Abstract
Soon after the discovery that insulin regulates blood glucose by Banting and Best in 1922, the symptoms and risks associated with hypoglycemia became widely recognized. This article reviews devices to warn individuals of impending hypo- and hyperglycemia; biosignals used by these devices include electroencephalography, electrocardiography, skin galvanic resistance, diabetes alert dogs, and continuous glucose monitors (CGMs). While systems based on other technology are increasing in performance and decreasing in size, CGM technology remains the best method for both reactive and predictive alarming of hypo- or hyperglycemia.
Prelude
This article is an extension of a presentation in a Workshop on Technology for Detection and Prevention of Hypoglycemia: Panel A: Prevention/Treatment, at the 2014 Diabetes Technology Meeting in Bethesda, Maryland (November 6, 2014). We started this presentation with a concise review of the current state of the art of hypoglycemia alarms by using the cartoon presentation (with apologies to Rube Goldberg) shown in Figure 1. While this is a “tongue-in-cheek” overview of glucose alarms, we will see components of this “system” in the technologies reviewed in this article.
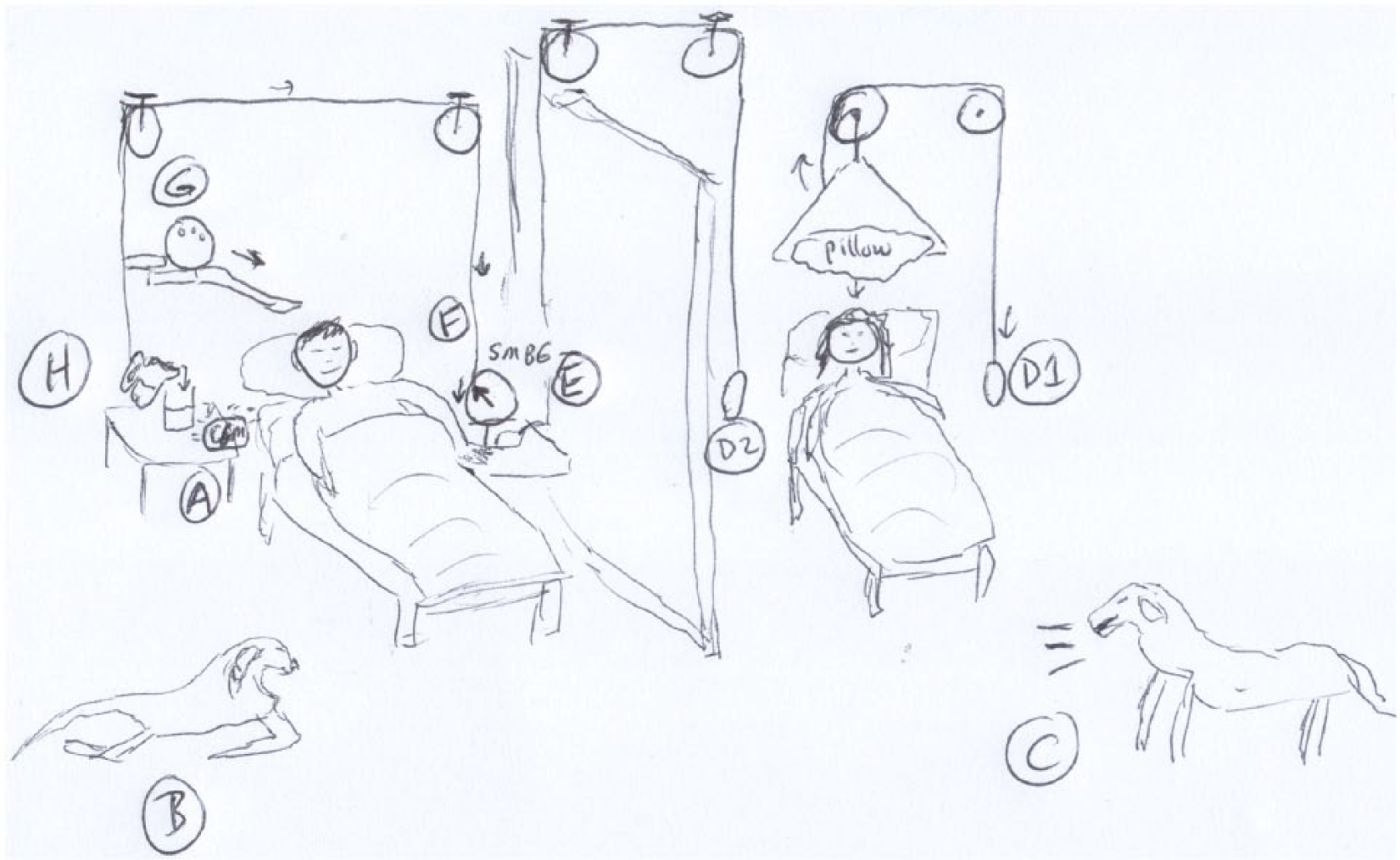
(A) CGM alarm sounds, (B) waking dog, who moves to adult room, (C) barking, and awaking adult, who either (D1) pulls lever placing pillow over head or (D2) pulls lever activating SMBG device (E). Low reading (F) causes lever to pull cord, moving bowling ball (G) to wake up child and (H) causing pitcher to pour glass of orange juice, so that the child can now alleviate the hypoglycemia.
