Abstract
Objectives:
The objective of this systematic review and meta-analysis was to synthesize evidence from randomized controlled trials (RCTs) evaluating the use of continuous glucose monitoring (CGM) to guide lifestyle choices, particularly nutrition, in the management of T2D.
Methods:
PubMed and Cochrane CENTRAL were searched from inception to June 6, 2025. Randomized controlled trials were included if their intervention involved the use of a CGM device and education or feedback intended to modify nutrition choices, either as part of a nutrition intervention or a multicomponent lifestyle intervention. Random-effects meta-analyses were performed, and certainty of evidence was rated in alignment with the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach.
Results:
A total of 21 RCTs involving 2734 adults in groups of interest were included, with 20 RCTs eligible for meta-analysis. Results from meta-analysis showed statistically significant improvements in HbA1c (MD: –0.46%, 95% CI: –0.71, –0.22), time in range (TIR) 70–180 mg/dL (MD: 7.18%, 95% CI: 2.77, 11.58), time above range (TAR) >180 mg/dL (MD: –7.32%, 95% CI: –12.98, –1.66), fasting glucose (MD: –7.86 mg/dL, 95% CI: –15.06, –0.65), body weight (MD: –2.06 kg, 95% CI: –3.74, –0.38), with moderate certainty of evidence, and for mean CGM glucose (MD: –11.57 mg/dL, 95% CI: –22.58, –0.56), and standard deviation (SD) glucose (MD: –4.06 mg/dL, 95% CI: –6.54, –1.58), with low certainty of evidence. No statistically significant differences were found for other outcomes, typically with low certainty of evidence.
Conclusions:
Findings from this systematic review and meta-analysis support the use of CGM as a tool to guide lifestyle choices with a focus on nutrition in the management of T2D, with significant benefits related to glycemia and body weight.
Introduction
For individuals living with diabetes, wearable devices are promising digital data sources that can support food and physical activity choices, thereby potentially allowing for more personalized self-care management. For example, continuous glucose monitoring (CGM) systems have been available for nearly two decades. 1 Based on evidence from clinical trials and real-world experiences, clinical guidelines have been established to set glycemic targets based on CGM data that can guide therapeutic decision-making 2 for clinicians and people with diabetes. 3 Furthermore, there is growing evidence, predominantly from studies in type 1 diabetes (T1D), that CGM-based measures, such as time in range (TIR), correlate with the risk of developing long-term microvascular complications, and therefore data from CGM may be used to supplement HbA1c as a measure of glycemic status for people living with diabetes.4-7 Historically use of CGM was limited to people with diabetes who require insulin. 8 However, more recently, there has been growing interest in expanding the use of CGM devices to include individuals with diabetes not using insulin, as well as adults with prediabetes or at-risk of developing type 2 diabetes (T2D)9,10 including those from traditionally underrepresented populations. 11 In all of these populations, the utility of the CGM lies heavily in its potential as a tool for behavior modification12,13 and to promote healthy lifestyle changes, such as adherence to evidence-based nutrition recommendations. 14
Guidance from professional organizations such as the American Diabetes Association (ADA) support the use of CGM to facilitate lifestyle choices as CGM technology can “. . .allow people with diabetes to evaluate their individual response to therapy and assess whether glycemic goals are being safely achieved. Integrating results into diabetes management can be a useful tool for guiding medical nutrition therapy and physical activity, preventing hypoglycemia, or adjusting medications.”
8
However, in previous research, details on the characteristics of the CGM-based interventions have often not been clearly described, including information on the type of devices used, the quality and quantity of CGM device training, and the relationship between the device use and changes in food choices and physical activity. For a technology such as CGM to be effective in helping to guide lifestyle changes, it is important that the person with diabetes and the professional recommending the device have (1) trust in the value of the information generated, (2) an understanding of the relationship between the real-time and retrospective glucose profiles and lifestyle choices, and (3) a commitment to continuously using the data for shared decision-making that leads to timely and appropriate behavior changes.15-17 Better insights into study characteristics could allow for more focused training for CGM users and could lead to more carefully designed interventions, eg, designs that help individuals and care providers optimize CGM use over time while also promoting evidence-based self-care behaviors.
Previous systematic reviews have evaluated the impact of incorporating CGM in the management of T2D and consistently demonstrated benefits in outcomes including improvement in HbA1c. 9 ,18-22 These reviews typically assessed the use of CGM broadly and included trials in which CGM was studied in a variety of contexts, from primarily informing medication titration to motivating lifestyle changes. However, there has been limited exploration of the use of CGM to guide specific aspects of diabetes management, such as nutrition. The objective of this systematic review was to synthesize the evidence from RCTs in which CGM was used in conjunction with education or guidance on nutrition or lifestyle changes in the management of T2D. We sought to be inclusive of the type of interventions in our review, including both structured nutrition interventions as well as multicomponent lifestyle education programs with a nutrition component.
Methods
Study Design
This systematic review and meta-analysis was registered on International Prospective Register of Systematic Reviews (PROPSERO) (CRD42024623086) and conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 23
Search Strategy
Searches were conducted in PubMed and Cochrane CENTRAL from inception to June 6, 2025. The strategy included a combination of medical subject headings and keywords targeting “type 2 diabetes” and “continuous glucose monitoring.” Additional terms for the intervention (eg, terms targeting “nutrition,” “lifestyle,” or “education”) were not included in the strategy given the expected variation in the terminology around nutrition education and lifestyle interventions. Rather, the search was designed to return any study evaluating CGM in T2D, and the relevance of the interventions with respect to their nutrition education component was evaluated during the screening process. The full search strategy is provided in Supplementary Materials Table A1 and Table A2. We also reviewed reference lists from previous systematic reviews and searched gray literature sources to ensure relevant trials were captured.
Study Selection
Two reviewers (MN and KF) independently screened articles in a two-stage process, first screening titles and abstracts followed by full-text articles. Screening was performed against predefined eligibility criteria (see section “Eligibility Criteria”), and reasons for exclusion at the full-text stage were documented. Disagreements were resolved through consensus, with adjudication by additional reviewers if necessary. The screening process was performed in Rayyan (Rayyan, Cambridge, MA), and references were managed with EndNote 21 (Clarivate, Philadelphia, PA).
Eligibility Criteria
Randomized controlled trials (RCTs) evaluating the use of CGM paired with lifestyle guidance with a focus on nutrition choices for T2D management were included. We included RCTs if their interventions included the (1) use of a CGM device, and (2) delivery of feedback or guidance intended to modify food or nutrition choices, either as part of a nutrition intervention or an element in a multicomponent lifestyle education program. For (1) the first criterion, we included any type of CGM device (ie, real-time, intermittently-scanned, or professional) and any duration of use (ie, single episode [used once], episodic [used during parts of the intervention period], or continuous [used throughout the entire intervention period]). For (2) the second criterion, we included a wide range of feedback types, including input from diabetes specialists, primary care physicians, dietitians, or other health care professionals (HCPs) in the form of handouts, live education sessions, or through an integrated platform. In addition to the main trial publications, we reviewed published protocols and registration records (eg, ClinicalTrials.gov), where available, to determine whether CGM was paired with nutrition or lifestyle guidance. Study selection criteria are presented in Supplementary Materials Table A3.
Eligible comparators were feedback or education alone (without CGM) or standard care. Exclusion criteria were studies where both arms received CGM as part of the intervention (eg, comparing personal and professional devices); studies conducted exclusively in participants with T1D, prediabetes, or gestational diabetes or in a mixed population in which results for the T2D population could not be isolated; and studies with nutrition components outside the scope of the review (eg, supplements, parenteral, and enteral nutrition).
