Abstract
Physical activity is important for patients living with type 1 diabetes (T1D) but limited by the challenges associated with physical activity induced glucose variability. Optimizing glycemic control without increasing the risk of hypoglycemia is still a hurdle despite many advances in insulin formulations, delivery methods, and continuous glucose monitoring systems. In this respect, the artificial pancreas (AP) system is a promising therapeutic option for a safer practice of physical activity in the context of T1D. It is important that healthcare professionals as well as patients acquire the necessary knowledge about how the AP system works, its limits, and how glucose control is regulated during physical activity. This review aims to examine the current state of knowledge on exercise-related glucose variations especially hypoglycemic risk in T1D and to discuss their effects on the use and development of AP systems. Though effective and highly promising, these systems warrant further research for an optimized use around exercise.
Introduction
Patients living with type 1 diabetes (T1D) are dependent on insulin replacement due to autoimmune destruction of β-cells. Additionally, they experience dysregulation in the response of other glucoregulatory hormones (e.g., glucagon and epinephrine), making glucose control challenging especially during exercise. 1
Exercise can be beneficial for patients with T1D with known improvements in cardiovascular fitness, vascular health, insulin requirements, body composition, and quality of life. 2 However, mainly because of the inability to reduce insulin at the time of exercise, exercise-induced hypoglycemia is frequent1,3-5 and remains the main limitation for exercise practice.6-9 Guidelines for minimizing exercise-related hypo- and hyperglycemic risks exist but their implementation require major individualization1,10,11 as numerous factors influence glycemia during exercise such as the type, intensity and duration of exercise, the timing in relation to meals, and glucose level at exercise start and should be taken into account to adjust insulin dosing and carbohydrate (CHO) intake. 