In this article, we first review glucose alarms based on skin conductance, followed by techniques based on electroencephalography (EEG) and electrocardiography (ECG) signals. The limited results concerning diabetic alert dogs are then covered, followed by continuous glucose monitoring (CGM) alarms, and multiple signal alarms. Finally, we discuss the evolution of alarm systems to artificial pancreas systems.
Background
Diabetes is a family of diseases characterized by insufficient or inefficient insulin delivery by the pancreatic beta cells, causing chronic hyperglycemia. Without treatment, patients suffering from diabetes are at an increased risk for long-term complications such as nephropathy, neuropathy, and retinopathy, thereby decreasing their life expectancy. Early on, physicians diagnosed diabetes by tasting their patients’ urine, motivating the development of urine glucose tests in the 1800s. 1 Although they made use of retroactive information, these urine glucose tests signified the first methods of analyzing hyperglycemia.
After the first clinical use of insulin to treat diabetes in 1922, 2 clinicians quickly realized the connection between insulin and diabetes and developed the first insulin-based therapies aimed at mitigating hyperglycemic complications. However, almost immediately after its discovery,3-5 insulin over-administration was linked to hypoglycemia, with short-term complications of sweating, tachycardia, coma, and death. The long-term effects of hyperglycemia and short-term effects of hypoglycemia constrain a narrow blood glucose range for optimal treatment of diabetes. However, because of the retroactive nature of urine glucose tests and their inability to detect hypoglycemia, more sophisticated alarm systems were needed to ensure adherence to the prescribed treatment.
The Center for Disease Control estimates that 29.1 million people in the United States are currently living with diabetes (with 5% diagnosed with type 1 diabetes), which generates $245 billion in health care costs annually. 6 This large impetus for improved glycemic control motivates the development of alarm systems that are able to detect and even predict instances of both hypoglycemia and hyperglycemia. Furthermore, these alarm systems provide peace of mind and an increased quality of life to those living with diabetes. This article reviews diabetes-related alarms based on a variety of biosignals, focusing on historical approaches, recent developments, and commercial devices.
Skin Conductance and Temperature
Scientists in the mid-1970s saw the need for a passive alarm system that would alert diabetic patients to nighttime hypoglycemia and avoid the “dead in bed” phenomenon. The first approaches to nighttime alarm systems involved readily available skin temperature and/or skin conductance (galvanic skin response) measurements. Although these devices did not directly measure blood glucose, they sought to record the depression in skin temperature and/or increase in perspiration associated with the onset of hypoglycemia. Figure 2 illustrates the depression in skin conductance observed during hypoglycemia. An early study linking a sudden drop in skin conductance to hypoglycemic events was published by Bolinger et al in 1964. 7 Patents described hypoglycemic-alarm systems equipped with electrodes for skin conductance as early as 1957 8 with others following in the 1970s and 1980s.9-11
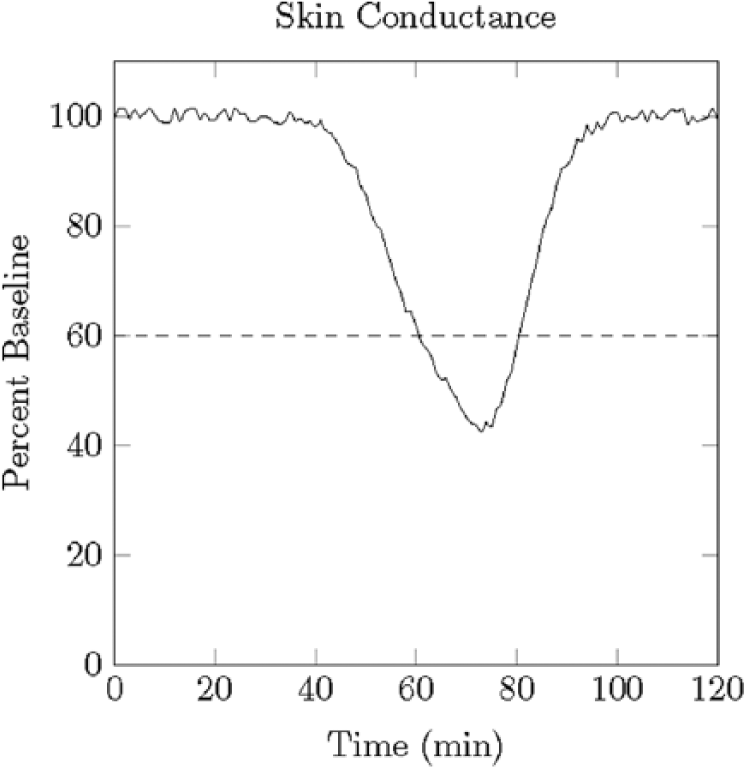
Illustration of the nadir in skin conductance associated with hypoglycemia.
