Abstract
Background:
This study aims to provide a better understanding of the ability of mobile health tools to offer glycemic control for patients with type 1 diabetes mellitus.
Methods:
Data gained from research articles searched in PubMed, Ovid (Medline), and CINAHL from 2005 to 2013 focused on interventions introduced to a type 1 diabetic population. Articles were screened to identify interventions that examined mobile health tools effect on glycemic control using %A1C as a proxy. Fourteen articles were included in this study. Descriptive data, %A1C difference, and statistical significance, if available, were extracted for comparison.
Results:
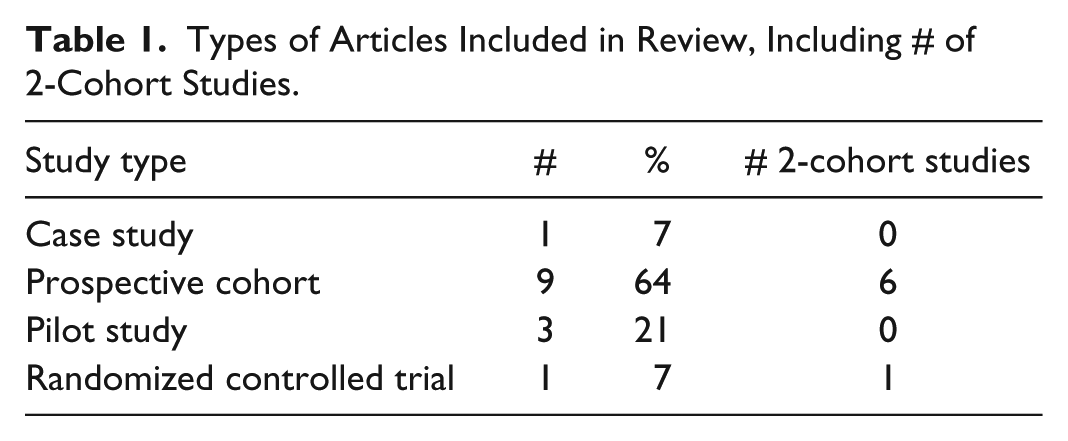
Five major categories were identified across the spectrum of interventions, including “Internet,” “Mobile,” “Mobile and Internet,” “Phone,” and “Videoconference and phone.” Seven of the 14 articles reported statistically significant decreases in measured outcomes. Seven studies examine a single cohort, and 7 examined a double cohort. Eleven of the 14 authors (79%) reported success with their intervention. Twelve studies reported a decrease in %A1C values in their intervention groups.
Conclusions:
Initial results for glycemic control through these tools appear promising, though inconclusive. Additional measures of mobile health tool efficacy should be assessed more directly. More rigorous study methods are also needed to improve the reliability of results.
Keywords
Diabetes is a chronic condition that continues to permeate the health care atmosphere as one of critical importance. Management of this condition and more specifically type 1 diabetes mellitus (T1DM) has drawn considerable attention as health care systems begin to renew their focus on costs and quality improvement with the ongoing implementation of the new health care law.1,2 At the same time telehealth, mobile health, and similar movements have all appeared in the health care sphere with increasing popularity as potential tools to aid persons with diabetes in the managing of their condition while lowering costs. 3 The rationale underlying the use of these tools is three-fold. First, these tools allow for enhanced communication with a provider. Second, patients are empowered to independently assess and control their disease state. 4 Finally, cost savings round out these benefits, demonstrating mobile health tools as one of the key technological instruments that attack chronic conditions in way that is affordable, effective, and accessible. 5 Broadly defined, these tools can be thought of as any use of mobile communications for health information and services. 6
Unfortunately, use of these tools as interventions in the disease states of person with type 1 diabetic persons has led to mixed results even as the incidence of T1DM in the United States remains among the highest in the world.7-10 Further studies specifically focusing on mobile tools impact on metabolic control outcomes have produced mixed results.11,12
T1DM merits particular attention, since it affects certain populations in much greater proportions than others. Several authors have documented the difficulty for pediatric populations to manage T1DM,13-15 while others have noted the unique challenges facing pregnant women diagnosed with T1DM.16,17 There is then a need to focus exclusively on the ability of these new digital tools to impact the aforementioned and general adult populations’ ability to manage T1DM with the aid of these tools. The objective then of this study was to review recent literature documenting the ability of mobile health tools to impact the glycemic control of type 1 diabetic patients.
