Abstract
Background
The quantitative assessment of the oculomotor system has emerged as a promising biomarker for neurodegenerative disorders. Although oculomotor impairments are commonly observed in multiple system atrophy (MSA) patients, the specific abnormalities and underlying neural structural changes remain poorly understood.
Objectives
To explore oculomotor abnormalities and associated brain changes in MSA, evaluating their potential as biomarkers for diagnosis and disease monitoring.
Methods
A total of 100 MSA patients and 50 healthy controls (HCs) were included in this study. All subjects underwent comprehensive evaluations, including clinical assessments, virtual reality (VR)-based ocular-tracking tasks and structural magnetic resonance imaging (MRI).
Results
Compared with HCs, MSA patients showed significantly impaired smooth pursuit (SP) with increased number of deviations (13.33 [29.33] vs. 5.67 [7.67], p < 0.001); reduced prosaccadic (PS) average velocity (194.80 ± 82.45 °/s vs. 263.07 ± 68.17 °/s, p < 0.001); and reduced antisaccade (AS) average velocity (165.82 ± 85.75 °/s vs. 257.05 ± 74.39 °/s, p < 0.001). A combination of PS and AS average velocities with SP number of deviations effectively distinguished MSA patients from HCs with an AUC of 0.814. PS average velocity was negatively correlated with UMSARS total scores (r = -0.354, p < 0.001), whereas AS accuracy was positively correlated with MoCA scores (r = 0.375, p = 0.001). Voxel-based morphometry revealed significant associations between these oculomotor parameters and atrophy in the cerebellum and frontal gyrus (p < 0.05, family-wise error correction).
Conclusions
Our study provides comprehensive insights into the VR-based quantitative oculomotor analysis and its association with regional brain atrophy in MSA, contributing to novel biomarkers identification and therapeutic targets exploration.
Plain Language Summary Title
Using Virtual Reality to Measure Eye Movement Problems and Brain Shrinkage in Multiple System Atrophy
Plain Language Summary
What is the research about? Multiple system atrophy (MSA) is a rare, progressive brain disease that causes problems with movement, balance, and other body functions. People with MSA often have trouble with eye movements, but we don't fully understand these issues or how they relate to brain changes. This study aimed to use a new virtual reality (VR) tool to measure eye movements and link them to brain shrinkage, to find better ways to diagnose and track the disease. Why is this important? MSA is hard to diagnose early because symptoms overlap with other conditions like Parkinson's disease. Current tests rely on doctors' observations, which aren't always precise. Eye movements involve many brain areas affected by MSA, so measuring them accurately could provide objective “biomarkers” for earlier detection and monitoring disease progression. How was the study done? We studied 100 people with MSA and 50 healthy volunteers. Everyone had clinical check-ups, VR-based eye-tracking tests (measuring how eyes fixate, follow moving objects, and make quick jumps), and brain MRI scans to measure brain volume in key areas. What did we find? People with MSA had worse eye movements than healthy volunteers, especially in following objects smoothly and making voluntary or intentional eye jumps. These problems helped distinguish MSA from healthy people. The eye issues were linked to disease severity, and corresponded to shrinkage in brain areas like the cerebellum and frontal regions. What does this mean? VR eye-tracking offers a simple, portable way to spot MSA changes that match real-life behaviors, unlike traditional tests. This could lead to better biomarkers for diagnosing MSA early and testing new treatments, improving care for patients. Future studies should compare MSA to similar diseases and track changes over time.
Introduction
Multiple system atrophy (MSA) is a progressive neurodegenerative disorder characterized by widespread neuronal loss and gliosis in the central nervous system, particularly affecting striatonigral, olivopontocerebellar, and central autonomic structures.1–3 Clinically, MSA presents a diverse spectrum of progressive autonomic, parkinsonian, cerebellar, and pyramidal symptoms. 4 It is classified into two subtypes based on the predominant motor phenotype: parkinsonian (MSA-P) and cerebellar (MSA-C). However, the overlap and variability of these clinical features pose significant challenges for early diagnosis. 5 Current diagnostic approaches rely heavily on clinical observations and patient-reported outcomes, which often lack the sensitivity and specificity needed to monitor disease progression or assess therapeutic efficacy in clinical trials.6–8 Consequently, identifying reliable and quantifiable biomarkers for MSA is critical for advancing future research. 9
Oculomotor assessment is an essential part of the neurological examination, as it involves neural circuits spanning the cerebrum, brainstem, and cerebellum. 10 In MSA, typical abnormal oculomotor features include gaze-induced nystagmus, mild to moderate hypometria of saccades, impaired smooth pursuit (SP) eye movements with decreased gain, and excessive square-wave jerks.11,12 Among these, saccadic abnormalities are a highly prevalent impairment in patients with MSA and are considered to hold potential as diagnostic biomarkers. The antisaccade (AS) task, which requires suppression of reflexive responses, largely depends on frontostriatal circuits, and their modulation by the superior colliculus and cerebellum.13,14 This task has been shown to correlate with disease progression in Parkinson's disease (PD), where nigrostriatal deficits disrupt frontostriatal circuits.15,16 Given MSA involves more extensive neurodegeneration in these pathways compared to PD, similar AS impairments may reflect MSA progression. However, these oculomotor impairments have been scarcely studied in MSA.
