Abstract
The role for the novel treatment approach of sodium-glucose cotransporter-2 (SGLT-2) in type 2 diabetes is increasing. Structured self-monitoring of blood glucose (SMBG), based on a less intensive and a more intensive scheme, may contribute to an optimization of SGLT-2 inhibitor based treatment. The current expert recommendation suggests individualized approaches of SMBG, using simple and clinically applicable schemes. Potential benefits of SMBG in SGLT-2 inhibitor based treatment approaches are early assessment of treatment success or failure, timely modification of treatment, detection of hypoglycemic episodes, assessment of glucose excursions, and support of diabetes management and education. The length and frequency of SMBG should depend on the clinical setting and the quality of metabolic control.
The first sodium-glucose cotransporter-2 (SGLT-2) inhibitors for the therapy of type 2 diabetes have been approved in the US and Europe. The role of the novel treatment approach is largely increasing. Although self-monitoring of blood glucose (SMBG) has been performed in numerous studies on SGLT-2 inhibitors, the potential role for SMBG in SGLT-2 inhibitor based treatment strategies has not been elaborated.
In 2010, two SMBG schemes of varying intensity across the type 2 diabetes continuum have been recommended by a European Expert Panel formed by experts in diabetology and endocrinology. 1 Based on this, recommendations for individualized SMBG strategies in glucagon like peptide-1 (GLP-1) based treatment approaches have been presented. 2
The aim of this article is to discuss individualized SMBG strategies for SGLT-2 inhibitor based treatment approaches also in light of SMBG schemes, which have been introduced in guideline and expert recommendations.1-3 We also aimed to present clinical scenarios for the need of SMBG. Potential scenarios may include early assessment of treatment success or failure, the need for timely modification of treatment, assessment of glucose excursions, and support of diabetes management and education.
SGLT-2 Inhibition - The Current Status
Recently, the role of the kidneys in glucose homeostasis has gained increasing attention. The kidneys play a significant role in carbohydrate metabolism. 4 Among others, an important task is the absorption of filtered glucose: With a glomerular filtration rate of 180 L per day and a plasma glucose concentration of 5 mmol/L (90 mg/dL), the kidneys filter approximately 162 g (900 mmol) of glucose per day, thereby contributing to maintenance of physiological fasting plasma glucose (FPG) levels. 4 In this adaptive system, SGLT-2 is responsible for 80–90% of renal glucose reabsorption. 4 In patients with type 2 diabetes, expression and activity of SGLT-2 are increased, thereby enhancing glucose reabsorption by up to 20%. 4 SGLT-2 inhibitors block the renal glucose reabsorption, thereby triggering glycosuria and decreasing blood glucose levels. The occurrence of treatment-induced glycosuria in patients receiving SGLT-2 inhibitors makes the assessment of glucose in the urine for control of glycemia improper.
Favorable effects on glycosylated hemoglobin A1c (HbA1c) levels, reductions in blood pressure and weight, and very low rates of hypoglycemia have recently been reported in reviews and meta-analyses5-8 which confirm preceding publications.9-18 Also a 3–6 mm Hg reduction in systolic blood pressure and a reduction in weight, both induced by the loss of glucose via the urine, have been observed.6-8,19 In rodent models, canagliflozin treatment for 6 to 12 months was demonstrated to improve measures of beta cell function. 20 SGLT-2 inhibition, however, may increase the risk of urinary tract and genital infections; further studies are needed to explore long-term efficacy and safety.6,21
Canagliflozin (US) and dapagliflozin (Europe) were the first two SGLT-2 inhibitors to be approved for treatment of type 2 diabetic patients. Empagliflozin is currently being reviewed for approval. Further substances such as ertugliflozin, ipragliflozin, and tofogliflozin are yet under development.