The first commercial devices that used skin conductance measurements for nighttime alarms were Erpic’s Diabalert and Teledyne’s Sleep Sentry. The Diabalert consisted of meter housing the alarm which was attached through a long wire to a skin surface electrode. Pickup tested the Diabalert on 7 patients in a clinical setting and found that 3 patients would have not been able to respond to an alarm while 2 did not show signs of sweating even though their blood glucose levels were 12.6 and 37.8 mg/dL. 12 The original Sleep Sentry resembled a bulky wristwatch and combined skin temperature and skin conductance measurements. Levandoski et al induced hypoglycemia in 17 type 1 diabetic and 10 healthy individuals and observed a sensitivity of 81% for the Sleep Sentry alarms. 13 Hansen and Duck collected data on 24 pediatric patients with type 1 diabetes for a total of 1444 at-home nights, resulting in 42 hypoglycemic events detected by the Sleep Sentry, 4 additional events detected by the patients or their parents without an alarm, and 150 alarms without hypoglycemia. 14 Furthermore, this initial study used a high current density, causing adverse skin reactions in 6 of the 24 patients. Clarke et al revealed failures of the Sleep Sentry to sound in 6 of 18 experiments despite blood glucose readings between 31 and 52 mg/dL. 15 In addition, none of the subjects under study reached the threshold for temperature drop and 80% of nonalarmers self-reported an increase in perspiration. 15 Heger et al studied a myriad of signals on 7 patients and reported that the Sleep Sentry either failed to sound or sounded too late to be of use to the patient in 3 cases. 16 Studies by Johansen et al on the use of the Teledyne Sleep Sentry for detection of nocturnal hypoglycemia in 22 patients for a total of 63 nights resulted in 22 alarm sounds with 6 instances of hypoglycemia. 17 Thus, this study concluded with the similar 3:1 ratio of false alarms to true alarms as found by Clarke et al.
These early devices were plagued by both false alarms, usually from perspiration unrelated to hypoglycemia, and missed alarms, possibly from hypoglycemia-associated autonomic nervous system failure (hypoglycemia unawareness). 18 Although clinicians and scientists were quick to point out flaws in early diabetic alarm systems,14,15,19 patients in these studies accepted the imperfections with 1 study noting that parents of pediatric patients were very hesitant to return the investigational devices. 14 The increased sense of security offered to patients and their loved ones outweighed the high rate of false alarms, speaking to the improved quality of life offered by alarm systems.
Currently, the Diabetes Sentry (Diabetes Sentry, Roanoke, TX) 20 is the only hypoglycemic alarm system on the market that exclusively uses this technology. The improved sensor quality and sleeker look are big improvements over the original Sleep Sentry. However, the superior monitoring offered by CGMs has limited the widespread use of the device. The Diabetes Sentry uses noninvasive sensors and is more affordable than alarm systems based on CGMs ($495 for the Diabetes Sentry as opposed to $3000 for Medtronic’s mySentry), but even Diabetes Sentry notes that this product is not an alternative to a CGM. 21 It is the authors’ opinion that ever-improving CGM technology coupled with the inability to extend the Diabetes Sentry to reliable daytime use will limit the future success of alarm systems based solely on this technology.
Electroencephalography