12 Because glucose fluctuations can occur during as well as for numerous hours after exercise (e.g., nocturnal hypoglycemia), intensive blood glucose (BG) monitoring is required to implement two major therapeutic actions: insulin dose modifications (mainly anticipated reduction) and/or additional CHO consumption.4,9,13 However, exercise reduces the accuracy of continuous glucose monitoring (CGM) systems further complexifying therapeutic decisions.14-18
Technological strategies aim to reproduce endogenous hormone secretion pattern of the healthy pancreas, decreasing hyperglycemia without increasing the risk of hypoglycemia. Currently, the artificial pancreas (AP) system is the most advanced technology to help reduce the risk of exercise-induced hypoglycemia. 3
The objectives of this review are (1) to summarize the current state of knowledge on physiological mechanisms of BG variability and known strategies to prevent hypo- and hyperglycemia around exercise, (2) to discuss how advances in the AP technology could be used to alleviate the burden of glucose management around exercise, and (3) to discuss remaining challenges to optimize this technology.
Physical Exercise: A Challenge for Patients Living With T1D
Glucose regulation will mainly depend on the balance between muscle glucose uptake versus hepatic glucose production and/or ingested glucose. Skeletal muscle uses glucose (glycogen stores and plasma glucose) as a source of energy during exercise and represents the major site for insulin-stimulated glucose uptake (Figure 1). 19 The main hormones implicated in glucose regulation are insulin (muscle glucose utilization and hepatic glucose production inhibition) and glucagon (hepatic glucose production) with the additional role of catecholamines hepatic glucose production in the cases of very intense exercise. 20
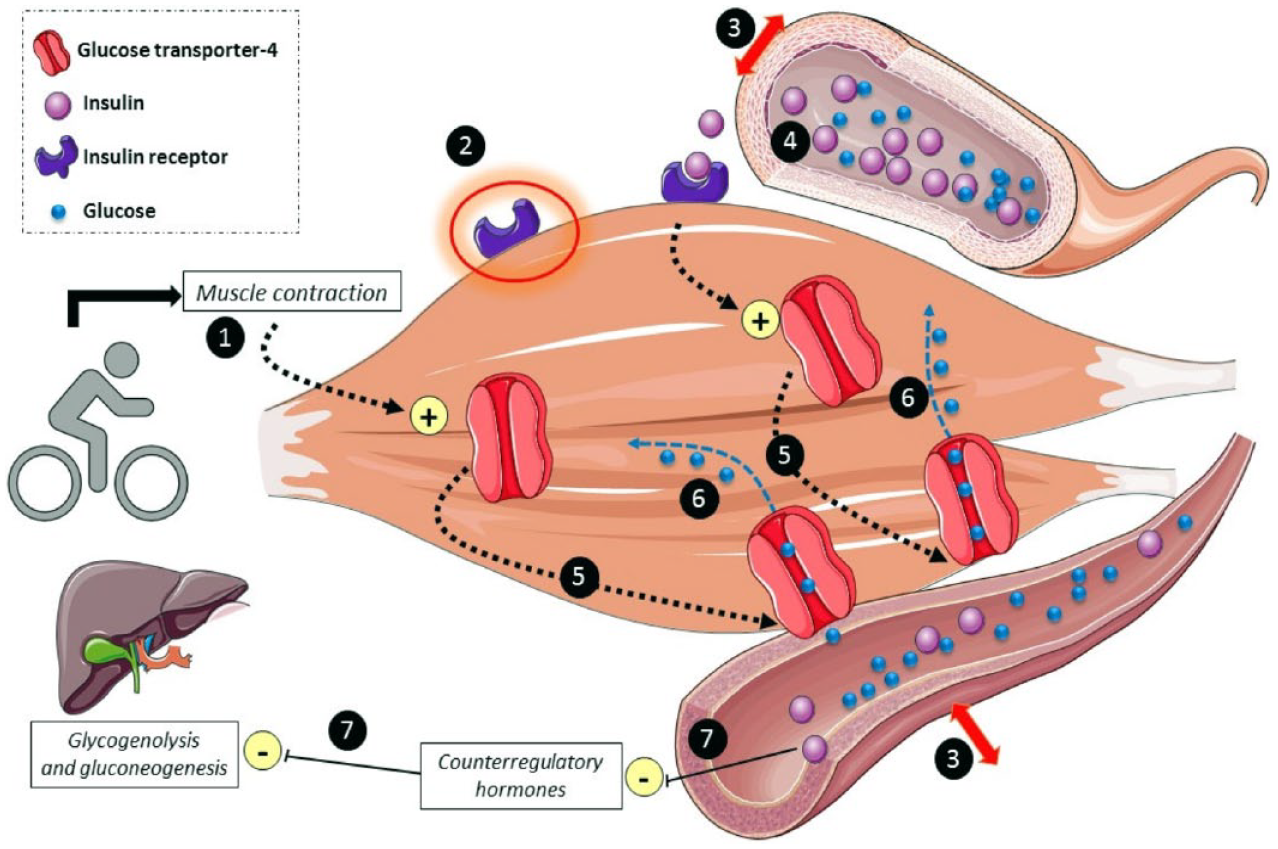
Underlying mechanisms inducing hypoglycemia in individuals with type 1 diabetes during low-to-moderate intensity exercise level exercise.
