Abstract
The last 25 years of CGM have been characterized above all by providing better and more accurate glucose values in real time and analyzing the measured glucose values. Trend arrows are the only way to look into the future, but they are often too imprecise for therapy adjustment. While AID systems provide algorithms to use glucose values for glucose control, this has not been possible with stand-alone CGM systems, which are most used by people with diabetes. By analyzing the measured values with algorithms, often supported by AI, this should be possible in the future. This provides the user with important information about the further course of the glucose level, such as during the night. Predictive approaches can be used by next-generation CGM systems. These systems can proactively prevent glucose events such as hypo- or hyperglycemia. With the Accu-Chek® SmartGuide Predict app, an integral part of a novel CGM system, and the Glucose Predict (GP) feature, people with diabetes have the first commercially available CGM system with predictive algorithms. It characterizes the CGM systems of the future, which not only analyze past values and current glucose values in the future, but also use these values to predict future glucose progression.
Keywords
Introduction
This year, continuous glucose monitoring (CGM) celebrates the 25th anniversary of its first commercial approval for clinical use. Since then, approximately 8.5 million people worldwide have used CGM, and the annual growth rate is currently around 25%. 1 Experts predict that with further development and lower manufacturing costs, CGM will replace blood glucose self-monitoring in people with diabetes (PwD) and type 1 diabetes (T1D) or type 2 diabetes (T2D) and will also have numerous indications outside of diabetes therapy (eg, prevention of T2D and exercise in healthy people).2-4 The increased uptake of CGM is not always matched with improved way of use and that there is still room for improvement in how CGM can be utilized and that this requires an improved feature set.
Retrospective Analysis
The first professional CGM, the MiniMed Continuous Glucose Monitoring System (CGMS; MiniMed Inc, Northridge, California), approved by the Food and Drug Administration (FDA) in 1999, measured glucose levels between 40 and 400 mg/dL and allowed for three days of retrospective data collection. This led to improved data analysis and pattern recognition and supports the user in making better therapeutic decisions. 5 This “look into the past of glucose values” through the ability to store glucose data, aggregate it into ambulatory glucose profiles (AGPs), and evaluate it according to international consensus recommendations (including average glucose, glycemic variability, percentage of time in, below, and above target range, glucose management indicator) is now a standard feature of all CGM systems on the market. 6 In addition, some CGM systems offer analytical tools for pattern recognition and decision support for therapy adjustment. For health care professionals, retrospective data analysis is still the most important function of CGM to assess the quality of therapy and to make recommendations to PwD on how to adjust their therapy.
Current Information
It is important for PwD and, if the tracking function is activated, for others to monitor their current glucose levels regularly. Most CGM systems also offer features that alert the user to potentially unwanted glucose events (such as hypoglycemia or hyperglycemia). Depending on the given CGM system, these glucose threshold alerts can occur when glucose levels are below or above certain thresholds, when they fall below certain predefined levels (pre-alarms), or when glucose levels rise or fall rapidly (rate-of-change alarms). These alerts allow the CGM user to counteract impending hypoglycemia or hyperglycemia.
Trend Displays of Future Glucose Levels
Most CGM systems provide trend arrows to give the user a graphical prediction of glucose levels in the near future. They show the direction of the glucose trajectory, ie, whether the glucose level will increase, decrease, or remain stable in the future. Trend arrows are based on past glucose data and do not include any other information (eg, insulin dose, food intake). As this is a purely mathematical value, it is currently only of guidance to CGM users for therapy control. Because of the many other influencing factors and the inertia of the trend arrow calculation, a currently measured glucose curve may, eg, indicate an increase, whereas the trend arrow is still pointing down. Nevertheless, trend arrows are often used by PwD.
