Abstract

Blood glucose data are frequently used in clinical decision-making. Therefore, it is critical that self-monitoring of blood glucose (SMBG) systems consistently provide accurate results. The new generation Accu-Chek® Guide blood glucose monitoring system (Roche Diabetes Care, Indianapolis, IN, USA) (system) utilizes innovative electrochemistry technologies that reduce the influence of environmental and endogenous factors on accuracy. When blood is applied to the test strip, several multi-frequency impedance measurements are taken in the first 2 seconds followed by fast-pulsed direct current (DC) amperometric measurements. Utilizing advanced algorithms, the information is analyzed to yield an estimate of the glucose concentration in less than 4 seconds.
We utilized the testing protocol defined in the ISO 15197:2013 criteria 1 to evaluate the accuracy and lot-to-lot variability in the system The accuracy of the system against more stringent accuracy measures was also evaluated.
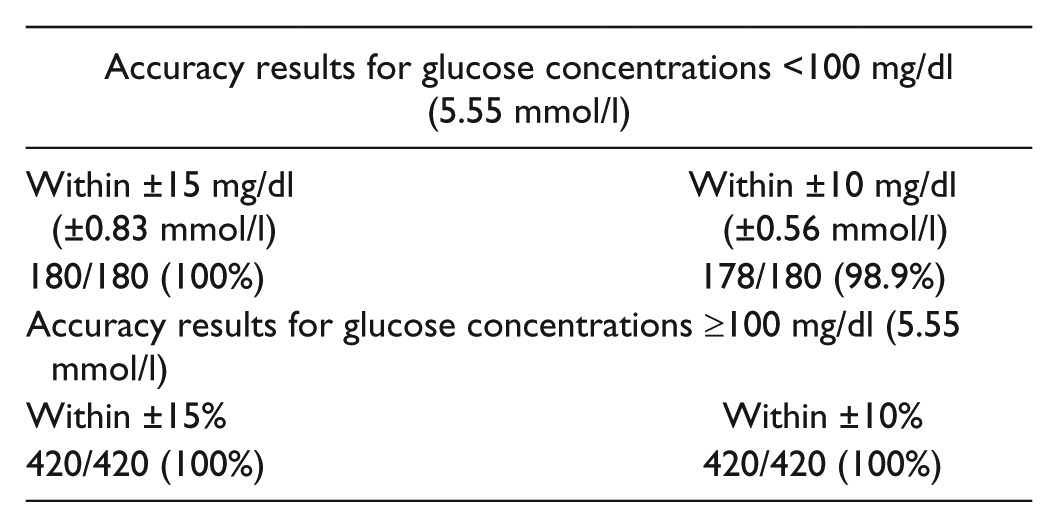
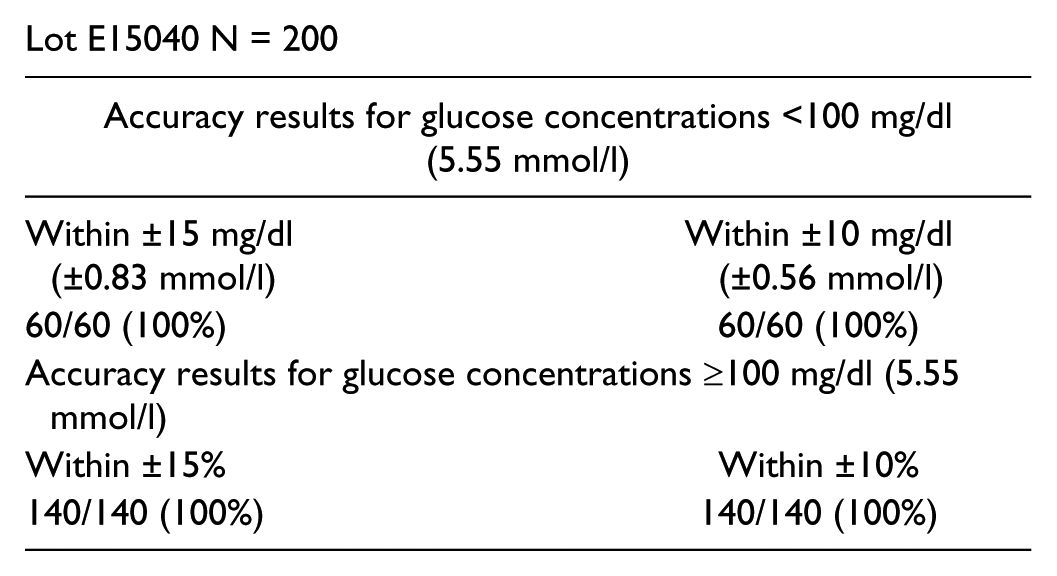
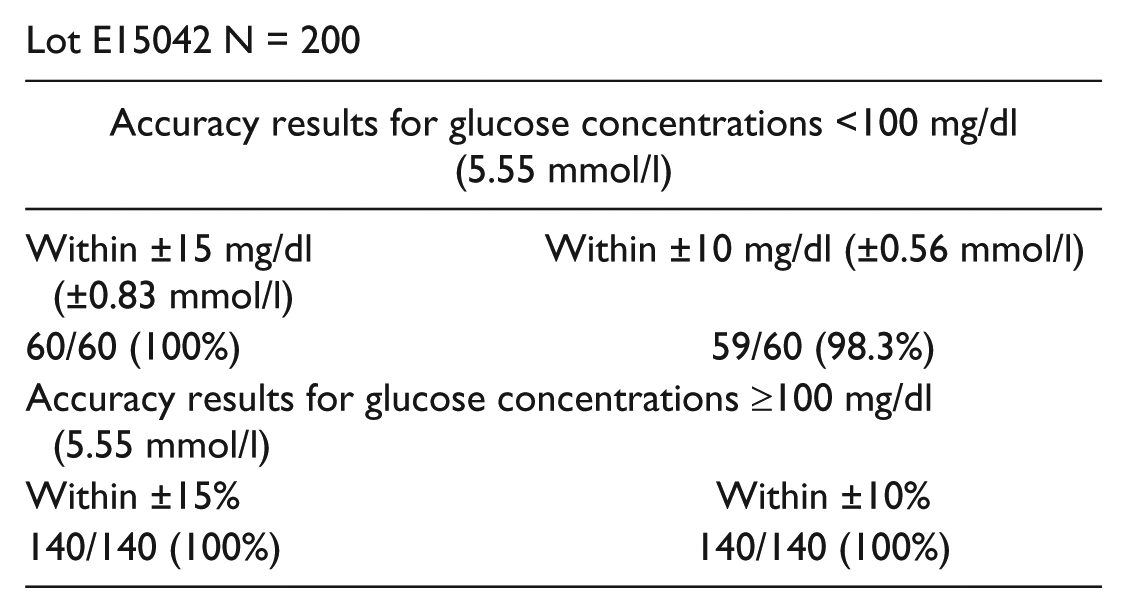
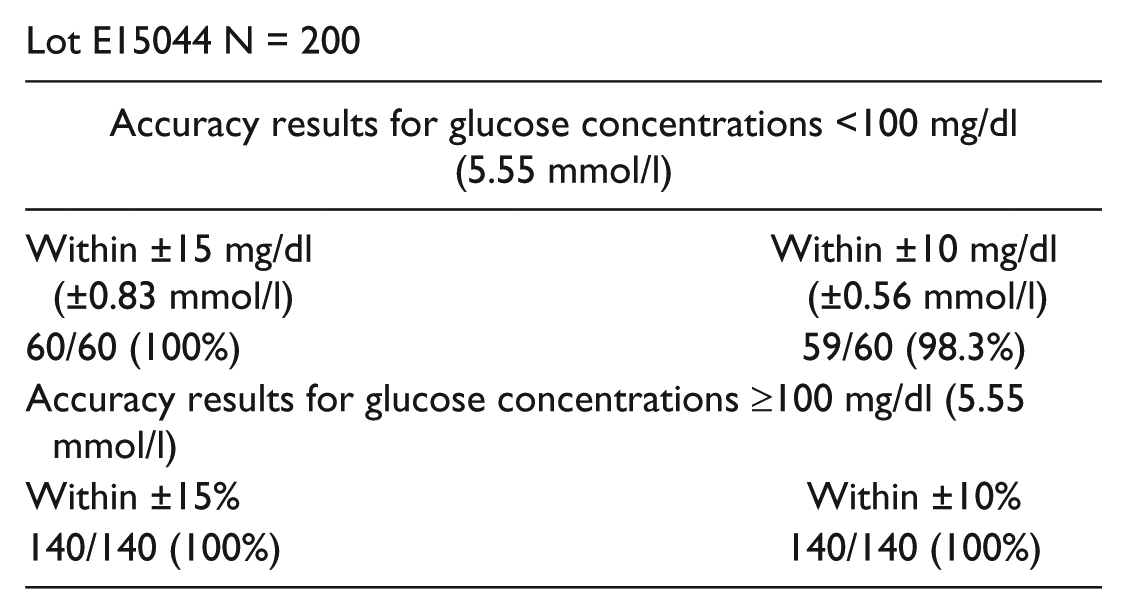
In comparison against current ISO criteria (DIN EN ISO 15197:2013), the system met the requirements in all 3 test strip lots (Table 1), and all test values (n = 600) were within Zone A of the Parke’s consensus error analysis 2 demonstrating consistent performance with negligible bias. The