Abstract

Scientific research needs a well-structured and robust study design and randomized controlled trials (RCTs) have been considered to be the gold standard to collect data that assess differing treatments. 1 The impact of the Hawthorne effect on clinical research outcomes have been studied in RCTs with evidence that research participation may modify trial outcomes.2,3 The Hawthorne effect describes the awareness of being in a study, and the possible impact on behavior of the participant. 3 The objective of this communication is to highlight the Hawthorne effect on psychobehavioral measures and the importance of a control group in clinical research, particularly for diabetes technologies.
This effect was analyzed in a large single-center RCT. Adolescents and young adults with type 1 diabetes on insulin pump, diagnosed with diabetes for at least a year, with glycated hemoglobin of ≤ 8.5% (69.4mmol/mol), and with impaired hypoglycemia awareness participated in a 6-month study and demonstrated a reduction in the incidence of severe and moderate hypoglycemia with low glucose suspension (LGS). 4 Participants in the control arm were on insulin pump alone while participants in the intervention arm were on sensor-augmented pump used in conjunction with LGS. The study also provided an opportunity to evaluate the fear of hypoglycemia (FOH) using a validated questionnaire. The Hypoglycemia Fear Survey 5 was administered at baseline and at the end of the study at 6 months. The validated versions of the survey were used in parents of children and adolescents < 18 years and in children aged 12 to 18 years, while the adult questionnaire was used for adults > 18 years. In both control and intervention arms, the participants had an identical number of research visits at similar time points and were reviewed by the same diabetes educator and/or doctor.
There was no difference in the FOH scores at baseline between parents, adolescent, and adult participants in both groups (P > .05). By the end of the study, there was a significant decline in the FOH scores from baseline in both control (P = .001) and intervention groups (P = .003) in parents. A similar trend was also noted in adult participants although there was no decline in FOH scores in the adolescent participants. However, it is important to note that there was no difference between the two groups at the end of the study at 6 months in the participants and their parents. (Table 1).
Fear of Hypoglycemia in Participants and Parents in a Randomized Controlled Trial.
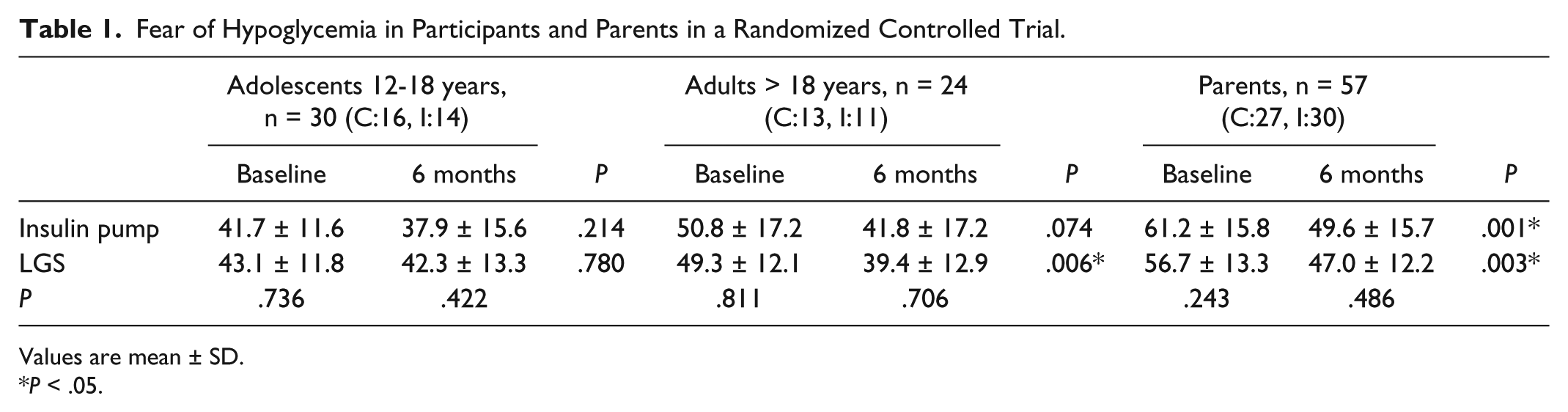
Values are mean ± SD.
P < .05.
In the absence of a control group, it could have been inferred that the significant reduction in the FOH in parents and adult participants was the effect of the intervention (LGS). However, the decline in FOH scores in the control group is important to acknowledge as it demonstrates a significant Hawthorne effect for this outcome. The visits in the control and intervention arm were similar, the participants were reviewed by the same health care professionals, and there was no specific diabetes education imparted except for pump and LGS commencement in the intervention group. The participants in the control group wore a blinded continuous glucose monitor during the study and did not have access to the study data during the visits. Thus, the control group had the same characteristics of the intervention group, except for the intervention applied to the latter. This decline in the FOH scores in the control group therefore appears to be by virtue of being in the study. FDA-mandated pivotal studies often do not require a control group, but this communication confirms the importance of a control group as part of the scientific design of trials that assess diabetes technology and perhaps especially for behavioral outcome measures.
Footnotes
Abbreviations
C, control; FOH, fear of hypoglycemia; I, intervention; LGS, low glucose suspension; RCT, randomized controlled trial.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partly funded by the Juvenile Diabetes Research Foundation (JDRF). Insulin pumps and glucose sensors were provided by Medtronic via an unrestricted grant. MBA was also supported by JDRF mentored clinical research fellowship.