Methods
A search of PubMed (2005-2013), CINAHL (2005-2013), and Ovid Medline (2005-2013) for research articles was conducted in October 2013 using the following search terms: self-management (Medical Subject Headings [MeSH]) and mobile health (MeSH) and type 1 diabetes mellitus (MeSH). These search terms and filters were used to find the most contemporarily relevant articles that embraced all methods of employing mobile health tools.
Inclusion and Exclusion Criteria
Inclusion criteria were any research article evaluating a diabetes self-care management technology that could be accessed in a mobile manner with a measured %A1C outcome for populations of persons diagnosed with T1DM. Studies were excluded if they did not have data on a patient population exclusively consisting of persons with T1DM, or which did not report outcomes including %A1C as a proxy for glycemic control. Previous work has identified %A1C as a viable proxy for glycemic variability, and this was continued here. 18
Study Selection and Data Extraction
The titles, abstracts, results, and citations were reviewed and screened based on the inclusion and exclusion criteria described earlier. Relevant data were individually collected from each screened article including patient sample, patient sample characteristics, intervention, intervention type, and length of intervention. The %A1C difference between start and end of study, descriptive statistics and statistical significance were also collected. The patient population characteristic data were sorted and then arrayed into a table, as was the intervention type. Data were analyzed for patterns based on differences in the intervention type used and the patient population measured. These population