Glycemic Control and Hypoglycemia in Monotherapy with SGLT-2 Inhibitors
Assessing efficacy and safety of SGLT-2 inhibitor based monotherapy, several studies lasting 12 to 26 weeks have been conducted. In type 2 patients inadequately controlled with diet and exercise, canagliflozin 100 and 300 mg was found to reduce HbA1c significantly as compared to placebo (−0.77%/−1.03% vs +0.14%). 22 At week 26, differences in least square (LS) mean changes in FPG of −2.0 and −2.4 mmol/L (−36 and −43 mg/dL) for canagliflozin 100 and 300 mg relative to placebo were observed. 22 Substantial reductions after a mixed-meal tolerance test were demonstrated with canagliflozin 100 (1-hour postprandial: LS mean change −2.7 mmol/L [–49 mg/dL]) and 300 mg (2-hour postprandial −3.6 mmol/L [−65 mg/dL]). 22 The rates of documented hypoglycemia were similar between canagliflozin 100 and 300 mg and placebo (3.6%, 3.0% and 2.6%, respectively). 22
In antidiabetic drug-naïve patients with inadequate glycemic control, significant reductions in HbA1c ranging from 0.58–0.89% were demonstrated with dapagliflozin 1 mg, 2.5 mg, 5 mg, and 10 mg.23,24 Significant reductions in FPG of 0.84, 1.34, and 1.6 mmol/L (15.2, 24.1, and 28.8 mg/dL) were found at week 24 with dapagliflozin 2.5 mg, 5 mg, and 10 mg. 23 Rates of hypoglycemia were in a similar range as in placebo groups.23,24
Treatment with empagliflozin 5 mg, 10 mg, and 25 mg was associated with an significant HbA1c reduction after 12 weeks of 0.4%, 0.5%, and 0.6%, respectively. 25 A decrease in FPG of 1.29, 1.61, and 1.72 mmol/L (23, 29, and 31 mg/dL) was associated with empagliflozin 5 mg, 10 mg, and 25 mg versus placebo (+0.04 mmol/L). 25 Rates of hypoglycemia did not differ between empagliflozin and placebo. 25
Glycemic Control and Hypoglycemia in Combination Therapies with SGLT-2 Inhibitors
Combination With Metformin
Add-on therapy with dapagliflozin was evaluated in adults with type 2 diabetes, having inadequate glycemic control with daily metformin (≥ 1500 mg per day). At week 24, an HbA1c reduction of 0.67%, 0.70% and 0.84% versus 0.30% was identified in patients receiving dapagliflozin 2.5 mg, 5 mg and 10 mg once daily versus placebo. 26 The long-term extension (102 weeks) of this study revealed HbA1c reductions of 0.48% (2.5 mg dose), 0.58% (5 mg), and 0.78% (10 mg) at 104 weeks versus an increase of 0.02% in placebo group. 27 In all dapagliflozin groups, at 102 weeks FPG reductions ranging from −1.07 to −1.47 mmol/L (−19 to −26 mg/dL) were seen, whereas increases were noted in placebo-treated patients. 27 No major episodes of hypoglycemia have been reported, with similar rates of hypoglycemia across all 4 treatment groups. 27
Efficacy and safety of canagliflozin versus glimepiride were assessed in type 2 diabetic patients with HbA1c levels 7.0–9.5% on stable metformin treatment. 28 Regarding lowering of HbA1c at 52 weeks, canagliflozin 100 mg was noninferior to glimepiride (LS mean difference −0.01% [95% CI –0.11 to 0.09]), while canagliflozin 300 mg proved to be superior to glimepiride (−0.12% [−0.22 to −0.02]). 28 In FPG, LS mean reductions versus baseline of 1.02, 1.35, and 1.52 mmol/L (18, 24, and 27 mg/dL) were seen with glimepiride, canagliflozin 100 and 300 mg, respectively. 28 Treatment with canagliflozin 100 mg and 300 mg was associated with significantly less hypoglycemic episodes as compared with the administration of glimepiride. 28 HbA1c reductions of up to 0.95% related to the dosage of canagliflozin were demonstrated also in a trial, which assessed patients inadequately controlled with metformin. 29 In the study, FPG was decreased by 0.89 to 1.5 mmol/L (16 to 27 mg/dL). 29 The incidence of symptomatic hypoglycemia was low and generally similar across active and placebo treatment groups. 29 In a 52-week phase 3 study, additional treatment with canagliflozin (triple therapy) significantly improved glycemic control in patients with type 2 diabetes which were inadequately controlled with metformin and pioglitazone. 30 Systolic BP and body weight were also reduced while a low incidence of documented hypoglycemia was found. 30
