Abstract
Background:
When a patient with diabetes measures blood glucose levels using a simple blood glucose meter, an error message or abnormal value may be displayed because of interfering substances, potentially leading to unnecessary medical treatment.
Methods:
Here, we tested 10 self-monitoring of blood glucose (SMBG) and point-of-care testing (POCT) devices to investigate under what conditions they fail to measure blood glucose levels or display abnormal values instead. We also evaluated the influence and linearity of several well-known interfering substances and hematocrit (Ht). Each adjusted sample was measured by each device twice and the values compared with the reference values.
Results:
Linearity was favorable in all but one model. The impact of the interfering substances and Ht has been confirmed in many SMBG and POCT devices. In particular, some models showed significant pseudo-high or -low levels for samples that contained ascorbic acid, which is used widely in infusion solutions. Three devices showed error messages to avoid presenting a false value.
Conclusions:
Since pseudo-high levels may result in the excessive administration of insulin and lead to adverse events, this report serves as a critical reminder of the importance of device choice for specific indications. Three new devices have safety functions that prevent the showing of false values.
This study aimed to evaluate the linearity and interference effect of hematocrit (Ht) and substances such as ascorbic acid and uric acid.
Strict blood glucose control is essential in diabetes treatment, and the usefulness of simple blood glucose meters has been widely accepted for their ability to provide measurement results more quickly and conveniently than clinical tests conducted in a laboratory.1-5 Devices that are being utilized in diabetes treatment include self-monitoring of blood glucose (SMBG) and point-of-care testing (POCT) devices.
The use of these devices has been widespread in Japan since they first became covered by the national health insurance; and various manufacturers now sell various models. All manufacturers currently manage SMBG accuracy and precision based on the guidelines described in the 2013 revised version of the ISO15197 6 (ISO15197:2013). 6 Previous reports have shown that the performance of SMBG devices released after 2013 is greatly improved compared to those of older models.7,8 However, the influence of Ht and various other interfering substances on measurement results has also previously been described.9-14
POCT devices are blood glucose meters used by health care professionals at the bedside. Compared to SMBG devices designed for daily monitoring, they provide more accurate and precise measurements; therefore, their use can be applied as an alternative to biochemical analyzers used in laboratories. As a result, the use of POCT devices in hospital wards has become required in recent years.15,16 However, despite the fact that POCT devices are also governed by international standards defined by Clinical and Laboratory Standard Institution (CLSI) POCT12-A3 guidelines, 17 the situation in Japan is that adjustment of the devices to comply with the CLSI guidelines is left to the discretion of each manufacturer, unlike overseas, where performance is strictly managed in accordance with the aforementioned standard. Similar to SMBG devices, substances such as ascorbic acid and uric acid reportedly affect the measurement results of POCT devices.18,19 Therefore, here we compared the accuracies of 10 of the latest SMBG and POCT devices using laboratory-measured values as a reference.
