Abstract
Background:
Among adults with diabetes, 19-34% will develop a diabetic foot ulcer (DFU), which increases amputation risk and health care costs, and worsens quality of life. Regular physical activity, when increased gradually, may help prevent DFUs. In this mixed-methods study, we examined the feasibility of a low-intensity, technology-based behavioral intervention to increase activity in adults at risk for DFUs.
Method:
Participants at risk for a DFU (n = 12; 66% female; mean age = 59.9 years) received four in-person exercise and behavioral counseling sessions over 2-3 weeks, supplemented with use of an activity monitor (to track steps) and text messages (to reinforce behavioral strategies) for an added 8 weeks. Pre- and postintervention assessments of accelerometer measured activity, daily mobility, and glycemic control (A1C) were completed. Treatment acceptability was assessed by questionnaire and via key informant interview.
Results:
The program appears feasible since all but one participant attended all four sessions, all used the activity monitor and all responded to text messages. Treatment acceptability (scale: 1 = very dissatisfied, 5 = extremely satisfied) was high; average item ratings were 4.79 (SD = 0.24). Participants increased their steps by an average of 881.89 steps/day (d = 0.66). A1C decreased on average by 0.33% (d = 0.23). Daily mobility did not change. Interview results suggest that participants perceived benefits from the intervention. Participant recommended improvements included providing more physical activity information, addressing pain, and intervention delivery in a podiatry clinic.
Conclusion:
Individuals at risk for a DFU might benefit from a minimally intensive, technology-based intervention to increase their physical activity. Future research comparing the intervention to usual care is warranted.
The World Health Organization reports that the prevalence of diabetes worldwide increased from 180 million in 1980, to 422 million in 2014. 1 Worldwide, the estimated economic burden due to diabetes is estimated to increase from $1.3 trillion in 2015 to $2.1-2.5 trillion by 2030 (32830 Bommer C. 2018) 2 . The management of diabetic foot ulcers (DFUs) significantly contributes to health care costs. In one year, adults with DFUs incur $9-13 billion in health care costs in excess of typical diabetes costs. 3 DFUs substantially increase risk for amputations, which are a major contributor to disability,4,5 cardiovascular disease 6 and mortality. 7 DFUs are also concerning to patients with diabetes. 8
Inadequate glycemic control increases DFU risk via peripheral neuropathy and/or peripheral artery disease. 9 Sensation loss, foot abnormalities and decreased skin integrity, in tandem with repetitive trauma produced by weight bearing activity, abnormal plantar pressures, and skin breakdowns contributes to DFU risk in individuals with peripheral neuropathy.9,10 One might assume that physical activity would increase DFU risk. Historically, care providers were advised that weight-bearing exercise may impose serious risks and that individuals with severe peripheral neuropathy should be encouraged to engage in non-weight-bearing activities.11-13 However, studies suggest that individuals who ulcerate have lower activity levels, and exhibit greater activity variability.14-16 For adults at risk for DFUs, gradually increasing activity may not increase their DFU risk, with the potential benefits of improving diabetes management 17 and reducing cardiovascular disease risk 18 and mortality.18,19 Accordingly, the American Diabetes Association’s stance regarding engagement in weight-bearing activities by individuals at risk for DFU has evolved. A 2010 joint position statement with the American College of Sports Medicine indicates that moderate walking does not increase risk of foot ulcers 20 and a 2016 American Diabetes Association position statement indicates individuals with peripheral neuropathy should engage in lower-body strengthening exercises to improve and maintain balance. 21
Supervised physical activity programs varying in frequency (1-6 visits/week) and duration (8-24 weeks) safely increased activity in adults at risk for DFUs.14,22-24 Supervised programs may promote activity initiation, but behavior change strategies may be necessary to promote maintenance. Lemaster and colleagues 14 incorporated behavioral strategies into telephone counseling following 8 supervised sessions. While overall steps did not differ between the intervention and a control, exercise bouts were greater in the intervention at the 6- and 12-month follow-ups. 14 Behavioral strategies were based on social cognitive theory, which proposes that the interplay of personal, behavioral and environmental determinants influence health behaviors like physical activity. 25
Self-determination theory addresses a weakness of social cognitive theory: little attention to types of motivation.26-28 Self-determination theory posits that motivation varies on a spectrum from intrinsic (ie, motivation that is internalized) to extrinsic (ie, motivation that is external to the individual). 29 Self-determination theory based strategies encourage ways to enhance intrinsic motivation for physical activity, and targeted use of extrinsic motivators.
