Abstract
Background:
There is a need for patients to be able to adjust their insulin doses accurately and independently during continuous subcutaneous insulin infusion (CSII) therapy in order to avoid glycemic excursions and improve glycemic control. Use of new technology has the potential to aid patients in visualizing their circadian patterns and improving their understanding of data provided by self-monitored blood glucose (SMBG) measurements.
Methods:
A 24-week crossover study was performed in 25 patients with type 1 diabetes mellitus using CSII and SMBG. Patients were randomized either to entering blood glucose data into handwritten logbooks or to using the Accu-Chek SmartPix information management system (IMS) coupled with instructions from a training manual to aid interpretation of the IMS readings. Patients analyzed these chart readings every 2 weeks, and outpatient visits were scheduled for both arms every 6 weeks.
Results:
There was a significantly lower mean overall blood glucose level with the IMS compared with use of a logbook (139 ± 16.2 versus 150 ± 19.8 mg/dl; Δ = 10.8 mg/dl; p < .01), and a significantly higher proportion of blood glucose values was in the target range compared with use of a logbook (43.6% versus 38.5%; p < .001). Hypoglycemic events were also significantly lower with the IMS compared with logbooks (3.7 fewer events/6 weeks; p < .05). There was no significant difference between groups in the daily frequency of SMBG measurements.
Conclusions:
The use of an IMS, coupled with an easily understood training manual, enables patients to improve glycemic control by performing accurate and timely self-adjustments to their insulin regimens.
Keywords
Introduction
Accurate adjustment of insulin doses during continuous subcutaneous insulin infusion (CSII) therapy is essential in order to achieve and maintain tight glycemic control in patients with type 1 diabetes mellitus (T1DM). 1 Detection of recurrent, reproducible variations in blood glucose levels outside the target range is critical to the success of self-management. 2 Although the practice of downloading information is becoming more common, glycemic data are usually collected as patients' handwritten entries in diabetes logbooks, recorded from self-monitored blood glucose (SMBG) measurements. The mechanics of keeping an accurate written logbook are, however, complex and conducive to errors in recording data, which need to be interpreted correctly to make accurate decisions on adjustments in insulin doses at specific points in time, as well as adjustments to fundamental insulin requirements. 3 Although patients can be trained to make these adjustments, it is quite often the treating physician who interprets the readings during consultations and makes the decisions on changes to the patient's regimen. However, these consultations are only scheduled at 6- to 12-week intervals, and an earlier opportunity to react to blood glucose excursions and make therapy adjustments should improve glycemic control and reduce the risk of diabetes-related complications.4–6
The Accu-Chek® Smart Pix (Roche Diagnostics GmbH, Mannheim, Germany) information management system (IMS) is a device that directly downloads data from metered SMBG measurements to a computer and produces graphical representations of patients' glycemic fluctuations, including variations in circadian blood glucose patterns. Training materials have been developed for patients using CSII to interpret the graphical displays, which enable them to independently make adjustments to their treatment algorithms (for example, basal rate, meal bolus insulin, and doses to correct for glycemic excursions) without the need for consultation with their physician.
The aim of this pilot study was to determine whether patients using CSII could make more effective adjustments of their insulin dose using the Accu-Chek Smart Pix IMS in conjunction with a training manual to interpret SMBG measurements compared with the use of a conventional diabetes logbook. The study aimed to assess whether use of the IMS would result in better metabolic control, as evidenced by improved glycosylated hemoglobin (HbA1c) levels, reduced frequency of hypoglycemia, and a greater proportion of blood glucose values within the target range.
Methods
This was a single-center, open, randomized, prospective, 24-week crossover study of 25 male (52%) and female patients aged 18 years or over with T1DM. The study was conducted from July 2007 to March 2009. Patients included in the study were familiar with the use of CSII therapy and were conducting regular SMBG measurements using a blood glucose meter compatible with the Accu-Chek Smart Pix IMS. Patients were also required to have access to a computer with appropriate software to download the information from the IMS. Patients were excluded from the study if they conducted fewer than four SMBG measurements per day or had problems using the software or hardware supplied. Other exclusion criteria included pregnancy (current or planned) and treatment with systemic glucocorticosteroids. A total of 28 patients were deemed eligible for the study; three patients withdrew (one violation of protocol, one pregnancy, one personal decision), resulting in data from 25 patients evaluated at study end.