During exercise, in healthy individuals, the combination of rapid insulin secretion suppression with increased counter-regulatory hormonal activation (e.g., glucagon, catecholamines, growth hormone, and cortisol) increases hepatic glucose production to match peripheral (muscle) glucose disposal resulting in minimal glucose varaitions.9,21 However, patients with T1D are unable to suppress injected exogenous insulin and also frequently to increase counter-regulatory hormonal increasing hypoglycemic risk. 1 Conversely, if insulin levels are too low (e.g., high glucose before exercise) and/or in the context of large catecholamine release (e.g., very intense exercise), hepatic glucose production will exceed muscle glucose utilization resulting in hyperglycemia.22,23
Exercise intensity is a major factor impacting BG regulation. 24 While low and moderate intensity exercise (e.g., jogging, running, the elliptical, swimming, and cycling) are more frequently associated with a hypoglycemic risk, very intense exercise (e.g., strength training, sprinting, interval training, circuit training, and some competitive sports) implies a dominant role for catecholamines with the increase in hepatic glucose production that could lead to hyperglycemia.22,23 Such hyperglycemia can persist during recovery.25-27
In addition, an exercise, even at a low intensity, will increase glucose disposal via insulin-mediated and noninsulin-mediated mechanisms for numerous hours especially at the time glycogen stores will be replenished explaining that exercise can also be associated with the risk of delayed, mainly nocturnal, hypoglycemia.3,28
Classical Strategies for Managing Hypoglycemia During Exercise
Strategies for minimizing hypoglycemic risk around exercise exist but it remains challenging to apply to a specific type, intensity, duration, timing, etc., exercise session29,30,31: the simplest strategy to apply is CHO supplements,32,33 especially for longer exercise, unanticipated exercise, and/or during postabsorptive periods (greater than or equal to three hours following meals). However, this strategy may not be optimal/desired by patients if weight loss is a goal of the exercise practice. 1 In addition, key aspects have not yet been well studied such as required CHO amount or optimal intake timing, and thus, it is hard to give precise advice to patients. Another strategy is adding high-intensity sprints in intervals or resistance training at beginning or end of exercise. 10 This approach takes advantage of the counter-regulatory hormones, especially the catecholamines that are secreted during high-intensity exercise. Also, we use a premeal bolus insulin-dose reduction4,34 for both multiple daily injections and continuous subcutaneous insulin infusion (CSII), when undertaken in the postprandial period (two to three hours postmeal). Recommendation for premeal bolus reduction is proportional to exercise duration and intensity: larger for longer duration and at higher intensity. 35 It should be noted that this approach can lead to postexercise hyperglycemic trend. For patients using CSII temporary basal rate, insulin reduction is recommended but this needs to be implemented more than 40 minutes before exercise to get a significant effect.3,5,36 For more security, many patients with T1D use sensor-augmented pump (SAP) therapy, which allows insulin delivery to be suspended automatically for up to two hours when the CGM detects a glucose level that has reached a prespecified threshold or, in more recent version, a prediction of an imminent hypoglycemia risk. This technology is the first step along the way to closing the loop of glucose control with the AP systems. However, even when one or combinations of these strategies are used, most patients still have wide exercise-related glucose fluctuations.
Artificial Pancreas Systems During Exercise: Evolution and Challenges
Artificial pancreas systems will automate CSII basal rate delivery by using a control algorithm that receives information from interstitial glucose sensors. It computes the optimal hormones (insulin: AP systems with single-hormone “SHAP” or insulin and glucagon: AP systems with dual-hormone “DHAP”) infusion rate to maintain glucose levels within a predefined target range, thus minimizing hypo- and hyperglycemia episodes 37 (Figure 2).
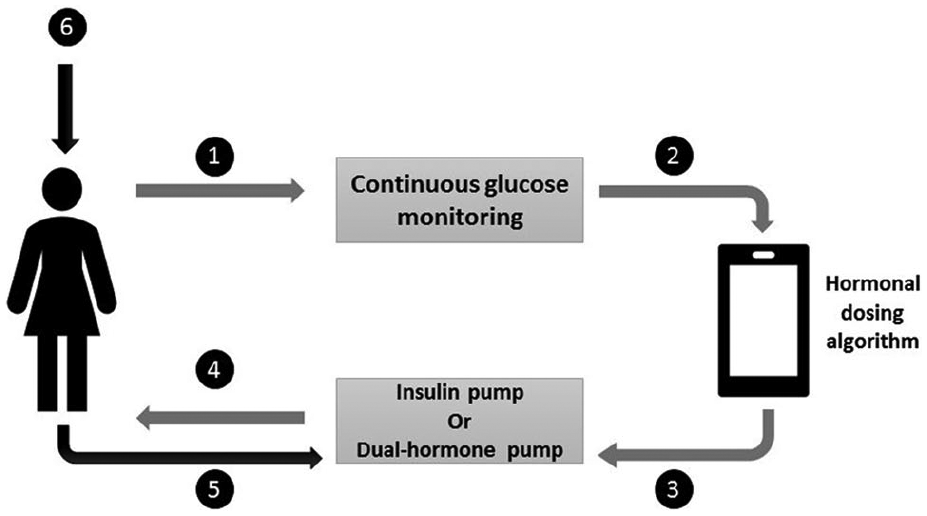
The three components of an artificial pancreas: glucose sensor, control system, and insulin pump.
