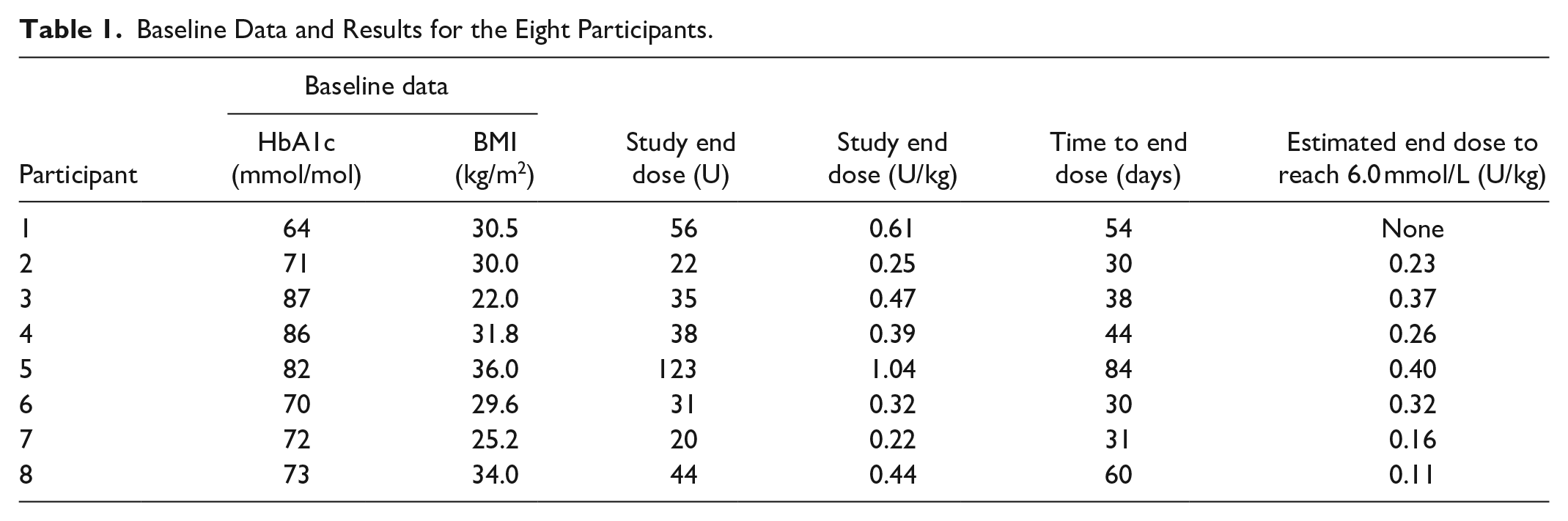
The median (range) study end dose was 37 U (20-123) or 0.4 U/kg (0.2-1.0). The dose estimated at two weeks was smaller than or equal to the study end dose in all cases (Table 1 and Figure 4) with median (range) 26.7% (0.0-75.8) error. One estimated end dose matched the study end dose with 0% error. An end dose estimation was not provided in one case due to unidentified response model.
Baseline Data and Results for the Eight Participants.
The clinician prescribed the suggested dose (75% of the estimated end dose) in three out of seven recommendations. In the remaining cases, the dose suggestion (75% of the estimated end dose) was lower than the current dose and the clinician recommended following the titration algorithm. In the following weeks, the titration algorithm was used until the study end dose was reached (a stepwise titration algorithm with dose change increments ranging from −4 to +8 U and a fasting glucose target of 72-108 mg/dL).
Secondary Objective Results
The error of the SMBG-based estimated end dose at two weeks (Figure 4) had a median (range) deviation of 10.5% (32.8% underestimation-172.7% overestimation). The SMBG-based dose suggestion at two weeks (75% of the total SMBG-based estimated end dose) was greater than the study end dose in two cases.
Before initiating insulin treatment, the median (range) time below range was 0.0% (0.0-0.0), in range was 20.0% (2.0-44.7) and above range was 80.0% (55.3-98.0) during the first four days in the study (Figure 5). Median (range) time the last four days below range was 0.0% (0.0-1.5), in range was 84.9% (66.0-99.0), and above range was 14.3% (1.0-34.0) (Figure 5). Seven out of eight participants obtained the consensus criteria for optimal time in range with more than 70% in range and less than 4% below range. 11 No SMBG values were below 54 or 72 mg/dL and no severe hypoglycemic events occurred.