The crossover design of the study is shown in
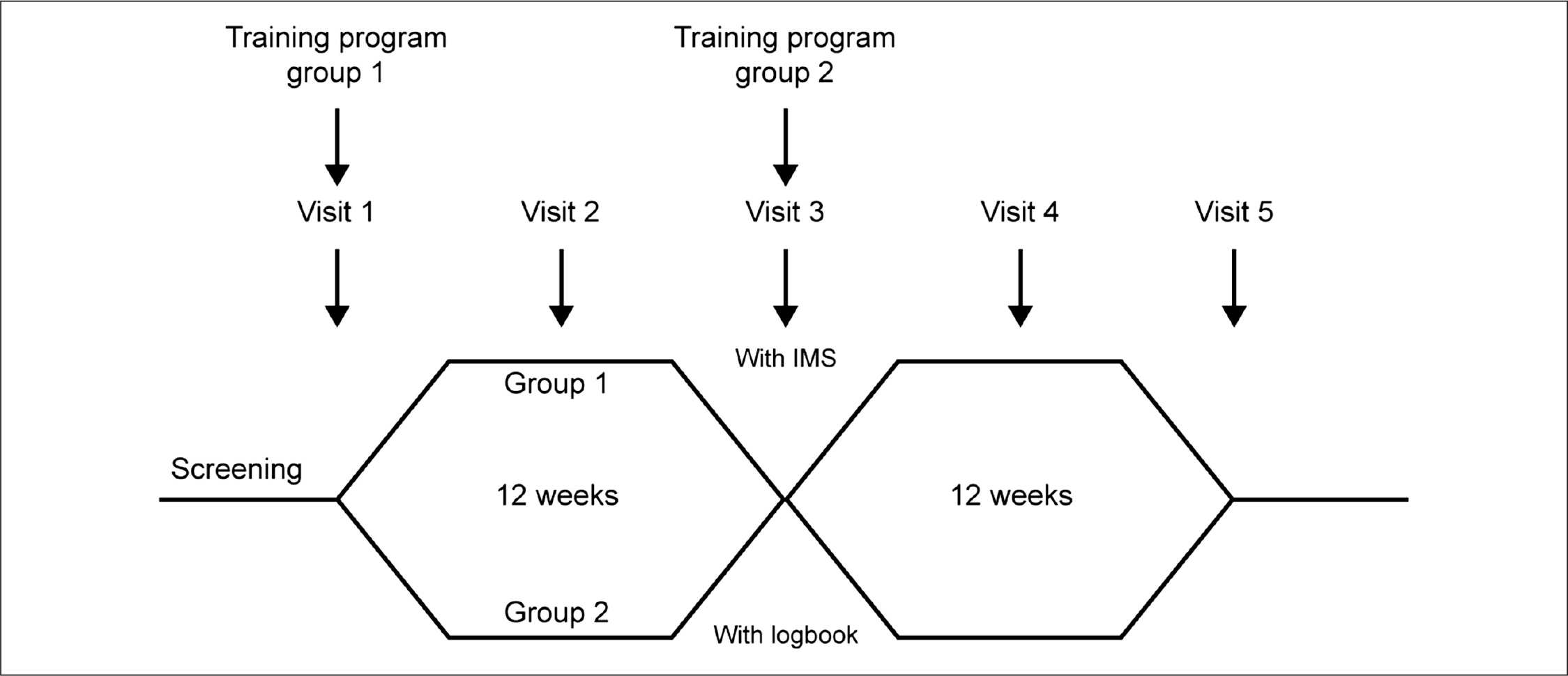
Study design.