While supervised programs could incorporate behavioral strategies into visits, intensive programs can be burdensome and costly, and typically not reimbursable. 30 Leveraging technology, such as activity monitors (ie, Fitbit®), text messaging, and global positioning systems (GPS), to increase physical activity, merit investigation. Using technology to deliver and reinforce behavioral strategies might be critical to implementing less intensive interventions. A recent review concluded that research is needed to examined the feasibility and efficacy of technologies to promote activity in people with diabetes. 31
This pilot examined the feasibility of a minimally intensive intervention that included supervised exercise, behavioral strategies (grounded in social cognitive and self-determination theory), and technology, to increase physical activity in adults at risk for DFUs. Feasibility was assessed via intervention component use, adverse events, self-reported intervention acceptability, retention rate, and improvements in steps, glycemic control, and daily mobility. Key informant interviews were conducted to further assess intervention acceptability.
Methods
Sedentary adults at risk for a DFU were recruited. Inclusion criteria included (1) ≥21 years of age; (2) diagnosis of diabetes; (3) peripheral neuropathy, defined as loss of protective sensation as identified by failure to detect a 10 gram Semms Weistein Monofilament on either foot at one of four sites tested 32 or a vibration perception threshold value of 25V or more at either foot’s hallux; 33 (4) glycated hemoglobin (A1C) >6.5% and <12%; (5) ability to speak and read English; (6) physician approval; and (7) internet access. Exclusion criteria included (1) self-reported >2 bouts of 20+ minutes of physical activity/week; (2) current foot ulcer; (3) proliferative retinopathy; (4) pregnant or planning a pregnancy; (5) inability to engage in activity without assistance; 34 and (6) significant medical illness, such as severe peripheral vascular disease defined as ankle brachial index <0.6 or cardiovascular autonomic neuropathy as evidenced by either resting heart rate above 100 bpm or orthostatic hypotension.35,36
Procedures
Participants were recruited via outpatient clinics, diabetes support groups, and community flyers. Study personnel conducted prescreenings to assess initial eligibility criteria, and then scheduled individuals for an in-person screening session.
Participants attended a two hour screening where they provided written informed consent and completed eligibility procedures. Participants were screened for peripheral neuropathy, vascular compromise and orthostatic hypotension. Foam impressions of the feet and barefoot first-step pedobarographic (EMED-X, Novel GmbH, Munich, Germany) measurements were taken for fabricating diabetic orthotics (TrueContour Therapeutic Insoles, Diapedia). 37 Participants completed a medical history questionnaire and the Revised Physical Activity Readiness Questionnaire 38 to assess safety for exercise. Height and weight were measured to calculate body mass index (BMI). Blood was drawn for assessing HbA1c. Participants received a GPS monitor (QStarz BT-Q1000XT) and tri-axial accelerometer (PAMSys™) to measure location specific physical activity for one week. Physician approval was obtained. Participants received $50 for completing the screening.
At least one week after screening, participants attended a 1 hour baseline session to receive orthotics, shoes, and a digital infrared thermometer to identify preulcerative inflammation.39-41 Participants were asked to complete temperature monitoring at six sites on each foot every morning and advised to reduce their activity and contact the study nurse, if the temperature difference between two corresponding sites on the left and right feet exceeded 4°F on consecutive days. Participants also received a Fitbit Zip and training in its use. They joined a private Fitbit group so that researchers could track their steps. Participants received $50 for completing the baseline.
Intervention
Supervised Exercise Sessions
Participants attended four 45-minute exercise sessions in the lab, over 2-3 weeks. Four sessions were selected to provide adequate time to create and implement a plan for gradually increasing activity. Sessions consisted of an exercise bout, modeled after studies that used supervised exercise with participants at risk for DFUs,14,22 and behavioral strategies instruction. A research associate, supervised by a physical therapist, conducted the exercise bout. Participants had their blood pressure and blood glucose monitored before exercising. After warming up, participants then engaged in moderate intensity treadmill walking (40-70% of heart rate reserve), followed by a cool-down period. The first exercise bout duration was based on the participant’s steps from the previous week. After eliminating outliers (days with ±2 times the week’s average), the duration of treadmill walking was calculated to equate approximately 0.5 of their daily step count. The second exercise session bout was the same duration as session one. Exercise bouts for sessions three and four were one minute longer to ensure gradual increases in activity. Foot temperatures were taken before and after the bout to reinforce temperature monitoring and assess for inflammation.
Participants then met with a clinical psychologist who introduced the behavioral strategies based on social cognitive theory and self-determination theory, and modeled after content used in the Diabetes Prevention Program (Table 1). 42 Between sessions, research assistants monitored participants’ steps and provided physical activity encouragement and reinforcement for behavioral strategies via text messages.
Overview of Supervised Exercise Sessions.
