Abstract
Advances in the development of innovative medical devices and telehealth technologies create the potential to improve the quality and efficiency of diabetes care through collecting, aggregating, and interpreting relevant health data in ways that facilitate more informed decisions among all stakeholder groups. Although many medical societies publish guidelines for utilizing these technologies in clinical practice, we believe that the methodologies used for the selection and grading of the evidence should be revised. In this article, we discuss the strengths and limitations of the various types of research commonly used for evidence selection and grading and present recommendations for modifying the process to more effectively address the rapid pace of device and technology innovation and new product development.
Keywords
Introduction
The global prevalence of diabetes in adults is expected to grow from 537 million (10.5%) in 2021 to 643 million (11.3%) by 2030 and 783 million (12.2%) by 2045. 1 It is estimated that about half of people with diabetes are undetected. 1 The direct costs of diabetes are expected to continue to grow. The International Diabetes Federation (IDF) estimates that total diabetes-related health expenditure will reach US$1.03 trillion by 2030 and US$1.05 trillion by 2045. 1
An estimated 40% to 60% of people with type 2 diabetes (T2D)—approximately 95% of all diabetes—have suboptimal glycemic control. 2 Globally, cardiovascular disease (CVD) affects approximately 32.2% of T2D adults, 3 and the global prevalence of diabetes-related end-stage renal disease (ESRD) rose from 19% in 2000 to approximately 30% in 2015. 4
Compounding the problem is the increasing shortage of health care providers (HCPs). The World Health Organization (WHO) has forecast a global deficit of approximately 18 million HCPs by 2030. 5 Together, these conditions create a “perfect storm” that threatens to overwhelm HCPs, health systems, and public/private insurers while underserving those in need of quality medical care.
Advances in the development of innovative medical devices and telehealth technologies create the potential to address these issues by improving the quality and efficiency of diabetes care through collecting, aggregating, and interpreting relevant health data in ways that facilitate more informed decisions among all stakeholder groups. Among the technologies directly related to diabetes management are continuous glucose monitoring (CGM), sensor-augmented insulin pumps (SAPs), automated insulin delivery (AID) devices “smart” insulin pens, and artificial intelligence decision support systems (AIDSS). 6
Many of the large medical societies periodically publish guidelines for utilizing these technologies in clinical practice. However, we believe that the methodologies used for the selection and grading of the evidence may be inappropriate. In this article, we discuss the strengths and limitations of the various types of research commonly used for evidence selection and grading and present recommendations for modifying the process to more effectively address the rapid pace of device and technology innovation and new product development. Although there are numerous wearable device technologies currently available that are relevant to a number of chronic conditions, such as CVD and chronic kidney disease, this article focuses primarily on diabetes devices and technologies as an example of how guideline developers may want to modify their assessment of medical evidence moving forward.
Current and Emerging Diabetes Technologies
Diabetes Management Devices
Continuous Glucose Monitoring
Large randomized controlled trials (RCTs) and real-world observational and retrospective studies have demonstrated the clinical efficacy of CGM use in individuals with type 1 and type 2 diabetes (T1D, T2D).7-16 As such, CGM has emerged as a recommended standard of care for individuals with diabetes who are treated with intensive insulin therapy.17-22
Continuous glucose monitoring systems continuously measure glucose concentrations in the interstitial fluid, which correlate with blood glucose levels. 23 Unlike traditional blood glucose monitoring (BGM), which provides only a single-point glucose measurement, CGM devices present data in numerical and graphical formats, displaying the current glucose level, glucose trends, and direction and velocity of changing glucose levels. However, each CGM manufacturer assigns different rate of change values. Importantly, all of the newest CGM systems provide programmable alarms and alerts that warn users of current and impending glycemic events.