Data Extraction and Coding
Data were extracted in structured forms by one of the two reviewers (MN and KF) with full quality review from the other. Disagreements were resolved through consensus. Extracted data elements included those related to study characteristics (eg, number of participants, location, follow-up); population characteristics (eg, health-related baseline measures and insulin use); intervention arms (eg, type of CGM device, duration of use, education materials, and delivery of nutrition-related feedback); comparator arms (eg, elements of standard care and self-monitoring of blood glucose [SMBG] frequency); co-interventions (eg, changes in medications and exercise); and outcome measures.
Intervention features related to feedback frequency, feedback communication style, CGM usage, CGM mode, and type of nutrition education were used to assess the “intensity” of the interventions. Ratings of high, moderate, or low intensity were first assigned to each feature. Then, “overall” intensity ratings across the individual features were assigned to (1) the interventions, and (2) the incremental comparison within the studies (ie, how intense the intervention was compared with the control). We assessed these two ratings separately given the variability in comparator groups across the included studies, with some studies comparing to a matched education or nutrition program and others comparing to continued standard care. The incremental comparison considers the additional support beyond usual care received by the comparator arm to capture the “net” difference between groups. These ratings were created with input from the expert panel. Additional details on the extracted features and rating system can be found in the Supplementary Materials.
In addition, studies were categorized based on level of diabetes management at baseline using outcome-specific thresholds. Outcome thresholds were: HbA1c, ≥8% (further from goal) versus <8% (closer to goal); time in range (TIR) 70-180 mg/dL, <60% time spent in range (further from goal) versus ≥60% time spent in range (closer to goal); time above range (TAR) >180 mg/dL, >40% time spent above range (further from goal) versus ≤40% time spent above range (closer to goal); body mass index (BMI, kg/m2), with obesity (further from goal) versus without obesity (closer to goal). The incremental intensity ratings and disease control categorizations were subsequently used in subgroup analyses as described later.
Outcomes
Main outcomes were HbA1c, TIR 70-180 mg/dL, TAR > 180 mg/dL, and BMI. Additional outcomes were weight, fasting glucose, CGM-measured mean glucose, TAR >240/250 mg/dL, time below range (TBR) <54/55 mg/dL, TBR <70 mg/dL, standard deviation (SD) glucose, percent coefficient of variation (%CV) glucose, total cholesterol, low-density lipoprotein (LDL), systolic blood pressure (SBP), calories, macronutrient intake, quality of life (QoL), and treatment satisfaction. Outcomes reported at any time point were eligible for inclusion in qualitative analysis, and outcomes reported at least 2 months were eligible for inclusion in meta-analysis.
Data Synthesis and Analysis
All outcomes included in meta-analysis were continuous. Change from baseline (CFB) values were used for analysis, and wherever required, the variance was imputed using standard methods. 24 Mean differences (MDs) with 95% confidence intervals (CIs) between the intervention and control arms were summarized as the study-level metric. For outcomes reported in at least 3 RCTs, a random-effects meta-analysis was conducted to account for clinical and methodological heterogeneity. 25 Analyses were conducted with CMA Version 4 (BioStat, Englewood NJ).
Heterogeneity was assessed using Cochran’s Q test and I2. 26 An I2 of 50% or greater was considered to indicate substantial heterogeneity. Analyses were conducted using data on change from baseline to the end of the intervention period (ie, end of the main period with CGM and feedback), except for four RCTs which only reported outcome data after an extended post-intervention follow-up period.27-30
The time points included in the analyses ranged from 2 to 12 months. Analyses using stricter time periods were also performed for the main outcomes (see Supplementary Materials Table A4). However, given the limited number of studies at more specific time points, we present results across the follow-up period in the primary analysis.
Subgroup analyses were performed to explore potential sources of heterogeneity. Subgroup results were performed for the main outcomes (ie, HbA1c, TIR 70-180 mg/dL, TAR >180 mg/dL, and BMI). The variables explored in subgroup analyses included: insulin use at baseline (none, <50%, or ≥50% using insulin), duration of CGM use (single episode, episodic, continuous), differential medication or exercise changes (yes or no), baseline disease control (further or closer to goal based on outcome-specific thresholds and based on HbA1c and BMI thresholds across all outcomes; see section “Data Extraction and Coding”), and intervention intensity (low, moderate, high; see section “Data Extraction and Coding”).
For all outcomes which were not amenable to meta-analysis, a qualitative synthesis was performed.
Quality Assessment
The risk of bias for each study was assessed using Cochrane’s revised tool for randomized trials (RoB2). 31 The methods for assessing the confidence or certainty in the cumulative evidence are aligned with the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Approach 32 and was applied for outcomes included in meta-analysis. The risk of bias and GRADE assessments were independently performed by two reviewers (MN and KF), with disagreements resolved through consensus or adjudication by additional reviewers if necessary.
Expert Input
The execution of this review was performed with input from a group of expert panel members, comprised of medical professionals, diabetes specialists, and dietitians. Meetings were held to discuss the clinical appropriateness of aspects related to methods, including outcome measures and grouping of time points and development of intensity ratings, as well as clinical interpretation of the findings that followed. This group was involved in drafting and reviewing the manuscript.
Results
Search Results
A total of 4984 records were identified, of which 25 records related to 21 unique RCTs were included in the review.27-30,33-53 Figure 1 presents the study selection process and reasons for exclusion at the full-text stage, which were primarily related to study design and intervention not of interest (eg, not including guidance or feedback targeted toward nutrition).
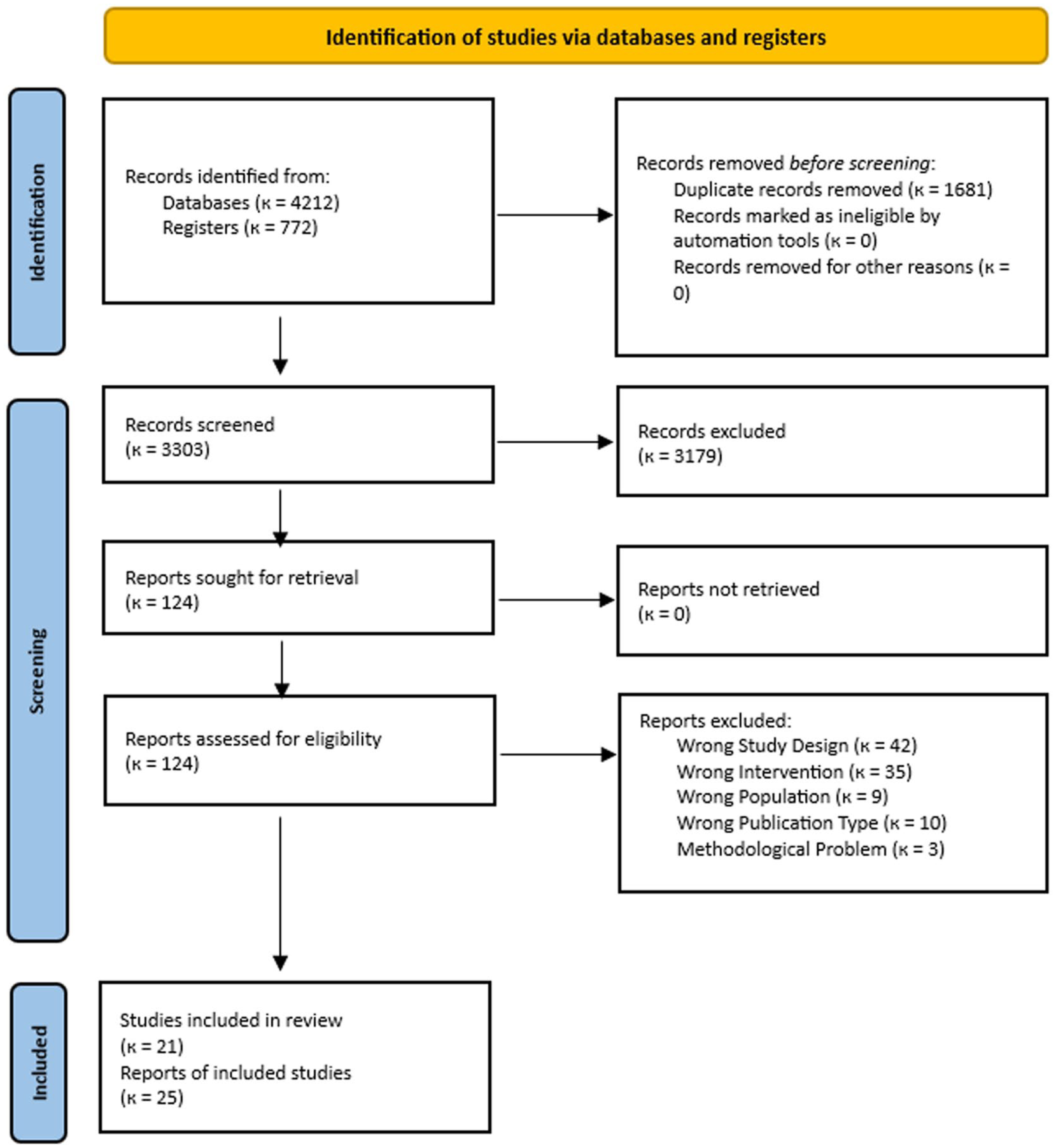
PRISMA flow diagram for study selection.