Combination with Sulfonylurea
Treatment with sulfonylureas (SU) has been reported to be associated with an increased risk of hypoglycemia.31-34 A systematic review on SU monotherapy and it’s use as an add-on to other glucose-lowering agents reported mild-to-;moderate hypoglycemic episodes to be significantly more frequent in SU treated patients than in the comparator groups (RR 2.41, 95% CI 1.41, 4.10). 35 Efficacy and safety of dapagliflozin (2.5, 5, or 10 mg/day) added to glimepiride (4 mg/day) were analyzed in patients with HbA1c levels of 7–10% previously treated with a monotherapy of glimepiride. 36 HbA1c decreased by 0.58%, 0.63%, and 0.82% with dapagliflozin 2.5 mg, 5 mg, and 10 mg, respectively (placebo: –0.13%). 36 Reductions from baseline in mean FPG of 1.18 mmol/L [21 mg/dL] (dapagliflozin 5 mg) and 1.58 mmol/L [28 mg/dL] (dapagliflozin 10 mg) versus 0.11 mmol/L [2 mg/dL] (placebo) were reported. 36 Hypoglycemic events were more frequent in the dapagliflozin groups (6.9-7.9%) compared with the placebo group (4.8%). 36
In a 52-week randomized trial with type 2 diabetic patients, who were inadequately controlled with a combination of metformin and sulfonylurea, canagliflozin was added versus the dipeptidyl peptidase-4 (DPP-4) inhibitor sitagliptin. 37 Addition of canagliflozin 300 mg achieved significantly greater reductions in HbA1c and FPG than sitagliptin 100 mg. 37 Treatment with canagliflozin 300 mg was related to LS mean reductions in HbA1c of 1.03%, FPG of 1.59 mmol/L (28.7 mg/dL), and 2-hour postprandial blood glucose of 3.25 mmol/L (58.5 mg/dL), respectively. 37 Within 52 weeks 43.2% of patients experienced ≥ 1 hypoglycemic episode. 37 In conclusion, an increased risk for hypoglycemia due to treatment with SU has to be considered.
Combination with Insulin
Evidence from several recent studies indicates an increased risk of hypoglycemia in insulin-treated patients with diabetes. In patients with type 2 diabetes treated for >5 years with insulin, a 25% prevalence of severe hypoglycemia has been reported. 38 In patients treated with newer basal insulin analogues, lower rates of severe hypoglycemic events requiring inpatient or emergency department care were seen as compared to users of other insulin formulations. 39 Add-on therapy with dapagliflozin 2.5, 5, or 10 mg was also explored in more than 800 patients with type 2 diabetes who were inadequately controlled with insulin (HbA1c 7.5–10.5%). 40 At 24 weeks, mean HbA1c was decreased by 0.79% to 0.96% with dapagliflozin compared to 0.39% with placebo. 40 Daily insulin doses could be reduced by 0.63 to 1.95 IU with dapagliflozin, whereas an increase by 5.65 IU became necessary with placebo. 40 In the pooled dapagliflozin groups, hypoglycemic episodes were seen with a rate of 56.6%, as compared to 52.8% in the placebo group. 40 At least, however, an increased risk of hypoglycemia has to be considered in patients who are treated with a combination of SGLT-2 inhibitors and insulin.
Rationale for SMBG in SGLT-2 Inhibitor Based Treatment Approaches
SGLT-2 inhibitor based treatment approaches are associated with considerable reductions in HbA1c and FPG. Low rates of hypoglycemic episodes were seen in monotherapies and comedication with metformin. More frequent hypoglycemic episodes, however, were recognized in combined treatment approaches with sulfonylurea and, particularly, insulin.