With immediate access to real-time glucose data and the “early warning” features, patients can act quickly to mitigate or prevent current and impending acute glycemic events. Through daily CGM use, patients can make more informed decisions about their insulin dosing, dietary intake, physical activity behaviors, and other areas of their daily self-management. CGM systems also have the capability to automatically transmit data to dedicated software for retrospective analysis and interpretation. Users also have the option to share their data with family and friends in real time, which adds an additional safeguard. Moreover, CGM is the only tool that can help to reach all the needed targets of glycemic control. 24
SAP and AID systems
With ongoing advances in CGM accuracy and reliability, manufacturers have integrated CGM technologies into two distinct insulin pump technologies, SAPs and hybrid closed-loop systems, often referred to as AID systems with distinct differentiation. When using an SAP system, CGM values are transmitted to the insulin pump but does not affect insulin dosing automatically. With AID systems, insulin dosages and delivery are regulated by a sophisticated controller algorithm that makes its determinations in response to real-time sensor glucose values, residual insulin action, and other inputs, such as meal intake and exercise announcement. Over the past five years, numerous trials have consistently shown that use of SAP and AID systems improve glycemic control, increase time in range, and reduce time in hypoglycemia and incidence of severe hypoglycemia in adults and children.25-31 Although current AID systems still require patients to manually input their carbohydrate intake to achieve adequate postprandial insulin coverage, more advanced AID systems that eliminate the need for carbohydrate counting and manually initiated prandial boluses are under development. 31
Smart insulin pens
A recent development in insulin delivery technology is the “smart” pen, which automatically tracks insulin dosing (timing and dosage) and provides dose-decision support through a bolus calculator. When connected to CGM, these devices facilitate a better understanding of each patient’s pattern of insulin use. Importantly, many smart insulin pens have the ability to transmit missed dose alerts when insulin is not delivered within a specified time frame. Smart insulin pens can accurately capture insulin data that can then be integrated with glucose and dietary (carbohydrate) data by software applications and transmitted to the patient’s HCPs.32,33
Diabetes-Related Devices
In addition to devices that are designed specifically for diabetes management, there is a growing number of wearable devices that monitor and report personal health data relevant to CVD and hypertension, which are common comorbidities of diabetes and significant risk factors for chronic heart failure, acute myocardial infarction, and stroke. 34 Devices, such as trackers of physical activity and energy expenditure, and calorie counters are additional tools.
Telehealth Technologies
Many of contemporary CGM and insulin delivery devices allow patients to automatically transmit real-time data to their health care team through cloud-based software for interpretation and analysis. When used in conjunction with virtual telehealth visits, these technologies enable patients to interact remotely with their clinicians through smartphones and other connected devices, which can increase their access to quality clinical care.
Use of virtual telehealth clinic visits, in conjuction with remotely monitoring CGM and insulin data, has been shown to improve glycemic control,35-38 reduce diabetes-related distress, 35 increase treatment satisfaction, 35 and enhance the adherence to prescribed therapy, 39 with increased time and cost efficiency compared with in-clinic diabetes care.37,40-43 Moreover, recent studies have shown that this approach to diabetes care addresses many of the challenges created by the coronavirus disease 2019 (COVID-19) pandemic.44-47 Finally, artificial inteligence decision support systems can safely and effectively help adjusting the daily doses of insulin 48 and other medications.
Telehealth technologies that support remote monitoring and physical examinations, such as retinal scanning, foot examinations, and blood pressure measurement are especially valuable in addressing the needs of patients who live in rural areas or low-income countries where even basic health care services are scarce.49,50 These technologies greatly expand the ability of health care systems to mitigate the projected shortage of HCPs.
Considerations for Evidence Selection
Systematic Reviews/Meta-Analyses of RCTs
Systematic reviews and meta-analyses (SRs/MAs) of well-designed RCTs are considered the highest level of scientific evidence. 51 Although SRs/MAs are well suited for evaluating the efficacy of medications and surgical procedures, they seldom consider the complexity and interplay of the various behavioral and attitudinal components that can impact the outcomes of interventions that utilize medical devices and technologies.
First, one must consider the limitations of RCTs themselves. Although RCTs provide essential information about the efficacy of a given intervention, pharmacologic or device-related, when applied within tightly controlled clinical conditions, findings from these studies may not reflect if, how, or to what degree individuals will integrate the intervention into their daily lives.
Unlike pharmaceutical trials that primarily assess the physiologic response to a given medication, medical device and technology studies often involve extensive modification of patient and/or clinician behavior as a critical component of the intervention. In addition, disparities among patient and/or clinician’s interpretation of the data obtained from devices (eg, self-monitoring of blood glucose [SMBG] and CGM) and subsequent responses might also lead to different outcomes.