Included Studies
Table 1 provides an overview of the 21 included RCTs. These trials randomized a total of 2734 adults in groups of interest with duration of T2D ranging from 4 to 18 years. Mean baseline HbA1c was >7% in all but one study,43,44 and mean baseline BMI typically indicated participants fell within the overweight (≥23 kg/m2 for Asian populations, ≥25 kg/m2 for other populations) or obese (≥27 kg/m2 for Asian populations, ≥30 kg/m2 for other populations) classification. Insulin use varied across the trials, with 7 trials conducted exclusively in noninsulin users, and others conducted in <50% using insulin (n=6) or ≥50% using insulin (n=6) (note 2 studies did not report insulin use). The trials typically compared CGM + nutrition or lifestyle feedback (using various delivery methods) to standard care (with emphasis on timely and frequent SMBG). Most trials used a personal CGM device, with a few trials using professional CGM. Duration of CGM use ranged from a single episode of 3 days to continuous use over 12 months.
Details of Included Studies.
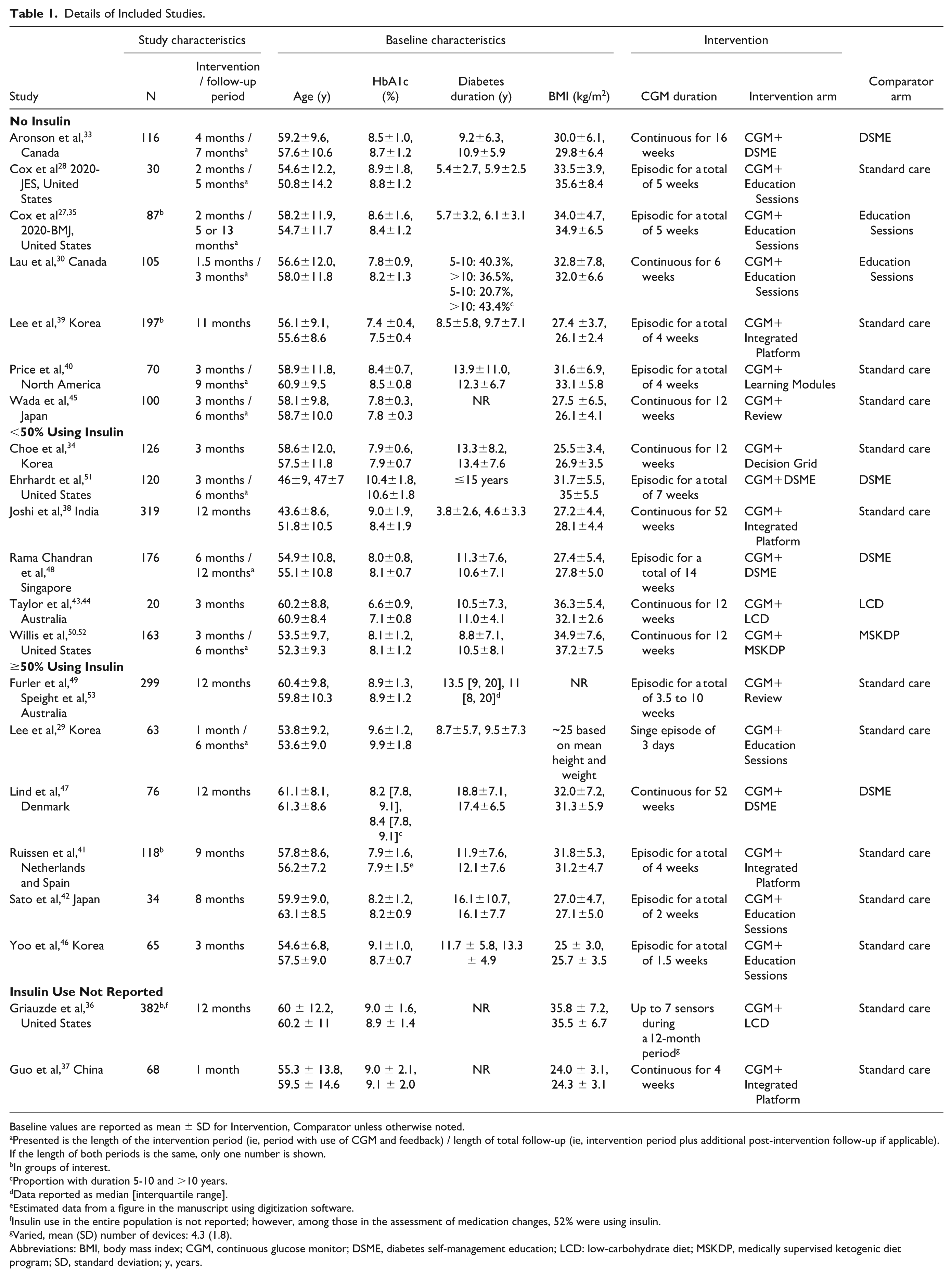
Baseline values are reported as mean ± SD for Intervention, Comparator unless otherwise noted.
Presented is the length of the intervention period (ie, period with use of CGM and feedback) / length of total follow-up (ie, intervention period plus additional post-intervention follow-up if applicable). If the length of both periods is the same, only one number is shown.
In groups of interest.
Proportion with duration 5-10 and >10 years.
Data reported as median [interquartile range].
Estimated data from a figure in the manuscript using digitization software.
Insulin use in the entire population is not reported; however, among those in the assessment of medication changes, 52% were using insulin.
Varied, mean (SD) number of devices: 4.3 (1.8).
Abbreviations: BMI, body mass index; CGM, continuous glucose monitor; DSME, diabetes self-management education; LCD: low-carbohydrate diet; MSKDP, medically supervised ketogenic diet program; SD, standard deviation; y, years.
Using the RoB2 tool, trials were generally rated as “low risk” or having “some concerns” for bias (Figure 2). The trials generally presented “low risk” of bias with respect to their randomization procedures, attrition, and analysis methods. The marks for “some concerns” were typically related to potential for reporting bias. Due to the inability to mask assignment to CGM devices, trials were not downgraded with respect to domain 2, despite limited information on the potential deviations from assigned interventions. Details on attrition are presented in Supplementary Materials Table A5.