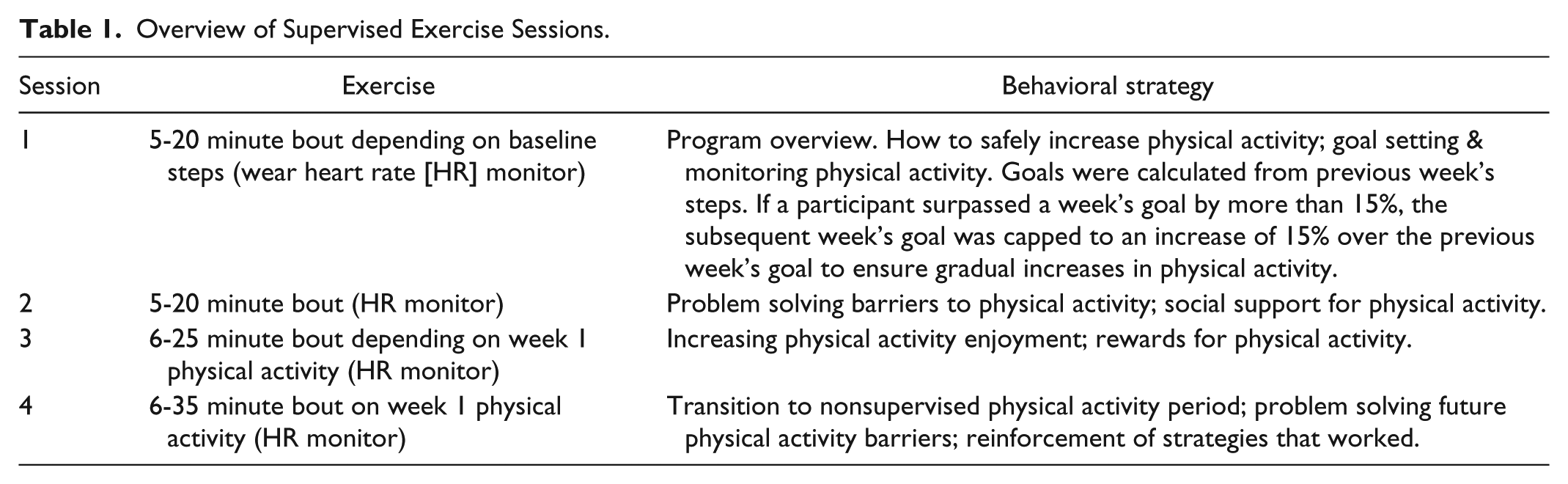
Remote Support Period
After the fourth session, participants began an 8-week period where they were asked to continue increasing their physical activity and were monitored remotely via the Fitbit. Participants received at least two tailored text messages/week to encourage activity and behavioral strategy use, and to problem solve barriers. Participants could access the private Fitbit social network for support. Research assistants posted strategies daily on the social network.
After the remote support period, participants completed assessments again and a key informant interview during a 1-hour session. At least one investigator conducted the key informant interview and inquired about participants’ intervention experience and use of GPS (see the appendix for the semistructured interview). Interview length ranged from 16 to 61 minutes (mean = 31.5, SD = 12.2). Interviews were audio-recorded and then transcribed. Participants completed a treatment acceptability measure and received $50 for completing the postintervention session. Procedures were approved by the institutional review boards of Rosalind Franklin University of Medicine and Science and DePaul University.
Measures
Demographics
Participants reported demographics (eg, gender, age, race/ethnicity) at the screening session.
Blood Draw
A phlebotomist drew 7ml of blood, which was sent to Quest Diagnostics Laboratory for A1C analysis.
Physical Activity Monitoring
Participants wore a pendant style tri-axial accelerometer (PAMSys, BIOSENSICS) for 1 week. This accelerometer provides an accurate assessment of total steps.43-45 Percentage of wear time was calculated; participants wore the monitor for 97% (SD = 3.0) of the time during baseline and 99% (SD = 1.0) during postintervention. On average, participants had 6.4 (SD = 1.2) days of valid data.
Daily mobility
Daily mobility was derived using an algorithm that partitions raw GPS trajectory data into meaningful segments such as stops and trips, which results in the number of places visited/day. 46 GPS data were combined with accelerometer data to assess physical activity locations. During the key informant interview, participants viewed maps in Google Earth© that highlighted the participant’s physical activity and nearby activity resources (eg, parks) to inquire about using GPS in an intervention (Figure 1).