subgroups were analyzed in accordance with the original article to retain the intent and analysis of the originating data. For this reason, pregnant and pediatric populations were analyzed separately as well as in conjunction with the general adult cohort studies.
Categorical data concerning type of intervention were separated into categories, “Mobile,” “Mobile and Internet,” “Internet,” and “Videoconferencing and phone,” as was appropriate based on the description of the intervention. A second interventional tag described the service offered by the device; these included “message service,” “diary,” and “phone service.”
Population samples were adjusted based on the author’s description of those patients who fully participated in the study, for the entire duration of the study. For example, if the author stated that 30 patients were included at the start of the study but 5 patients dropped out during the study, the sample population (n) would be listed as 25. The mean duration of the intervention was recorded if given and rounded appropriately to give an integer.
Results
Comprehensive literature searches identified 54 articles (Figure 1). The titles and abstracts of these articles were read and 14 articles were ultimately determined to be potentially relevant. The distribution of articles included a predominance of cohort studies, with 3 prospective, or pilot studies, 1 randomized controlled trial, and 1 case study (Table 1). Six studies7-10,19-24 focused on a pediatric population and 1 study focused on a population of pregnant women. 25 The median study duration was 6 months (range, 1-9 months). It is worth noting that only 7 of these studies employed a methodology that compared an interventional group to a control group (Table 1). By each article’s author’s own determination of intervention success, 11 of the 14 authors (79%) considered their interventions successful. The studies deemed unsuccessful by the author’s determination were for failing to produce a statistically significant effect or the desired effect at all.21,26,27 Seven of the 14 studies reported statistically significant declining %A1C values.19,20,23,25,26,28,29
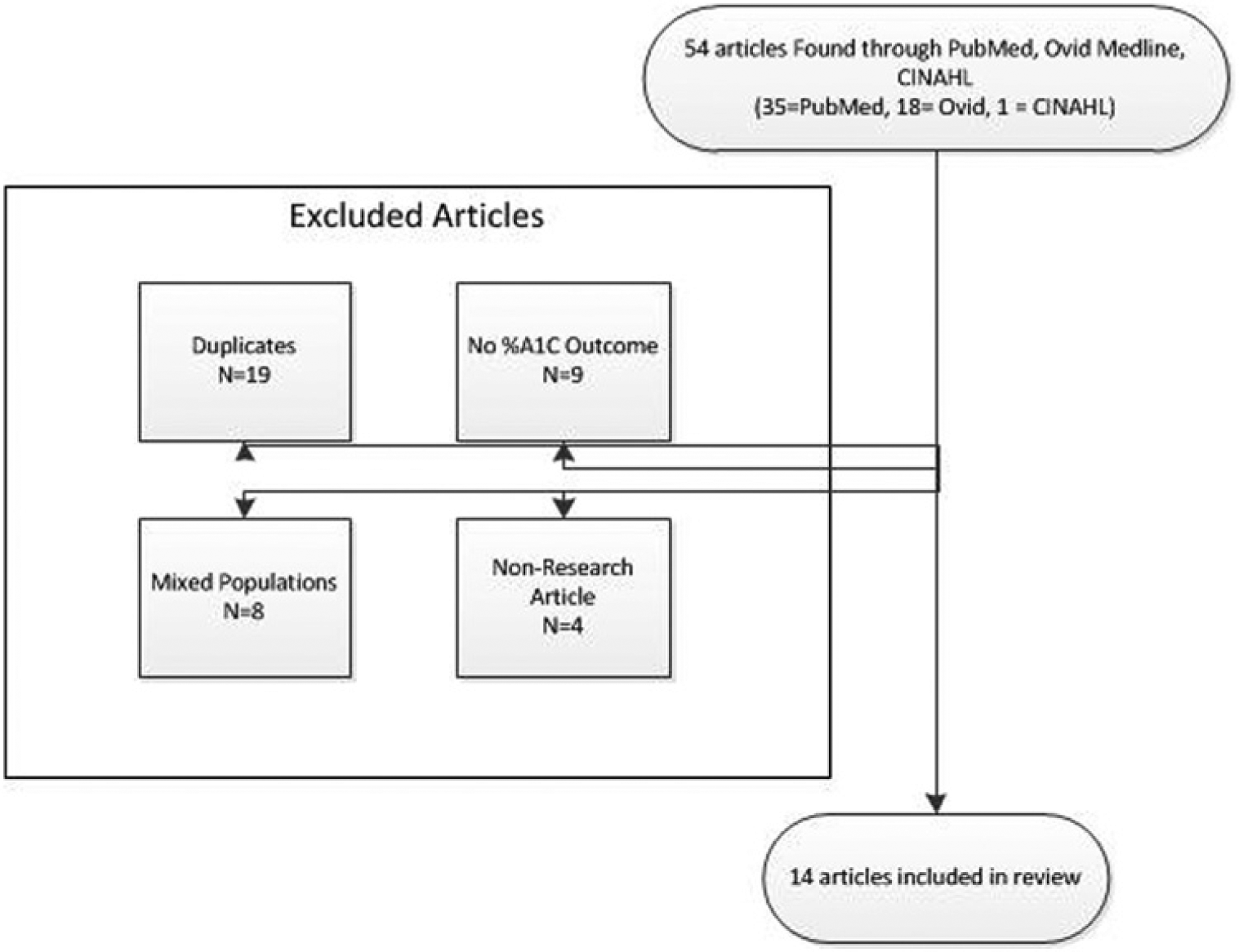
Search strategy flow diagram.
Types of Articles Included in Review, Including # of 2-Cohort Studies.
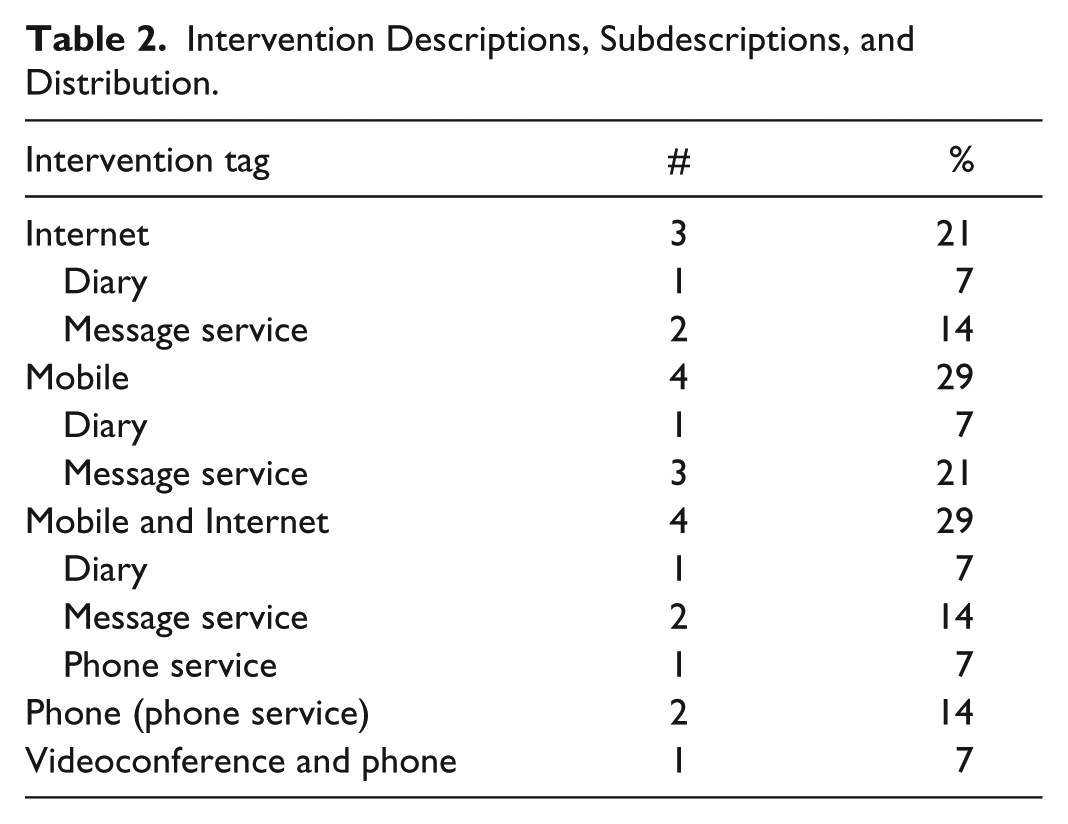
Five intervention approaches were observed in these studies: Internet based,23,26,30 Mobile based,25,28,31,32 Mobile and Internet based,20,22,27,33,34 a telephone service,21,24 and an optional videoconference and phone service (Table 2). 19 The type of service offered further categorized these interventions into 3 broad categories of “message service,” “phone service,” and “diary” (Table 2). The videoconference with phone service article was excluded from this second categorization since its service is self-descriptive.
Intervention Descriptions, Subdescriptions, and Distribution.
Single-Cohort Studies With %A1C Outcome
There were 127 subjects involved in single-cohort studies using %A1C as an outcome. Among these 7 articles, 4 reported statistically significant results. All of these reported a decrease in the mean %A1C value (Table 3). The highest of these changes was −3.2%, the lowest was −0.33%, and the median was −0.82%.
Single Cohort Studies With %A1C Change and Population Characteristics.
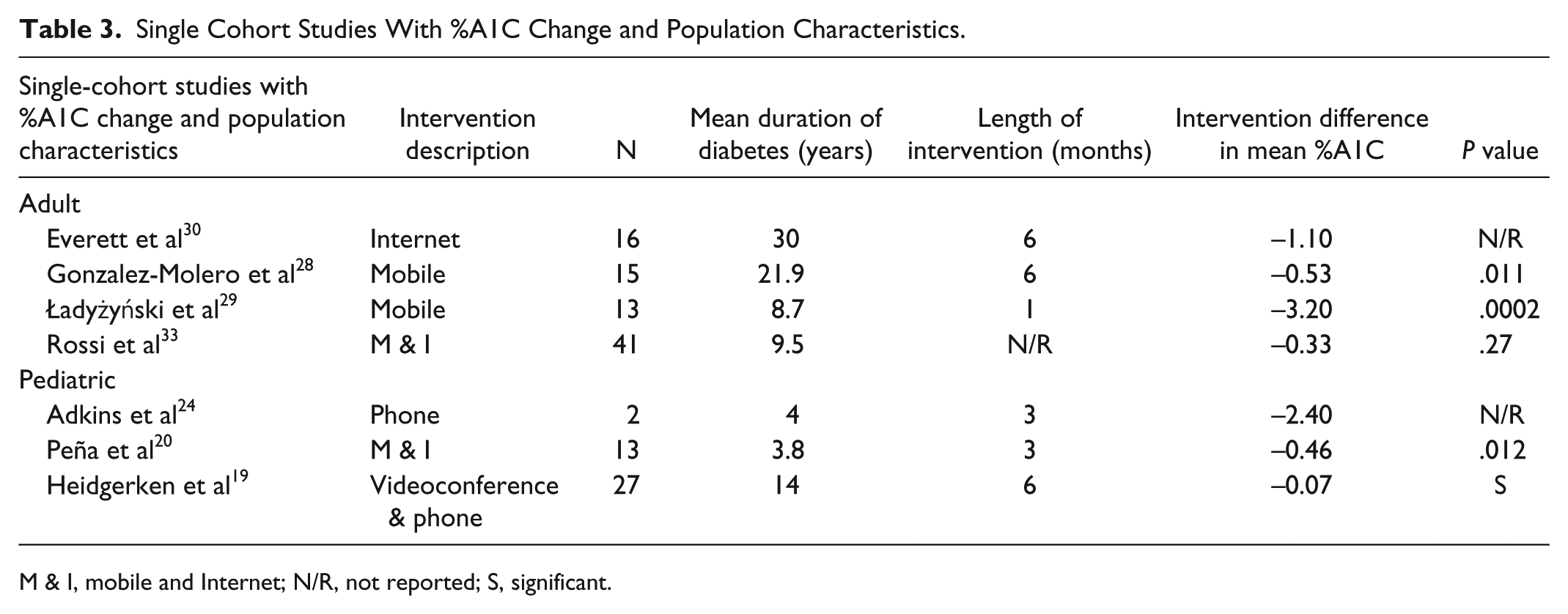
M & I, mobile and Internet; N/R, not reported; S, significant.