In recent decades, there has been impressive advancement with AP systems technology to improve diabetes management.38,11 Device developments have progressed to such a degree that AP systems show improvements in time-spent-in-target BG levels compared to CSII systems when tested in small sample groups under free-living conditions. 39 A recent meta-analysis 39 indicated that, compared to SAP, a daily mean three hours time-in-target range improvement with 2.5 hours less in hyperglycemic and 30 minutes less in hypoglycemic range.
Despite these advances, perfecting the system to deal with situations that induce a rapid change in BG (e.g., during exercise and/or postmeal) is still a challenge.21,40-43 Moreover, it is important to note one limitation when reviewing the efficacy of AP studies during exercise. Artificial pancreas algorithms in different clinical trials are based on distinct designs which dictate how the glucose is controlled. The most commonly used ones are based on model predictive control, proportional-integral-derivative control, and fuzzy logic control.44,45 Technical differences among AP algorithms are beyond the scope of this review, but in the future with enough accumulating data collected during exercise with each controller type, clearer comparisons might become possible.
Overview of the AP Systems and Challenges
As detailed above, patients with T1D are faced with an arduous process to achieve optimal glucose control (minimally glucose fluctuations) and this is even more pronounced during exercise. Likewise, the automation of these decisions with an AP system will ideally necessitate timely and accurate information about these factors in association with exercise. 40
The variability in exercise modalities is the most important challenge to creating a system that will be able to adapt to all types and duration of activity for a wide variety of individuals, with a broad range of BG levels at the start of exercise and then response during exercise. The design of a complex sensor that recognizes not only changes in BG but also the start, type, intensity, and end of the exercise would be paramount.39,40
There are additional limitations imposed by the components of AP systems, namely CGM accuracy and rapid insulin analogs pharmacokinetics. (1) Continuous glucose monitoring accuracy is dependent on several factors during exercise such as the physiological delay needed to equilibrate glucose levels between blood and interstitial compartments, intrinsic sensor delays (depending on sensor type) as well as changes in body temperature and blood flow around sensor insertion site.46-48 Continuous glucose monitoring systems have many algorithms to adjust for these factors in addition to filtering signal noises. While CGM accuracy has significantly improved with newer sensor generations, it is not yet optimal. This often translates into lower a mean tendency toward overestimation of actual BG levels when dropping rapidly or underestimation with fast rising levels depending on the type of exercise.14-18 (2) When CGM accuracy is added to the pharmacokinetics of available subcutaneous rapid insulin analogs, a large inertia is created in the system making automated glucose control during exercise extremely challenging: during moderate exercise, the previously injected insulin will continue to be absorbed from subcutaneous depots increasing hypoglycemia risks. On the other hand, when BG levels are increasing rapidly during high intensity exercise, additional insulin boluses or dosing might be needed but face delays to be absorbed and to exert an effect in a timely manner.1,3 Therefore, additional algorithms that can detect and discriminate the type of practiced exercise to predict direction and extent of glucose changes might be needed for optimal functioning of AP systems.49,50 Low-to-moderate intensity exercise is the most frequently practiced type of exercise by patients living with T1D and was thus included in most of the conducted studies testing the efficacy of AP systems during exercise with only few reports tackling high-intensity sessions.
A fully automated AP without additional inputs was tested by Elleri et al. 51 In this study, 12-pediatric subjects used a SHAP for 36 hours that included 40 minutes morning and 20 minutes evening moderate-intensity exercise sessions. Unfortunately, SHAP did not eliminate the risk of exercise-induced hypoglycemia, and five hypoglycemic episodes (<54 mg/dL) were recorded. Comparable results were found in adult individuals with T1D. 52
Thus, with the current rapid acting insulins and glucose sensors, an AP system that is based solely on CGM measurements probably cannot act fast enough to minimize exercise-induced hypoglycemia. 53
To improve AP systems performance around exercise, researchers use four types of approaches: (1) automated exercise detection or manual exercise announcement, (2) dual-hormone closed-loop strategy, (3) CHO supplementation, and (4) combinations of two or more of these approaches (Figure 3).