system also met tightened accuracy criteria of ≥95% of blood glucose (BG) values within ±10 mg/dl of laboratory reference value at glucose levels < 100 mg/dl and within ±10% of laboratory reference value at BG levels ≥ 100 mg/dl (Table 1). No discernable hematocrit effect was observed.
System Accuracy Versus ISO 15197 2013 Requirements and Tightened Accuracy Targets.
All Lots N = 600


Lot E15040 N = 200
Lot E15042 N = 200
Lot E15044 N = 200
The enhanced accuracy demonstrated in our study is largely achieved by reducing the influence on the BG result of factors such as temperature, ambient humidity, hematocrit, and endogenous interferences present in the blood. A notable improvement in the system is a novel, spill-resistant container, which presents and retains the individual strips in an easy-to-handle manner, which may significantly reduce contamination and wastage of test strips due to accidental spillage. Other improvements, which are less visible to the user, are embodied in the technology of the test strip. For example, the proprietary flavin adenine dinucleotide (FAD)-dependent glucose dehydrogenase enzyme used in the chemistry eliminates any effects from maltose, maltose derivatives or excess galactose. The reagent formulation also significantly reduces inaccuracy when a patient fails to close the container for extended periods in high humidity.
The complex measurements are made possible by a new generation, complete, battery-powered, high-precision “meter-on-chip” based on the ADuCM350 part from Analog Devices Inc (Waltham, MA, USA). This technology was recently developed for portable device applications such as body-worn devices for monitoring vital signs and chemical sensors, and is designed for high precision potentiostatic, current, voltage and impedance measurement capabilities. Through an advanced and proprietary encoding system, information for every lot manufactured is communicated to the meter whenever the user inserts a test strip without requiring any additional action.
The system was developed on a foundation of electrochemical technology that provides notably improved accuracy over previous Accu-Chek product generations. 3 Our study demonstrated that the system met and exceeded current ISO accuracy requirements.
Footnotes
Acknowledgements
The authors wish to thank Christopher G. Parkin, CGParkin Communications, Inc, for assistance in preparing this manuscript.
Abbreviations
BG, blood glucose; DC, direct current; SMBG, self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RLB, LJK, and AMS are owners and directors of Rainier Clinical Research Center (RCRC). RCRC is an independent clinical research center, which receives grants and funding from various pharmaceutical and device companies to conduct trials. No conflict of interest is reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Roche Diabetes Care, Indianapolis, IN, USA.