Two-Cohort Studies With %A1C Outcome
Among the comparative studies reporting %A1C values there were 295 subjects involved. There was no study population that had more than 50 people in either the control or the interventional group. Among these, only 3 reported a statistically significant finding for comparing the 2 groups %A1C differences. It is also worth noting that only 1 of all these studies had a randomized subject base. 27 Amongst the control population, the range of %A1C difference from start to end was −5.10% to 0.98%, with a median value of −0.04%. The intervention groups’ range of %A1C difference values is −1.40% to 0.40%, with a median value of −0.68% (Table 4).
Two-Cohort Studies With %A1C Change, Population Characteristics, and
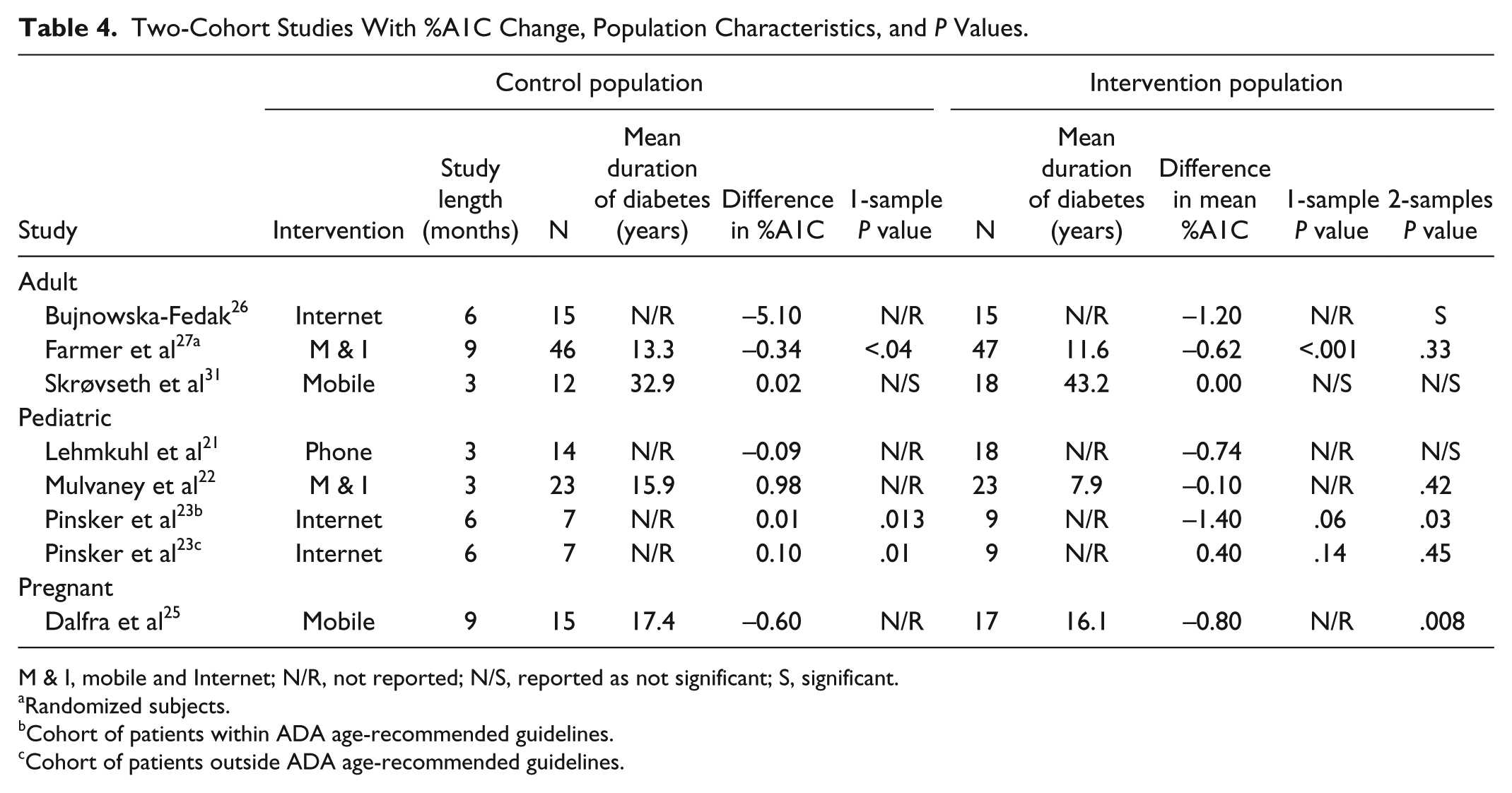
M & I, mobile and Internet; N/R, not reported; N/S, reported as not significant; S, significant.
Randomized subjects.
Cohort of patients within ADA age-recommended guidelines.
Cohort of patients outside ADA age-recommended guidelines.
Pediatric Population Subgroup Analysis
There were several studies that analyzed an exclusively pediatric population.19-24 Three of these studies found a statistically significant decrease.19,20,23 The range of %A1C difference in the pediatric intervention populations was −2.80% to 0.40%, with a median of −0.60%.
Pregnant Population Subgroup Analysis
One study examined the effect of a mobile health management intervention in a population of type 1 diabetic pregnant women. 25 Using a mobile intervention, Dalfra et al’s 25 intervention population experienced a statistically significant 0.8% decrease in %A1C from start to end of the study, compared to the control group’s 0.6% decrease in %A1C (Table 4).
Discussion
This systematic review focused on the ability of mobile health tools to grant patients with T1DM greater glycemic control. These tools took a variety of forms, and provided a number of different services to aid diabetic patients. The majority of authors determined their intervention to be successful. This coincides with the majority of studies that demonstrated a decrease in %A1C. However, not