In type 2 diabetes, SMBG is a beneficial approach to assess blood glucose responses related to medications, nutrition, intercurrent illnesses, and lifestyle modifications. It also supports the prevention of acute and chronic complications.1,41 SMBG also enables the visualization of hypoglycemic episodes42,43 and is considered as a potential tool for identifying risk scenarios for hypoglycemia. 44 Its value with view on the detection of postprandial glucose excursions and glycemic variability is increasing.45,46
A 14-year follow up of more than 500 patients with type 2 diabetes found 2-hour blood glucose levels but not FPG to predict cardiovascular events and all-cause mortality. 47 The role of SMBG in type 2 diabetes is also supported by the growing evidence for the association between glycemic variability and an increased risk of endothelial dysfunction, cognitive impairment,48,49 micro- and macrovascular complications, and mortality.50,51 Improvement of blood glucose excursions, mainly in the postprandial phase, have been hypothesized to be beneficial for the outcome. 51 Glycemic variability can be visualized very well by SMBG.52,53
The St Carlos Study explored more than 160 newly diagnosed type 2 diabetic patients. The group with SMBG-based educational and pharmacological intervention achieved significantly greater reductions in median HbA1c (6.6% to 6.1%;
The STeP (Structured Testing Protocol) Study, which enrolled 483 poorly controlled (HbA1c ≥ 7.5%), insulin-naïve people with type 2 diabetes, revealed significantly greater reductions in mean HbA1c in the structured testing group compared to the active control group.
46
At 1 year, the SMBG group showed a significantly greater mean reduction in HbA1c (−1.2% vs −0.9%;
Due to a Cochrane review, SMBG in non-insulin treated patients with type 2 diabetes is associated with a mean HbA1c reduction of 0.3% at 6-month follow-up in patients with a diabetes history of > 1 year, a nonsignificant mean reduction of HbA1c (0.1%) at 12-month follow-up in patients with a diabetes history of > 1 year, and a significant mean reduction of HbA1c (0.5%) at 12-month follow-up in patients with newly diagnosed type 2 diabetes. 54
The UK Prospective Diabetes Study (UKPDS) demonstrated a 1% reduction in HbA1c to be associated with a 37% decrease in risk for microvascular complications and a 21% decrease in the risk of any end point or death related to diabetes. 55 According to the UKPDS risk engine, an HbA1c reduction of 0.3% can be assumed to translate into an approximately 0.38% reduction in coronary heart disease. 56
The rationale for the potential role of SMBG in SGLT-2 inhibitor based treatment approaches includes 5 aspects:
1. Early assessment of treatment efficacy
In patients with HbA1c baseline levels < 8%, reductions in HbA1c related to SGLT-2 inhibition have been reported to be less pronounced than in patients with baseline HbA1c levels ≥ 9%. 57 SMBG may be superior in assessing early glycemic effects and also potential treatment failure as compared to HbA1c. In patients treated with SGLT-2 inhibitors, reductions in FPG are reported to be apparent as early as week 1 of treatment. 23 HbA1c, however, reflects glucose concentrations over the previous 4 to 8 weeks, 58 whereas SMBG provides day-to-day information. 59
2. Early modification of treatment
Treatment escalation in type 2 diabetes frequently is delayed. 60 Of patients 60–70% experience long-term increases in HbA1c above 7.0%. 60 Appropriate usage of structured SMBG, however, is promoting more timely/aggressive treatment changes in non-insulin treated type 2 diabetes.45,46 In insulin-treated patients, SMBG provides guidance on making changes to ongoing therapy (ie, altering the dose, timing, or frequency of basal insulin or making changes in therapy regimen), and supports immediate therapeutic decisions, such as adjusting the dose of rapid-acting insulin related to a meal. 61
3. Detection of hypoglycemic episodes
In monotherapy settings22 -25 and in combination with metformin,26-29,62 SGLT-2 inhibitor based treatment approaches are characterized by very low rates of hypoglycemia. Addition of SGLT-2 inhibitors to sulfonylurea or insulin, however, increases rates of hypoglycemia remarkably.36,37,40 Because SGLT-2 inhibitors are often prescribed in addition to sulfonylureas or insulin, we may expect increased rate of hypoglycemic events that could be prevented by comprehensive SMBG.
4. Assessment of glucose excursions (glycemic variability)
Glycemic variability is suggested to substantially contribute to diabetic complications independently of HbA1c levels.51,63,64 SMBG has been evidenced to be suitable in detecting postprandial glycemic excursions.52,53,65 Despite the evidence suggesting SGLT-2 inhibitor based treatment approaches to reduce postprandial glycemic excursions, 14 glucose excursions still remain present.
