Abstract
Background:
The aim of this study was to compare glycemic control between SMBG-continued and -discontinued subjects with type 2 diabetes in the SMBG-OHA study.
Method:
Of the 96 subjects from the SMBG-OHA study, 59 were recruited for the 24-week, comparison follow-up study. The study outcomes were the differences in change in HbA1c levels at 24 weeks between the 2 groups, and change in SMBG frequency in SMBG-continued subjects.
Results:
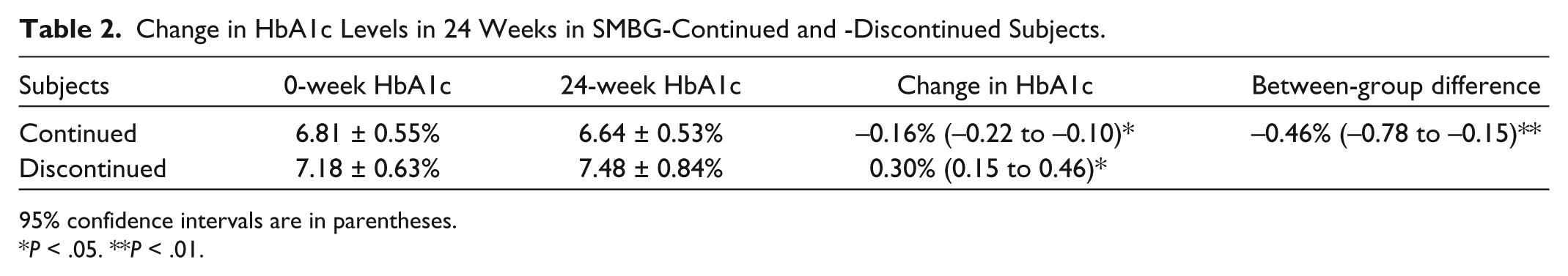
Although health insurance does not cover the cost of SMBG, 22.0% of subjects continued SMBG of their own will after the SMBG-OHA study was completed. HbA1c levels were maintained from 6.81 ± 0.55% to 6.64 ± 0.53% in SMBG-continued subjects. Conversely, HbA1c levels were increased from 7.18 ± 0.63% to 7.48 ± 0.84% in SMBG-discontinued subjects. HbA1c levels were significantly different by 0.83 ± 0.25% (95% CI: −1.33 to −0.36). The difference in change in HbA1c between the groups was −0.46% (95% CI: −0.78 to −0.15). SMBG frequency was decreased from 2.02 ± 1.06 to 1.53 ± 0.86 times a day.
Conclusions:
HbA1c levels were maintained in SMBG-continued subjects but increased in SMBG-discontinued subjects. The study implied that almost once daily SMBG is helpful to maintain glycemic control in non-insulin-treated type 2 diabetes.
Keywords
Self-monitoring of blood glucose (SMBG) is a useful tool for patients with diabetes to maintain glycemic control. Patients are able to prevent hypoglycemia and adjust medications, medical nutrition therapy, and physical activity. 1 SMBG is also beneficial for the health care provider to assess the effectiveness of management plans on glycemic control. 2 Therefore, the International Diabetes Federation (IDF) recommends SMBG for achieving glycemic goals in non-insulin-treated type 2 diabetes. 3 However, its effect on glycemic control is still inconclusive in patients treated less frequently with insulin injection, oral hypoglycemic agents (OHA), or medical nutrition therapy alone.4,5 In the diabetes glycemic education and monitoring study, no significant improvement in glycemic control was found in non-insulin-treated type 2 diabetes not using SMBG with or without instructions in incorporating findings into self care. 6 Conversely, a recent review concluded that SMBG is useful for glycemic control in such patients. 7
We also have reported that SMBG is helpful for glycemic control in OHA-treated type 2 diabetes (SMBG-OHA study).
8
In the study, participants were randomized to 3 groups: no SMBG-performed group (no SMBG group); SMBG-performed group by fingertip blood sampling (fingertip group); and SMBG-performed group by palm blood sampling (palm group), and change in HbA1c at 24 weeks was compared among the 3 groups. HbA1c levels of all subjects at 24 weeks were decreased more in the fingertip (–0.23 ± 0.10%) and palm groups (–0.16 ± 0.06%) than in the no SMBG group (0.31 ± 0.07%) (
Although SMBG is useful for glycemic control in conjunction with comprehensive self-care education, skills training, and ongoing support for patients,9,10 it is important to ask whether or not improved HbA1c levels by SMBG were maintained after patients discontinued SMBG. In the ROSSO-in-praxi follow-up study, 228 of 327 participants completed 2-year follow-up, and 88% of participants have continued to perform SMBG for 2 years.