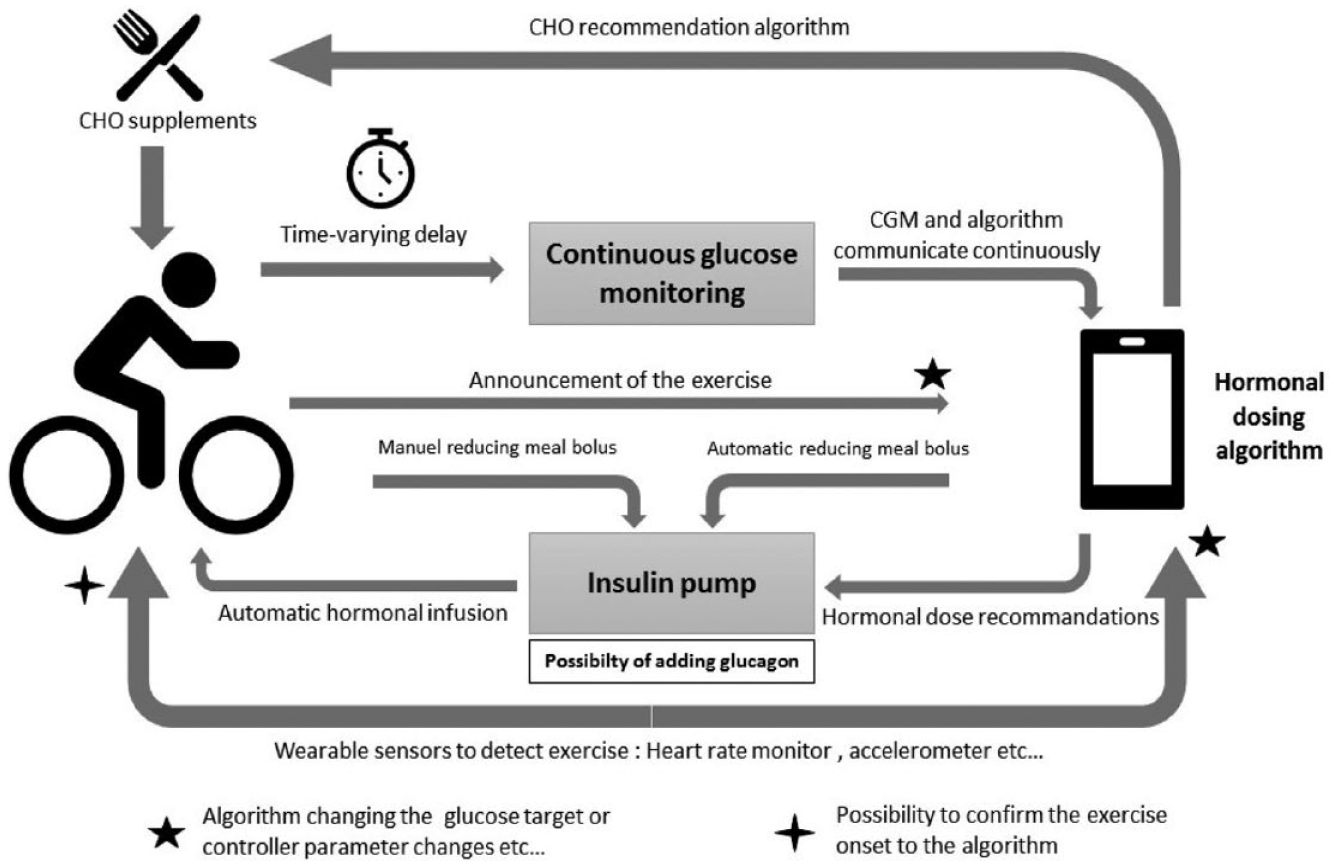
Challenges and solutions related to the use of the artificial pancreas systems during exercise.