5. Support of diabetes management and diabetes education
SMBG-based visualization of changes in blood glucose due to medications, life style changes, physical activities and food intake facilitates diabetes education. Patients’ awareness of the disease as well as adherence to lifestyle modification and treatment may be improved. 42
Recommendations for Schemes of SMBG in SGLT-2 Inhibitor Based Treatment Approaches
Two schemes for a periodical performance of SMBG are recommended for SGLT-2 inhibitor based therapies: scheme 1 is aimed for less intensive testing (Figure 1), scheme 2 (Figure 2) for more intensive testing.
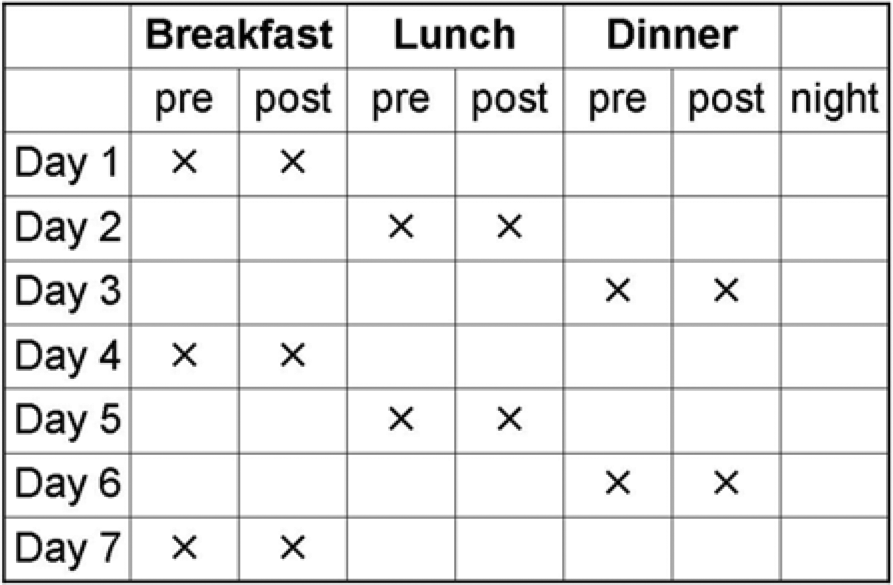
Scheme 1: Example of less intensive, paired testing in type 2 diabetes. The scheme focuses on paired meal testing (pre- and postprandial) once per day to identify trends of glucose levels in response to food intake. Frequency/duration of testing, according to individual aspects: 1 paired meal testing monthly, 1 week paired testing monthly/3-7 days paired testing weekly. 1