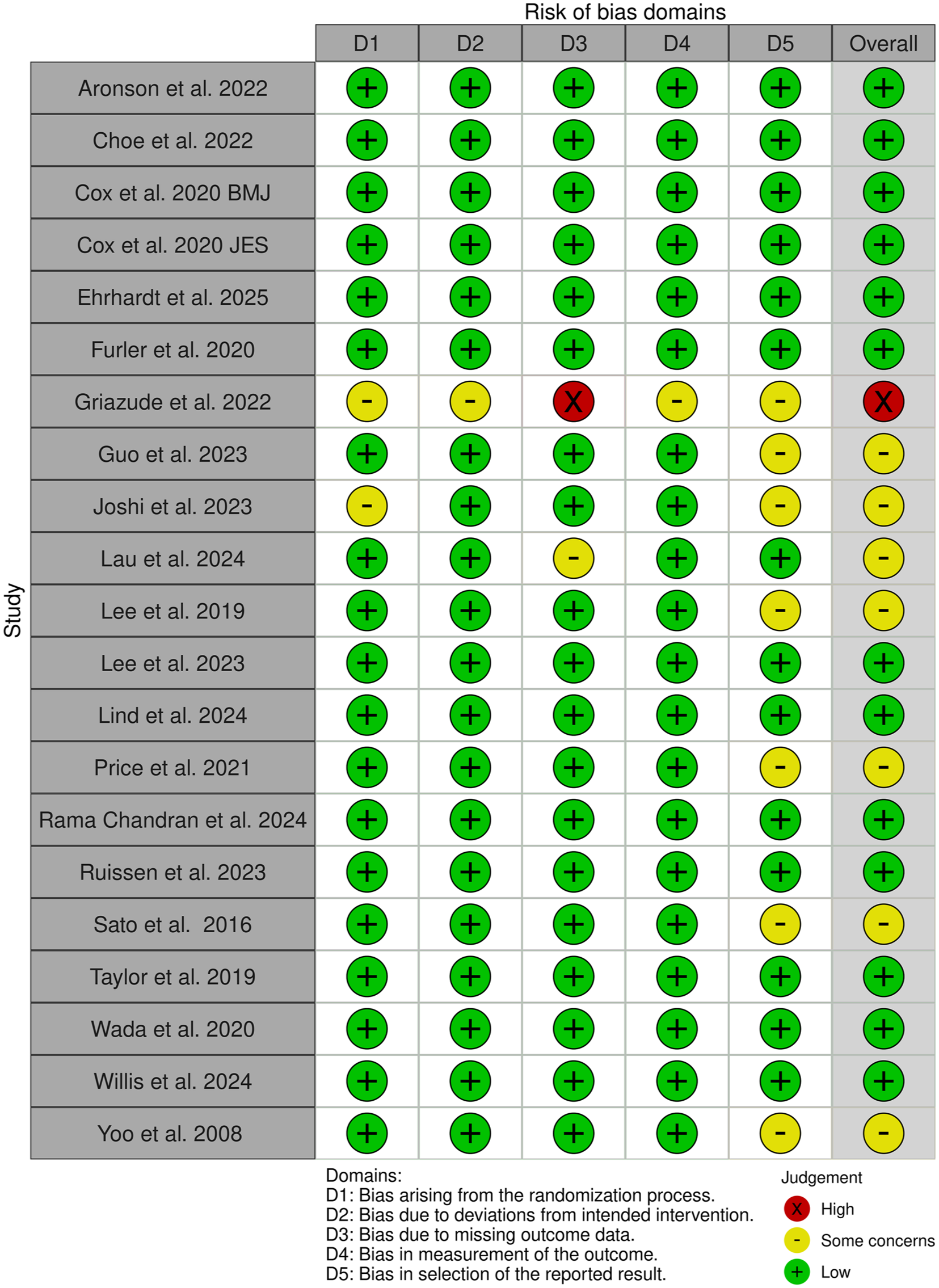
Risk of bias assessment summary: study level.
Meta-Analysis
Of the 21 included RCTs, 20 described in 24 publications,27-30,33-36,38-53 were eligible for inclusion in this meta-analysis. The reason for excluding one trial from meta-analysis was duration of follow-up of <2 months. 37 Table 2 presents results from meta-analysis. Statistically significant differences were observed for mean changes in HbA1c (Figure 3), body weight, fasting glucose, TIR 70-180 mg/dL, and TAR >180 mg/dL, with certainty of evidence rated as moderate and for mean changes in mean CGM glucose and SD glucose, with certainty of evidence rated as low.
Results From Meta-Analysis.
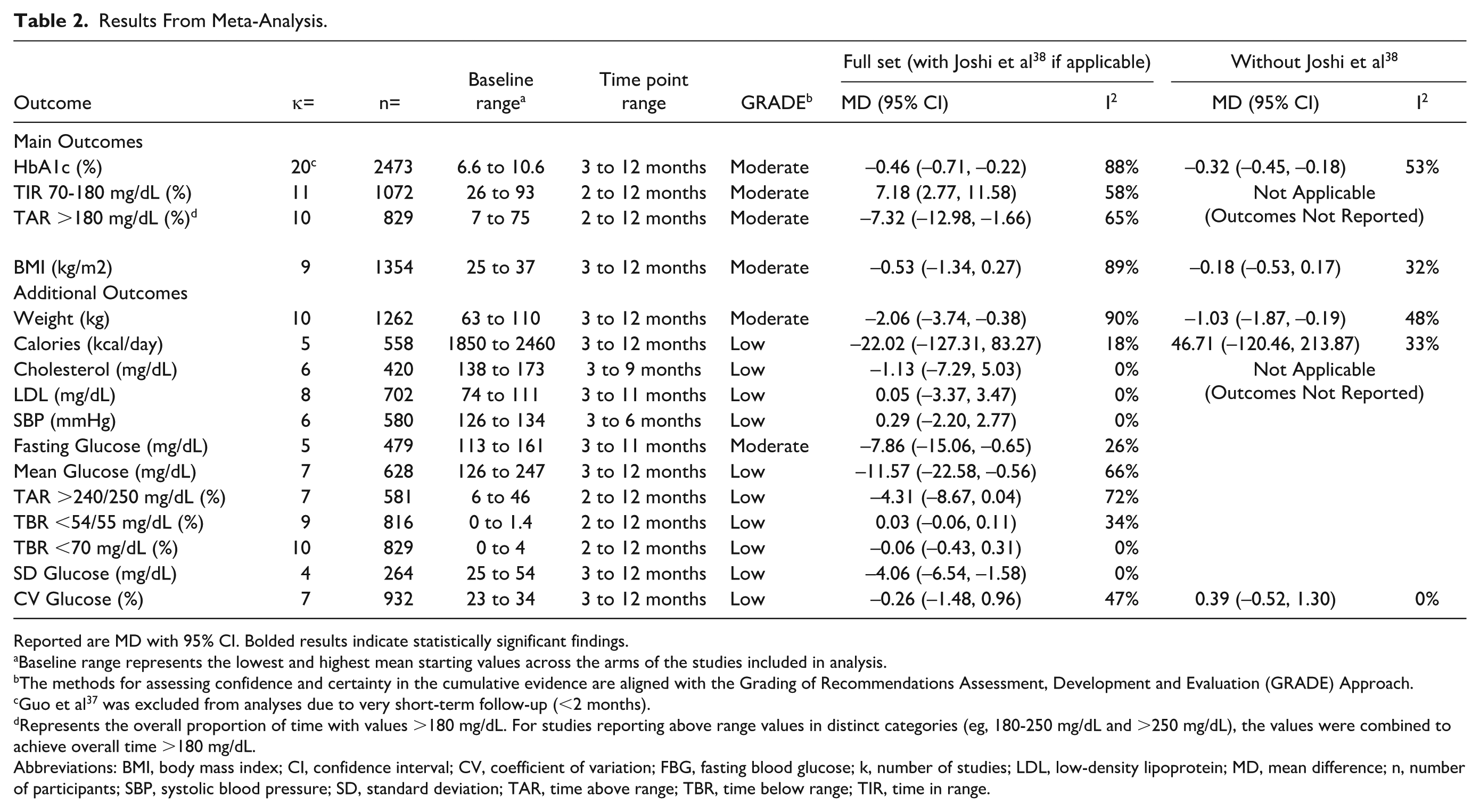
Reported are MD with 95% CI. Bolded results indicate statistically significant findings.