Methods
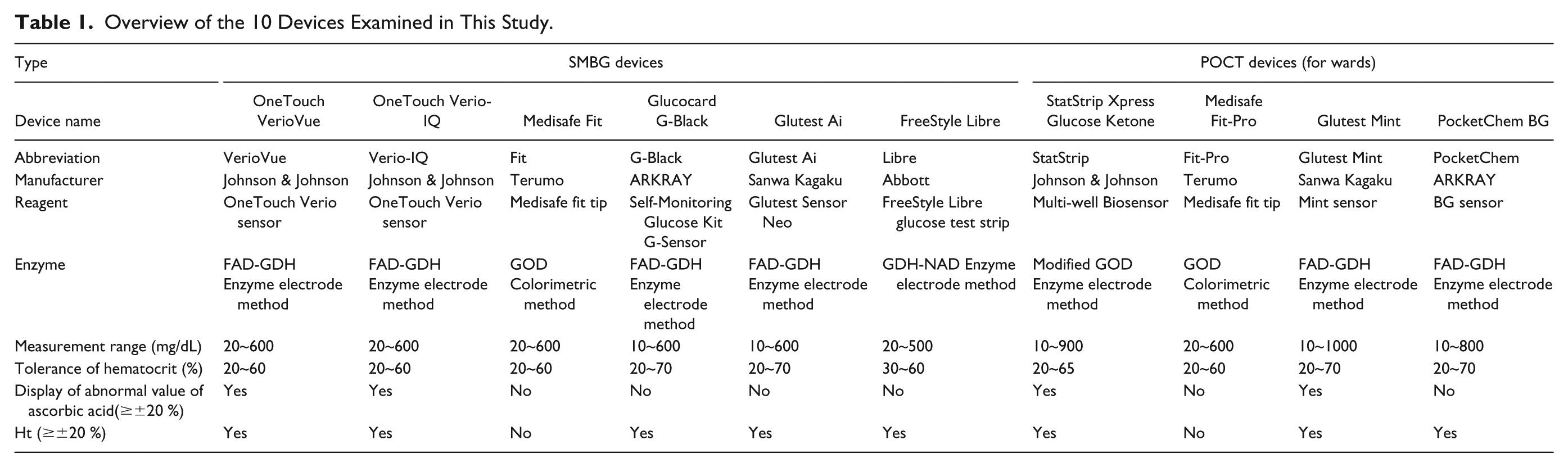
This study tested 6 SMBG devices and 4 POCT devices (Table 1). The description of each model is summarized in Table 1. The Libre is equipped with a glucose monitoring system that allows for the continuous measurement of blood glucose levels for up to 14 days as well as a blood glucose measurement function using electrodes; here we tested only its blood glucose measurement function using blood glucose test strips.
Overview of the 10 Devices Examined in This Study.
Samples were adjusted by glucose spiking of venous blood obtained from healthy individuals.
Influence of Interfering Substances
Each of the following was dissolved in a physiological saline solution (Otsuka, Japan): uric acid, ascorbic acid, propranolol, pralidoxime methiodide (PAM), acetaminophen, and furosemide. Interfering substances were added to the low-glucose concentration samples to obtain even lower concentrations, and each measurement was performed in duplicate by the 10 examined devices. The influence of each coexisting substance on the measured values was evaluated in accordance with ISO15197:2013 requirements, namely, “that there should be a difference of 10 mg/dL (<100 mg/dL) or 10% (≥100 mg/dL) between the mean blood glucose level in the normal samples and the mean blood glucose level in the samples containing additional interfering substances.”
Linearity
The samples were adjusted by glucose spiking of venous. After the measurements were completed, plasma separation was immediately performed using a centrifuge and plasma glucose concentrations were measured using a glucose analyzer (TBA-120FR/FIA; Canon, Japan) in the laboratory. The TBA-120FR uses the hexokinase method. The data obtained from the laboratory tests were used as reference values in the examination of linearity.
Influence of Ht
Samples with five different Ht values were prepared with serial Ht values of 20-60% in 10% increments. The Ht values were measured using an Ht analyzer (Coulter LH780; Beckman Coulter, Brea, CA, USA). All measurements were performed in duplicate on each of the 10 examined devices starting with the samples with high Ht values.
The influence of Ht on the measured value was evaluated according to the ISO15197:2013 requirements, namely under conditions showing a 10% (≥100 mg/dL) difference between the mean blood glucose levels in samples with a normal Ht (42 ± 2%) and the mean blood glucose levels in samples with Ht values within the operating range of the measuring device.
This study received approval from the medical ethics review board of PL Hospital on February 21, 2018 (control number: 8-17-193).
Results
Influence of Interfering Substances
Figures 1 and 2 show the influence of interfering substances for the 10 examined devices.