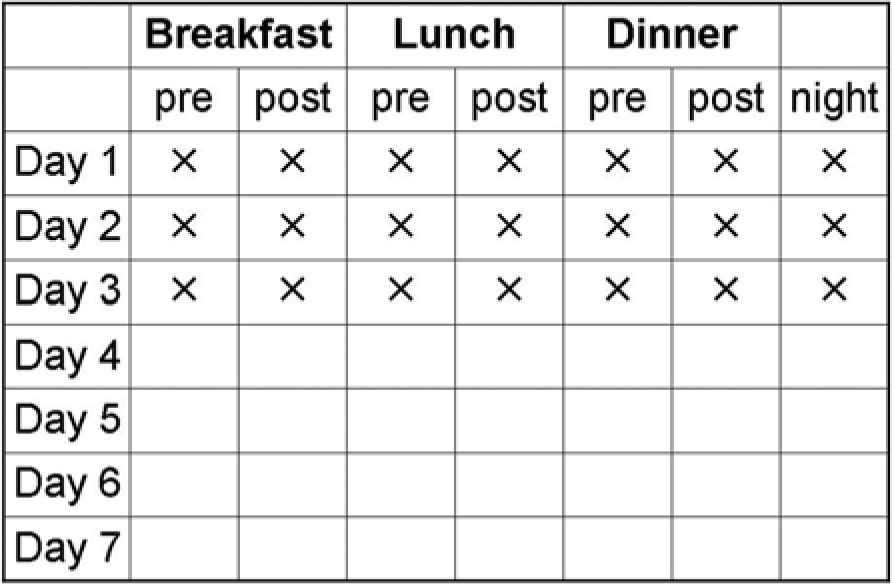
Scheme 2: More intensive testing. Seven tests per day over a minimum of 3 days up to 7 days for educational purposes, or for adjustment of therapy, or evaluation of diet. The scheme focuses on the dynamics of glucose levels per day to identify the variability of glucose levels. Frequency/duration of testing: minimum 3 days monthly. 1
The recommendations also consider the recommendations of the IDF Guideline on “Self-Monitoring of Blood Glucose in Non-Insulin Treated Type 2 Diabetes.” 3 The IDF Guidelines on Postmeal Glucose Management include postprandial targets of < 9.0 mmol/L (< 160 mg/dL) taken 1 or 2 hours after a meal. 66
The schemes are recommended for various treatment scenarios (Figure 3):
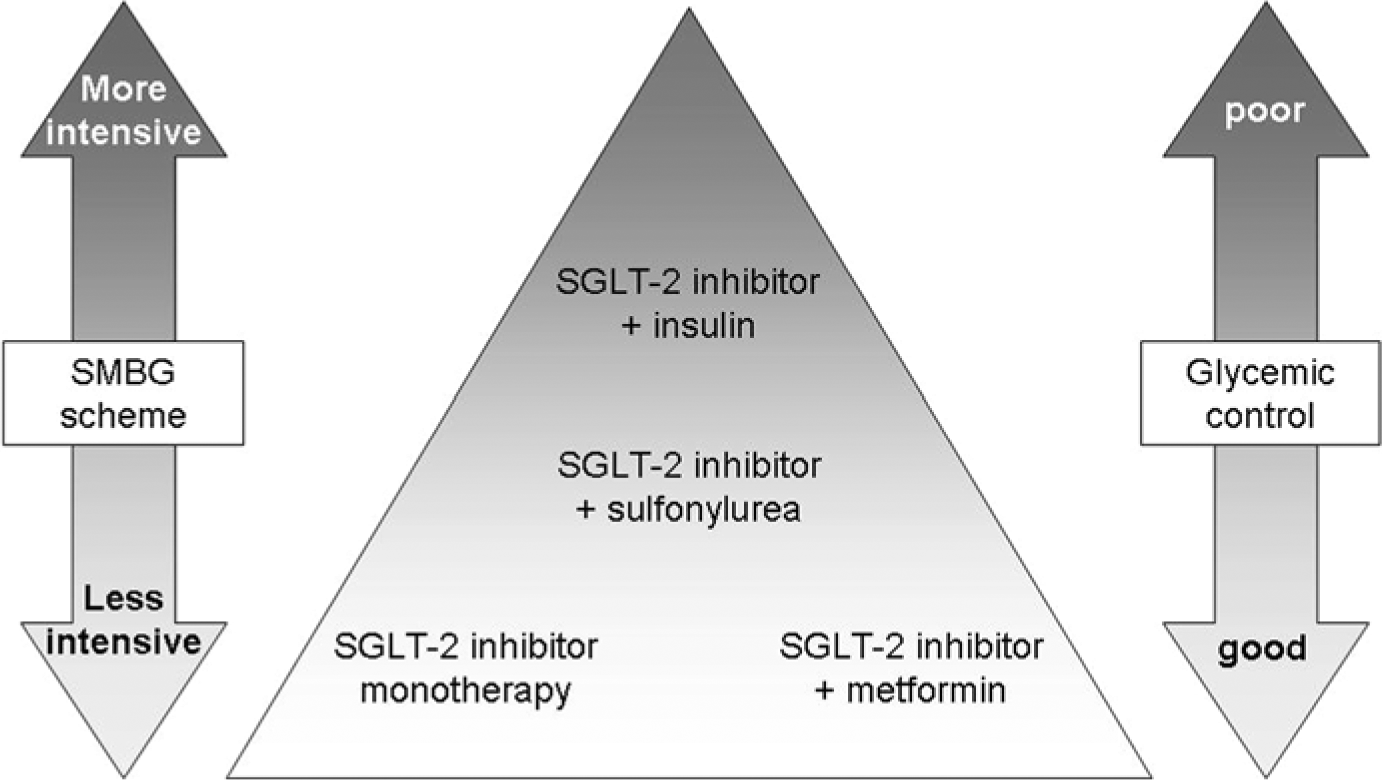
Allocation of scheme 1 and scheme 2 to different clinical scenarios. Increasing intensity of testing with treatment escalation and deterioration of glycemic control. Declining intensity with treatment de-escalation and improvement of glycemic control.