Baseline range represents the lowest and highest mean starting values across the arms of the studies included in analysis.
The methods for assessing confidence and certainty in the cumulative evidence are aligned with the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Approach.
Guo et al 37 was excluded from analyses due to very short-term follow-up (<2 months).
Represents the overall proportion of time with values >180 mg/dL. For studies reporting above range values in distinct categories (eg, 180-250 mg/dL and >250 mg/dL), the values were combined to achieve overall time >180 mg/dL.
Abbreviations: BMI, body mass index; CI, confidence interval; CV, coefficient of variation; FBG, fasting blood glucose; k, number of studies; LDL, low-density lipoprotein; MD, mean difference; n, number of participants; SBP, systolic blood pressure; SD, standard deviation; TAR, time above range; TBR, time below range; TIR, time in range.
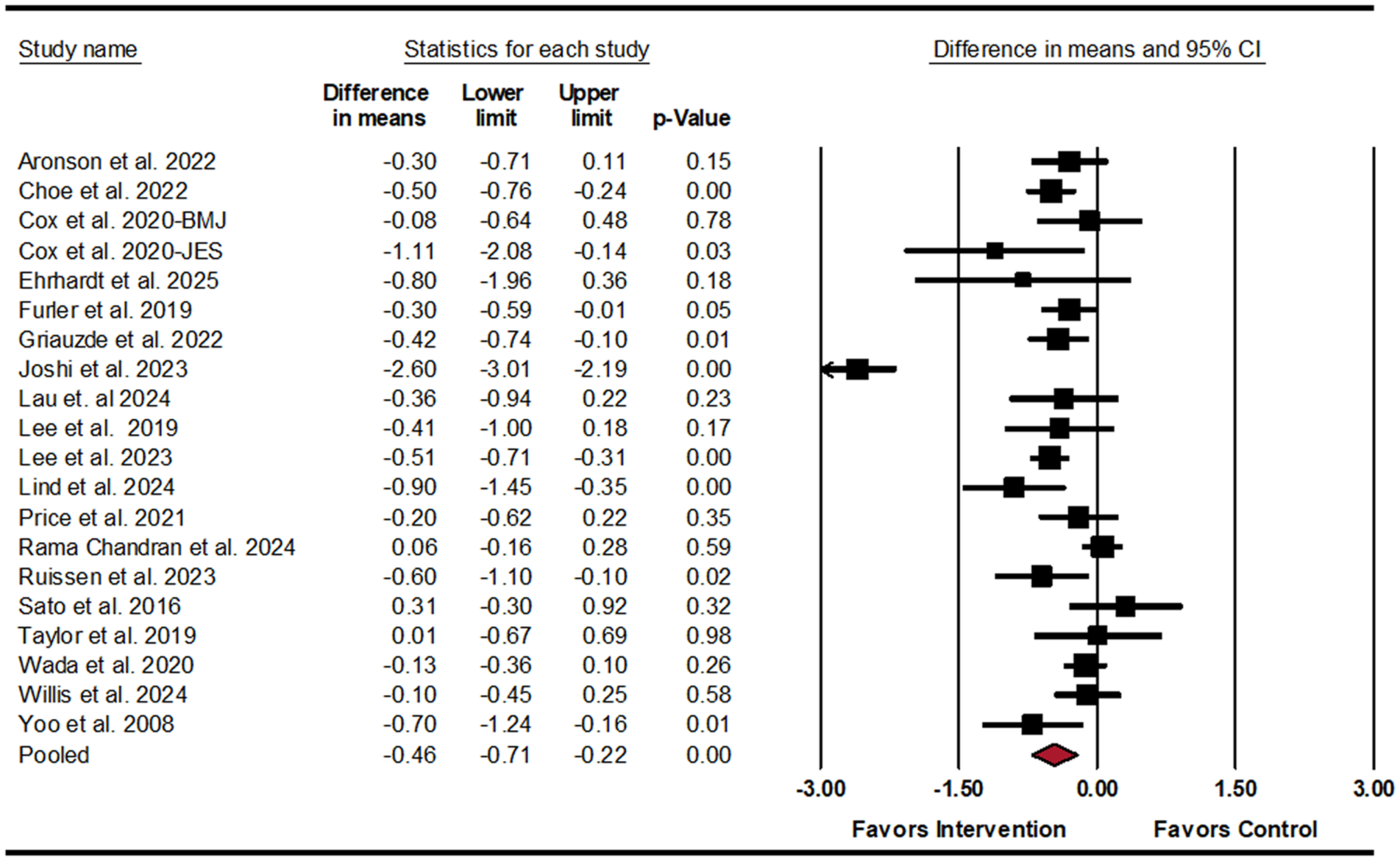
Forest plot for mean difference in HbA1c (%).
The results indicate that on average, compared with the control group, participants in the intervention groups experienced a 0.46% (95% CI: –0.71, –0.22) greater reduction in HbA1c, 2.06 kg (95% CI: –3.74, –0.38) greater reduction in body weight (or approximately 2.5% reduction), 7.86 mg/dL (95% CI: –15.06, –0.65) greater reduction in fasting glucose, 11.57 mg/dL (95% CI: –22.58, –0.56) greater reduction in mean CGM glucose, and 4.06 mg/dL (95% CI: –6.54, –1.58) greater reduction in SD glucose. In addition, on average, compared with the control group, the intervention group spent 7.18% (95% CI: 2.77, 11.58) more time per day in the goal glucose range and 7.32% (95% CI: –12.98, –1.66) less time per day above the goal glucose range. No statistically significant differences were found for other outcomes, with certainty of evidence typically rated as low.
Substantial heterogeneity was observed for most analyses (Table 2). For the assessment of the incremental comparison intensity (see section “Data Extraction and Coding”), only two trials were rated as “high,” of which one trial 38 appeared to be the primary driver of heterogeneity in multiple analyses. Heterogeneity was generally reduced by removing Joshi et al 38 from meta-analysis where applicable (Table 2). There was no change in statistical significance after removing Joshi et al, 38 although the effects were less pronounced, with HbA1c reduction of 0.32% (95% CI: –0.45, –0.18) and weight reduction of 1.03 kg (95% CI: –1.87, –0.19).
Subgroup analyses were conducted for the main outcomes (ie, HbA1c, TIR 70-180 mg/dL, TAR >180 mg/dL and BMI) (Table 3). Results revealed trends toward greater benefits in RCTs with participants further from goal at baseline based on the outcome-specific and HbA1c thresholds. However, based on the BMI threshold, there were trends toward greater benefits for TIR 70-180 mg/dL and TAR>180 mg/dL for those closer to goal. Subgroup results by incremental comparison intensity had trends suggesting greater benefits in RCTs with more intense comparisons for HbA1c and TIR 70-180 mg/dL, but not BMI or TAR>180 mg/dL. Subgroup results by intervention intensity showed trends toward greater benefits for HbA1c with more intense interventions but not for the other outcomes. Subgroup results for differential medication changes (ie, whether there was evidence of more or less change to medications between groups) showed trends toward greater improvements for HbA1c in studies with evidence of change, with all studies indicating the intervention group required less medication than the control group. Most studies did not report data to inform differential exercise changes. Overall, the current evidence base limits conclusions with respect to the factors explored in subgroup analyses.