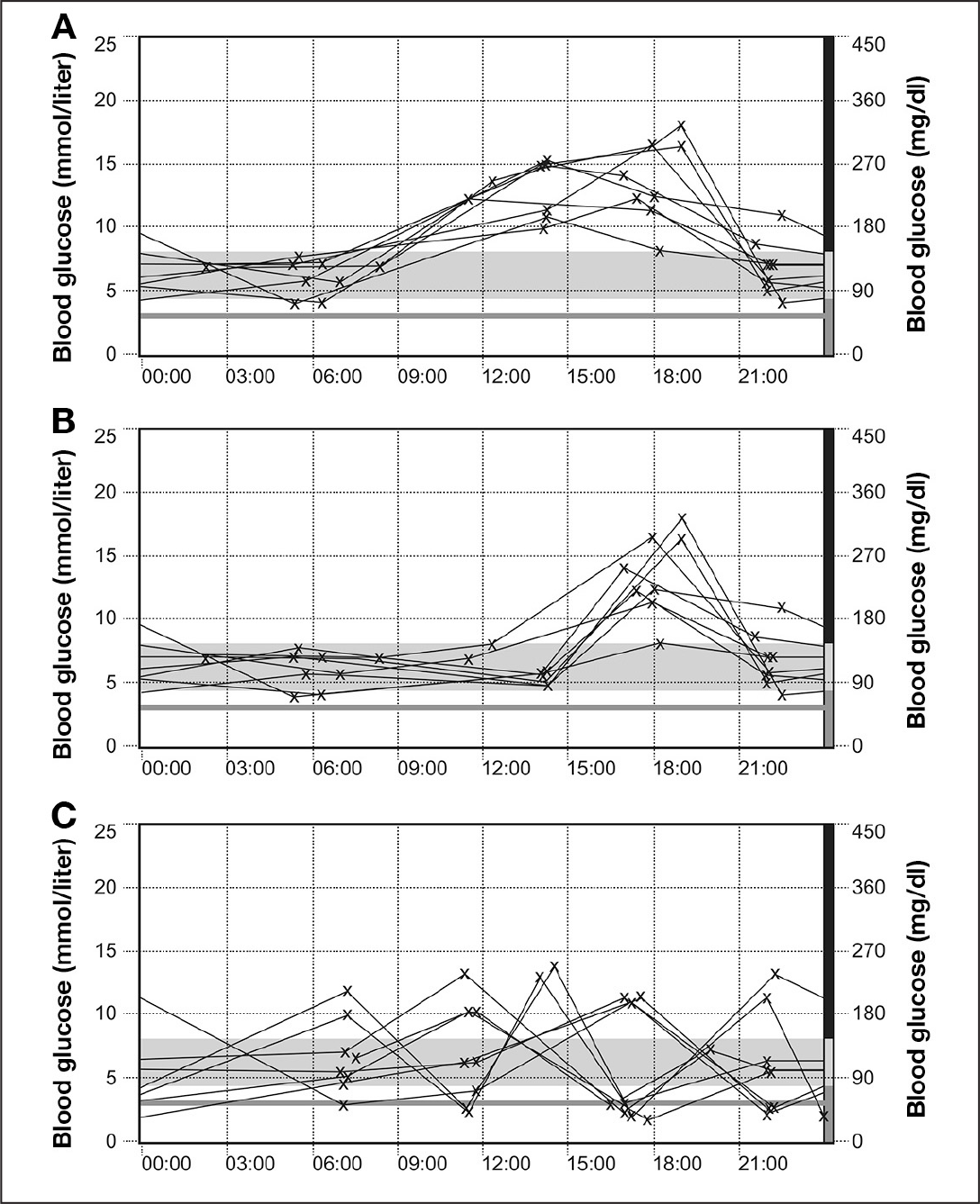
Accu-Chek Smart Pix IMS day graphs depicting periods of (A) hyperglycemia: treatment suggestion, increase your insulin bolus at breakfast and lunch; (B) hyperglycemia: treatment suggestions, (i) if you eat a meal at this time, increase the insulin bolus for this meal, (ii) if you do not eat a meal, increase your basal rate; and (C) hypoglycemic events: treatment suggestion, reduce the size of insulin bolus used to correct for potential high blood sugar.