Automated Exercise Detection Strategy
Biometric variables such as heart rate, near-body temperature, skin temperature, energy expenditure, heat flux, accelerometry, and galvanic skin response have been shown to correlate with changes in BG during various types of exercise. 49 Clinical outcomes of this study indicated that skin temperature, heart rate, and energy expenditure were the best variables to provide information that could help the AP systems to improve glucose levels during different exercise intensities. 49
In a recent experiment, Turkosy et al 54 challenged their multimodule multivariable (inputs: energy expenditure, galvanic skin response, and the subcutaneous glucose value from the CGM) adaptive SHAP under various types and intensities of exercise (HIIT, high-intensity interval training; MCT, moderate continuous training; and RT, resistance training) without requiring any manual information to be entered by the user. The authors note that, with their AP systems, regardless of the intensity of the exercise, there is a consistent decline in BG in both exercise and recovery period. The percent of time spent in significant hypoglycemia (<55 mg/dL) was low despite the lack of any manual exercise announcement (%time in hypoglycemia: HIIT, 0.0% ± 0.0%; MCT, 1.73% ± 5.03%; and RT, 1.03% ± 4.37%). The main limitation of this study is the absence of a control arm, so the performance of their AP systems cannot be compared with any standard of care (e.g., CSII).
To reduce the burden on patients with T1D, Jacobs et al
55
added a heart rate monitor to their SHAP to detect exercise (45 minutes at 60% maximal heart rate) automatically without requiring any manual information to be entered by the user. When heart rate exceeded 125% of resting levels, this triggered a change in glucose target from 111 to140 mg/dL and subsequent insulin infusion by the AP systems. A decrease in time-spent-in-hypoglycemia (<70 mg/dL) was significantly reduced with heart rate signal integration to SHAP 0.5% ± 2.1% vs 7.4% ± 12.5% (
The use of exercise detection that will use additional AP system inputs may be beneficial in the anticipation of hypoglycemia risk. However, any AP systems that are developed using current CGM and CSII methods will be limited by slow pharmacokinetics of insulin and lagging CGM glucose response when BG declines rapidly. Thus, turning off/reducing insulin infusion upon sensing the start of exercise may be insufficient to completely prevent hypoglycemia, 5 except maybe if exercise is performed shortly after a meal. 56
Dual-hormone (Insulin and Glucagon) AP Systems Strategy
The addition of glucagon to AP systems can be used in two ways: (1) preventive approach where glucagon mini-boluses are recommended when total insulin infusion suspension is not enough, aiming to prevent hypoglycemia and (2) the AP systems recommend more important/aggressive insulin rates aiming for lower mean BG with occasional glucagon delivery to avoid hypoglycemia both in the case of insufficient insulin infusion suspension as well as accidental excessive insulin delivery. This approach was used to reduce hypoglycemic risk both during exercise as well as delayed risk during the subsequent night. 57
We conducted a randomized crossover trial to compare SHAP, DHAP, and the CSII during 24 hours including exercise session (60 minutes at 60% VO2max) in adults. During the exercise session, hypoglycemia (<60 mg/dL) was common with CSII treatment visits (
Also, our team has compared DHAP against CSII in 15 adults with T1D during an overnight period following an evening exercise session (30 minutes at 60% VO2max). We have shown that DHAP significantly reduced hypoglycemic risk by up to eightfolds (20-fold reduction overnight) while increasing the percentage of glucose-in-target time by 16% (equivalent to 3.8 hours/day). 59
Glucagon addition may provide a solution to prevent exercise-induced hypoglycemia. However, the requirement of a pump allowing to separate insulin and glucagon infusion, leading to increased costs and complexity of such devices, in addition to required long-term safety assessment of chronic glucagon use is a significant barrier.60,61 The balance between benefits and inconvenient (costs and risks) thus remains to be established.
Carbohydrate Supplementation Strategy
The proactive snacking strategy in combination with SHAP would provide a simple and effective means to decrease exercise-induced hypoglycemia in active individuals with T1D.