The median (range) study duration was 44 days (30-84) and all participants reached the fasting glucose target within 12 weeks. Titration algorithm deviations due to low CGM readings occurred 19 times in total. Most deviations were due to low CGM readings and were done according to the clinician’s advice.
Continuous Glucose Monitoring Performance
During the first two weeks, a median (range) of 7% (2-58) of CGM data were missing, whereof one participant was missing more than the 30% consensus criteria. 11 The poorest dose prediction with a 75.8% underestimation was for the participant where 58% of data were missing. For the participant where dose estimation was not possible, 10% of the data were missing. Correlation between the dose estimation error and missing data during the first two weeks was 0.51, ie, in general, the dose estimation accuracy decreases with increased missing data. In the full study period, a median (range) of 9% (1.5-17.4) of CGM data were missing. Delay in CGM data availability at visit 2 was 6 to 16 hours.
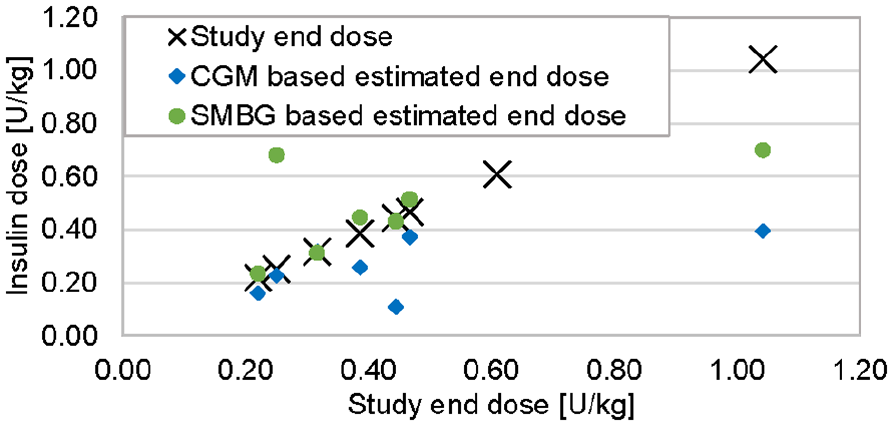
A visualization of the primary outcome. Comparison of the estimated end dose to reach 6.0 mmol/L based on insulin data, and continuous glucose monitoring or SMBG data after two weeks, compared to the study end dose for the eight participants.
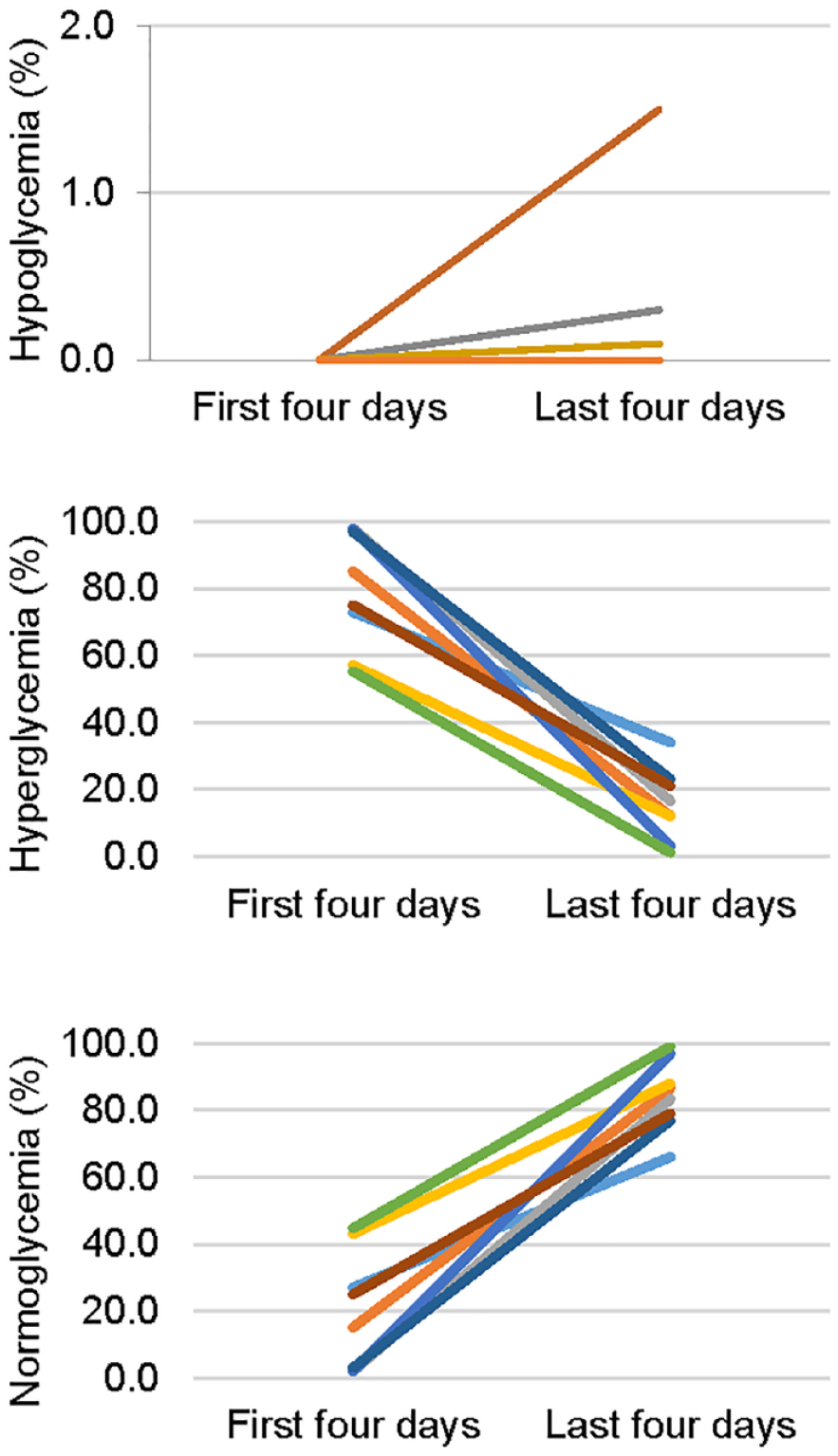
Time in hypoglycemia (below 70 mg/dL), hyperglycemia (above 180 mg/dL), and normoglycemia (between 70 and 180 mg/dL) during the first and last four days of the study for the eight participants, as measured by percentage of continuous glucose monitoring data points in each range.
Discussion
This feasibility study demonstrates that a novel approach for insulin initiation in people with T2D based on two weeks of CGM and insulin data is feasible, and doses are not overestimated. The key results on dose estimation accuracy showed that in all participants, the estimated dose was safe and no hypoglycemic events occurred.