11
Compared with the baseline value, participants who performed SMBG daily (n = 46) demonstrated an overall HbA1c reduction of 0.28 ± 1.21% on average at the end of follow-up. Participants who performed SMBG several times a week (n = 79) or irregularly as needed (n = 76) showed a nearly constant HbA1c with slight reduction of 0.11 ± 0.68% and 0.09 ± 0.70%, respectively. In contrast, participants who discontinued SMBG (n = 46) demonstrated a marginal increase in HbA1c of 0.07 ± 1.19%. Significant differences in change in HbA1c were not observed among the groups. However, borderline tendency was demonstrated between those groups with daily and no performance of SMBG (
Here, we conducted a follow-up study to examine the constant effect of SMBG on glycemic control in subjects of the SMBG-OHA study. In the follow-up study, subjects could continue or discontinue performing SMBG of their own will.
Methods
Patients and Methods
The SMBG-OHA follow-up study was a 24-week, single-center cohort study to evaluate the efficacy of SMBG on glycemic control in OHA-treated type 2 diabetes. Participants of the SMBG-OHA study in Kyoto University Hospital continued or discontinued SMBG of their own will after the SMBG-OHA study was completed. Participants were not instructed about how and when they did SMBG and how to interpret and act on their SMBG data. The study protocol was approved by the Institutional Review Board of Kyoto University Hospital, and registered on the University Hospital Medical Information Network in Japan (UMIN000001525). Written informed consent was obtained from all subjects.
Measurements
The primary endpoint was the difference in change in HbA1c at 24 weeks between SMBG-continued and -discontinued subjects. The secondary endpoints were the difference in HbA1c levels at 24 weeks between SMBG-continued and -discontinued subjects, and change in SMBG frequency in SMBG-continued subjects.
Statistical Analysis
HbA1c outcomes were assessed blindly for statistical analysis. Paired
Results
Subjects
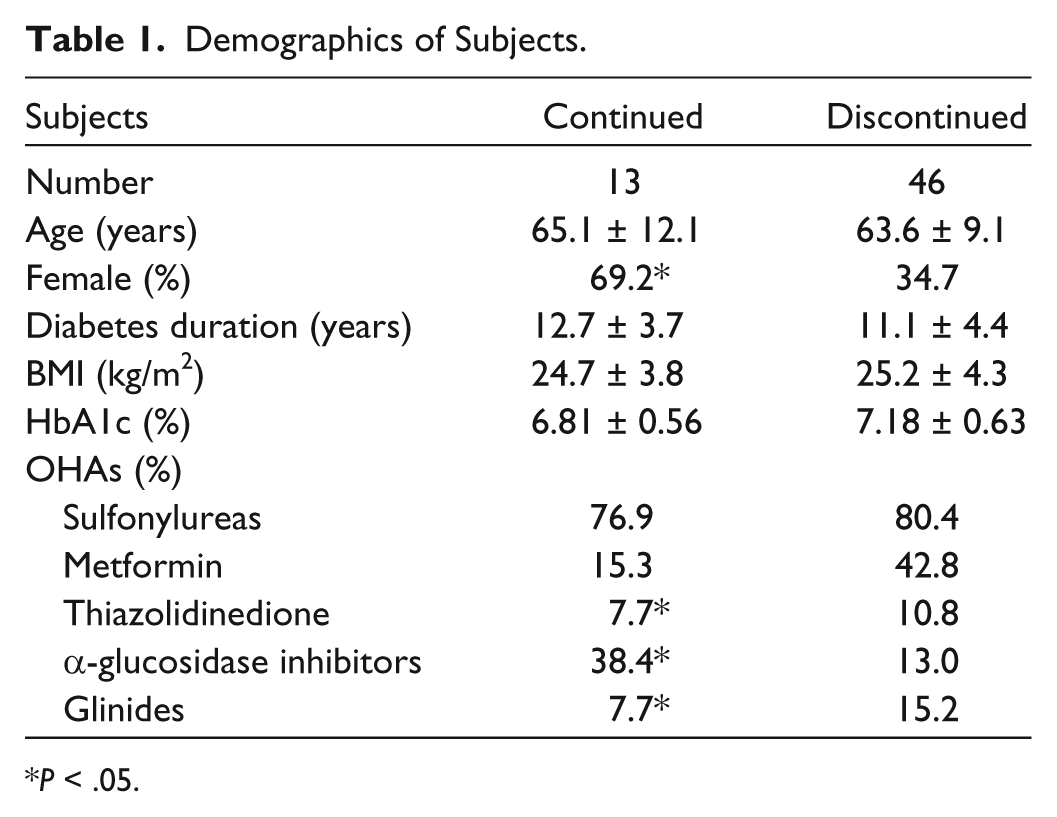
Of the 96 subjects who completed the SMBG-OHA study, 59 were enrolled into the follow-up study: 32 subjects in the fingertip group and 27 subjects in the palm group. Eight of 32 subjects (25.0%) in the fingertip group continued SMBG, and 5 of 27 subjects (18.5%) in the palm group continued SMBG. There was no significant difference in the ratio of SMBG-continued subjects between the fingertip and palm groups. Finally, 13 subjects were allocated to the SMBG-continued group and 46 subjects to the SMBG-discontinued group. Average age (mean ± SD) of SMBG-continued and -discontinued groups was 65.1 ± 12.1 years and 63.6 ± 9.1 years, respectively; ratio of females was 69.2% and 34.7% (
Demographics of Subjects.