Diabetes alarm systems based on EEG attempt to detect the decrease in cognitive function associated with hypoglycemia. Time series generated by EEGs are traditionally analyzed in the frequency domain and separated into delta, gamma, alpha, and beta bands, listed in order of increasing frequency. In the 1950s, Ross and Loeser were the first to simultaneously record EEG time series and blood glucose samples, 22 while Regan and Browne-Mayers 23 described the increase in delta and gamma frequencies and decrease in alpha frequencies associated with hypoglycemia, which has been supported by many other groups. 24 Figure 3 illustrates the differences in euglycemic and hypoglycemic EEGs. In a study of 13 diabetic patients, Pramming et al found that no changes in EEG frequencies were observed above 54 mg/dL and significant changes occurred at 36 mg/dL. 25 These changes appear to be independent of age, sex, awareness status, and duration of diabetes.25-27 In 1996, Heger et al made numerous measurements on 7 healthy and 18 insulin-dependent diabetic subjects and concluded that changes in EEG frequencies were the earliest and most significant; however, this group also noted that problems with power consumption, miniaturization, and sensor numbers limited this technology for everyday use. 16
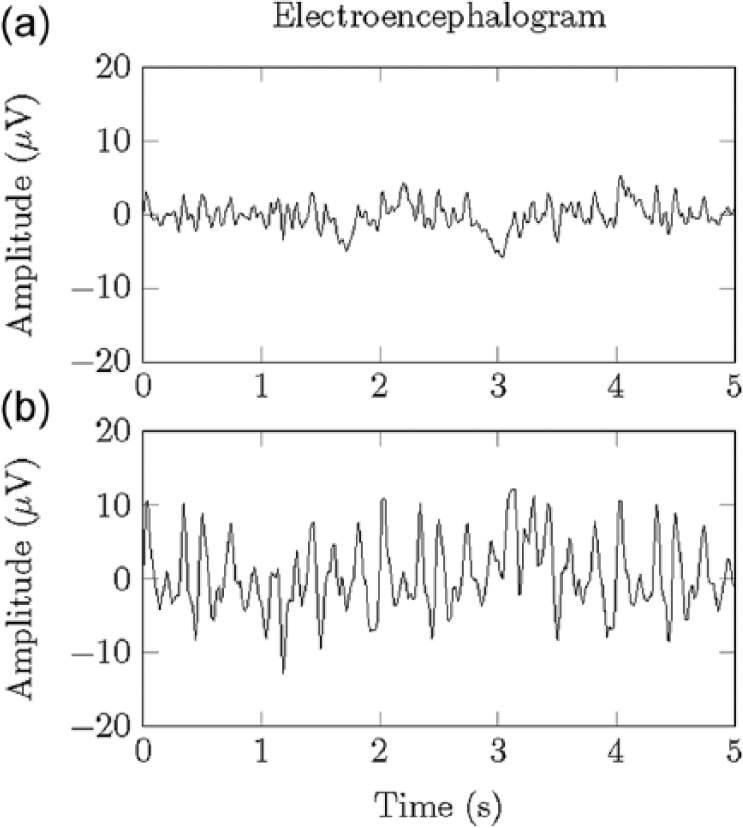
Illustration of an electroencephalogram signal during (a) euglycemia and (b) the increased alpha frequencies associated with hypoglycemia.
Unlike simple thresholds commonly used in skin conductance and skin temperature alarms, scientists have recently begun exploring more sophisticated analysis of EEG time series. Laione et al tested an experimental alarm system based on spectral analysis and artificial neural networks (ANNs) on 8 patients, reporting an average sensitivity and specificity of 71.1% and 71.5%, respectively, when subjects remained immobilized with their eyes shut during measurements. When the ANN was trained and tested on the same subject, the algorithm obtained an accuracy, false-negative, sensitivity, and specificity percentages of 85.2%, 14.8%, 60%, and 100%, respectively, giving evidence supporting subject-specific algorithms. 28 Fabris et al used a multiscale entropy approach to analyze EEG time series of 19 patients with type 1 diabetes, further supporting the claim that hypoglycemia is associated with a decrease in signal complexity. 29 Improvements in sensor technology and processing power have enabled these techniques to become more and more pragmatic.
Faster signal processing and improved electrode technology have only recently allowed EEG signals to be used in commercial diabetes alarms. Hypo-Safe A/S (Hypo-Safe A/S, Lyngby, Denmark) is currently pursuing an alarm system that makes use of a miniature EEG device implanted under the skin, behind the ear.30-33 For a very nice description of the Hypo-Safe device and the algorithm behind it, see. 27 Briefly, the most current algorithm extracts relevant features using a bank of infinite impulse response filters and feeds this information to an ANN-based classifier. Persistence of hyperglycemia detection over 10 minutes is used to signal an alarm, with detection of deep sleep rejecting an alarm. The use of 1 small, inconspicuous sensor coupled with its ability to detect hypoglycemia in those with hypoglycemia unawareness during both day and night are major advances in diabetes alarms. However, since many ordinary activities (eg, deep sleep and chewing) must be filtered by supervised classifiers 27 and some patients with a functional glucagon response do not exhibit EEG changes,34,35 further studies on the specificity and sensitivity of the fully commercial device will ultimately determine the success of this product.