Group 2 was asked to record daily readings of their SMBG measurements according to their usual practice in conventional diabetes logbooks in a tabular or graphic fashion, making daily entries in a row at the same time each day, along with entries regarding medications, insulin doses, exercise, hypoglycemic episodes, and food consumption. Patients in this group continued to analyze and interpret their data according to their accustomed procedures.
Outpatient visits were scheduled for both groups every 6 weeks, at which time blood glucose values were read and analyzed using Accu-Chek Camit Pro software, a precursor of the Accu-Chek 360° software (Roche). Mean blood glucose values and the number of hypoglycemic events (blood glucose values <54.1 mg/dl) since the last visit were recorded, and the percentage of blood glucose values within the target range (72–144 mg/dl) was calculated. Glycosylated hemoglobin levels were measured at visits 1, 3, and 5. The groups crossed over at 12 weeks, at which point group 1 converted to using logbooks to record their meter readings and, after receiving the same training that group 1 had been given, group 2 used the IMS for the next 12-week period of the study.
The study protocol complied with the Declaration of Helsinki and regulations for observational studies in Germany, and full ethical approval was obtained by local ethics committees. All patients signed and dated written informed consent forms, and data were collected in case report forms, where anonymity was maintained.
Statistics
Statistical evaluation was performed on between-group differences for HbA1c measurements, mean blood glucose levels, frequency of blood glucose monitoring, proportion of patients achieving blood glucose targets and proportion of blood glucose values within the target range using the t-test for paired samples (SPSS MS Windows 16.0.2). Data are presented as mean ± standard deviation (SD).
Results
Baseline characteristics for the patients are presented in
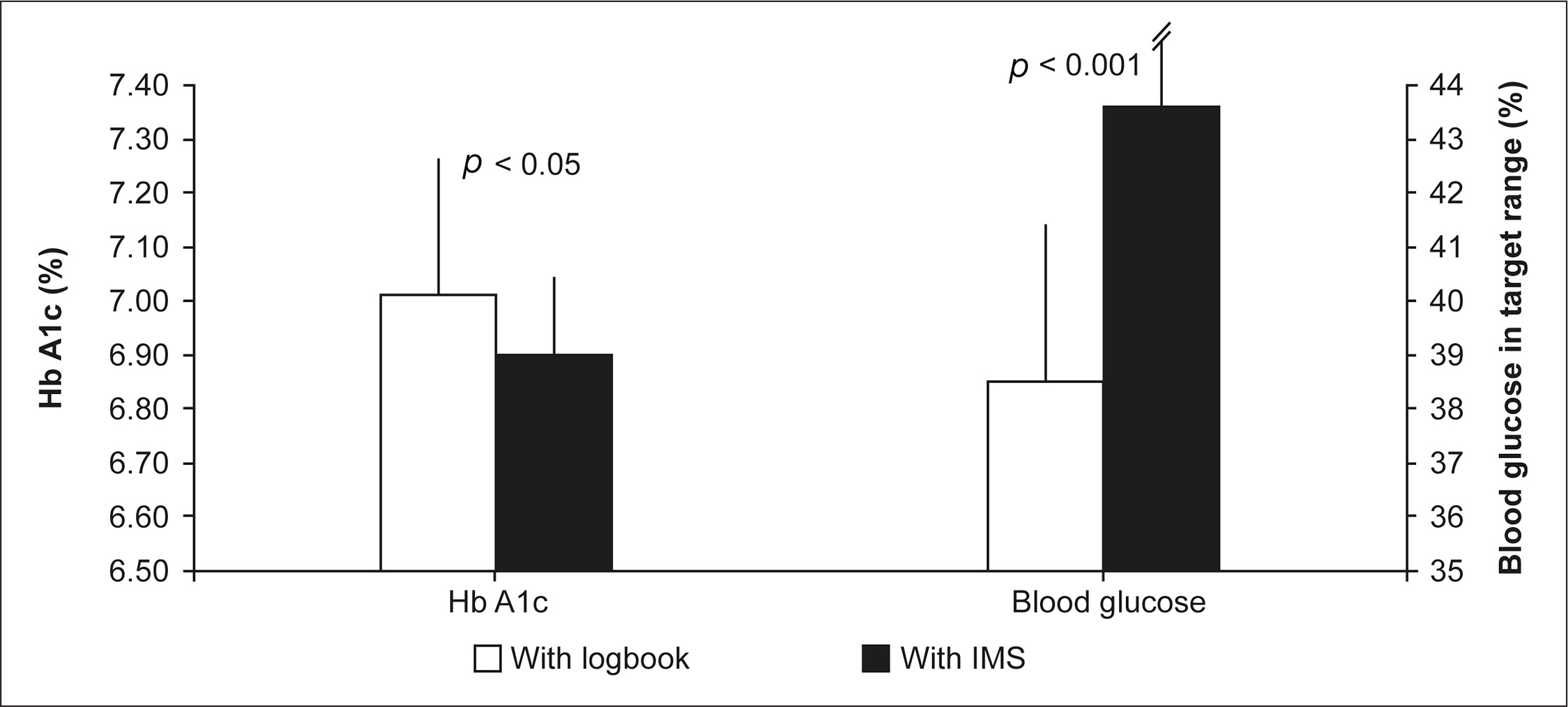
Differences between groups in HbA1c levels and proportion of patients achieving glycemic targets. Error bars represent the standard deviation.