Because the mechanism of action involves a unique combination of technology performance, behavioral responses, and attitudinal issues, the efficacy of the intervention is dependent on behavior change as well as the device/technology. For example, whereas studies have shown the clinical benefits of CGM in lowering glycated hemoglobin (HbA1c), the glycemic improvements are more pronounced among individuals who frequently review and utilize their glucose data.52-63
Moreover, RCTs do not necessarily reflect the effects of the intervention within populations that were excluded from the trials. For example, most of the large RCTs investigating the efficacy of CGM exclude individuals with a history of severe hypoglycemia or impaired hypoglycemia awareness.9,10,13,15 Thus, findings from many RCTs often cannot be generalized to a larger diabetes population, in some instances, to the specific subpopulation that could mostly benefit from the interventions. This limitation could easily be addressed by modifying the inclusion/exclusion criteria, so that these subpopulations can be assessed.
Another critical limitation of SRs/MAs for assessment of medical devices and technologies is the delay between the time that studies are conducted, results are assimilated, and the report is published. By the time enough studies for a robust report have been completed, subsequent improvements in accuracy, reliability, and convenience will render the results outdated and potentially misleading. Given the rapid pace of technology innovation, this time lag can prevent inclusion of the most recent generation devices and technologies.
An example of this is the 2012 report from the Cochrane Collaboration, which assessed the clinical benefits of CGM in adults and children. 64 Based on findings from the 12 studies assessed, the authors concluded that the clinical efficacy of CGM was “limited.” Importantly, only four of the 22 studies included in the Cochrane analysis were published after 2009.26,60,65,66 All of the CGM devices covered in the Cochrane report had already been replaced with more accurate and usable devices prior to its publication.
In addition, because SRs/MAs incorporate results from diverse clinical studies, they often group all CGM devices into a single category of technology without differentiating between the strengths and limitations of the individual systems. This becomes problematic when the studies included in the SR/MA overrepresent a specific CGM device. For example, in the case of an SR/MA that is heavily weighted with studies of a specific CGM device that is less accurate than the other devices evaluated, the aggregated outcomes will, by default, lead to a less favorable assessment of efficacy of CGM as a category. Moreover, patients’ perceptions of the accuracy and reliability of their CGM device significantly diminishes their persistence in using their device, 67 which, as discussed earlier, can negatively impact clinical outcomes.
Given the distinct differences between medications and medical technologies, it is becoming increasing clear that the dependence upon RCTs and SRs/MAs, alone, fails to address the significant influences of patient attitudes and behaviors on CGM outcomes in real-world settings. Other types of evidence should be considered in the guideline development process.
Real-World Evidence
Because the benefits of medical devices and telehealth technologies for chronic disease management are primarily dependent on patient behavior (Is the device being used? Is it being used correctly?), different approaches to evaluating the effects of these tools are needed. As such, there is growing appreciation for the value of real-world evidence (RWE) in assessing the effectiveness of diabetes devices and telehealth technologies when used within various populations for everyday self-management. Generated by prospective, retrospective, and observational studies, RWE can fill in the knowledge gap between how individuals respond to a given intervention within a controlled setting compared with patterns of response and clinical outcomes in real life. Recognizing the importance of real-world study designs, many payers and regulatory agencies now require manufacturers to submit RWE in conjunction with findings from their RCTs when reporting the safety, effectiveness, and cost-benefit parameters of new medications and medical devices.68-71
Although the use of observational and prospective real-world studies is not new, a key driver behind the rapidly growing emphasis on RWE is the “digitization” of health care. This has led to the creation of large databases that accumulate comprehensive, longitudinal medical information for millions of individuals. Available data include demographic information, medical records, laboratory data, pharmacy prescriptions, hospitalizations, emergency department utilization, and outpatient care. Importantly, robust safeguards must be in place to protect patient privacy.
However, as with RCTs and SRs/MAs, real-world studies utilizing large database records have important limitations. Although these databases provide detailed clinical and demographic information, they include a risk for prescription bias and provide little or no information about patients’ socioeconomic status or education/digital literacy level. Moreover, they do not capture attitudinal or behavioral information, both of which are relevant to studies involving medical devices and technologies. For example, whereas data regarding prescription and acquisition of a CGM system are readily available, patient-specific factors, such as their reasons for choosing one device over another, are not captured. Nor can we determine patients’ persistence in monitoring or use of their glucose data in daily self-management. Did they utilize their glucose data to make treatment decisions and how did they make treatment decisions? Did they use the information appropriately? Therefore, although large database analyses can provide strong indications of effectiveness by comparing clinical endpoints (eg, HbA1c, hospitalizations) before and after acquiring the device or technology, the underlying reasons that explain why device use was effective cannot be determined.