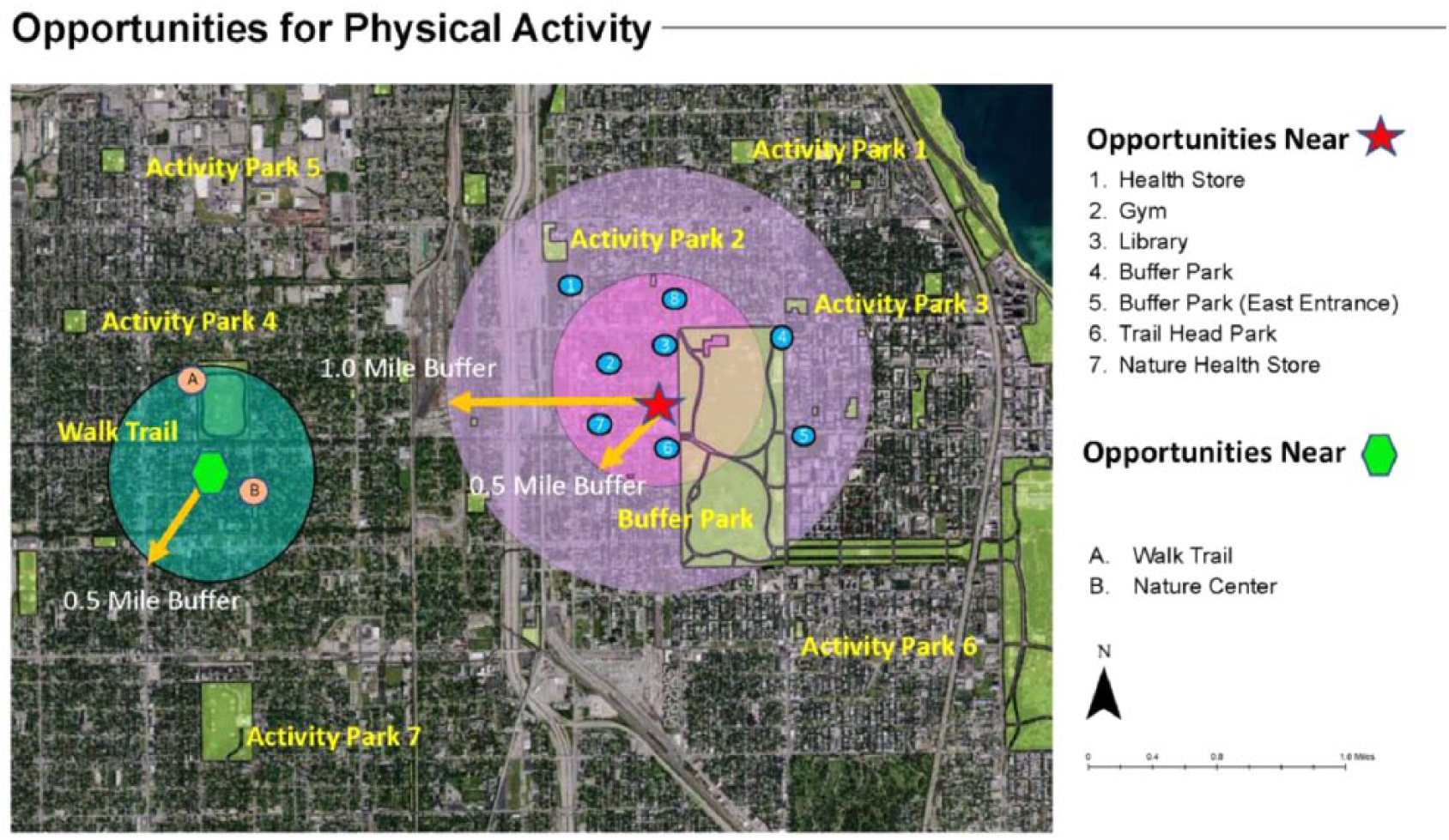
Personalized community resources map for physical activity opportunities. The star and hexagon reflect two commonly visited locations.
Feasibility
Retention rate was compared to studies that used supervised exercise for adults at risk for a DFU.14,22-24,47,48 Percentage attendance to supervised exercise sessions was calculated and compared to studies that reported attendance.14,22,47,48 Treatment acceptability was captured using a modified version of the diabetes measurement and evaluation tool 49 that assessed participants’ satisfaction with the intervention on a scale from 1 = very dissatisfied to 5 = very satisfied (coefficient alpha = 0.91). Adverse events were categorized as “unsure if related,” “unrelated,” “possibly related,” or “definitely related.”
Analytic Plan
Descriptive statistics were used to examine treatment acceptability, supervised exercise sessions attendance, retention, and adverse events. Linear mixed models were used to estimate the effect of the intervention on steps, glycemic control, and mobility. Covariates considered for inclusion in the analyses included age, gender, BMI, and the month the participant began the intervention (to control for seasonal variation in activity). Models included a random intercept due to participant variability at baseline in outcomes. Model testing began with all covariates and use of an identity covariance structure. Fit indices (ie, –2 restricted log likelihood, Akaike’s information criterion, and Schwarz’s Bayesian criterion) were compared between models that removed covariates and used other covariance structures to find the best fitting model. This pilot examined intervention feasibility to inform intervention development; thus, no formal power analysis was conducted. Since the study was underpowered, effect sizes were calculated from estimated marginal means.
