Abstract
In type 1 diabetes (T1D), even short bursts of physical activity can affect blood glucose.1,2 Fear of hypoglycemia is a barrier to exercise participation, and prevention of early and overnight hypoglycemia following activities is a major concern. 3 In our development of closed-loop artificial pancreas systems, we have observed that physical activity can lead to hypoglycemia despite suspension of insulin delivery. 4 Thus, to inform and improve upon these systems, 5 we compared changes in glycemia following 2 sprinting bouts of differing durations.
After signing informed consent, 12 recreationally active, healthy adults (7 F, 5 M) with T1D (42.2 ± 15.4 years, mean ± SD) participated. On day 1, subjects were fitted with a Dexcom G4®Platinum CGM (Dexcom®, San Diego, CA), an ActiGraph wGT3X-BT activity monitor (ActiGraph, Pensacola, FL), and a Polar® heart rate monitor (Polar Electro Oy, Finland). On Days 2 and 4, subjects consumed a Glucerna® Hunger Smart™ Shake containing 14-16 grams carbohydrate for breakfast, for which they took a usual dose of rapid-acting insulin. Three hours later, when insulin and glucose levels were stable, they performed a supervised 10- or 60-second sprint in randomized order at a local track.
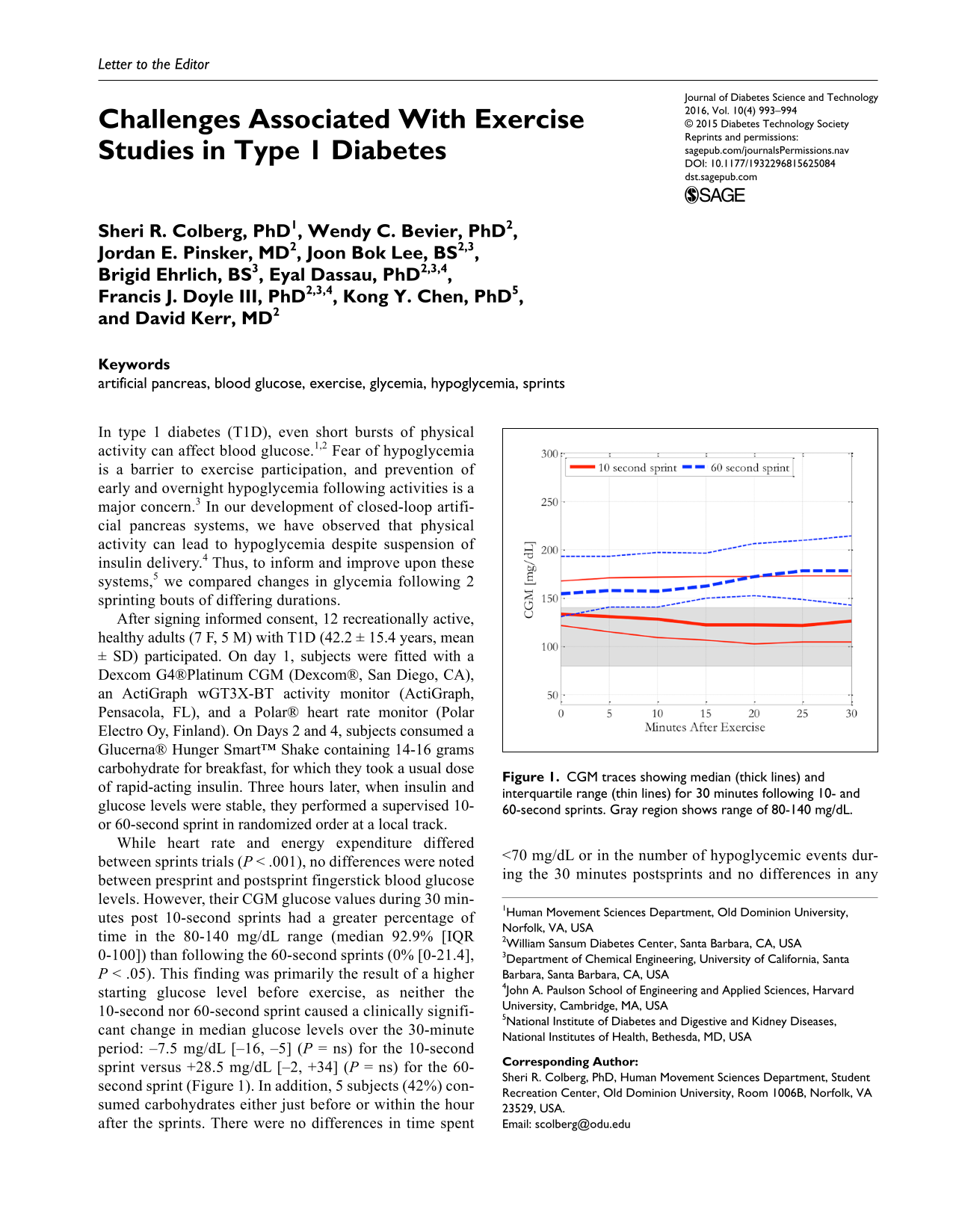
While heart rate and energy expenditure differed between sprints trials (P < .001), no differences were noted between presprint and postsprint fingerstick blood glucose levels. However, their CGM glucose values during 30 minutes post 10-second sprints had a greater percentage of time in the 80-140 mg/dL range (median 92.9% [IQR 0-100]) than following the 60-second sprints (0% [0-21.4], P < .05). This finding was primarily the result of a higher