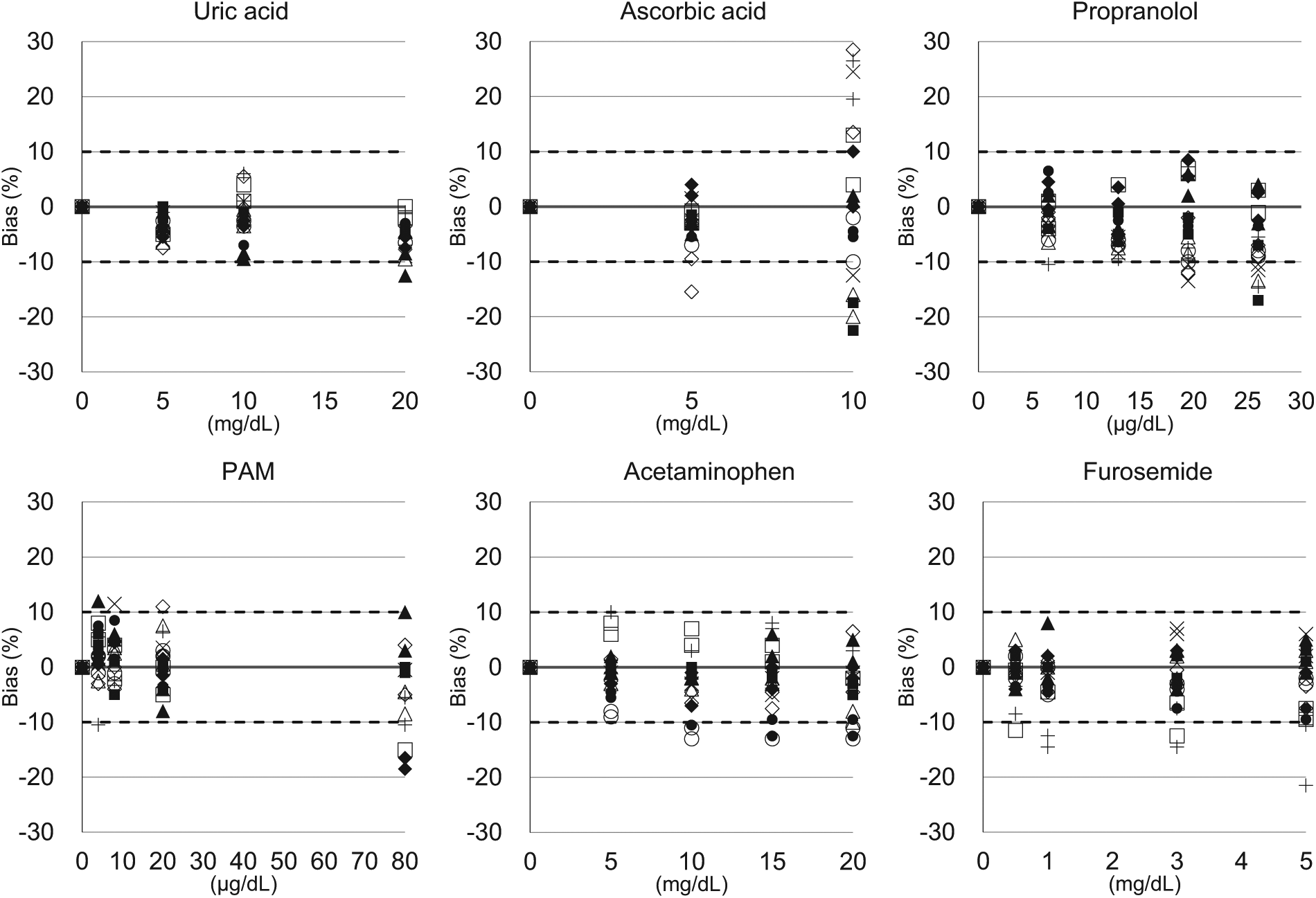
Effects of interfering substances on self-monitoring of blood glucose (SMBG) and point-of-care testing (POCT) devices. •VerioVue; 〇Verio-IQ; ■Fit; □G-Black; ◆Glutest Ai; ◇Libre; ▲StatStrip; △Fit-Pro; ×Glutest Mint; +PocketChem.
