Abstract

Keywords
Sensor-augmented pump therapy (SAP) has demonstrated to improve HbA1c and reduce the frequency of hypoglycemia in patients with type 1 diabetes (T1D). Different levels of automation of insulin infusion in response to sensor readings have been introduced.1-3 Our aim was to prospectively evaluate the additional benefits provided by SAP with predictive low-glucose suspend function (PLGS) in T1D patients experienced in the use of SAP with low-glucose suspend feature (LGS).
Thirty-six patients, aged 35 ± 14 years, 69% females, diabetes duration (median [IQR]) 26 years [14-33], time on SAP with LGS (median [IQR]) 20 months [15-44], 56% with history of severe hypoglycemia, already using the MiniMed-VEO®, with the LGS activated, were simultaneously upgraded to MiniMed-640G® with PLGS activated. After 6 months, while routine clinical care was maintained, all the patients were reevaluated. One month of data from both SAP systems were downloaded and glycemic variability measures were calculated. Adult patients were asked to complete a set of questionnaires (Gold and Clarke score, Hypoglycemia Fear Survey [HFS], Diabetes Quality of Life [DQoL], Diabetes Treatment Satisfaction [DTS], and Pittsburgh Sleep Quality Index [PSQI]). All patients signed an informed consent form. The protocol was approved by the Local Research Ethics Committee.
No changes in HbA1c were found at the end of follow-up (both 7.1 ± 0.6%). No severe hypoglycemia episodes or diabetic ketoacidosis episodes were reported. The changes found after the upgrade from LGS to PLGS are summarized in Table 1. In the patients with impaired awareness of hypoglycemia, 45% (n = 14), Gold Score was reduced from 5.6 ± 1.1 to 4.7 ± 1.6 (P = .022), and 3 out of the 14 patients (21%) regained hypoglycemia awareness. Of the adults, 65% (n = 20) reported being less worried about hypoglycemia, mainly at night, with PLGS and all of them agreed to keep using SAP with PLGS.
Changes After the Upgrade From Low-Glucose Suspend Function (LGS) to Predictive Low-Glucose Suspension (PLGS).