Psychosocial Measures
Because effectiveness of interventions involving medical devices is dependent upon whether and to what degree patients persist in using their device, it is important to identify and assess the presence and impact of the various factors that can influence treatment adherence. Suboptimal adherence to diabetes self-management regimens leads to poor clinical outcomes,72,73 increased health care service utilization, 74 and higher overall health care costs.75-78 Conversely, optimal adherence is associated with improved glycemic control and decreased health care resource utilization.74,79 As reported by Boye et al, there is a significant association between higher adherence and lower health care costs, specifically those associated with acute diabetes-related events such as severe hypoglycemia and diabetic ketoacidosis (DKA). 79
Treatment satisfaction
Among the key factors influencing adherence is treatment satisfaction.80,81 A recent study of 135 T2D adults (age 60 years) reported that patients who were satisfied with their diabetes treatment showed higher levels of adherence to their self-management regimens. 82 In an earlier study, Chantelau et al reported higher treatment satisfaction among those who were able to choose their intensive insulin regimen (multiple daily injection [MDI] or insulin pump). 83 A commonly used tool for assessing treatment satisfaction is the Diabetes Treatment Satisfaction Questionnaire (DTSQ), which is available in two versions: DTSQs for baseline status and DTSQc for changes in treatment satisfaction. 84 Other tools that are specific to satisfaction with glucose monitoring and insulin delivery methods include the Glucose Monitoring System Satisfaction Survey (GMSS), 85 Continuous Glucose Monitoring Satisfaction Scale (CGMSS), 86 and Insulin Delivery Satisfaction Survey (IDSS). 87
Diabetes-related distress/depression
Treatment adherence is also impacted by the presence of diabetes-related distress and/or depression. Diabetes-related distress affects up to 50% of individuals with diabetes88-90 and is significantly associated with poor glycemic control,90-94 high prevalence of complications,94,95 diminished adherence to prescribed self-management regimens,90-92,96 and, in men, all-cause mortality.
95
Similarly, depression is one of the common comorbidities associated with diabetes and other chronic diseases.97,98 Clinically significant depressive symptoms are twice as common in individuals with T2D than those without diabetes.
99
Earlier studies have shown that individuals afflicted with depression have suboptimal glycemic control
100
and have poorer quality of life (QoL),
101
higher utilization of health care resources,
102
more complications,103,104 and higher risk of mortality.
104
Importantly, a 2008 meta-analysis of 43 published reports that included 17 319 individuals showed a significant association between depression and poor treatment nonadherence in patients with diabetes.
105
Diabetes-related distress is often assessed using the DDS tool, a validated 17-item questionnaire that uses a Likert scale to grade each item from 1 (
Quality of life
Quality of life is impacted by several factors, including an individual’s physical health, psychological health, socioeconomic status, social relationships, and personal beliefs.108,109 Several studies have shown a strong association between QoL and adherence to diabetes self-management.110-113 Patients with a higher level of adherence tend to have a higher QoL and vice versa. 114 Commonly used tools for assessing QoL include the EQ-5D-5L survey 115 and Hypoglycemic Confidence Scale (HCS). 116
Cost-effectiveness analyses
Cost-effectiveness analyses are performed to assess the benefits and costs of an intervention by comparing it with standard care or other alternatives to determine whether the value of the intervention outweighs its cost. This information is critical for both HCPs and payers in the development and recommendation of optimal treatment strategies and in setting policies for insurance coverage.
Limitations of the GRADE System
Most methodologies for evidence selection and grading used by medical associations are based upon a hierarchy of evidence types, ranging from expert opinion and consensus as the lowest level of evidence to SRs/MAs of RCTs as the highest level. The exception is the Grading of Recommendations Assessment, Development Evaluation (GRADE) approach, which assesses the magnitude of the benefits, harms and burdens of the intervention and its comparators, quality of the evidence, underlying values and preferences of the patient population to whom the recommendation applies, 117 as well as the cost, feasibility, and acceptability of the intervention 118 (Table 1).