Transcripts were imported into NVivo; a descriptive thematic analysis was used to analyze key informant interview data. 50 The lead author read all transcripts to create a coding structure focused on identifying semantic content in themes, rather than interpretation. Two transcripts were randomly selected and coded independently by the lead author and two coauthors. After reviewing the coding, the coding structure was revised slightly to distinguish between temperature monitoring accountability versus physical activity accountability and additional detail was added to improve coding reliability. Three transcripts were coded independently, followed by coding review meetings. Remaining transcripts were divided amongst coding pairs. Coders achieved 89.25% agreement on average. 51
The lead author reviewed the codes and organized them by themes in three areas: (1) intervention benefits; (2) intervention improvements; and (3) potential usefulness of GPS. At least 4 participants (36%) had to discuss content for it to be considered a theme. The other coders reviewed themes to ensure consistency; no changes were made and no new themes emerged. Quotes and the percentage of participants who contributed content in support of the theme provided evidence of theme validity. Participant ID (PID) numbers are listed after quotes.
Results
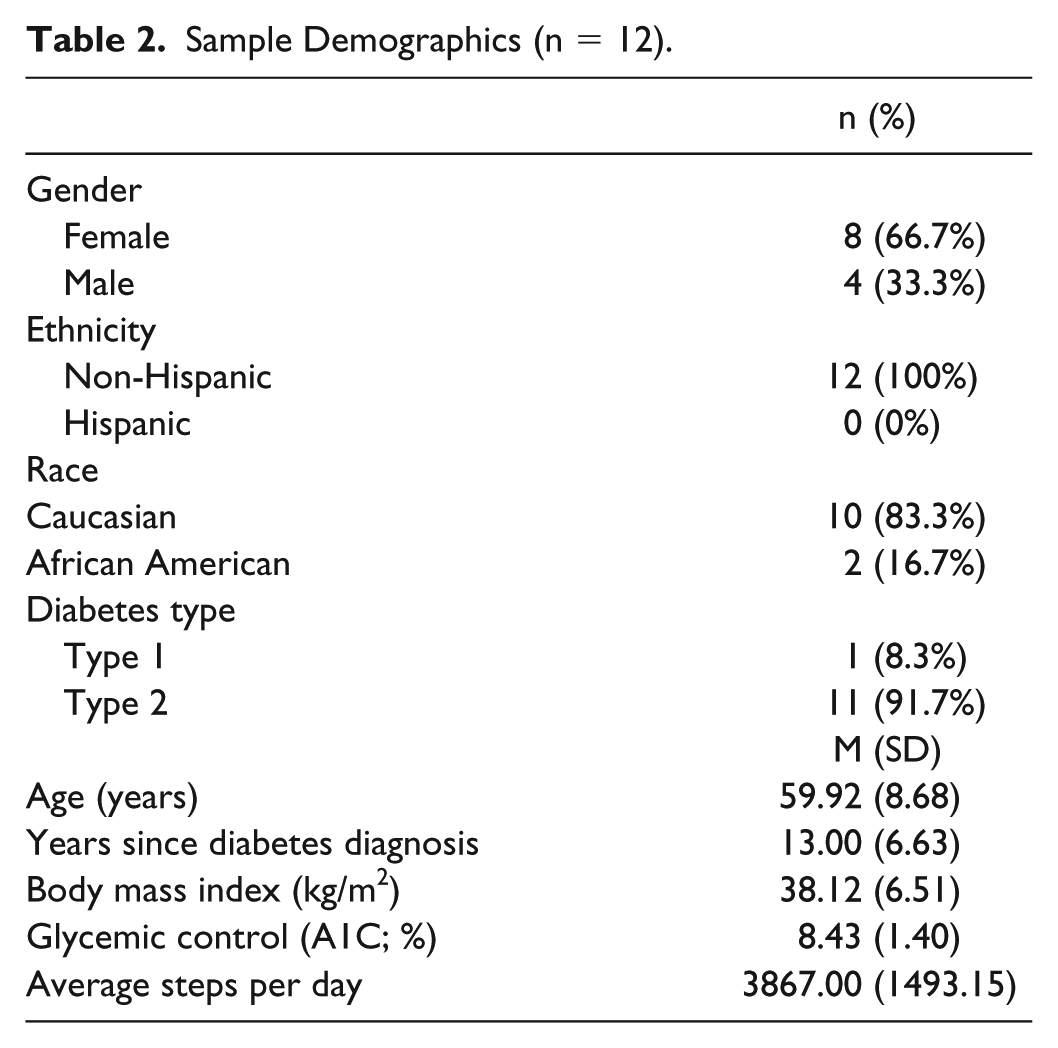
Participants (n = 12) were on average 59.92 years of age (SD = 8.68) and had diabetes for an average of 13.00 years (SD = 6.63; Table 2). Of the participants, 11 (91.67%) completed the postintervention assessment, which is comparable to other physical activity studies in adults at risk for DFUs (mean = 87.8%; range 75.9-100%). All but one participant attended all supervised exercise sessions, which resulted in an average attendance rate of 97.9%. Percentage attendance was greater than other studies (mean = 81.9%; range = 67.8- 95%), likely due to the lower number of sessions. On average, participants uploaded Fitbit data for 7.67 weeks of the intervention; all responded to text messages.