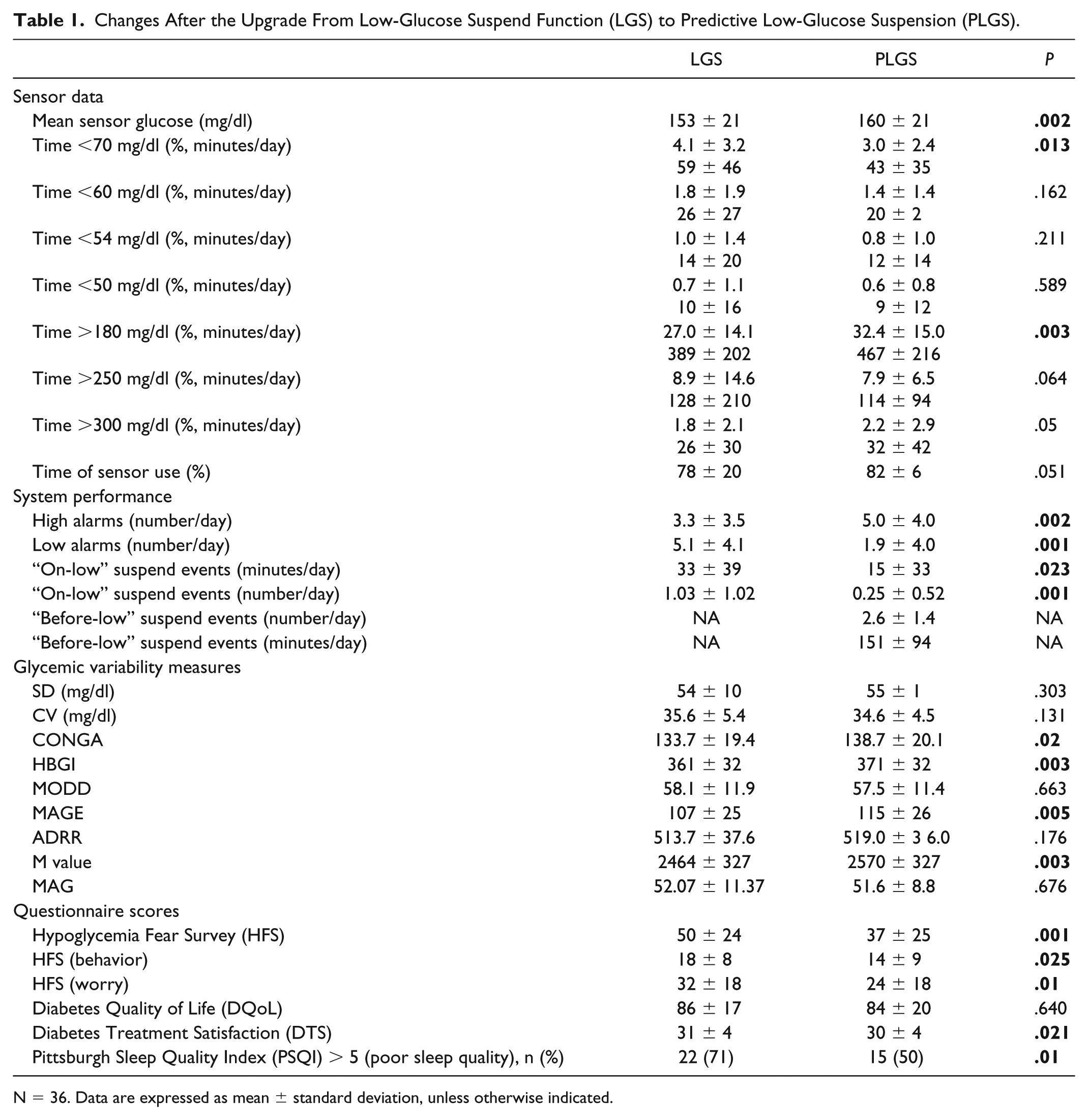
N = 36. Data are expressed as mean ± standard deviation, unless otherwise indicated.
Few studies have specifically addressed the advantages of PLGS in patients already experienced with the use of LGS. Biester et al 4 showed a 59% reduction in time <70 mg/dl in 6 children, as a result of an unintentional study protocol deviation. Zhong et al 5 retrospectively analyzed anonymized data from 851 users who had switched from SAP with LGS to SAP with PLGS, finding a 40% reduction in the time spent <70 mg/dl. Gómez et al 6 found a reduction in the frequency of severe hypoglycemia.
Our study has several strengths. The analysis of glycemic variability and patient-reported outcomes give a comprehensive assessment of the advantages and weaknesses of the PLGS. Testing the system in patients already benefiting from automation against hypoglycemia, with an optimal glycemic control and a low rate of hypoglycemia, has challenged the possibilities of the new system. Also, the impact of diabetes technology on sleep quality has been previously very scarcely studied.
In summary, we present a prospective study evaluating the benefits of SAP with PLGS compared to SAP with LGS, showing a reduction in hypoglycemia frequency along with an improvement in patients’ fear of hypoglycemia and the sleep quality, while an optimal and stable glycemic control was maintained.
Footnotes
Abbreviations
ADRR, Average Daily Risk Range; CONGA, Continuous Overall Net Glycaemic Sction; CV, coefficient of variation; DQoL, Diabetes Quality of Life; DTS, Diabetes Treatment Satisfaction; HBGI, High Blood Glucose Index; HFS, Hypoglycemia Fear Survey; IQR, interquartile range; LGS, low-glucose suspend; MAG, Mean Absolute Glucose; MAGE, Mean Amplitude of Glycaemic Excursions; MODD, Mean Of Daily Differences; PLGS, predictive low-glucose suspend; PSQI, Pittsburgh Sleep Quality Index; SAP, sensor-augmented pump therapy; SD, standard deviation; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