GRADE Approach to Rating Quality of Evidence. 119

Abbreviation: GRADE, Grading of Recommendations Assessment, Development Evaluation.
Strengths
Because the GRADE approach simplifies the process by eliminating reliance on SRs/MAs, the most recent studies of new medications and devices can be graded and incorporated into guidelines and updates. The approach also offers clear guidance for determining confidence in the outcomes reported. The process is further simplified by designating the two levels of recommendations, strong or weak. 119 A strong recommendation is given when an intervention is clearly beneficial or detrimental to the patient. A weak recommendation is attributed in situations where the beneficial aspects of the intervention must be weighed against the patient’s individual circumstances and preferences.
Limitations
However, although we endorse this approach for assessing pharmaceutical trials, we believe a shift in our current research paradigm is needed to fully address the complexities of device- and technology-driven interventions. For example, in the recent Australian Evidence-Based Clinical Guidelines for Diabetes, 120 we had a number of concerns regarding the evidence used to develop the CGM recommendations. Among these is the timeliness of the studies reviewed. The most recent study used to assess CGM devices with alarms was the HypoDE study, 121 published in 2018, which assessed the efficacy of an earlier-generation CGM device (Dexcom G5 Mobile). 122 An additional limitation was that only one small study, published in 2016, 123 was used to determine guidelines for the use of CGM devices without alerts due to the rigid criteria for evidence inclusion. 124 Again, the CGM device evaluated in this study is based on early generation technology. Importantly, both types of systems (with and without alerts) received only a “conditional” recommendation.
Recommendations for Modifying Current Methodologies for Evidence Selection and Grading
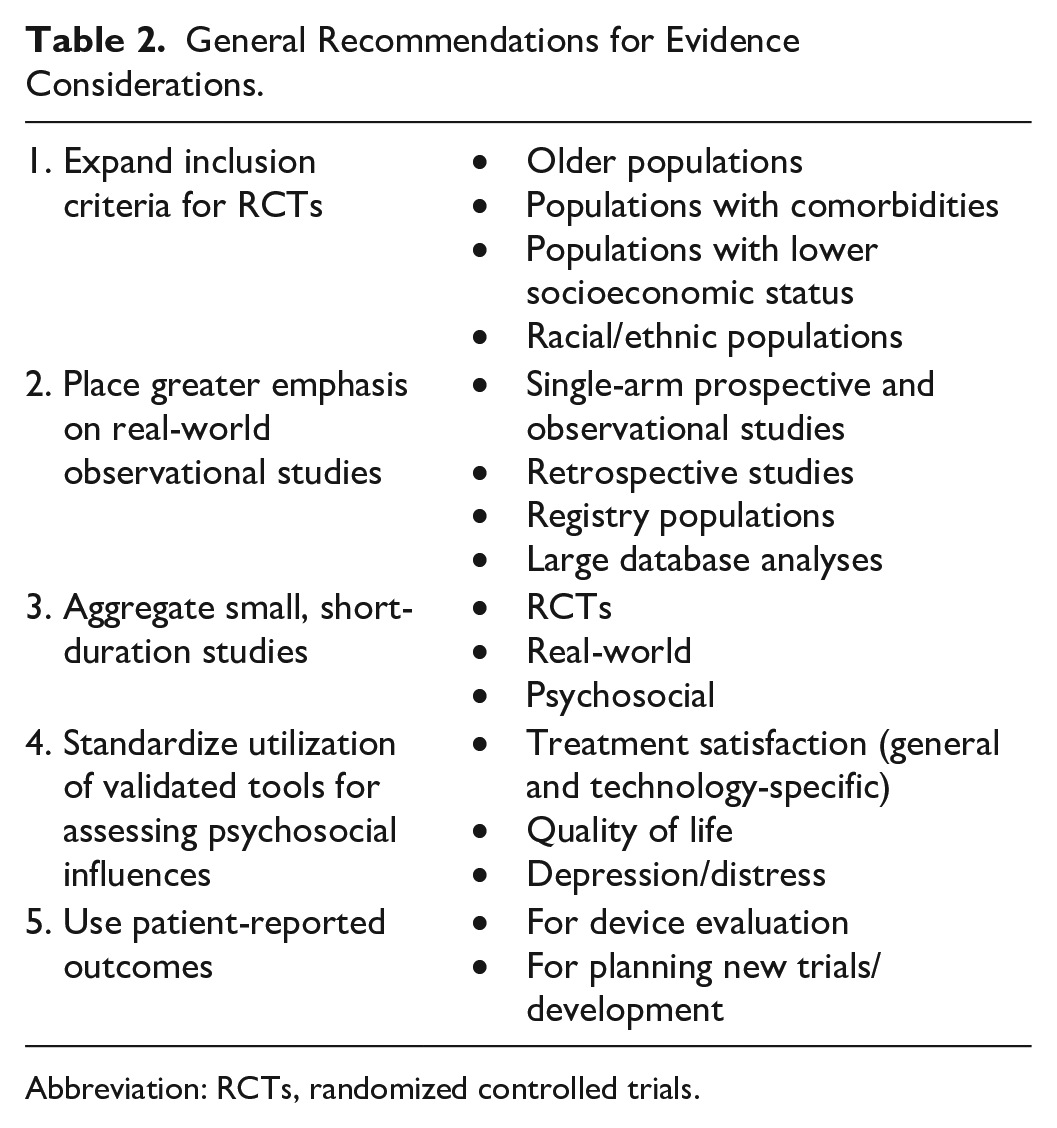
As discussed, current eligibility criteria for evidence used for clinical guideline development does not accommodate rapidly evolving medical devices and technologies. The Guideline Taskforce compiled the following recommendations for professional societies to consider for the rapid development of future guidelines for these tools (Table 2).
General Recommendations for Evidence Considerations.
Abbreviation: RCTs, randomized controlled trials.
Expand Inclusion Criteria for RCTs
Manufacturers and technology developers need to consider expanding their research programs to include all subgroups within a given population, particularly those who would benefit most from the intervention. For example, older adult patients account for nearly half of all individuals with diabetes 125 and are at highest risk for severe hypoglycemia.125-132 Yet these patients are often excluded from RCTs investigating CGM and AID systems, device technologies that have been proven effective in reducing severe hypoglycemia in younger adults.133-139 Other patients who have often been excluded from RCTs are those with problematic hypoglycemia or multiple comorbidities. Randomized controlled trials that assess the efficacy of these technologies across the entire diabetes population, including racial/ethnic populations, are needed for both clinical guidance and policy decision-making among payers.