Prediction of Glucose Levels
To improve usage of CGM systems further, it would be desirable for users not to be disturbed by alarms relatively frequently, which leads to a kind of “alarm inertia.” Such alarms often show up in unfavorable situations (eg, sleep). Instead, attempts are increasingly being made to develop prospective prediction models for the future direction of glucose levels, which are also of help with the prevention of hypoglycemia and hyperglycemia.7-9
To this end, the glucose levels measured by CGM systems are analyzed using computational operations, which are mostly models powered by artificial intelligence (AI) or machine learning. These algorithms are particularly suited to solving problems that are highly nonlinear and where the variables evolve dynamically depending on various influencing factors. In addition, attempts are made to improve the prediction by integrating other influencing factors, such as therapy factors (eg, insulin dose, diet, and exercise), insulin sensitivity, stress, menstruation, concomitant diseases, and medications. 8
Predictive approaches can be used by next-generation CGM systems that empower users to proactively prevent glucose events, such as hypoglycemia or hyperglycemia, from occurring in individuals without the use of an automated insulin delivery (AID) system. In AID systems, such predictive models can help to improve glycemic control by better accounting for the influence of other factors and personalized patterns. Overall, this development will reduce the importance of trend arrows and alarms in CGM systems in the future, which is certainly in the interest of CGM users. In a recent survey of 3,544 PwD in Germany, Austria, and Switzerland, in which they were asked about which aspects of CGM were most important to them when choosing a CGM system, respondents considered the prediction of hypoglycemia to be the most important reason (Figure 1). 10
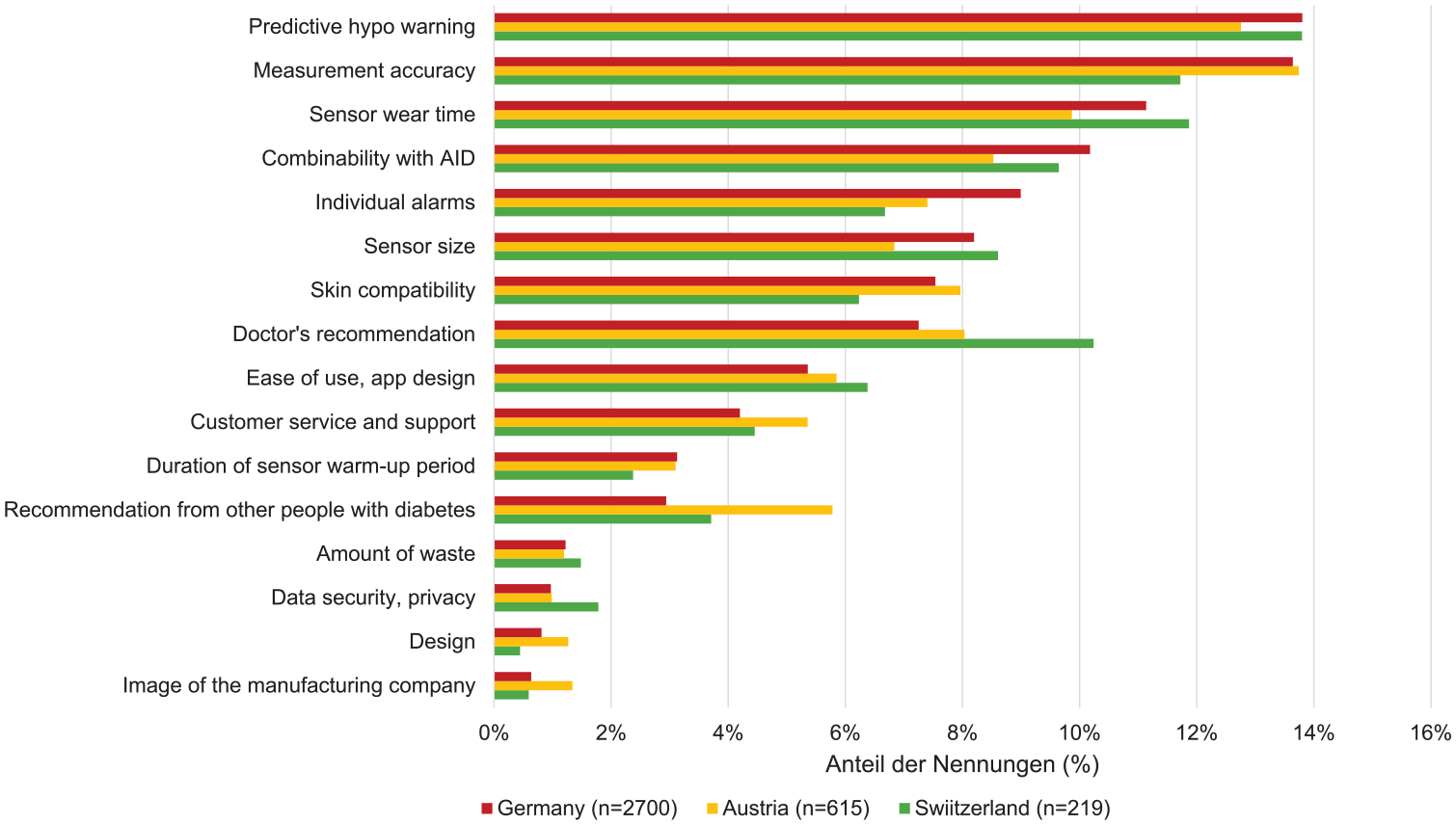
Main reasons for choosing a CGM system (n = 3544 patients with diabetes).