starting glucose level before exercise, as neither the 10-second nor 60-second sprint caused a clinically significant change in median glucose levels over the 30-minute period: –7.5 mg/dL [–16, –5] (P = ns) for the 10-second sprint versus +28.5 mg/dL [–2, +34] (P = ns) for the 60-second sprint (Figure 1). In addition, 5 subjects (42%) consumed carbohydrates either just before or within the hour after the sprints. There were no differences in time spent <70 mg/dL or in the number of hypoglycemic events during the 30 minutes postsprints and no differences in any variables over 1, 3, and 6 hours after sprinting. All subjects had already eaten lunch within 2 hours after each study session.
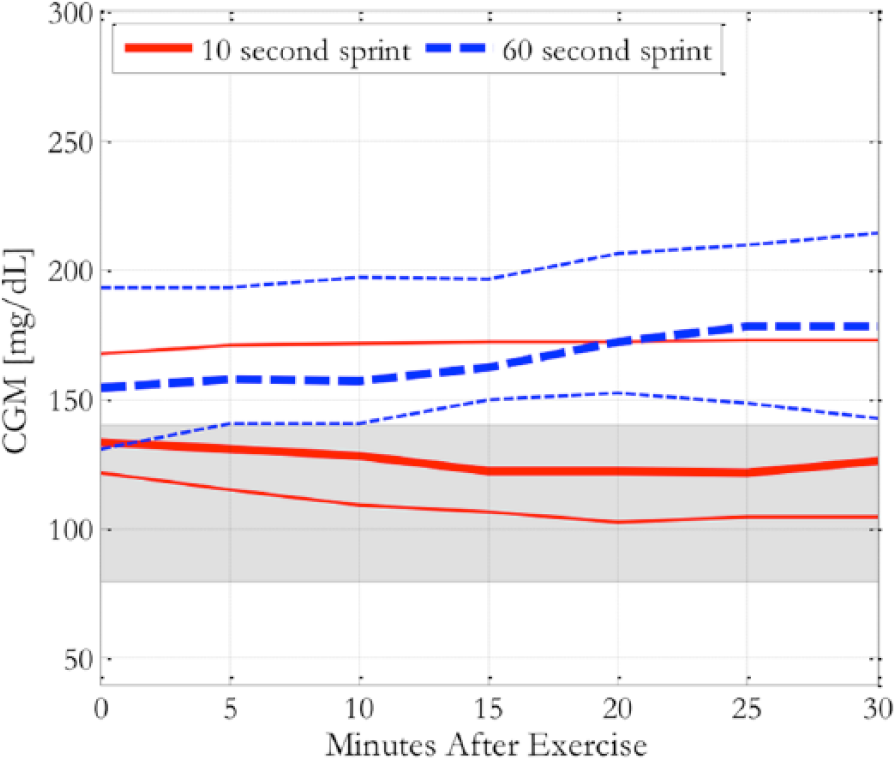
CGM traces showing median (thick lines) and interquartile range (thin lines) for 30 minutes following 10- and 60-second sprints. Gray region shows range of 80-140 mg/dL.
With a small number of subjects, our results suggest that short duration sprints may lead to a drop in blood glucose in persons with T1D, but also highlight the challenges in studying exercise in this group as the need for postexercise interventions (carbohydrate intake) is common. Future studies on exercise in people with T1D under free-living conditions should consider standardized preexercise glucose levels and ideally postexercise carbohydrate intake.
Footnotes
Acknowledgements
We gratefully acknowledge support from the Investigator-Initiated Study Program of LifeScan, Inc, and product support from Dexcom, Inc.
Abbreviations
CGM, continuous glucose monitoring; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to acknowledge financial support from National Institutes of Health Grants R01 DK085628 and DP3 DK094331.