Electrocardiography
Electrocardiography (ECG) reveals a variety of changes associated with the autonomic nervous system response during episodes of hypoglycemia. In the 1950s, studies of healthy subjects injected with insulin revealed an increase in heart rate, increase in systolic pressure, and decrease in diastolic pressure during hypoglycemia.36,37 These early approaches suffered from their nonspecificity to hypoglycemia, with exercise, stress, and other stimuli setting off false alarms. 16 Beginning in the mid-1990s, scientists have found correlations between a lengthened QT interval and hypoglycemia.38-42 Figure 4 illustrates typical ECG waveforms during euglycemia and hypoglycemia. Usually associated with nocturnal hypoglycemia and “dead in bed” syndrome,43,44 lengthened QT intervals are more indicative of hypoglycemia detection than earlier ECG metrics, although these correlations are dependent on both the methods used to calculate the QT interval45,46 and heart rate correction. 47 Additional changes in ECG-related parameters in response to hypoglycemia include increased QT dispersion, 48 an altered T-wave morphology,41,49-51 and reduced spatial QRS-T angle. 49 Furthermore, changes in ECG- and EEG-related parameters in response to hypoglycemia have been shown to occur simultaneously. 41
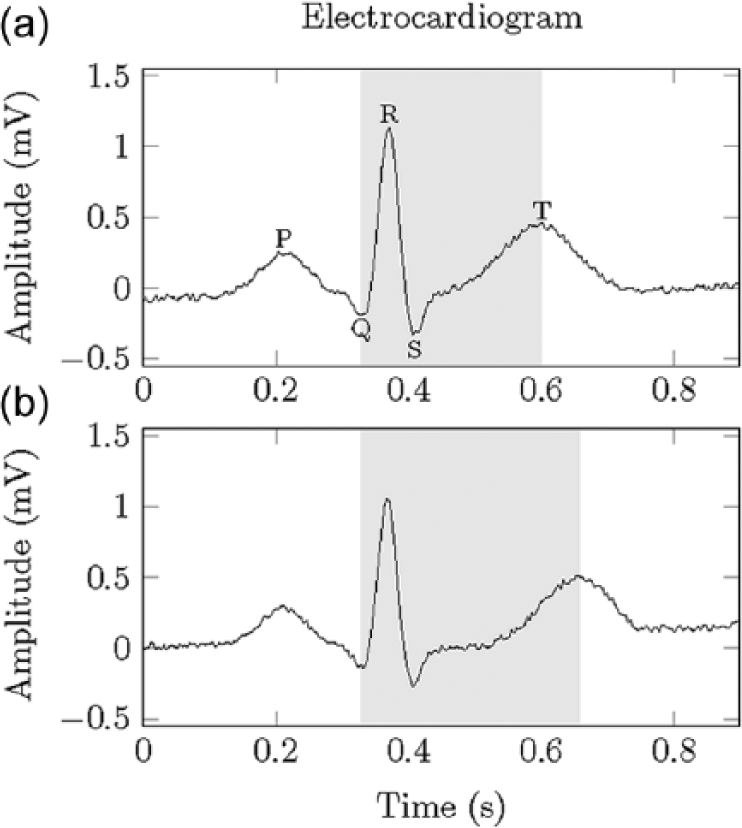
Illustration of (a) an electrocardiogram signal during euglycemia and (b) the lengthened QT interval associated with hypoglycemia.
In addition to hypoglycemia detection, ECG-related parameters have also been correlated to hyperglycemia. A reduced heart rate variability52-54 is the most widely studied ECG-related parameter for hyperglycemia detection, but altered QT interval,53,55,56 QT dispersion,53,55 and PR interval55,57 have also been reported as indicators of acute hyperglycemia. Studies by Nguyen et al 57 showed that different subsets of ECG-related parameters were associated with hyperglycemia and hypoglycemia, giving rise to the possibility that ECG-based alarm systems could detect both kinds of events.
Spurred on by advances in signal processing techniques and sensor technology, experimental alarm systems have attempted to use ECG-related parameters for hypoglycemia detection. Harris et al developed the Hypoglycemia On-line Monitoring Ensemble (HOME) to track QT interval changes during sleep. 58 Alexakis et al automatically extracted features and tested 2 subject-specific classifiers based on ANNs and linear discriminant analysis (LDA) on 11 type 1 diabetic patients. 59 The ANN alarm system was reported to achieve an average accuracy, sensitivity, false alarm rate, specificity, and false negative rate of 85.07%, 96.64%, 5.40%, 82.99%, and 3.22%, respectively, and the LDA classifier produced slightly less desirable results. Nguyen et al tested an alarm system based on Bayesian neural networks on 25 patients with type 1 diabetes, resulting in a sensitivity and specificity of 83.46% and 63.88%, respectively 60 (further iterations of this approach are found in Nguyen et al61,62 and Nuryani et al 63 ).
Although experimental devices that utilize ECG-related parameters seem promising, commercial alarms based on changes in ECG measurements have had limited success. No longer marketed, the HypoMon® (AiMedics Pty Ltd, Sydney, Australia) used heart rate, corrected QT interval, and skin impedance for nocturnal hypoglycemia detection. Skladnev et al evaluated the performance of the HypoMon on 52 subjects, which resulted in sensitivity and specificity metrics of 73% and 68%, respectively. 64 The HypoMon entered both the Australian and UK markets, but was later recalled due to a large amount of false negatives. 65 Possible explanations for the low performance of commercial ECG-based alarm systems include the effects of various medications, electrolyte abnormalities 66 as well as hypoglycemia unawareness. 67
Diabetic Alert Dogs
Various volatile organic compounds (VOCs) in exhaled breath (eg, acetone) have been found to correlate with glucose levels.68-70 Furthermore, VOCs have shown correlations with hyperglycemia, 71 plasma triglycerides and free fatty acids, 72 blood ketones, 73 and exercise. 74 This possible wealth of information could allow for more advanced diabetes alarm systems with a single, noninvasive sensor. However, even with the recent development of portable acetone sensors (eg, Prabhakar 75 ) and the previous successes of FDA-approved breath analysis (eg, ethanol for screening blood alcohol), it is unlikely that breath analysis will be integrated into continuous monitoring systems due to the nondiscreet nature of these sensors.