HbA1c Findings
HbA1c levels were maintained in SMBG-continued subjects, from 6.81 ± 0.55% to 6.64 ± 0.53% (Table 2). Conversely, HbA1c levels were increased from 7.18 ± 0.63% to 7.48 ± 0.84% in SMBG-discontinued subjects. HbA1c levels were significantly lower by 0.83 ± 0.25% (95% CI: −1.33 to −0.36,
Change in HbA1c Levels in 24 Weeks in SMBG-Continued and -Discontinued Subjects.
95% confidence intervals are in parentheses.
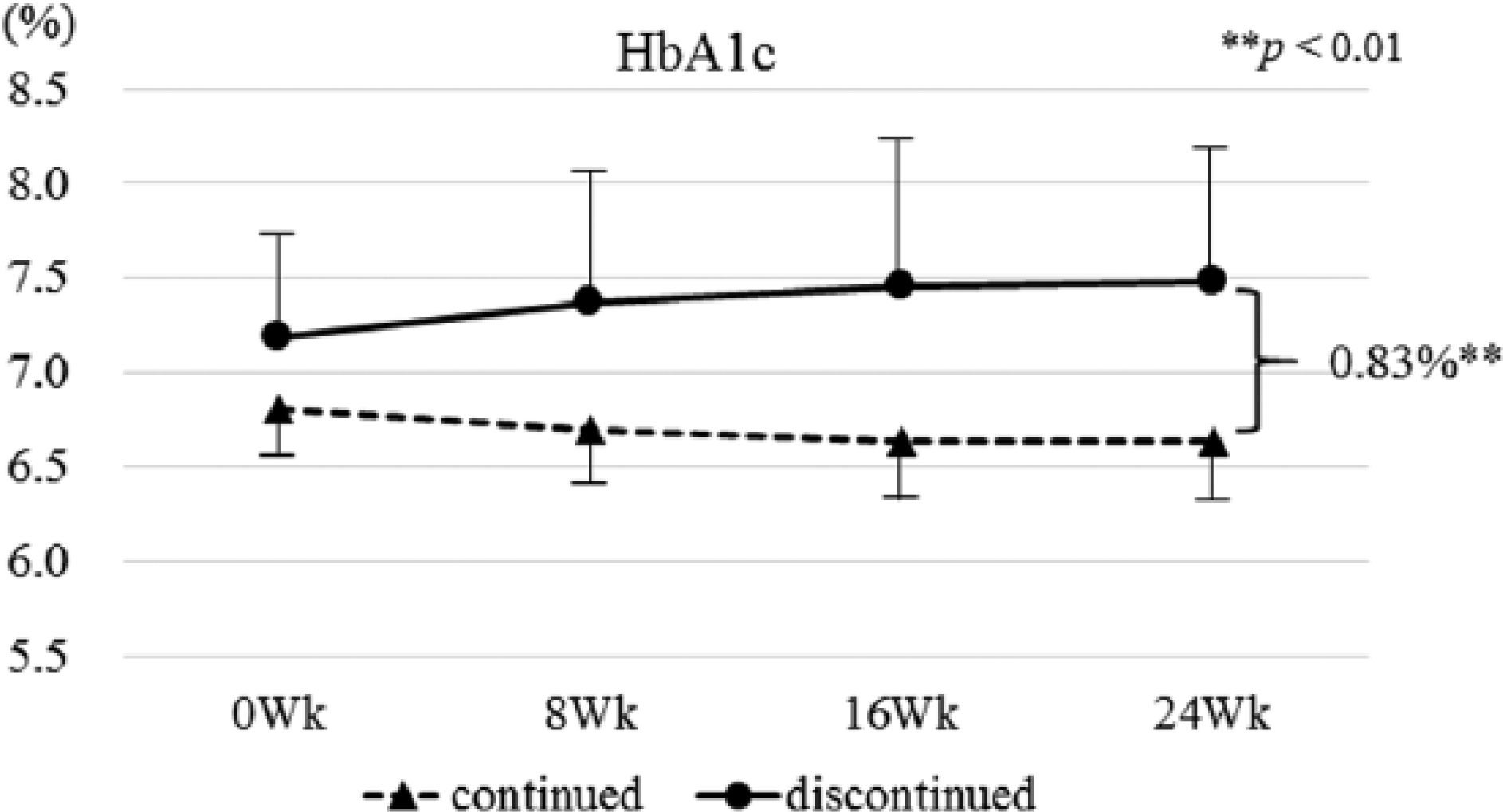
HbA1c levels during 24 weeks in SMBG-continued and discontinued subjects. Independent sample Student’s
Interestingly, 5 of 37 subjects (13.5%) in the no SMBG group of the SMBG-OHA study newly started SMBG (data not shown) after the study was completed. Their HbA1c levels were decreased from 7.12 ± 0.64% to 6.86 ± 0.70% (
SMBG Frequency
SMBG was regularly performed in SMBG-continued subjects of the fingertip and palm groups. SMBG frequency was 1.48 ± 1.00 times a day in the fingertip group and 1.60 ± 0.68 times a day in the palm group (Table 