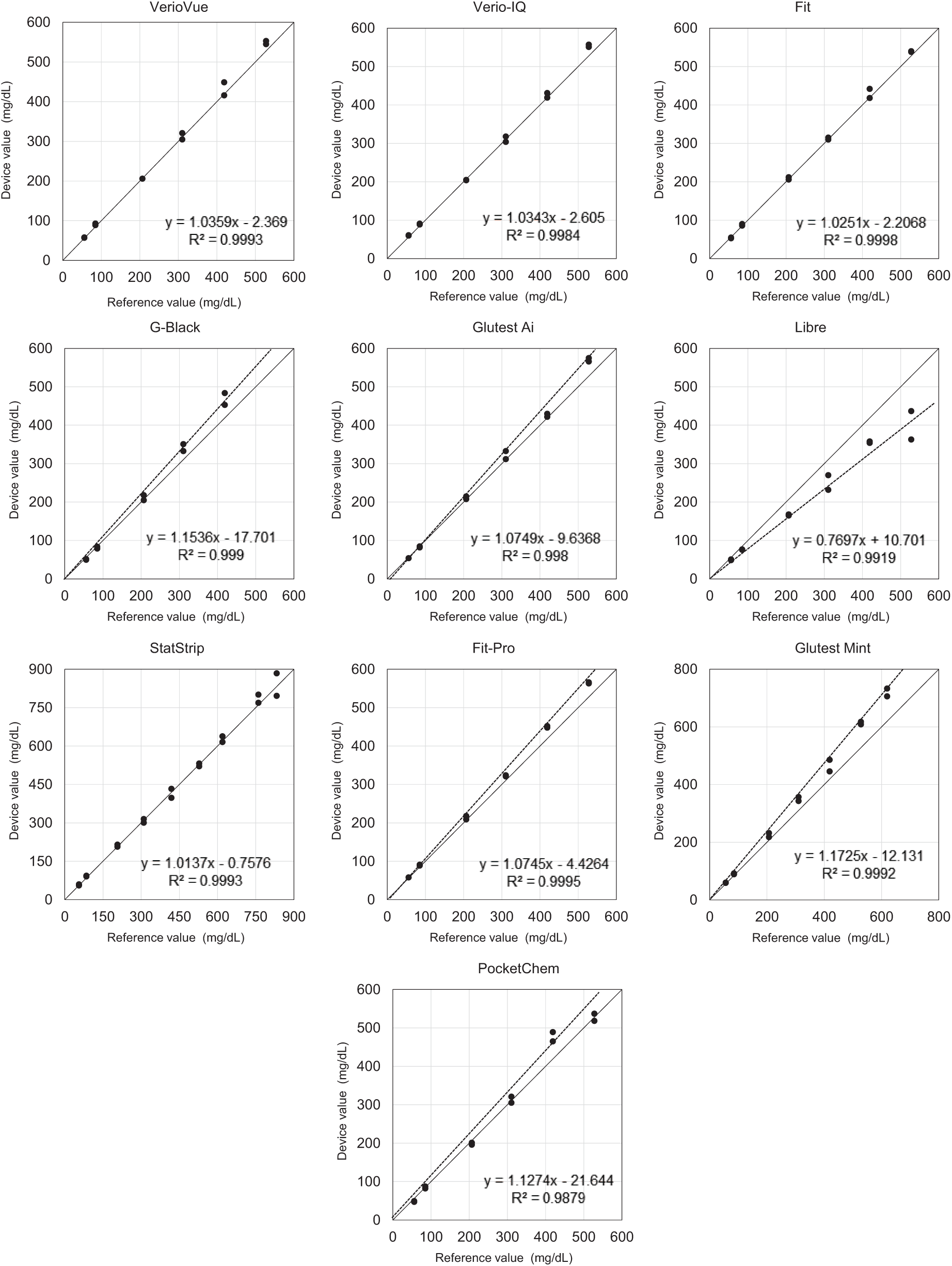
Linearity of the self-monitoring of blood glucose (SMBG) and point-of-care testing (POCT) device measurements versus the TBA-120FR/FIA laboratory-grade glucose analyzer.
Uric acid
Up to a uric acid concentration of 20 mg/dL, no impact was found, however, at a concentration of 20 mg/dL, the StatStrip deviated toward lower values.