In 12 patients with T1D, Patel et al
60
demonstrated that a simple snacking intervention (15 and 30 g CHO for BG >150 and <150 mg/dL, respectively) without announcing exercise could prevent drops in BG around exercise. Without snacking, BG levels decreased by 53±10 mg/dL vs modest increase by 10±13 mg/dL in the snacking strategy (
Over three days, AP experiments including two sessions (aerobic, resistance, or interval exercise) tested on six subjects, SHAP suggested the consumption of CHO depending on both glucose level and trend. 62 There were only five hypoglycemia episodes with an average of three hypoglycemia alarms per day per subject. The average CHO consumption was 14±7.8 g per alarm. Around exercise session, CHO amounts are suggested at higher glucose level (80 mg/dL). The authors demonstrated that most hypoglycemia can be predicted, and that low glucose levels can be raised back to the desired levels by consuming an appropriate CHO amount.
It is important to note that snack consumption may be counter-productive given the increased prevalence of the metabolic syndrome in patients with T1D who frequently practice exercise for weight loss or maintenance objectives.1,3,63
Artificial Pancreas Systems With Combination of Several Strategies
Many studies have attempted to improve the efficiency of the AP systems by combining multiple strategies (glucagon addition, exercise detection, and exercise announcement).
Jacobs et al tested DHAP and exercise announcement at the start of a 45-minute exercise at 60% of their maximum heart rate.
64
In random order, 21 adults with T1D experienced three sessions: (1) DHAP with exercise announcement; (2) DHAP without exercise announcement; and (3) SAP. Time spent in hypoglycemia during DHAP + announcement was reduced by 2.8% vs unannounced exercise strategy (
Our group compared the efficacy SHAP vs DHAP during two types of exercise matched for energy expenditure (continuous exercise vs interval exercise).
65
Exercise was announced 20 minutes prior to its start, and this led to the change of the target glucose level from 95 to 150 mg/dL until the end of the exercise session. Lower doses of glucagon were needed during interval exercise (0.093 ± 0.068 mg) than during continuous exercise (0.126 ± 0.057 mg) (
Another study was performed in 20 patients with T1D comparing DHAP vs SHAP vs SAP vs usual care for 16 days (four days each arm including 45 minutes at 60% VO2max).
66
Both AP systems had an integrated algorithm for exercise detection that received input from heart rate monitor and accelerometer. Mean time in hypoglycemia was the lowest with DHAP during the exercise period: 3.4% ± 4.5% vs 8.3% ± 12.6% for SHAP (
Conclusion
Studies designed to specifically evaluate the use of AP systems during exercise are limited, but many studies have included periods of structured exercise as part of their experiment protocols that extend to 24 hours or more (summarized in Table 1). AP systems showed better performance than CSII or SAP in many exercise conditions, though complete hypoglycemia prevention is not yet achieved. The major hindrance is related to delays in insulin absorption and interstitial glucose sensing probably making exercise announcement necessary at this stage. Adding biometric variables to detect and classify the type, intensity, and energy expenditure of physical activities can also improve AP systems performance. Yet, the number of wearable devices used should be kept to a minimum and/or integrated to insulin pumps, glucose sensor, or AP controller. Glucagon addition is also a promising avenue, but stable formulations and long-term safety assessment of chronic glucagon use should be first achieved. Ideally, future AP systems, especially with the advances in deep learning artificial intelligence efforts, should be able to predict the effects of specific physical activities (type, intensity, and duration) and changing variables during group sports, in addition to detecting and discriminating exercise-concurrent activities such as meals, sleep, and stress.
Summaries of Main Clinical Trials on Artificial Pancreas Efficiency During Physical Activity in Type 1 Diabetes.