Key results on time below, above, and in range indicate that titration to target using this approach is feasible within the time frame of 12 weeks (Table 1 and Figure 5). Stepwise titration algorithms with steps larger than 4 U of insulin are usually used for once weekly adjustments. Twice weekly titration was considered safe for three reasons: (1) use of CGM with hypo-alarm, (2) clinicians had access to the CGM data and frequent contact with participants, and (3) the fasting glucose target was widened from 72 to 90 mg/dL often used in treat-to-target trials from 72 to 108 mg/dL.
We observed a correlation between missing data in the first two weeks and dose estimation performance. Some participants expressed problems with calibration, activating a new sensor, and connectivity problems between the transmitter and the smart phone. Improved new generation CGM devices solve some of these problems. Retrospective analysis of the only case, where estimating the end dose was not possible at two weeks, shows an 11-hour lag from CGM data. The analysis indicates that this may somewhat be mitigated by increased outlier robustness in the dose estimation method and including the full 14 days of data.
In terms of absolute error, we observe that the performance of dose estimation using SMBG data is more accurate than using the CGM-based titration glucose level, in all except one case (Figure 4). However, the dose is overestimated in three cases when using SMBG data, whereof one is highly overestimated. In the case of the large overestimation, prebreakfast SMBG values were highly affected by elevated glucose following the previous day’s dinner, while the titration glucose level appeared in the middle of day, before dinner. This may have caused the prebreakfast SMBG to be a poor indicator for the dose response. In this study, the target for the CGM-based titration glucose level, ie, the lowest glucose of the day, was set to the upper limit of the prebreakfast SMBG target range. This may be too conservative and should potentially be set to a lower value due to the differences between titration glucose level and prebreakfast SMBG.