Subgroup Results for Main Outcomes.
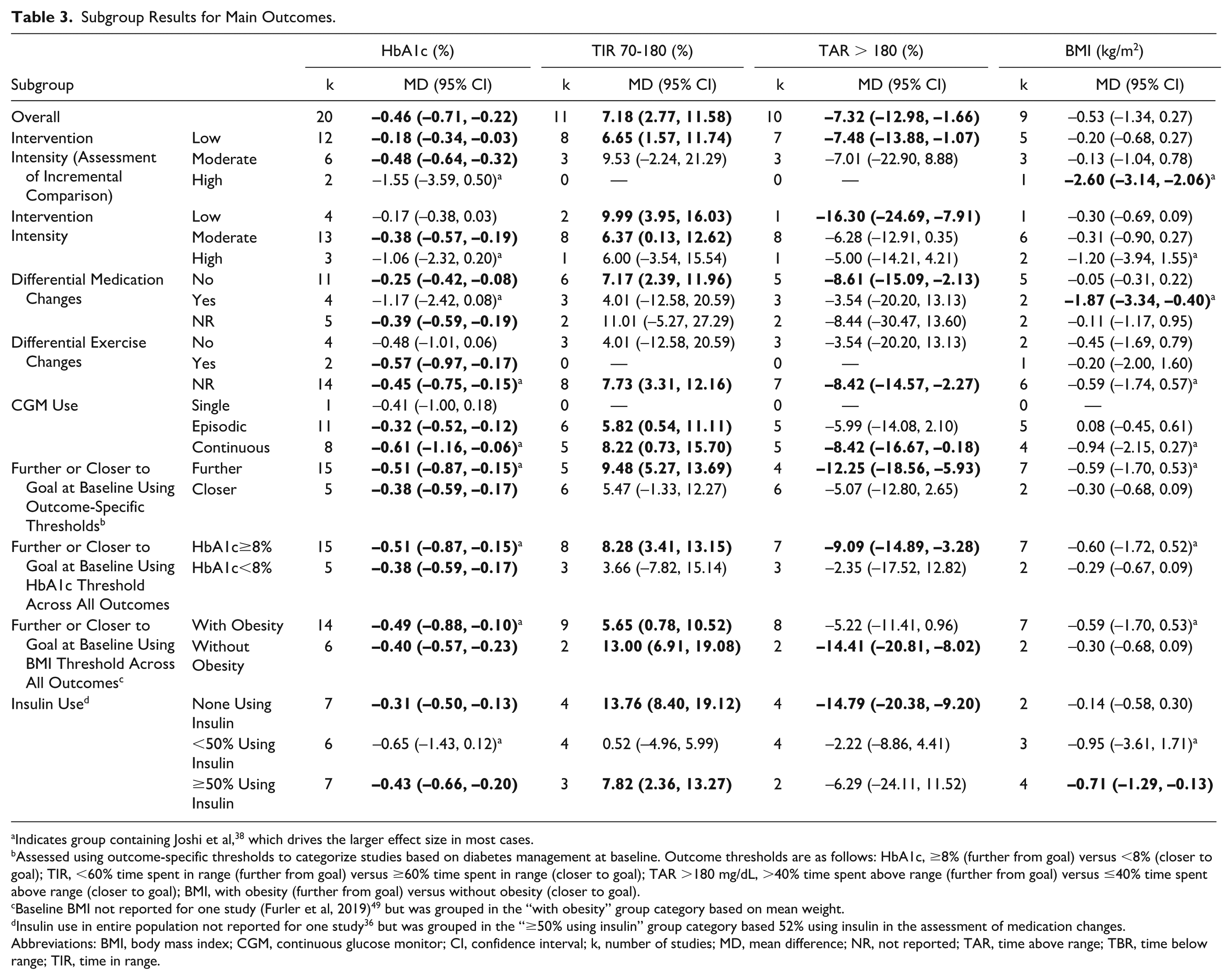
Indicates group containing Joshi et al, 38 which drives the larger effect size in most cases.
Assessed using outcome-specific thresholds to categorize studies based on diabetes management at baseline. Outcome thresholds are as follows: HbA1c, ≥8% (further from goal) versus <8% (closer to goal); TIR, <60% time spent in range (further from goal) versus ≥60% time spent in range (closer to goal); TAR >180 mg/dL, >40% time spent above range (further from goal) versus ≤40% time spent above range (closer to goal); BMI, with obesity (further from goal) versus without obesity (closer to goal).
Baseline BMI not reported for one study (Furler et al, 2019)49 but was grouped in the “with obesity” group category based on mean weight.
Insulin use in entire population not reported for one study 36 but was grouped in the “≥50% using insulin” group category based 52% using insulin in the assessment of medication changes.
Abbreviations: BMI, body mass index; CGM, continuous glucose monitor; CI, confidence interval; k, number of studies; MD, mean difference; NR, not reported; TAR, time above range; TBR, time below range; TIR, time in range.
Baseline values across the included studies showed wide ranges, particularly for glycemic measures. HbA1c ranged from 6.6% in Taylor et al 43 to 10.6% in Ehrhardt et al 51 ; TIR 70-180 mg/dL ranged from 26% Ehrhardt et al 51 to 93% in Taylor et al 43 and TAR>180 mg/dL ranged from 7% in Taylor et al 43 to 75% in Ehrhardt et al. 51 Analyses excluding the studies with the lowest and highest baseline values generally showed similar results. For HbA1c, estimates were –0.49 (95% CI: –0.74, –0.23) excluding Taylor et al, 43 –0.46 (95% CI: –0.70, –0.21) excluding Ehrhardt et al, 51 and –0.48 (95% CI: –0.73, –0.22) excluding both, with baseline HbA1c values ranging from 7.4 to 9.7% among remaining studies. For TIR 70-180 mg/dL, results were 7.21% (95% CI: 2.56, 11.86) excluding Ehrhardt et al, 51 8.30% (95% CI: 4.21, 12.38) excluding Taylor et al, 43 and 8.43% (95% CI: 4.08, 12.78) excluding both, with remaining baseline TIR values ranging from 42% to 70%. For TAR>180 mg/dL, estimates were –6.84% (95% CI: –12.97, –0.72) excluding Ehrhardt et al, 51 –8.86% (95% CI: –14.33, –3.38) excluding Taylor et al, 43 and –8.54% (95% CI: –14.55, –2.53) excluding both, with baseline TAR ranging from 28% to 58%.
Qualitative Synthesis
Results were summarized qualitatively for macronutrient intake, QoL, and treatment satisfaction. These outcomes were not included in meta-analysis due to limited reporting (ie, <3 studies) or variation in data reporting. Evidence on intake of macronutrients or treatment satisfaction was limited. Of the 21 included studies, 5 studies reported on carbohydrate and fat intake, and 4 studies reported on protein intake, with inconsistent findings. Regarding treatment satisfaction, 8 studies reported on various scales also with inconsistent findings.