Baseline Characteristics
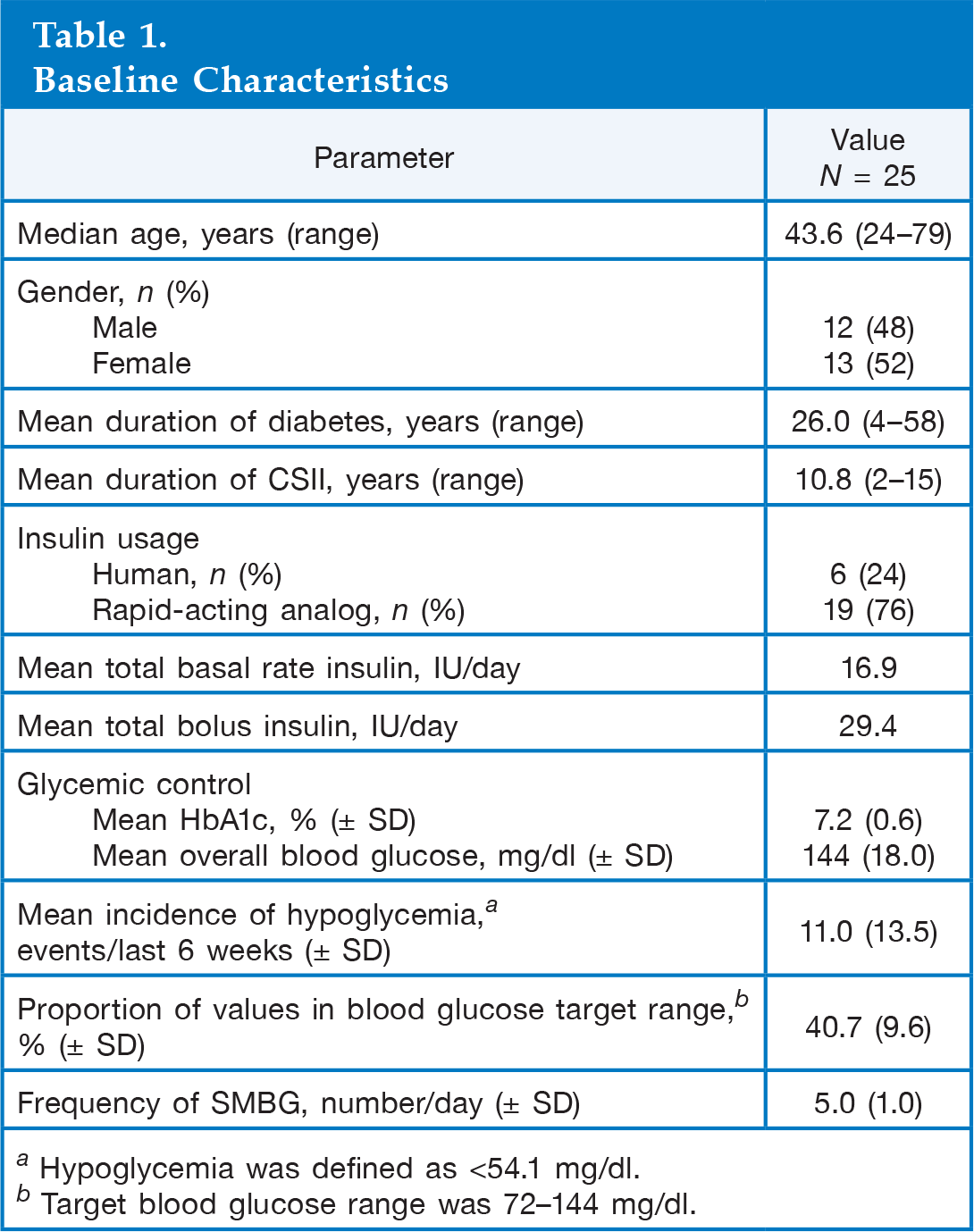
Hypoglycemia was defined as <54.1 mg/dl.