For the participant with the largest study end dose of 1.0 U/kg, the dose estimated at two weeks was underestimated by 62%. This is expected as for participants needing large doses to reach the fasting glucose target, the dose needed to observe a dose response is expected to be higher. Furthermore, the linear dose response was expected in the range of 0.4 to 0.8 U/kg. We observed a strong correlation between BMI and study end dose, which may therefore be an indicator for the dose load needed to observe a dose response.
The use of CGM was a great advantage when making dose adjustment decisions as the clinicians had information about low glucose between SMBG measurements. The clinicians furthermore had the opportunity to identify need for bolus insulin at the end of study, which would not have been possible using SMBG data only. Identification of the need for additional medication may potentially be automated in a future version.
The open CGM may have affected participants’ lifestyle. If the users changed lifestyle toward more healthy diet and exercise, this would have decreased the study end dose and influenced the accuracy of the estimated end dose. However, for safety reasons, the study was designed with an open CGM and still we found the estimated end dose safe.
The study was designed to last for a maximum of 84 days. HbA1c at the end of study would therefore not be reflective of the glycemic outcomes. Instead, CGM data were used for outcome evaluation and compared with the consensus CGM targets. 11
Standard of care titration algorithms and other dose guidance systems, such as the Intelligent Dosing System and the Diabetes Insulin Guidance System, are iterative approaches that adjust doses on a day-by-day or week-by-week basis. The novelty of this approach is the intermittent intensive data capture and the potential to provide a roadmap for the titration. The expected implications of this are decreased fear of hypoglycemia and improved perception of need for insulin by patients, thereby improving glycemic outcomes in T2D treatment. The method can be used in combination with iterative titration approaches.
In its optimal implementation, the method requires the use of connected glucose measuring devices and insulin pens and is therefore more expensive than standard of care. However, considering the high socioeconomic cost of diabetes and the great need for motivation and adherence support, the periodic use of such a solution may be economically feasible.
Conclusion
This study was a feasibility study with eight participants, and statistical significance was not expected. The results are specific to IDeg and may therefore not apply for other insulins. The concept of a study end dose defined in this study depends on the titration method, the type of basal insulin, and the intrapatient variability in pharmacodynamic and pharmacokinetic profiles of the insulin.
To the best of our knowledge, this is the first clinical study specifically on the use of CGM data to initiate basal insulin treatment in T2D and the use of a dose response model to estimate the dose needed to reach a fasting glucose target. An optimized version of this treatment approach has the potential to provide a roadmap of the treatment, which may facilitate understanding and perception of medication need, and reduce fear of hypoglycemia and complexity related to insulin initiation.
In this paper, we have shown a method for safe estimation of the end dose of basal insulin titration in T2D. We find that the dose prediction at two weeks should be improved and are working on methods for doing this. We believe that this method will become a useful tool for initiation guidance for basal insulin. Such a system could be implemented using connected insulin pens and calibration-free CGM technologies for seamless support to the clinicians and people with T2D.
Footnotes
Acknowledgements
We would like to thank the people who participated in the study. We also thank diabetes nurse Susanne Margrethe Myrup Houe and others at Steno Diabetes Center Copenhagen and Hvidovre University Hospital.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.B.A., H.B., and M.L.J. are full- or part-time employees of Novo Nordisk A/S. K.N. serves as advisor to Sanofi, Medtronic, Abbott, and Novo Nordisk; owns shares in Novo Nordisk; has received research grants from Novo Nordisk and Roche Diabetes Care; and has received fees for speaking from Medtronic, Roche Diabetes Care, Rubin Medical, Sanofi, Novo Nordisk, Zealand Pharma, and Bayer. S.S. has served on advisory boards for Roche Diabetes Care and Medtronic.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is funded by Novo Nordisk A/S and Innovation Fund Denmark through the Industrial PhD project 5189-00033B, and the Danish Diabetes Academy supported by the Novo Nordisk Foundation.