Ascorbic acid
When the ascorbic acid contained in the samples was present at low doses (5 mg/dL, 10 mg/dL), the SMBG devices (VerioVue Lifescan, Verio-IQ, G-Black, and Glutest Ai as well as the POCT devices StatStrip and Glutest Mint) met the ISO15197:2013 requirements. However, the SMBG devices Fit and Libre as well as the POCT devices Fit-Pro and PocketChem tended to show a considerable deviation toward lower or higher values.
In addition, as shown in Table 2, when the ascorbic acid concentration was 200 mg/dL, the Fit and Fit-Pro showed abnormally low values (<20 mg/dL) versus abnormally high values for the G-Black (≥600 mg/dL), Libre (≥500 mg/dL), Glutest Ai (582.5 mg/dL), and PocketChem (342.5 mg/dL). Further, at 400 mg/dL, the PocketChem continuously showed an abnormally high value of 505.5 mg/dL.
Effects of High Concentrations of Ascorbic Acid on the Measured Values.
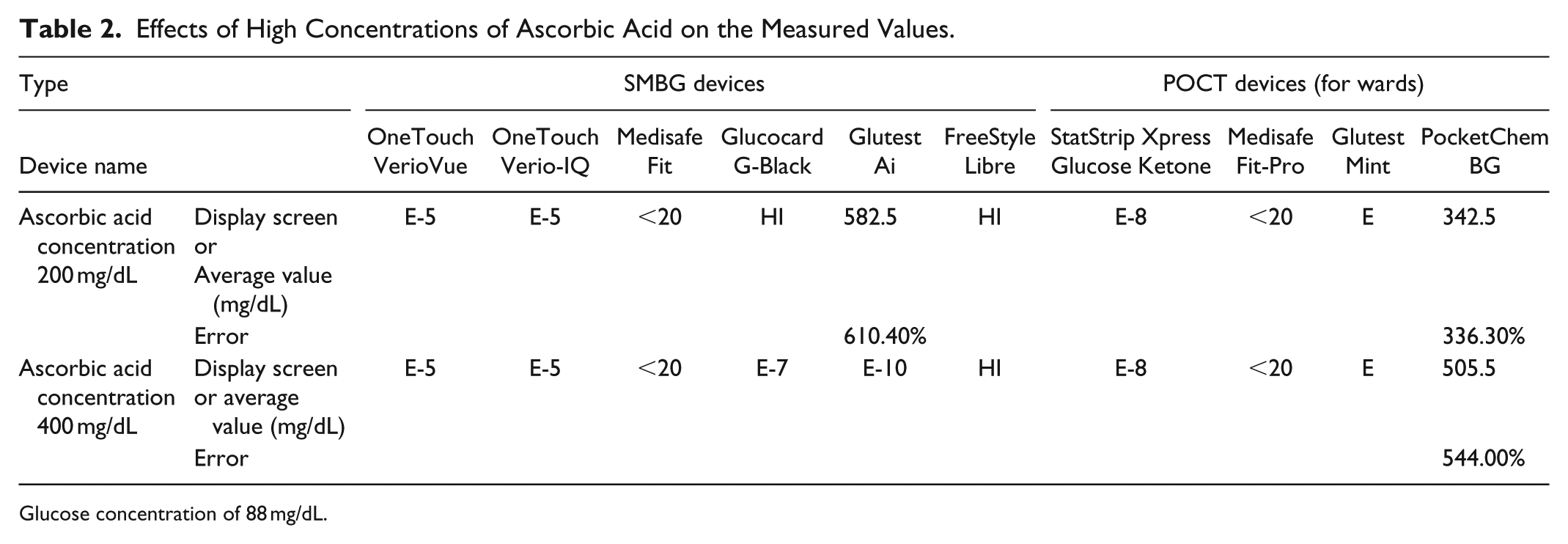
Glucose concentration of 88 mg/dL.
Propranolol
The device models that met the ISO15197:2013 requirements at all propranolol concentrations were the SMBG devices VerioVue, Verio-IQ, G-Black, Glutest Ai, and Libre as well as the POCT device StatStrip. The SMBG device Fit as well as the POCT devices Fit-Pro, Glutest Mint, and PocketChem showed a deviation toward lower values when the samples contained propranolol.