Place Greater Emphasis on Real-World Observational Studies
Given the behavioral and attitudinal influences that are inherent in device- or technology-based interventions, guideline developers should consider placing greater emphasis on the value and utility of real-world observational studies. As discussed above, although RCTs provide needed evidence to demonstrate the
Aggregate Small and Short-Duration Studies
Small and short-duration studies are often excluded when selecting evidence for guideline development. As a result, it may take years before critical findings from these studies are formally considered. An example of this is how long it took medical organizations to establish a target for postprandial glucose management due to a lack of RCTs demonstrating the importance of this metric. Recognizing that an RCT could not be conducted due to ethical restrictions, developers began looking at the small but numerous single-arm studies,144-151 which showed both the clinical consequences of excessive postprandial excursions and the benefits of reducing and minimizing the frequency of those excursions. As a result, all major diabetes organizations have established targets for postprandial glucose excursions as an important component of glycemic control.
Standardize Utilization of Validated Tools for Assessing Psychosocial Influences
Because adherence to prescribed use of devices is critical to achieving desired outcomes, investigators should increase use of validated tools to assess the presence and influence of behavioral and attitudinal factors that can potentially impact adherence and outcomes. It would also be beneficial if the research community could come together to standardize a list of assessment tools to be used in all trial designs to facilitate direct comparisons across all studies.
Use Patient-Reported Outcomes
Patient-reported outcomes (PROs) measures are used to assess the impact of a medical treatment or intervention on a number of factors, including symptoms or treatment side effects, such as functional outcomes, or multidimensional constructs, such as health-related quality of life (HRQoL) or health utility. 152 Moreover, findings from well-defined and reliable PRO instruments can be used to guide technology development and support claims in medical product labeling. 153 Because use of PROs helps to assess the magnitude of the benefits of a given medication or technology, findings from these reports can provide meaningful information to guideline developers when assessing both the product’s effectiveness and user preference.