Data on QoL were mixed, with some RCTs indicating benefits but not across all time points and domains evaluated. Guo et al 37 observed improvements in the psychological condition, social relationship, and treatment influence subscales of the Diabetes Specific Quality of Life (DSQL) scale in the intervention group after 4 weeks, although no significant changes were noted in the physiological condition subscale. 37 Cox et al 28 2020-JES reported improvements in the psychological dimension of the World Health Organization (WHO)-QoL scale at 5 months, but not in the physiological dimension. Rama Chandran et al 48 documented improvements in the EQ-5D utility values at 12 months favoring the intervention group, with no changes observed at 2 or 6 months and no differences between groups in the EQ-VAS at any time points. Lind et al 47 reported significant improvements in the WHO Five Well-Being Index (WHO5) and SF-36 General Health scale at 12 months in the intervention group compared with control. In contrast, Ruissen et al 41 found no significant changes from baseline to 9 months in either group on the WHO-QoL scale, and Furler et al 49 found no differences between groups at 12 months on the WHO5 scale. 53
Discussion
This systematic review and meta-analysis examined the role of CGM in guiding lifestyle choices with a focus on nutrition in the management of T2D and demonstrated a statistically significant HbA1c reduction of -0.46% compared with education or standard care without using CGM, with moderate certainty of evidence. The analysis also showed statistically significant improvement in TIR 70-180 mg/dL, and reduction in TAR > 180 mg/dL, fasting glucose, and body weight, with moderate certainty of evidence, and reduction in mean CGM glucose and SD glucose, with low certainty of evidence. No statistically significant differences were observed for other outcomes included, typically with low certainty of evidence. Overall, these findings support the use of CGM as a tool for guiding lifestyle and nutrition choices in T2D.
Our review included a heterogeneous group of studies, with differences in baseline glycemic metrics, duration of diabetes, and use of insulin. The studies also varied in terms of the duration of CGM use, continuous versus episodic CGM use, and the type and intensity of the nutritional/education interventions (including the frequency of education, duration and length of sessions, and use of additional digital health technologies). It is also noteworthy that time points in the meta-analysis ranged from 2 to 12 months, as longer intervention periods may be reasonably expected to show greater improvements in outcomes. To explore this, we conducted analyses in 3-month intervals, but the small number of studies, particularly in the later time periods, limited our ability to identify meaningful trends over time.
Subgroup analyses were also performed to explore potential sources of heterogeneity. Overall, data to support evidence of differential effects were limited, but allowed observation of some trends. With respect to insulin use, we observed trends toward reductions in HbA1c regardless of baseline insulin therapy. This finding aligns with other recent meta-analysis results, which indicated similar effects in insulin-requiring individuals and those using only oral agents. 19 Our analysis also highlights the minimal early use of CGM in T2D, with only 1 study enrolling participants with less than 5 years duration of diabetes. 38 This study showed greater benefits on key glycemic and body composition outcomes compared with other studies for various reasons, including the intensity of the intervention. The findings emphasize the need for additional research on the impact of CGM combined with lifestyle and nutrition education earlier in the course of T2D and prior to insulin initiation, as CGM can serve as a tool to increase engagement with education and improve overall disease understanding and management in those with a recent diagnosis. 54
Results from subgroup analyses also showed trends toward greater glycemic benefits among those with higher baseline HbA1c levels. This finding was initially observed in the MOBILE study subanalysis, which specifically showed greater benefits for individuals on insulin with baseline HbA1c levels (>10.0%). 55 Additionally, the IMMEDIATE study included in this analysis demonstrated that DSMES, either with or without CGM, led to greater glycemic reduction in a noninsulin-requiring population with higher baseline HbA1c levels (>9.0%). 33 Consistent with 2025 ADA guidelines, 8 subgroup results also support the consideration of CGM wear as continuous rather than episodic, when possible, with trends toward greater reductions in HbA1c with continuous wear compared with episodic wear or single use.
A unique aspect of this meta-analysis was the evaluation of CGM as a tool to guide lifestyle choices with a focus on nutrition. Nutrition is a key component in managing T2D, and guidance supports a variety of dietary approaches based on the needs of the individual. 14 CGM can serve as a tool for individualizing eating plans by providing real-time insights into the relationship between food and glucose levels. We sought to include data on the type of education on nutrition or food choices provided in studies included in our review in an effort to characterize how CGM has been paired with guidance of this nature in the studies. We observed considerable differences across the interventions in terms of the type, frequency, and personalization of nutrition education. Most studies integrated a nutrition element within a multicomponent lifestyle intervention, while a few evaluated a specific nutrition intervention, such as use of CGM to guide a low- or very-low-carbohydrate eating pattern. We also observed considerable differences in the comparator groups across the studies, with some studies comparing to a matched nutrition or education programs and others comparing to standard care. To explore the impact of this variation on the observed treatment differences, we developed an “intensity” rating framework to collect and rate key features of the interventions and incremental comparisons within the studies for subsequent investigation in subgroup analyses. We were able to see trends toward greater benefits with more intense interventions and comparisons for HbA1c, for which we had the most studies, but not consistently across the other main outcomes. Overall, this was an exploratory approach that was limited by the lack of reporting on key intervention features in the studies as well as the inability to isolate certain aspects of the interventions such as exercise. Future studies and more detailed reporting on intervention and comparator features will allow for closer examination of these factors and their effects on key outcomes.
One of the two “high” intensity studies was determined to be a driver of heterogeneity in most analyses. 38 Joshi et al 38 evaluated a multicomponent intervention that incorporated data from CGMs, fitness trackers, and other digital health technologies to inform personalized recommendations for food, activity, and sleep using artificial intelligence (AI) technology. The heterogeneity observed in the analysis showcases how outcomes may differ when the “intensity” or comprehensiveness of interventions vary. Although resource limitations may hinder implementation of certain aspects of a “high” intensity intervention, a “minimum” for interventions involving nutrition guidance should be considered to achieve meaningful outcomes. It is noteworthy that in the subgroup of “moderate” intensity comparisons, there was a significant HbA1c reduction of 0.48%. This finding is particularly relevant for clinics with limited resources.
A standardized definition for nutrition education or intervention is lacking within the included studies, which likely influences the findings of this analysis. “Nutrition Intervention” is the third step in the Academy of Nutrition and Dietetics’ Nutrition Care Process, a systematic method that dietetics and nutrition professionals use to provide personalized nutrition care to meet the specific needs of an individual. 56 It is driven by the other Nutrition Care Process steps, Nutrition Assessment and Diagnosis, and the purpose is to plan and implement actions intended to positively change or improve a nutrition-related problem, such as hyperglycemia. 56 The interventions across the included studies varied in terms of intensity and personalization, with only a few aligning with the Academy of Nutrition and Dietetics criteria. Therefore, standardizing methods used to better describe what type of content is covered and how the nutrition education is delivered (eg, nutrition intervention with a credentialed nutrition and dietetics practitioner or another care provider), is crucial in CGM studies and will allow for better insights into the potential impact of CGM when used to guide personalized nutrition interventions. Standardization will also support consistency in data analysis, comparisons across studies, and replication of interventions in different populations.
There is also a need to develop standards for comparison, including creating appropriate attention control groups beyond standard of care. This will include matching groups for nutrition education, frequency of interactions with professionals and offering alternative forms of self-monitoring. Through our review, we noted considerable differences in the control groups, with some receiving standard care, while others initiated a matched education or nutrition program. Although we attempted to reflect these differences in our “intensity” ratings by assessing the incremental comparison of the studies (ie, how intense the intervention was compared with the comparator), the use of standardized comparators would better enable comparison of treatment effects across studies.
Future Research Directions
Continuous glucose monitoring has transformed diabetes care and is considered a standard of care in managing insulin-treated diabetes. 8 More recently, there is growing interest in offering CGM to individuals with noninsulin treated T2D and prediabetes. 57 Furthermore, in 2024, several over-the-counter CGMs were cleared in the United States. As these grow in popularity, a major challenge will be to make clinical sense of 96 to 1440 interstitial glucose readings per day in otherwise healthy individuals. 58
Although there is robust clinical evidence that diabetes education and medical nutrition therapy can reduce the risk of progression from prediabetes to T2D in clinical practice, referral rates are low and for those that are referred, the number of participants completing programs is suboptimal. 59 As CGM provides personal insights into prevailing glucose levels, the value of this technology for individuals with or at risk of T2D needs to be further investigated. 3 This includes evaluating the level of user’s understanding of the link between food choices and glucose profiles and the ability of an individual to change behavior.