Although breath analysis sensors are of limited use for diabetes alarm systems, some believe that diabetic alert dogs may be trained to associate changes in a patient’s breath with acute complications from diabetes. Although anecdotal evidence abounds, scientific evidence supporting the use of dogs for detecting hypoglycemia is limited. Numerous case studies of dogs detecting hypoglycemic events have been reported (see Chen et al, 76 O’Connor et al, 77 Tauveron et al, 78 and Wells et al 79 for examples), however, these reports provide little or no evidence on the sensitivity and specificity of these “alarms.” Organizations such as Dogs4Diabetics® Inc and Canine Hope for Diabetics claim that dogs use scent to detect hypoglycemia. However, in a 2013 study, Dehlinger et al presented 3 dogs trained to detect hypoglycemia with cotton balls rubbed on the skin of diabetic patients during periods of euglycemia and hypoglycemia. 80 Even though this same methodology was used to train the dogs for hypoglycemia detection, the authors reported an average sensitivity and specificity of 55.5% and 52.8%, respectively. Despite a lack of scientific evidence supporting the use of these “alarm systems,” many patients report an increased feeling of safety when they work with a diabetic alert dog. Furthermore, some groups are working on alarm systems that would allow dogs to remotely signal a patient’s loved ones or emergency services during a hypoglycemic attack. 81 Due to the poor sensitivity and specificity and the increased patient burden, the continued use of diabetic alert dogs remains unclear.
Continuous Glucose Monitors
The alarm systems described previously aim to detect changes associated with symptoms of hypo- and/or hyperglycemia, classifying a patient’s state as either hypoglycemic, euglycemic, or hyperglycemic. However, other alarm systems seek to monitor signals that are related to current blood glucose levels. These signals have the ability to estimate the current blood glucose level over a wide range and possibly give more information than a signal capable of giving only simple classification. These CGMs can be divided into noninvasive and subcutaneous sensors. The next sections will evaluate the evidence from the scientific literature regarding the alarm components of these devices.
Noninvasive Glucose Monitoring
There are many different ways to obtain a noninvasive estimate of blood glucose and we refer the interested reader to a number of detailed reviews (eg, Vashist, 82 Tura et al, 83 Ciudin et al, 84 So et al 85 ) for more details. Here, Tura et al’s definition of a noninvasive glucose sensor—that which does not lead to blood collection and which does not pierce the skin with a solid object—is used. 83 Many attempts to develop commercial devices for real-time noninvasive glucose measurements, such as those based on near infrared spectra, have been unsuccessful due to interference from other blood metabolites, inter- and intrapatient differences in tissue characteristics, and miniaturization of technology. Furthermore, either the poor accuracy or the short lifetimes of these devices have limited the amount of information on the alarm components of these devices. Therefore, the alarm component of the GlucoWatch® (Cygnus Inc, Redwood City, CA) is the only noninvasive device that is evaluated.
The GlucoWatch was the first commercial device that monitored glucose noninvasively and in real-time. Cygnus received the CE mark in 1999 and FDA clearance for the GlucoWatch in 2001, with an updated model approved in 2002. This device made use of reverse iontophoresis, a process by which an electric current brings interstitial glucose to the surface of the skin and then measures the amount of glucose via an electrochemical sensor.86,87 Whereas autonomic nervous system failure undermined the performance of skin temperature/perspiration and ECG devices, regular autonomic nervous system response proved problematic for the reverse iontophoresis technology. Thus, skin conductance and skin temperature sensors were added both to filter erroneous measurements and provide more information to the alarm system if a large skin temperature change or sweat accumulation was detected. 88
Many scientists, clinicians, and type 1 diabetics initially flocked to the GlucoWatch because of its promise to forever change diabetes care with continuous, noninvasive measurements and nighttime alerts. Cygnus determined that 100 mg/dL (5.6 mmol/L) was the optimal tradeoff between sensitivity and specificity of their threshold alarm. 89 In studies of 40 children, Chase et al found the ratio of total alarms to hypoglycemic events was 1.5. The DirecNet Study Group reported that 31% of the glucose values were within 15 mg/dL of the reference value and the sensitivity and false alarms were 23% and 51%, respectively, for threshold alarms set at 60 mg/dL. 90 In a later study, the DirecNet Study Group found the use of the “down alert” alarm improved the sensitivity from 24% to 88% and increased the false positive rate from 6% to 56% during insulin induced hypoglycemia. 91 Furthermore, the sensitivity improved from 23% to 77% and the false positive rate increased from 60% to 77% during overnight studies. 91 Gandrud et al studied 45 subjects at a diabetes camp, reporting 20 low-glucose alarms with reference measurements, of which 10 were true positives.93Although its noninvasive glucose monitoring was conceptually appealing, the GlucoWatch was plagued with high false alarm rates, an excessive warm up period of 2-3 hours, inability to operate under temperature changes and increased perspiration level, and the tendency to cause skin irritation in some patients. The GlucoWatch was discontinued on July 31, 2007. 82
Continuous Subcutaneous Glucose Monitors (CGMs)
High sensor accuracy and alarm sensitivity has propelled the use of CGMs to warn of glycemic excursions. CGMs make use of small enzymatic sensors inserted beneath the skin to measure interstitial glucose. An oxidation-reduction reaction produces a measureable current that is calibrated with a blood glucose measurement. For more information on the chemistry and design of these sensors, see the review by McGarraugh. 92 The first commercial CGM, the MiniMed (Medtronic, Northridge, CA), was approved by the FDA in 1999, but alarms did not appear on commercial devices until the mid-2000s with the introduction of the Guardian (Medtronic, Northridge, CA) and the STS™ (DexCom, San Diego, CA) CGMs. In studies of the Guardian in 71 subjects, Bode et al reported a 67% sensitivity, 90% specificity, and 47% false alerts for hypoglycemic (≤ 70 mg/dL) alarms and a 63% sensitivity, 97% specificity, and 19% false alerts for hyperglycemic (≥ 250 mg/dL) alarms. 93 Furthermore, this study revealed that while a CGM with a threshold alarm decreased the duration of hypoglycemia, the occurrence of hypoglycemia was unchanged.