Target blood glucose range was 72–144 mg/dl.
Differences in Target Parameters after 24 Weeks
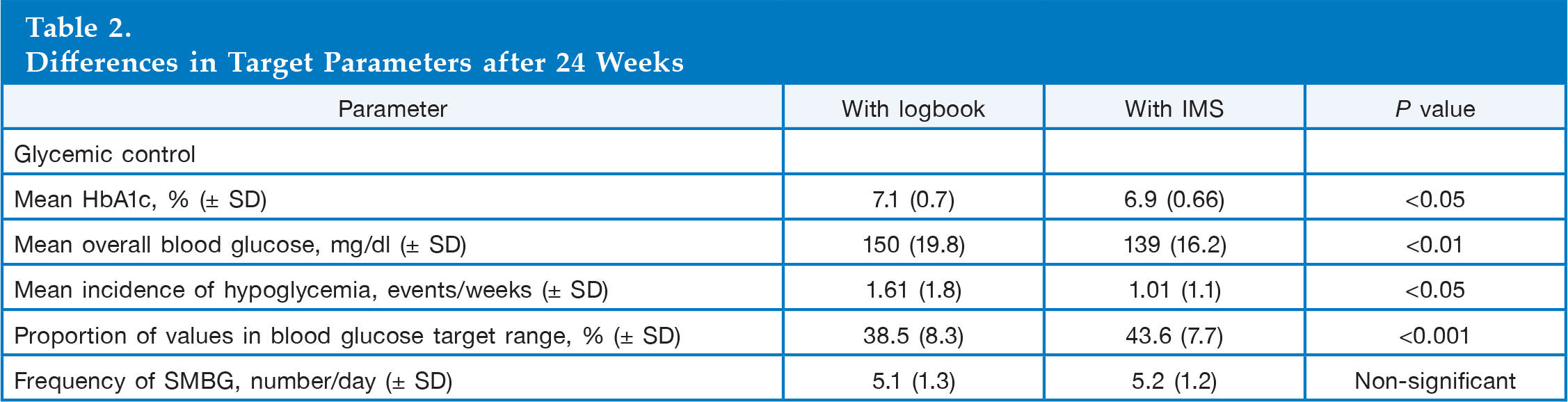
In addition, a significantly higher proportion of patients achieved the mean blood glucose levels in the target range, 72–144 mg/dl, using the IMS compared with logbooks (43.6% versus 38.5%, respectively; p < .001;
The number of hypoglycemic events occurring per week was significantly lower when the patients used the IMS rather than their written logbooks (1.01 versus 1.61; p < .05), and there were significantly fewer hypoglycemic events in the 6 weeks prior to study visits with the use of the IMS compared with logbooks (3.7 fewer events; p < .05;
No significant difference was observed in the daily frequency of SMBG measurements when the IMS was used compared with use of a logbook.
Conclusions
The results of this study show that use of the Accu-Chek Smart Pix IMS by patients with T1DM is associated with improved glycemic control and reduced incidence of hypoglycemia compared with the use of logbooks. The use of the instrument with its accompanying manual and consolidating training provided patients with a means to visually identify problematic points in their circadian glycemic fluctuations over a typical 2 week period.