3). Overall, SMBG frequency of SMBG-continued subjects was 1.53 ± 0.86 times a day. SMBG frequency of both groups was significantly decreased during the follow-up period, but there was no significant difference in SMBG frequency between the fingertip group and the palm group. In addition, SMBG frequency was 1.04 ± 0.80 times a day in newly SMBG-started subjects of the no SMBG group.
SMBG Frequency.
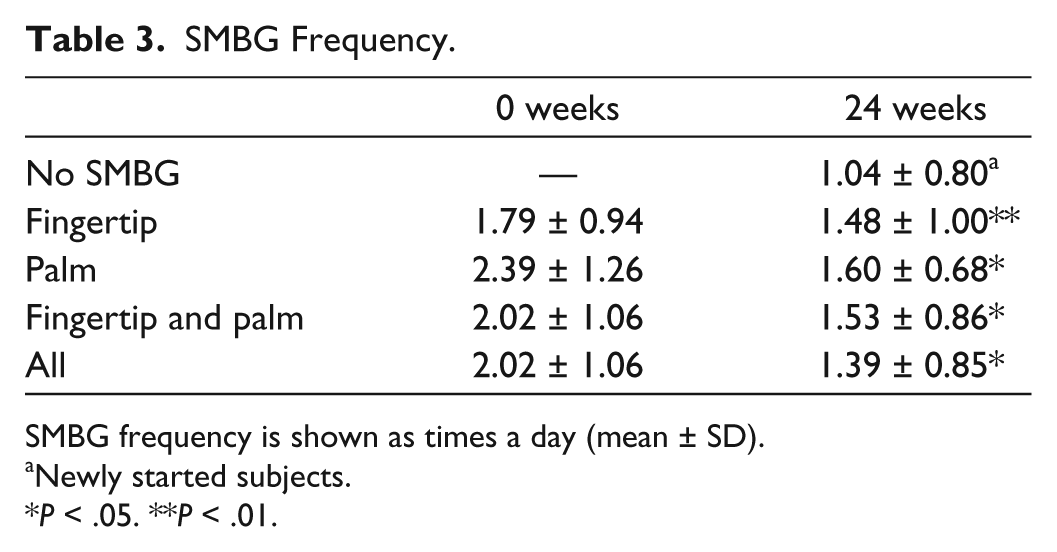
SMBG frequency is shown as times a day (mean ± SD).
Newly started subjects.
Discussion
In the present study, we showed that SMBG is useful for maintaining glycemic control in OHA-treated type 2 diabetes patients. HbA1c levels of SMBG-continued subjects were 6.64 ± 0.53%, which achieved the HbA1c target of <7.0%.12,13 However, in SMBG-discontinued subjects, HbA1c levels worsened from 7.18 ± 0.63% to 7.48 ± 0.84%, which were the baseline levels of original registration in the SMBG-OHA study. Thus, to keep glycemic control, it is better for patients to continue SMBG routinely.