all of these values were statistically significant. In addition, the predominant use of prospective studies in place of randomized trials fails to demonstrate with any meaningful certainty the efficacy of these tools. It is worth noting that the overall impression of these tools’ ability to aid diabetic patients with their conditions remains positive.
While certain studies focused on the effect of these interventions to aid specific subpopulations within the type 1 diabetic population, very little evidence of success was found. Thus, the muddied picture of how these tools significantly affect persons with T1DM is not made any clearer by examining 1 subpopulation in comparison to another. Further work in this area is needed to remedy this.
Certain limitations must be kept in mind when considering this study. First, a concerning number of results with nonsignificant
Looking ahead, there is significant space for research of increased breadth and depth in this area. A cursory glance at the “health” section of Apple’s App Store or the shelves of a pharmacy store reveals a plethora of mobile applications and tools designed to aid persons with diabetes in the management of their condition. Yet there is little evidence to guide physicians or consumers on which tools are best. Furthermore, while existing systematic reviews have addressed the impact of mobile health tools and telemedicine on T1DM in addition to type 2 diabetes mellitus, 11 as well as the advantages of monitoring and communicating with patients remotely, 36 there is still a need for focused research exclusively on T1DM that assesses glycemic outcomes. Further research must establish this evidence and provide transparency for consumers and providers interested in these mobile health tools so a better understanding of their efficacy may be established.
Footnotes
Acknowledgements
The author wishes to thank Dr. Suzanne Boren for her advice and guidance when constructing this review.
Abbreviations
ADA, American Diabetes Association; MeSH, Medical Subject Headings, T1DM, type 1 diabetes mellitus.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