Sample Demographics (n = 12).
Treatment acceptability was high (mean = 4.79, SD = 0.24; Table 3). The lowest rated item was satisfaction with session location (mean = 4.18, SD = 1.08). One participant developed an adverse event, a University of Texas grade 1B DFU, 52 which was deemed probably related to study participation; it resolved, and the participant resumed activity. Six other adverse events were deemed unrelated to study participation.
Mean Treatment Acceptability Ratings (n = 11).
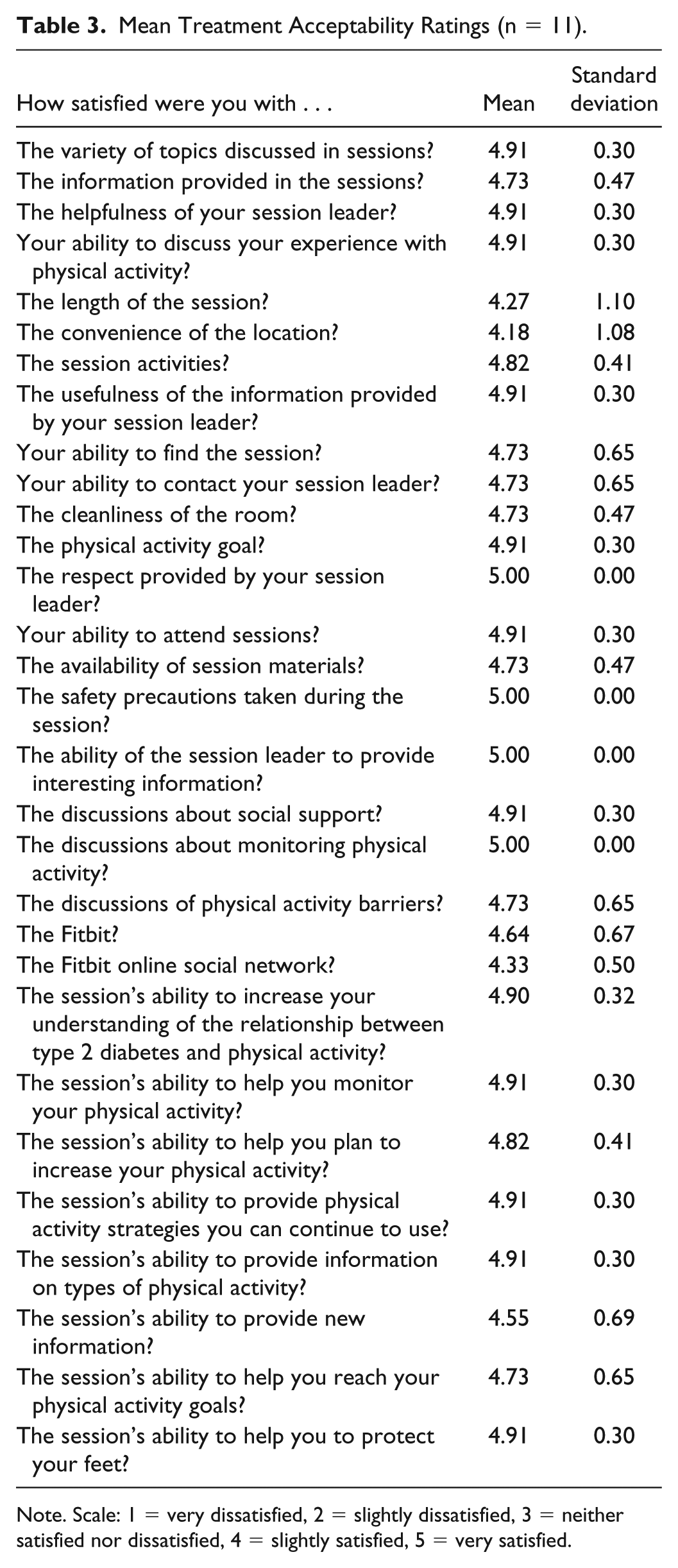
Note. Scale: 1 = very dissatisfied, 2 = slightly dissatisfied, 3 = neither satisfied nor dissatisfied, 4 = slightly satisfied, 5 = very satisfied.
For steps and glycemic control, the best fitting model included the covariates age, gender, BMI and the month participants began the intervention, and a diagonal covariance structure. Participants’ daily steps increased from an average of 3825.31 steps/day (SD = 1503.84) to 4707.2 steps/day (SD = 1151.63; d = 0.66). Participants’ average glycemic control improved from 8.47% (SD = 1.34) to 8.14% (SD = 1.54; d = 0.23). The best fitting model for mobility included gender and BMI and a compound symmetry covariance structure; a square root transformation was used because mobility was not normally distributed. Mobility did not change (baseline: mean = 1.63; SD = 1.28; postintervention: mean = 1.63, SD = 1.29).