Practical Prediction App in Combination With a Novel Continuous Glucose Monitoring System
This issue of JDST features the Accu-Chek SmartGuide Predict app, which is an integral part of a novel AI enabled CGM system that predicts glucose levels day and night for PwD. It is the first commercially available CGM system to include a specific glucose prediction app. With the Low Glucose Predict (LGP) functionality, the app can warn PwD before low glucose episodes occur, and with the Glucose Predict (GP) function, it visualizes an easy-to-read curve of prdicted glucose excursions within the next 2 hours. In addition, the Night Low Predict (NLP) feature can be used to estimate the likelihood of hypoglycemic events at night before going to bed.
A detailed description of the novel CGM system can be found in the Technology Report in this issue by Glatzer et al. 11 Herrero et al 12 and Mader et al 13 report in two studies on the measurement accuracy of the CGM system and the quality of the prediction. Good measurement accuracy and prediction performances were observed over the entire glucose measurement range, especially for low glucose values. Important questions of clinical practice are discussed in the article by Glatzer et al, 14 such as who benefits from the CGM system and the Prediction app, how prediction is set up in practice, and what therapeutic measures should be initiated in the event of an increased risk of glucose imbalance.
For PwD and their families, nocturnal hypoglycemia is particularly stressful and frightening. Despite the use of CGM, the number of nocturnal hypoglycemic episodes is still too high, and frequent alarms disturb sleep. A review article in this issue of JDST about this topic presents the importance of nocturnal hypoglycemia and discusses possible solutions, such as better predictive tools. Because there are fewer confounding factors (eg, exercise and eating) at night, the prediction of hypoglycemia at night is particularly satisfactory. 15 Nocturnal hypoglycemia is also significant because the episodes last longer compared with daytime hypoglycemia and the risk of glucose levels <54 mg/dL (TB54) and time below range (TBR) is increased. 16
Based on the results of a survey, Ehrmann et al 17 were able to show that the extended glucose predictions are considered important by PwD. Respondents reported that knowing about future glucose levels and being notified about upcoming hypoglycemia events was useful to them, made them feel less anxious, and reduced the burden of diabetes.
Conclusion
Predicting future glucose levels is an important evolution of CGM systems. The Accu-Chek SmartGuide Predict app is part of the first commercially available CGM system with this feature for PwD with TD1 or TD2, in a sense it represents a paradigm shift on how CGM is being used. It aims at empowering PwD in preventing undesirable glucose events such as (nocturnal) hypoglycemia and thus offers a solution to a clinically relevant problem that has not yet been fully solved.
Footnotes
Acknowledgements
The authors are grateful to Dr Guido Freckmann, Professor Oliver Schnell, Dr Ralph Ziegler, and Timor Glatzer for giving advice and input in the course of writing the editorial.
Abbreviations
AGP, ambulatory glucose profile; AI, artificial intelligence; AID, automated insulin delivery; CGM, continuous glucose monitoring; GP, glucose predict; LGP, low glucose predict; NH, nocturnal hypoglycemia; NLP, night low predict; PwD, people with diabetes; T1D, type 1 diabetes; T2D, type 2 diabetes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BK is head of the research institute of the diabetes academy Bad Mergentheim (FIDAM). BK has received speakers’ honoraria or consulting fees from Abbott, Bayer, Berlin Chemie, Dexcom, Embecta, Emperra, Lilly, Novo Nordisk, Roche, Sanofi, and Ypsomed. LH is a shareholder of the Profil Institut für Stoffwechselforschung GmbH, Neuss, Germany, and a consultant for several companies that are developing novel diagnostic and therapeutic options for diabetes treatment.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Roche Diabetes Care GmbH.