Pralidoxime methiodide
The accuracy and precision of most of the tested devices met the ISO15197:2013 requirements. However, when PAM was 6.5 μg/mL, the StatStrip showed a deviation toward higher values in contrast, when PAM was 80 μg/mL, the SMBG devices VerioVue and Verio-IQ displayed an error message that prevented measurements, while the G-Black and Glutest Ai showed a deviation toward lower values.
Acetaminophen
The accuracy and precision of most of the tested devices met the ISO15197:2013 requirements. However, the SMBG devices VerioVue and Verio-IQ tended to show a deviation toward lower values when the samples contained acetaminophen.
Furosemide
Our findings confirmed that the accuracy of virtually all of the tested devices met ISO15197:2013 requirements. However, the blood glucose levels displayed by the POCT device PocketChem tended to deviate toward lower values in the presence of furosemide.
Linearity
Figure 2 shows the linearity of the 10 devices examined in this study. The SMBG device G-Black as well as the POCT devices Glutest Mint and PocketChem showed slightly higher values. Meanwhile, the SMBG device Libre (blood glucose measurement function) tended to show considerably low values.
Influence of Ht
Figure 3 shows the influence of Ht on the 10 examined devices. At a glucose concentration of 144 mg/dL, the findings were as follows: when Ht was 20%, the SMBG device Fit and the POCT devices Fit-Pro and PocketChem showed a deviation toward higher values; when the Ht value was 60%, the Fit, and Fit-Pro showed a deviation toward 20% lower values. In addition, in samples containing glucose 362 mg/dL, the findings were as follows: the Fit, Libre, and Fit-Pro showed a deviation toward higher values when the Ht value was low, while the Fit and Fit-Pro showed a deviation toward lower values when the Ht value was high.
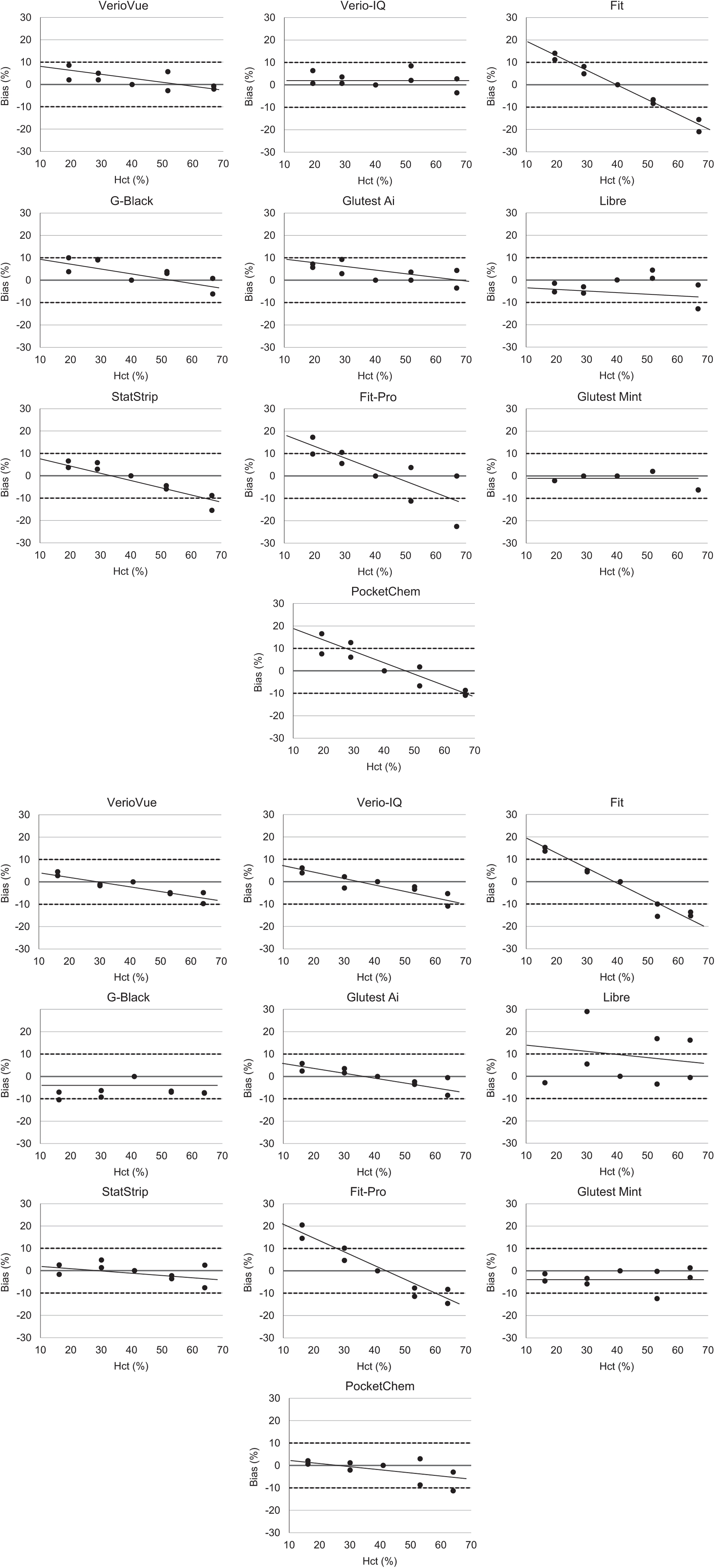
Effect of hematocrit on self-monitoring of blood glucose (SMBG) and point-of-care testing (POCT) device measurements. Glucose concentration of 144 mg/dL. Glucose concentration of 362 mg/dL.
Discussion
New ISO standard 2013 require accuracy criteria with at least 95% of results within ±15 mg/dL compared to reference values at blood glucose concentrations <100 mg/dL and within ±15%. Linearity was examined in the 10 devices tested here, and our findings showed generally favorable results in most device models except the Libre, which confirmed that most of the SMBG and POCT devices met the ISO15197:2013 requirements for accuracy. The Glutest Mint showed slight (17%) higher values compared to reference values. Regarding the Libre blood glucose test strip, BG electrodes were previously reported to show lower values. 10 In addition, previous reports in academic conferences held in Japan and other countries showed that sensors designed for measurements in interstitial fluids also showed abnormally lower or higher values.20,21 For this reason, SMBG has been recommended for the confirmation of hypoglycemia when interstitial fluids are used for the measurement of glucose levels; however, caution is needed and one should be aware that the values measured by SMBG devices themselves are low as shown here.