Table 4 includes key informant interview themes and quotes, which are distinct from the quotes below.
Key Informant Interview Results: Themes and Participant Quotes.

Intervention Benefits
Intervention provided ways to help increase physical activity
All participants (100%) described useful intervention features for increasing activity. Participants (5; 45%) noted that the supervised exercise sessions were helpful for learning skills and for walking. One participant commented, “You helped me behave a lot more just by the whole mind change thing . . . you gave me more like a positive instead of like a negative. Like you told me to reward myself, which I never thought of” (PID22). Nine participants (81%) discussed the benefits of goals and tracking physical activity and 9 (81%) identified accountability and encouragement provided by the text messages or the Fitbit. A participant stated, “[Text messages] kept me connected, even though I wasn’t there. . . . If I had any questions, I could always ask” (PID14). One benefit that 4 (36%) participants identified was how physical activity helped them feel better. One participant commented, “I feel better, it’s the initial getting me to go is kinda like, ‘okay, really? I gotta go walk . . .’ but you know, once I get out there, I feel good, it relieves stress and you know it’s just all around helpful to be active, which I didn’t realize that before” (PID 22).
The shoes and/or insoles were beneficial
Most participants (90%) liked the shoes and/or insoles, which supported activity. A participant stated, “I’m in heaven walking. I could walk 10 miles with those things on” (PID8). Seven participants (63%) noted that it took time to achieve a comfortable fit. One participant commented, “The orthotics that you made at first I had a little problem because . . . one of them was just a little uncomfortable, but after using them for a while . . . I did better with them” (PID2).
Accountability provided by temperature monitoring
Nine participants (81%) discussed how the temperature monitoring helped them take care of their feet. One participant stated, “The temperature monitor . . . I think it’s necessary because if it wasn’t there, I wouldn’t know” (PID11).
Intervention Improvements
Resolve technology problems
Most problems related to the Fitbit. Five participants (45%) described problems syncing their Fitbit and four (36%) mentioned that it did not always register activity. One participant noted, “But I did watch my active minutes, and that’s when I saw when I was folk dancing it didn’t register active minutes” (PID1). Participants (4, 36%) also described problems with the Fitbit battery.
More information on physical activity
Four participants (36%) described wanting additional physical activity information. Participants desired information on the amount of physical activity required before starting the program and more information on where and how to be active.
Address typical physical activity barriers
Seven participants (63%) stated that pain is a major barrier to physical activity. One participant commented, “Get rid of my pain, then I could do physical activity I wanted” (PID14). Eight participants (72%) described weather as a barrier.
Connection to a health care provider
Participant comments (9; 81%) reflected the importance of their relationship with a provider. One participant commented, “I trust her. . . . She always recommends me for something that was good for me. And I take Dr. [name removed] recommendations very seriously” (PID 1).
Potential Usefulness of GPS
GPS would be helpful
Most participants (10, 90%) reported no concerns about the GPS. They identified two ways in which GPS could be used in a physical activity intervention. Six participants (54%) described how GPS could identify physical activity opportunities. One participant commented, “If we had additional information about some of the places of where it would be good for walking . . . and if you had actually a map of how to walk indoors . . . that would be useful” (PID18). Participants (7, 63%) described how GPS could be used to prevent overexertion and/or DFUs. A participant described, “My nephew’s wife invited us to this bike ride . . . and I was almost gonna go to it, but she didn’t realize there was a lot of uphill. . . . I have to be careful, so I do, that would be helpful, yea, [to receive information on] the level of the terrain” (PID1).
GPS would not be helpful
Nine participants (81%) described reasons GPS would not be useful. Six participants (54%) described how GPS would not help them identify physical activity opportunities because they are familiar with their area.
Discussion
Adults at risk for a DFU might benefit from a less intensive, technology-based, behavioral intervention to increase physical activity. Gradual increases in activity appeared to increase physical activity and improve glycemic control. The average increase in steps was about 300 steps lower than what was observed in a 12-week supervised exercise program, 22 and the average improvement in glycemic control was 0.2% lower than what was demonstrated in two other supervised exercise programs.24,47 Nonetheless, the benefits observed using a less intensive intervention are noteworthy, especially since other supervised exercise programs have not observed improvements in steps 14 or glycemic control.48,53
Delivering the intervention in the setting where patients at risk for DFUs receive foot monitoring and care could enhance dissemination. Connection to health care providers was a common theme during the key informant interviews, and intervention location was the lowest rated treatment acceptability item. Individuals at risk for DFUs attend podiatry visits in the US at least semiannually; 54 though accessibility to podiatrists varies in other countries. While fall prevention physical activity interventions have been implemented in podiatry clinics (eg, Spink et al), 55 no activity interventions for adults at risk for DFUs have occurred in podiatry clinics. If efficacious, an intervention delivered in clinics that treat patients at risk for DFUs has high sustainability potential, which is critical for the development of cost-effective interventions. 56
One participant in the present study developed a DFU, which is not unexpected since 19-34% of adults with diabetes will develop a DFU and 65% of patients with a healed DFU will reulcerate within 5 years of healing. 57 The heightened risk of ulceration in this population may explain why physical activity studies in adults with diabetes often exclude adults at risk for a DFU, which further highlights the need for developing tailored activity interventions that directly address a patient’s ulceration risk. Participants endorsed benefit from two synergistic DFU prevention measures: the custom-made orthotics and shoes, and the temperature monitoring device. Specialty shoes and orthotics are recommended for reducing DFU risk in adults with peripheral neuropathy. 58 Since some participants required adjustment of their orthotics, housing the intervention in a clinic where patients receive podiatric care would help streamline the fit process. Temperature monitoring devices are affordable, though more research is needed to support the benefits of temperature monitoring. Emerging research on the use of wearable sensors assessing plantar pressures may provide another avenue for remotely monitoring patients.59,60 Including intervention components that routinely and easily assess DFU risk may be critical to include in physical activity interventions to assuage provider and patient concerns, and prevent a DFU.