Initiation of SGLT-2 inhibitor based monotherapy and combination with metformin:
SMBG scheme 1 is suggested to facilitate diabetes education, understanding and behavioral changes and to assess rapidly treatment efficacy.
Duration/frequency of testing: 1–3 days weekly for 2-4 weeks
Stable/good metabolic control: 1–3 days monthly
Unstable/poor metabolic control: 3 days to 1 week monthly
Initiation of a combination therapy using SGLT-2 inhibitors as an add-on to sulfonylureas:
SMBG scheme 1 is recommended to detect hypoglycemic episodes, glycemic variability and to facilitate diabetes education, understanding and behavioral changes as to assess rapidly treatment efficacy.
Duration/frequency of testing: 1–3 days weekly for 4 weeks
Stable/good metabolic control: 1–3 days monthly
Unstable/poor metabolic control: 3 days to 1 week monthly
Scheme 2 may be considered in case of poor glycemic control and in patients with higher risk for hypoglycemia. Switch to scheme 1 as soon as the individual metabolic goals are reached.
Initiation of a combination therapy implementing SGLT-2 inhibitors as an add-on to insulin:
SMBG scheme 1 is recommended to depict hypoglycemic episodes and glycemic variability. SMBG will also help to assess rapidly treatment efficacy and to reduce promptly insulin dose when needed. It is recommended to facilitate diabetes education, understanding, and behavioral changes. The frequency and length of the performance of SMBG scheme 1 should also depend on the type of insulin administration.
Duration/frequency of testing: 3–7 days weekly up to continuous paired testing
Stable/good metabolic control: 3–7 days weekly up to continuous paired testing
Unstable/poor metabolic control: continuous paired testing
Performance of SMBG scheme 2 may be considered in those with intensive insulin therapy according to the level of glycemic control and the variability of glucose. Scheme 2 may also be performed in patients with higher risk for hypoglycemia.
Conclusions
The role for SGLT-2 inhibitors in the treatment of type 2 diabetes is increasing. The new class of treatment is reported to be efficacious in lowering HbA1c, preprandial and postprandial blood glucose, weight, and blood pressure. In monotherapy and as an add-on to metformin, SGLT-2 inhibitor based treatment is characterized by a very low risk of hypoglycemic episodes. In combination with sulfonylurea and, particularly, insulin, higher rates of hypoglycemia have been observed.
In SGLT-2 inhibitor based treatment approaches, SMBG may be used to visualize effects on preprandial glucose, postprandial glucose and glucose variability. SMBG may be useful for 5 clinical settings: (1) early assessment of treatment success or failure, (2) timely modification of treatment, (3) detection of hypoglycemic episodes, (4) assessment of glucose excursions, and (5) support of diabetes management and diabetes education.
The length and frequency of SMBG should depend on the clinical setting and the quality of metabolic control. Local resources and economic aspects will also contribute the performance of SMBG in the patients. A less and a more intensive scheme for SMBG meet the needs for the assessment of glycemic level in SGLT-2 inhibitor based treatment. The performance of simple, clinically meaningful and standardized SMBG strategies may have the potential to further optimize diabetes management and education without increasing the frequency of testing per se.
With regard to the further development of useful SMBG patterns, an evaluation of the presented SMBG schemes is desirable.
Footnotes
Abbreviations
DPP-4, dipeptidyl peptidase-4; FPG, fasting plasma glucose; GLP-1, glucagon-like peptide-1; HbA1c, glycosylated hemoglobin; LS, least square; SGLT-2, sodium-glucose cotransporter-2; SMBG, self-monitoring of blood glucose; UKPDS, UK Prospective Diabetes Study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors are members of an expert panel of Bayer HealthCare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The expert panel was supported by an unrestricted educational grant of Bayer HealthCare.