Preliminary work suggests that new CGM metrics may allow for stratification of individuals into subgroups that will increase our understanding of the heterogeneity of T2D including the impact of psychosocial, behavioral and environmental influences on outcomes. 60 This will also necessitate capturing, a priori, more granular information related to food and lifestyle choices including timing of meals, diet quality, macronutrients, portion sizes and physical activity, although capturing food choices can be challenging. 61 It is also important to assess the impact on psychological functioning such as QoL, depressive symptoms, and empowerment. Continuous glucose monitoring metrics will continue to evolve and be incorporated into clinical guidelines to include options for different target ranges to fit various patient populations. Furthermore, CGM metrics of time in range or above a specific threshold may be supplemented by area under the curve, which incorporates both time and degree of hyperglycemia. In addition, it is inevitable that other analytes will be added including real-time measurements of ketones. 62
The cost of using CGM can be a barrier to equitable use. For individuals not using insulin, health economic research is required to determine the optimum cost-effective “dose” of CGM including the duration and frequency of use for different subgroups (ie, at-risk vs prediabetes vs T2D). This includes a need to capture variables such as the number of doctor visits, medication changes and costs, and hospitalizations. Additional research is also needed to determine the optimal referral times and follow-up frequency with an educator or care provider when people with diabetes are using CGM to motivate and maintain nutrition and other behavior changes that improve glycemic outcomes.
Continuous glucose monitoring has been an enabling technology for people with diabetes. In the future as CGM becomes more mainstream in primary care, the onus for the diabetes research community is to create the evidence necessary to help professionals and patients to gain the maximum return on their investment of time and resources.
Limitations
We observed considerable differences in the interventions as well as certain population characteristics that contributed to the heterogeneity in analyses. In addition, many studies incorporated education on nutrition within a multicomponent program which targeted changes to other factors (eg, activity and medications) in addition to diet. To understand the potential impact of concurrent changes in these aspects, we conducted subgroup analyses by evidence of differential exercise or medication changes, although data were limited, particularly on exercise. Lastly, data were sparsely reported for macronutrients, QoL, and treatment satisfaction, limiting the understanding for these important outcomes. Beyond these limitations, our study has several notable strengths. While previous reviews have examined the use of CGM in a broader context, we focused our review on studies with a nutrition component. This focus allowed for a more targeted characterization of the study interventions. We subsequently incorporated these characteristics into our “intensity” ratings and performed subgroup analyses to understand how varying intensity may affect outcomes, providing valuable insights for future CGM-guided nutrition research.
Conclusions
This systematic review and meta-analysis provided a comprehensive overview of how CGM has been used to inform lifestyle choices with a focus on nutrition in RCTs. The current evidence demonstrates benefits for several outcomes including HbA1c, fasting glucose, TIR 70-180 mg/dL, and TAR >180 mg/dL, weight, mean CGM glucose, and SD glucose, with indications of potential benefits for other outcomes, which may strengthen as the evidence grows.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251384318 – Supplemental material for Continuous Glucose Monitoring to Guide Lifestyle Choices With a Focus on Nutrition in the Management of Type 2 Diabetes: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-dst-10.1177_19322968251384318 for Continuous Glucose Monitoring to Guide Lifestyle Choices With a Focus on Nutrition in the Management of Type 2 Diabetes: A Systematic Review and Meta-Analysis by Raveendhara R. Bannuru, Natalie J. Bellini, Richard M. Bergenstal, Elizabeth A. Beverly, Elena A. Christofides, Daniel Cox, Nicole Ehrhardt, Katherine Fazioli, Amy Hess Fischl, David Kerr, Otis W. Kirksey, Ka Hei Karen Lau, Thomas W. Martens, Meredith Noble, Prajakta Khare Ranade, Viral N. Shah, Holly J. Willis and Nuha A. ElSayed in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
ADA, American Diabetes Association; AI, artificial intelligence; BMI, body mass index; CGM, continuous glucose monitoring; CI, confidence interval; CV, coefficient of variation; DSME, diabetes self-management education; DSQL, diabetes specific quality of life; EQ-VAS, EuroQol Visual Analog Scale; GRADE, grading of recommendations assessment, development and evaluation; HCPs, health care professionals; LCD, low-carbohydrate diet; LDL, low-density lipoprotein; MD, mean difference; MSKDP, medically supervised ketogenic diet program; NI, nutrition intervention; PRISMA, preferred reporting items for systematic reviews and meta-analyses; QoL, quality of life; RCT, randomized controlled trial; SBP, systolic blood pressure; SC, standard care; SD, standard deviation; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range; TIR, time in range; WHO5, WHO-5 well-being index; WHO-QoL, World Health Organization quality of life.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RRB has declared no conflicts of interest. NJB has served as a co-investigator for Tandem (employer); is part of a speaker’s bureau for Abbott Diabetes, OmniPod, and Proventio Bio/Sanofi; and is an advisor for Medtronic Diabetes and Lifescan Diabetes Institute. RMB has received research support, has acted as a consultant, or has been on the scientific advisory board for Abbott Diabetes Care, Ascensia, CeQur, DexCom, Eli Lilly, embecta, Hygieia, Insulet, MannKind, Medtronic, Novo Nordisk, Onduo, Roche Diabetes Care, Tandem Diabetes Care, Sanofi, United Healthcare, Vertex Pharmaceuticals, and Zealand Pharma. RMB has declared no personal conflicts of interest all funding for these activities are to his nonprofit institution. EAB has declared no conflicts of interest. EAC has received funding (to institution), received consulting fees, received travel funding, is part of a speaker’s bureau, or served on advisory board or an instructor/speaker at a CME for Abbott Nutrition, Eli Lily, Novo Nordisk, Recordati, Corcept, Ascendis, and received travel funding, is part of a speaker’s bureau, or served on advisory board or an instructor/speaker at a CME for Xeris. DJC is a co-investigator on an Abbott grant and has a contractual agreement with Dexcom to provide CGM supplies at reduced rates for an NIH grant. NE is a consultant/advisor for Bayer, Dexcom, Novo Nordisk, Medtronic, and has received researcher/educational grants from Dexcom, Novo Nordisk, Boehringer Ingelheim. KF has declared no conflicts of interest. AHF has declared no conflicts of interest. DK has served as a consultant for Sanofi, Evidation Health, and Better Therapeutics and has stock options for Glooko. OK is part of a speaker’s bureau for Novo Nordisk. KHKL has declared no conflicts of interest. TWM has received (to institution) research funding from Abbott Diabetes Care, Dexcom, Insulet, Lilly, Medtronic, Novo Nordisk, Sanofi, and Tandem, speaker’s fees from Abbott Diabetes Care, Dexcom, Lilly, Novo Nordisk, Sanofi, and consulting fees from Dexcom, Sanofi, and Lilly. MN has declared no conflicts of interest. PJK has declared no conflicts of interest. VNS has served as part of an advisory board for Sanofi, Embecta, Biomea Fusion, and Novo Nordisk, has received personal fees from Embecta, Insulet, Tandem Diabetes Care, insulet, Sequel Med Tech, Dexcom, Genomelink, and Lumosfit for speaking or consulting, and has received research grants from Novo Nordisk, Alexion, Tandem, Insulet, Enable Bioscience, Zucara Therapeutics, and Lilly. HJW has received funding (to institution) from Abbott Diabetes Care and Dexcom and served as a consultant and on an advisory board or an instructor/speaker at a CME for Abbott Diabetes Care, and Dexcom. NAS has declared no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by an unrestricted educational grant to the American Diabetes Association from Abbott Diabetes Care. The Funder was not involved in the design, conduct, analyses, interpretation or publication of this study.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