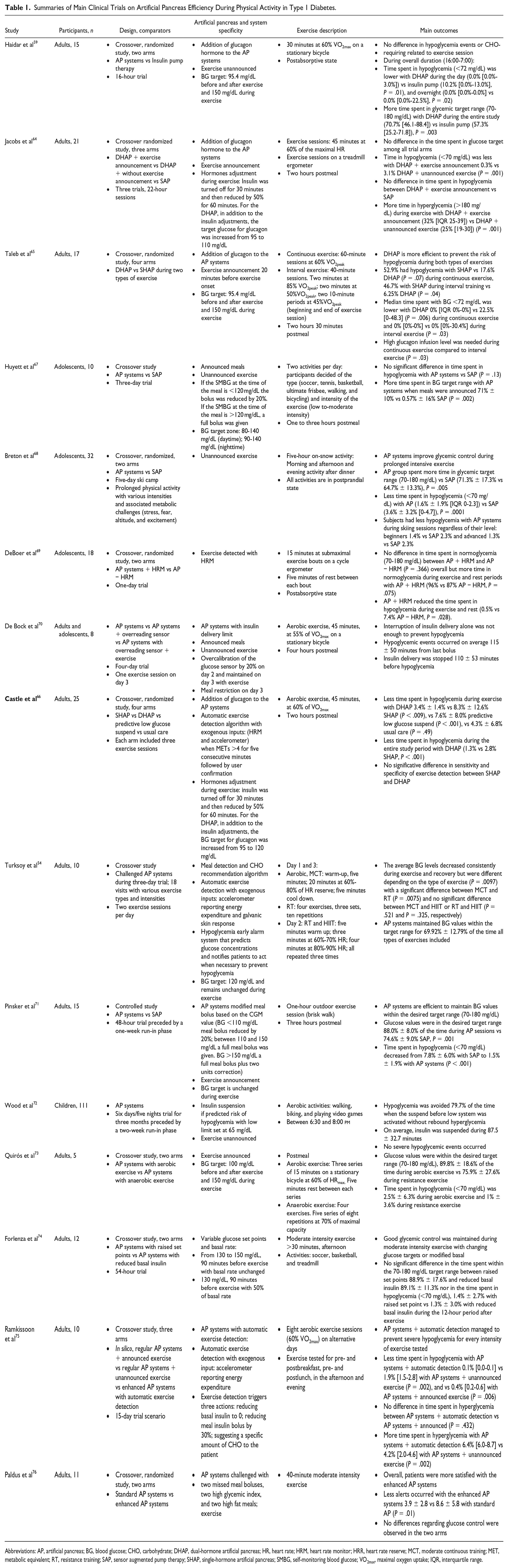
Abbreviations: AP, artificial pancreas; BG, blood glucose; CHO, carbohydrate; DHAP, dual-hormone artificial pancreas; HR, heart rate; HRM, heart rate monitor; HRR, heart rate reserve; MCT, moderate continuous training; MET, metabolic equivalent; RT, resistance training; SAP, sensor augmented pump therapy; SHAP, single-hormone artificial pancreas; SMBG, self-monitoring blood glucose; VO2max, maximal oxygen uptake; IQR, interquartile range.
Footnotes
Acknowledgements
NT is the recipient of scholarship of CIHR (Canadian Institutes of Health Research) and FRSQ (Fonds de Recherche Santé Québec) scholarships. RRL is holding the J-A DeSève diabetes research chair and a Diabetes Canada program grant (DI-5-17-5282-RR) that both supported this work.
Authors’ Contributions
ST, NT, and RRL conceived the literature review design and content. ST drafted the manuscript which was critically reviewed by NT, JM, EN, MR, and RRL. All authors revised and agreed upon the final submitted manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ST NT, JM, EN, and MR have no conflicts of interest to declare. RRL has the following to declare: Research grants from Astra-Zeneca, E Lilly, Merck, NIH, Novo-Nordisk, Sanofi-Aventis; Consulting/advisory panel with Abbott, Amgen, Astra-Zeneca, Boehringer, Carlina Technology, Eli Lilly, Janssen, Medtronic, Merck, Neomed, Novo-Nordisk, Roche, Sanofi-Aventis; Honoraria for conferences received from Abbott, Astra-Zeneca, Boehringer, E Lilly, Janssen, Medtronic, Merck, Novo-Nordisk, Sanofi-Aventis; consumable gift (in Kind) from Abbott, Animas, Medtronic, Roche; unrestricted grants for clinical and educational activities from Eli Lilly, Lifescan, Medtronic, Merck, Novo Nordisk, Sanofi; patent for T2DM risk biomarkers, catheter life & artificial pancreas; Purchase fees related to artificial pancreas from Eli Lilly.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Diabetes Canada program grant (DI-5-17-5282-RR). ST is supported by a fellowship grant from SFD (Société Francophone du Diabète) and FRM (Fondation de Recherche Médicale).