Current commercial CGM devices have even higher sensor accuracies, especially in the hypoglycemic range. Although the performance of threshold alarm systems on these later generation devices have not received as much attention as the sensor accuracies, improved sensor accuracy in the hypoglycemic range is almost certain to directly translate to a more accurate hypoglycemic alarm. An updated calibration algorithm was shown to improve sensitivity in the hypoglycemic range from 54.9% with Paradigm REAL-Time System to 82.3% with the Paradigm Veo. 94 The DexCom G4™ PLATINUM has also shown greater accuracy in the hypoglycemic range than the DexCom SEVEN PLUS. 95
While other alarm systems have shown improvements in alarm sensitivity/specificity over time, CGMs with threshold alarms have also shown improvements in clinical outcomes. We refer the readers to DeSalvo and Buckingham 96 for a recent review on the current and future use of CGMs in diabetes management. In studies of the STS system, Garg et al reported that insulin requiring type 1 and type 2 diabetics given real time access to CGM readings and threshold alarms spent 21% and 23% less time in the hypoglycemic and hyperglycemic ranges, respectively. 97 Deiss et al showed that the Guardian® REAL-Time CGM System with threshold alarm systems was able to reduce HbA1C levels in type 1 diabetics. 98 In a study of 322 adults and children using the SEVEN (DexCom, San Diego, CA), MiniMed Paradigm REAL-Time (Medtronic, Northridge, CA), or FreeStyle Navigator (Abbott Diabetes Care, Alameda, CA) in a home environment, the JDRF also showed improved time in range and HbA1C levels. 99
Predictive alarms are one of the most exciting aspects of CGM-based alarm systems, allowing patients to take preventative, rather than corrective, action. Although exact details are retained as trade secrets, commercial devices use linear predictions based on the previous rate-of-change of CGM readings to issue a predictive alarm. 100 For an overview of predictive CGM-based alarm systems prior to 2010, we refer the interested reader to Bequette. 101 Since that time, many experimental predictive alarms based on data driven models have been developed. With a hypoglycemic threshold of 60 mg/dL and a 30-minute prediction horizon, adaptive time series based on CGM only102,103 demonstrated a sensitivity and specificity of 89% and 78%, respectively. Future improvements of this alarm system have included activity 104 and insulin 105 information. Other approaches for short-term CGM predictions make use of neural networks106-108 or support vector machines. 109
Multiple Signal Alarms
In addition to devices that only measure 1 variable, there have been experimental attempts to integrate multiple signals for more robust alarms. A patent from 2003 mentions the use of both ECG and EEG signals for hypoglycemia detection, 110 but no commercial device has employed this technology. Attempts to augment CGM-based alarm systems with ECG-derived signals,111,112 skin temperature/conductance signals,88,104,113 and energy expenditure signals 104 have also been reported. While adding multiple signals can potentially add information and provide more information to alarm systems, the increased patient burden must be taken into account to ensure the effective translation from the clinic to the home.