As demonstrated in this study, pattern analysis can highlight points of high and low readings at the same testing time of each day so that reasons for glycemic excursions can be identified and a strategy put in place to address them. Analysis of glycemic data using information from handwritten diaries is a more arduous task that could discourage patients and impede their independence in managing their disease. 3 Advances in technology have made glycemic information more accessible to patients,8,9 and this has played a major role in enabling patients to make informed decisions on their insulin requirements. 10
Subjects were highly self-motivated in both arms of this crossover study. All patients in the analysis were compliant with the study protocol, which required regular SMBG measurements and use of diaries or the IMS. This further highlights the success of the IMS and training in improving glycemic regulation. The independent adjustments using data derived from the IMS were considered to be accurate by the study team and were effective in improving disease management, even after a short period of time.
Our study shows that use of the Accu-Chek Smart Pix IMS and accompanying training manual is highly educational in instructing patients on how to interpret their SMBG measurements and to adjust their insulin doses, even after a relatively short training period. The fact that there was no significant difference in the number of daily SMBG measurements associated with the use of the IMS suggests that the device's visualization of SMBG information, coupled with advice on therapy changes, may give patients greater ability to manage their glycemic status compared with the use of a conventional logbook.
The IMS displays aid the patient in visualizing points of time in which glycemic excursions may occur, thus enabling appropriate adjustments to insulin doses. There were significantly fewer hypoglycemic events with use of the IMS compared with use of data from a logbook, and a significantly higher proportion of patients were able to keep their blood glucose levels within the target range of 72–144 mg/dl, which is also of note in such a short time period of using the instrument.
One could expect influence resulting from the training sessions on IMS and manual at the beginning of the IMS period. However, from the HbA1c values for group 1 (which was using the IMS in the first part of the study), no carryover effect was visible at the end of the study, i.e., after switching to the logbook method, HbA1c values increased back to the baseline range (data not shown). This observation could point toward an immediate role of using an IMS together with the manual, as patients might already be well informed on how to act, but the IMS together with the manual might have supported them to recall their knowledge and act appropriately.
The main limitations of our pilot study are its very small size, short duration (only 12 weeks of Accu-Chek Smart Pix IMS use), and the fact that the population was limited to one center, which means that a Hawthorne effect cannot be discounted for either group. 11 Although the study did not have a placebo group, its crossover design allowed for comparison of the IMS and logbooks in the same patients.
The encouraging results from this pilot study warrant further investigation in a larger cohort over a longer time period. The study highlights a number of interesting points that are worthy of a more extensive analysis. For example, a more detailed investigation could include a comparison of glycemic control in group 1 patients in the second phase of the study, when they switched to logbooks, with that of group 2 patients using logbooks, who did not receive training until the crossover point. Such an analysis could differentiate between the influence of the training and the visualization of blood glucose data on glycemic control. A record of the frequency and types of therapy interventions carried out by the patients when in the different arms would also be of value.
Although we assume that patients carried out more therapy adjustments when using the IMS compared with the use of diaries, these data were not collected and would be of interest. In addition, a survey of patient opinion on the convenience and ease of use of the device in this highly motivated group of patients could indicate whether an IMS might help with the management of less compliant patients, such as adolescents and children.
In conclusion, this study shows that the graphical representation of data by an IMS, when accompanied by an explanatory manual that is easily understood, can be a valuable aid to help patients make accurate and timely self-adjustments to their insulin regimens.
Footnotes
Abbreviations:
This work was supported by an unrestricted grant from Roche Diagnostics Deutschland GmbH.
Andreas Reichel has received consulting fees from Roche Diagnostics Deutschland GmbH, is a member of advisory boards, and has received lecture fees for speaking at invitation from Novo Nordisk, Sanofi-Aventis, Eli-Lilly, and Roche Diagnostics. Annette Moritz is an employee of Roche Diagnostics Deutschland GmbH.
Acknowledgments:
Support for third-party editorial assistance for this manuscript, furnished by Dr. Ann Parkin, MediTech Media Ltd., was provided by Roche Diagnostics Deutschland GmbH.