In the follow-up study, SMBG frequency was 1.48 ± 1.00 times a day in the fingertip group and 1.60 ± 0.68 times a day in the palm group. The average of SMBG frequency in SMBG-continued subjects was 1.53 ± 0.86 times a day, and HbA1c levels were stably less than 7.0%. In the SMBG-OHA study, reduction of HbA1c levels was negatively correlated to SMBG frequency. 8 Twice daily SMBG improved HbA1c levels by about 0.5%, and once daily SMBG was the minimum required for maintenance of HbA1c levels. These results indicated that at least once daily SMBG is helpful to maintain glycemic control in non-insulin-treated type 2 diabetes.
It has been reported that SMBG had a positive or negative impact on quality of life, well-being, and patient satisfaction.14,15 An SMBG frequency of ≥1 per day is significantly related to higher levels of distress, worry, and depressive symptoms in non-insulin-treated patients. 16 The ZODIAC-17 study also showed that OHA-treated type 2 diabetes patients experienced some worsening of their personal health perception. 17 Franciosi et al reported that an SMBG frequency ≥ 1 time per day was significantly related to higher levels of frustration, worries, and depression symptoms in non-insulin-treated patients because educating patients about how to interpret and act on their SMBG data was little instructed by primary care physicians. 18 Conversely, the ROSSO-in-praxi follow-up study demonstrated that daily SMBG had beneficial long-term effects on glycemic control, weight, and quality of life. 19 In addition, structured SMBG (SteP trial) can significantly improve glycemic control without decreasing general well-being. 20 This recent evidence indicates that when health care providers assess SMBG data carefully with patients and support their diabetes through timely self-management, patients’ quality of life (QOL) and well-being are not distressed.
Another negative aspect of SMBG is cost. Annual diabetes cost has increased all over the world. In Japan, 1 test strip is about 1.2 dollars and a lancet is about 10 cents. If a patient does SMBG twice a day, it costs at least 78 dollars a month. However, SMBG once or twice per day was found to be cost-effective. 21 It was associated with a significantly reduced number of hospitalizations and reduced overall duration of hospitalization for patients with diabetes. Nonetheless, low SMBG frequency minimizes diabetes-associated cost. Recently, we reported that color record in SMBG has a favorable effect on self-management performance without any influence on psychological stress and results in improved glycemic control in less frequently insulin-treated type 2 diabetes patients. 22 To promote improvement in self-management and glycemic control, a simpler, more efficient, and more economical method of SMBG, such as color record, should be considered as a next step in OHA-treated type 2 diabetes.
There are limitations to our study. First, although continuation of SMBG tests was dependent on subjects’ will, the study population was small. The main reason why the participants did not continue SMBG was the cost. However in the SteP trial 20 and the ROSES trial, 23 patients are willing to test more frequently when they can see the cause and effect of their health behaviors and medications. Therefore, further studies involving a larger number of patients are needed to investigate if patients are willing to test more frequently at their own expense. Second, subjects were recruited from only a single center, so that regional differences were not included in our study. Third, psychological aspects and satisfaction of subjects were not evaluated in the study. Hence, it should be evaluated if characteristics of patients, such as education levels and family income, affect SMBG performance and glycemic control, and questionnaires such as the Summary of Diabetes Self-Care Activities Measure (SDSCA) and Profile of Mood States (POMS) should be addressed in the next study.
Conclusion
The SMBG-OHA follow-up study indicated that glycemic control is maintained by continuing SMBG in non-insulin-treated type 2 diabetes. However, when SMBG was discontinued, glycemic control was worsened. The study also showed that almost once daily SMBG is helpful to keep the effect of SMBG on glycemic control. Thus, to maintain or improve glycemic control in OHA-treated type 2 diabetes, continued SMBG is recommended. However, when patients stop SMBG, health care providers should provide timely and supportive care to maintain their glycemic control stability.
Footnotes
Acknowledgements
Parts of this study were presented as a poster presentation at the 8th International Conference on Advanced Technologies & Treatments for Diabetes (ATTD), Paris, France (February 18-15, 2015). Clinical Trial Number: UMIN000001525.
Abbreviations
IDF, International Diabetes Federation; OHA, oral hypoglycemic agents; POMS, Profile of Mood States; QOL, quality of life; SDSCA, Summary of Diabetes Self-Care Activities Measure; SMBG, self-monitoring of blood glucose; UMIN, the University Hospital Medical Information Network in Japan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