From Alarm Systems to an Artificial Pancreas
The high sensor accuracy and improved clinical outcomes offered by CGM systems have established enough confidence in CGM systems to actually act on CGM-based alarms and alter insulin delivery. This promise to move from alarm system to artificial pancreas is what has allowed CGM alarm systems to eclipse alarm systems based on the other reviewed technology. For a recent review of current progress in the development of artificial pancreas systems, see Shah et al. 114
Hypoglycemia Minimizer
While threshold alarms rely on patients to take corrective action, hypoglycemia minimizers (HMs) in the form of low glucose suspend (LGS) systems automatically suspend insulin delivery when patients are unable to respond to hypoglycemia. For an early review of LGS systems, see Pickup. 115 Both the Paradigm® Veo™ (Medtronic, Northridge, CA), which obtained the CE Mark in 2009, and the MiniMed® 530G (Medtronic, Northridge, CA), which obtained FDA approval in 2013, suspend insulin delivery for up to 2 hours at the onset of hypoglycemia. The LGS component of the Paradigm Veo has been shown to significantly reduce exposure to hypoglycemia,116-118 especially during the night,119,120 without increasing exposure to hyperglycemia.116,117,120 A recent retrospective analysis of the LGS feature of the MiniMed 530G in > 20 000 patients over a 40-week period also shows decreased exposure to hypoglycemia without increase in hyperglycemia. 121
Just as highly accurate threshold alarms paved the way for LGS systems, predictive alarms have led to predictive low glucose suspend (PLGS) systems. Unlike LGS systems, PLGS systems can reduce the occurrence as well as the area under the curve of hypoglycemia. Simulation studies have been used to illustrate the benefits of a PLGS system over an LGS system. 106 Medtronic has recently released the MiniMed™ 640G system with SmartGuard™ in Australia that is able to predict glucose 30 minutes in advance and suspend insulin delivery. 122 Although more ambulatory data is needed to determine the effectiveness of this system, many experimental systems give evidence for improved glycemic control with PLGS systems. An algorithm utilizing a voting scheme to predict hypoglycemia 123 successfully prevented 84% of nocturnal hypoglycemic events in an in-patient study. 124 A Kalman filter-based algorithm successfully prevented hypoglycemia in 73% of subjects in an in-patient study. 125 With updated tuning parameters, this algorithm reduced the occurrence of nocturnal hypoglycemia by almost 50% in an out-patient setting. 126 A larger study of the same PLGS system reduced median hypoglycemia area under the curve by 81% and extended hypoglycemia by 74% without significant increase in hyperglycemia or morning ketosis. 127 An experimental algorithm based on time series forecasting has also been evaluated by inducing hypoglycemia through exercise in a small number of patients, resulting in a reduction in occurrence of hypoglycemia. 128 Other HM approaches have incorporated insulin 129 or historical CGM 130 information and smoothly attenuate insulin delivery, rather than completely suspend/resume insulin delivery. For a more information on hypoglycemia prediction algorithms and HMs, we refer the readers to Bequette. 131
Hypoglycemia/Hyperglycemia Minimizer
Experimental systems that act on predicted hyperglycemic alarms to deliver more insulin have only been recently developed. In addition to reducing patient exposure to hypoglycemia, these hypoglycemia/hyperglycemia minimizers (HHMs) or control to range systems (CTRs) seek to decrease the mean glucose level. The sCTR and eCTR systems presented in Patek et al 132 and clinically evaluated in Breton et al 133 increased time in the target range while reducing the exposure to hypoglycemia compared with standard open-loop continuous subcutaneous insulin infusion therapy. In an in-patient safety study, Finan et al report that patients using their HHM spent 69.6% and 30.2% of the time in the 70-180 mg/dL and >180 mg/dL ranges, respectively. 134 While acting on hypoglycemic alarms via LGS or PLGS systems can be fairly aggressive and reduce hypoglycemia exposure to near zero, acting on hyperglycemic alarms in an HHM must be more cautious.
Safety Components of an Artificial Pancreas
With the development of the artificial pancreas, a closed-loop system that determines the insulin administration which maintains euglycemia, alarm systems will be necessary to ensure patients can take the minimal action necessary to maintain the system and manually control their glucose level in the presence of system malfunctions. From this viewpoint, the patient’s interaction with the artificial pancreas system is analogous to an operator’s interaction with a chemical plant: under normal circumstances, no action is required, but severe circumstances require manual action to maintain a desired state. Current experimental systems have made use of traffic lights 129 and other safety layers 135 to indicate alarms of differing severities during closed-loop control. Patients may be alarmed if an insulin set failure is detected, the CGM signal has dropped or is too variable, the insulin available in the pump is low, the patient is close to the total daily dose (TDD) limit, or if the system needs to be charged. Furthermore, other experimental safety layers incorporate LGS or PLGS suspend systems.135,136 We refer the interested reader to Bequette 137 for more details on some of the safety layer features necessary to a fully closed-loop system.
Summary
Diabetic alarm systems potentially afford more effective diabetes management and an increased quality of life to those living with diabetes. While some signals are only capable of classifying a patient’s state as normal and hypo- and/or hyperglycemic, others can estimate blood glucose concentrations. Furthermore, some signals are only suitable for detection of altered glycemic states while other signals can predict these undesirable events. Although standalone alarm systems will become obsolete with the development of the artificial pancreas, alarm systems will form the core safety components and have paved the way for insulin administration algorithms of various complexities.
Footnotes
Acknowledgements
We wish to recognize Bruce Buckingham for his pioneering work in glucose alarms, and acknowledge our many discussions with him on a number of collaborative projects.
Abbreviations
CE, Conformité Européene; CGM, continuous glucose monitor; CTR, control to range; ECG, electrocardiograph; EEG, electroencephalography; FDA, Food and Drug Administration; HHM, hypoglycemia/hyperglycemia minimizer; HM, hypoglycemia minimizer; LDA, linear discriminant analysis; LGS, low glucose suspend; PLGS, predictive low glucose suspend; TDD, VOC, volatile organic compound.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding from the NIH (NIDDK 1R01DK102188-01) and JDRF (17-2013-471 and 22-2011-647) is gratefully acknowledged.