Summary
Advances in digital technologies have led to the development of innovative tools that enable people with diabetes to safely achieve their desired levels of glycemic control. Education in how to use these technologies is essential. Moreover, clinicians’ ability to remotely access and evaluate patient data using connected devices and interact with patients through virtual telehealth visits significantly increases patient access to quality health care. As artificial intelligence capabilities continue to develop, both access and efficiency will further improve.
However, appropriate, evidence-based guidelines for effectively using these technologies must be established. As discussed here, criteria for technology studies should differ from those that evaluate pharmacotherapies. Therefore, appropriate, evidence-based approaches for effectively evaluating these technologies must be established so that useful, practical, and timely guidelines can be developed. This will require that all individuals involved in guideline development have extensive clinical experience in the relevant medical devices and technologies. In addition, guideline development panels should include representatives from all aligned medical organizations to avoid discordance in recommendations. Finally, given the rapid rate of innovative technology development, we recommend that guideline development panels meet (in person or virtually) every three to six months to review current evidence and determine whether and when guidelines require updating.
Footnotes
Abbreviations
AID, automated insulin delivery; AIDSS, artificial intelligence decision support systems; BGM, blood glucose monitoring; CGM, continuous glucose monitoring; CGMSS, Continuous Glucose Monitoring Satisfaction Scale; CVD, cardiovascular disease; DADS, Diabetes Anxiety Depression Scale; DKA, diabetic ketoacidosis; DTSQ, Diabetes Treatment Satisfaction Questionnaire; ESRD, end-stage renal disease; EXSCEL, Exenatide Study of Cardiovascular Event Lowering; GMSS, Glucose Monitoring System Satisfaction Survey; GRADE, Grading of Recommendations Assessment, Development Evaluation; HbA1c, glycated hemoglobin; HCL, hybrid closed loop; HCP, health care provider; HCS, Hypoglycemic Confidence Scale; HFS-II, Hypoglycemia Fear Survey-II; HRQOL, health-related quality of life; IDF, International Diabetes Federation; IDSS, Insulin Delivery Satisfaction Survey; MDI, multiple daily injection; PROs, patient-reported outcomes; QoL, quality of life; RCT, randomized controlled trial; RWE, real-world evidence; SAP, sensor augmented insulin pump; SMBG, self-monitoring of blood glucose; SRs/MAs, systematic reviews and meta-analyses; T1D, type 1 diabetes; T2D, type 2 diabetes; WHO, World Health Organization.
Cardiovascular Outcome Trial (CVOT) Guideline Taskforce
Participants of the CVOT Taskforce, in alphabetical order, are as follows:
Frank Brosius, American Society of Nephrology (ASN); Antonio Ceriello, Italian Online Guidelines, Associazione Medici Diabetologi (AMD); Michael Cheung, Kidney Disease: Improving Global Outcomes (KDIGO); Cosentino Francesco, European Society of Cardiology (ESC); Jennifer Green, American Diabetes Association (ADA); Linong Ji, Peking University People’s Hospital; Monika Kellerer, Deutsche Diabetes Gesellschaft (DDG); Susan Koob, Preventive Cardiovascular Nurses Association, (PCNA); Mikhail Kosiborod, American College of Cardiology (ACC); Nikolaus Marx, European Society of Cardiology (ESC); T. Prashant Nedungadi, American Heart Association (AHA); Lars Rydén, European Society of Cardiology (ESC); Helena Rodbard, American Association of Clinical Endocrinologists (AACE); Oliver Schnell, Forschergruppe Diabetes e.V.; Wayne Huey-Herng Sheu, Taichung Veterans General Hospital; Eberhard Standl, Forschergruppe Diabetes, e.V.; Per Olav Vandvik, MAGIC Evidence Ecosystem Foundation (![]() ).
).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sponsors of the Guideline Workshop are listed in alphabetical order: Abbott GmbH, AstraZeneca Pharmaceuticals LP, Bayer AG, Boehringer Ingelheim International GmbH, Eli Lilly and Company, and Novo Nordisk Pharma GmbH. The sponsors had no influence on the workshop, its proceedings and discussions, or on the creation of this article.