Participants suggested intervention improvements that could be tailored to patient need. Pain was identified as a barrier to physical activity. While physical activity may reduce pain in participants at risk for a DFU,22,23 adding pain intervention strategies might be necessary to encourage activity. 61 Participants desired more information about exercise. Expanding aerobic exercise content and providing resistance training may be beneficial since both are recommended for diabetes management 20 and greater variety in exercise could promote intrinsic physical activity motivation.62,63
Most participants identified reasons why GPS derived recommendations would and would not be useful for encouraging activity and preventing DFUs. As highlighted in social cognitive theory, the environment influences health behaviors like physical activity, and strategies for targeting the environment are recommended for increasing physical activity. 64 Despite walking more, the number of places participants visited did not increase. Providing tailored community physical activity resources might have encouraged participants to explore other activity options, which could further bolster intrinsic motivation for physical activity. Using GPS for identifying tailored community, physical activity resources demonstrates promise among adolescents,65,66 but evidence in adults is limited. One of the advantages of using GPS to promote physical activity in adults at risk for DFUs, is the potential for providing real-time intervention to support physical activity increases, while monitoring situations when increased activity could trigger a DFU. Pilot work that provides adults at risk for a DFU with hands on experience of how GPS could assist with safely increasing their physical activity is warranted.
Study strengths include the collection of objective physical activity data and the use of quantitative and qualitative data to understand intervention benefits and challenges. While intervention adherence and follow-up retention were high, the small sample size and lack of a control condition tempers the conclusion that the intervention increased physical activity and improved glycemic control. The use of rolling recruitment resulted in having few participants active at the intervention at the same time, which limited the extent to which participants could receive support from others via the private social network. The mostly white sample limits generalizability to a diverse population. We incorporated behavioral strategies based on self-determination theory to support physical activity maintenance, but the lack of follow-up beyond the postintervention assessment impedes our understanding of whether intervention-related changes were maintained.
Conclusion
Regular physical activity is critical to glycemic control 17 and to risk reductions in cardiovascular disease and mortality.18,19 While supervised physical activity programs have increased physical activity in adults at risk for DFUs, they may not be cost-effective and could be burdensome. 30 A less intensive physical activity intervention that incorporates short-term, supervised exercise sessions, behavioral strategies, wearable devices, and text-messaging appears feasible. Research examining whether GPS might be useful in promoting physical activity, and whether delivery in clinics that routinely monitor patients’ foot health could improve reach and sustainability of the intervention, are needed. A randomized controlled trial that includes a comparison condition like usual care, could provide efficacy data for this multicomponent intervention. A multiphase optimization strategy study design could be used to determine which components best increase physical activity in adults who are at risk for DFUs, though cost-effectiveness research on the relative benefits of each component would be critical to widespread implementation.
Footnotes
Appendix
Acknowledgements
The authors wish to thank the following individuals for their support in the conduct of the investigation: Rachel Domijancic, Christopher Girgis, Matthew Marti, Emily Mosher, and Jacquelyn Ortiz. Christopher Girgis’s effort was supported by NIH training grant T35DK074390 from the National Institute of Diabetes and Digestive and Kidney Diseases. The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Diabetes and Digestive and Kidney Diseases or the National Institutes of Health. Last, we are tremendously grateful for our participants’ time and effort in support of this study.
Abbreviations
A1C, glycemic control; BMI, body mass index; CBC, complete blood count; CMP, comprehensive metabolic panel; DFU, diabetic foot ulcer; GPS, Global Positioning System; PAMSys, PID, participant ID, physical activity monitor.
Data Sharing
Deidentified data can be requested from the corresponding author, KLS. Email:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a DePaul University–Rosalind Franklin University of Medicine and Science Collaborative Pilot Project to KLS, RTC, EM, and SH.