Ascorbic acid is used in various cases such as in burn treatment (blood levels: 10 mg/dL) and cancer treatment (blood levels: 400 mg/dL); some cases may show blood levels of 400 mg/dL. The ranges of therapeutic concentrations defined by the CLSI (EP07-A2) are as follows: acetaminophen, 20 mg/dL; furosemide, 1-3 mg/dL; and propranolol hydrochloride, 30 µg /dL. However, the values are likely to be higher in cases of renal and hepatic dysfunction; for this reason, we conducted tests using concentration ranges that included higher values.
The results of POCT devices, which are used in hospital wards, are not to be trusted simply because they are POCT devices; instead, caution is needed because of the possible effect of interfering substances depending on the device model.
Cases of fatal accidents due to the effect of maltose 22 and hypoglycemia symptoms due to the effect of PAM 13 have been reported. In those reported cases, the measured values displayed by the simple glucometers were much higher than the actual blood glucose levels (approximately 300-400 mg/dL higher); this seemed to be due to the fact that the therapeutic strategy was based on the false high values. Particular caution is needed in device models that, due to the effects of interfering substances, display inaccurate measured values that deviate from the actual blood glucose levels. Our findings showed that some devices displayed considerable false highs or lows in tests using samples containing ascorbic acid, those abnormal values pose a risk of excessive insulin administration. In previous measuring instruments using the electrode approach, false high values were commonly found due to reducing substances such as ascorbic acid and uric acid, which increased the number of electrons generated and delivered to the electrodes. In our study as well, false high values similar to those of previous reports23,24 were found at high concentrations of the reducing substances except in the StatStrip and OneTouch VerioVue. In addition, in the Fit, which uses the colorimetric method, peroxidase enzyme activity was inhibited by ascorbic acid, pigment generation was suppressed, and the device displayed abnormally low values. Meanwhile, in new measuring instruments using the electrode approach, such as the POCT device StatStrip and the SMBG device OneTouch VerioVue, functions allowing for the detection of the effect of interfering substances and noises as well as an error display function have been added, to ensure that measured values are not displayed when incorrect. At drug concentrations that are assumed to be found in actual patients, such an error was not observed with the VerioVue, but errors may have been displayed to specific patients because of their abnormal blood conditions (abnormal flow of electricity due to interfering substances).
In addition, in April 2017, a case reported overseas showed measurement results of 400 mg/dL, which was suspected to be a false high associated with the administration of ascorbic acid. 25 Thus far, reports on the effect of ascorbic acid have shown a value of approximately 20 mg/dL, while reports on measuring instruments’ abnormally high or low values (compared to 400 mg/dL) in cancer treatment as well as reports on device models with new error displays were the first of such reports in Japan. Further, in this study of the latest devices, the effects of other interfering substances as well as that of Ht were studied.
In addition, because SMBG devices and POCT devices use whole blood to measure blood glucose levels, conventional SMBG devices showed increased plasma volumes and elevated blood glucose levels when the Ht value was low, while blood glucose levels tended to be lower when the Ht value was high. 26 Simple glucometers on the market in recent years use Ht correction, but the details of the corrections differ among manufacturers and have not been made available to the general public. Our study showed that in the SMBG devices Fit and Libre as well as the POCT devices Fit-Pro and PocketChem, the measured values of blood glucose were affected by high or low Ht values. Caution is needed when using such device models. Ascorbic acid and Ht were added to Table 1 among the items on important points based on the results of our study and will be considered new important points to consider.
The Japan Diabetes Society also recently highlighted the need for accuracy control and maintenance of POCT devices, which used to be performed by each hospital’s laboratory testing department. It is also important that hospital staff members have a profound understanding of the effects of interfering substances and Ht and that they use the latter only after carefully reading the package insert.
POCT devices are used in hospitals and essentially required to provide an accurate equivalent to that of automated analyzers used in the clinical laboratory. In other countries, strict compliance to the international CLSI “POCT12-A3” standards is required for most POCT devices; however, in Japan, POCT devices conforming to the CLSI guidelines are actually those in which Ht and most interfering substances have less of an effect on measured values. In the same way as SMBG devices, which are subjected to strict accuracy control by the ISO15197:2013 guidelines, stricter accuracy control and conformity to the CLSI guidelines could potentially be performed on POCT devices in Japan in the future.
Conclusions
Here we evaluated the measurement accuracy of 6 SMBG devices and 4 POCT devices. Our results showed good linearity for all but one device. Meanwhile, our findings confirmed the various effects of interfering substances and Ht. The measurement errors depended on device type and were found in both SMBG and POCT devices. Particularly as an effect of ascorbic acid, some devices showed considerably aberrant false highs and lows that could cause medical accidents. As an alternative to automated analyzers in the clinical laboratory, POCT devices are simple glucometers that can also be used diagnostically in hospital settings. In our study, the only POCT device (StatStrip) in Japan that complies with the international CLSI POCT12-A3 standard did not show any abnormally high (false highs) or low (false lows) values in the presence of ascorbic acid. As in other countries, stricter accuracy control of POCT devices may be needed in Japan. Further, in actual clinical settings, it is commonly assumed that the measured values will be affected by various circumstances and that there will be cases of false lows, false highs, or error messages; therefore, in such cases, measurements may have to be performed in a clinical laboratory.
Footnotes
Abbreviations
CLSI, Clinical and Laboratory Standard Institution; FAD-GDH, flavin adenine dinucleotide dehydrogenase; GDH-NAD, nicotinamide adenine dinucleotide dehydrogenase; GOD, Glucoseoxidase; Ht, hematocrit; PAM, pralidoxime methiodide; POCT, point-of-care testing; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