Recent virtual reality (VR)- based quantitative eye movement assessments present several advantages over traditional approaches. 17 First, this approach enables sensitive detection of clinically significant oculomotor signatures with high objectivity and reproducibility, independent of clinician experience required for bedside assessment. 18 Moreover, VR delivers stimuli in an immersive three-dimensional environment that minimizes external interference and elicits natural gaze behaviors, providing a closer approximation to real-world dynamics than the static screens used in traditional quantitative assessments. Additionally, the intuitive interface and portability of VR systems facilitate efficient and accessible operation in diverse settings. 19
Despite these advantages, applications to MSA remain unexplored. Moreover, growing evidence suggests that oculomotor parameters reflect specific disruptions within affected cerebral networks.20,21 Combining oculomotor analysis with neuroimaging could help clarify the anatomical substrates of these abnormalities and provide deeper insights into their neural mechanisms. However, current evidence remains limited by small cohort sizes and restricted oculomotor paradigms in prior studies.21,22
To bridge these gaps, our study comprehensively investigated oculomotor alterations and their neural underpinnings in a large MSA cohort, combining VR-based eye-tracking systems, clinical assessments and structural MRI. We aimed to evaluate their potential as reliable biomarkers.
Methods
Participants study
In this cross-sectional study, we recruited 100 patients with MSA (60 MSA-C and 40 MSA-P) from the Department of Neurology, Xiangya Hospital of Central South University between February 2023 and February 2025. The inclusion criteria required (1) independent availability of complete eye-tracking assessment; (2) a diagnosis of clinically established or clinically probable MSA according to the 2022 Movement Disorder Society Criteria, 23 which include autonomic failure, parkinsonism, or cerebellar ataxia in conjunction with supportive clinical and imaging findings. Diagnoses were established by two experienced neurologists, with an independent blind diagnosis from a third movement disorders expert included in cases of discrepancy. Spinocerebellar ataxia 1, 2, 3, 6, 7, 17 and dentatorubral-pallidoluysian atrophy (DRPLA) were genetically excluded. Additionally, 50 gender- and age-matched healthy controls (HCs) were recruited from patients’ family members and the Physical Examination Center at Xiangya Hospital. All HCs, who had no history of neurological disorders and were able to independently complete the full eye-tracking assessment, underwent standardized screening including review of medical history, basic neurological and imaging examinations.
The exclusion criteria for all participants were as follows: the history of epilepsy, stroke, brain surgery, or psychiatric disease (e.g., schizophrenia, bipolar disorder), unrelated intracranial organic lesions; peripheral vestibulopathy and ophthalmic diseases; other systemic conditions or diseases that could have caused ocular symptoms affecting eye tracking; the history of alcohol or drug abuse; MRI contraindications.
The study received approval from the Ethics Committee of Xiangya Hospital of Central South University, and all participants provided informed consent before their inclusion in the study.
Clinical assessments
All patients underwent clinical assessments by two experienced neurologists. Disease severity was assessed using the Hoehn and Yahr Parkinson's disease staging scheme (H&Y stage) and the Unified Multiple System Atrophy Rating Scale (UMSARS), which includes activities of daily life (UMSARS-I), motor symptoms (UMSARS-II), and chore-based disability (UMSARS-IV). 24 Cognitive function was comprehensively evaluated using the Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA) for global cognitive performance, 25 while the Frontal Assessment Battery was used for the assessment of frontal executive function (FAB). 26
Eye movement recording
Eye movements were recorded binocularly using the National Medical Products Administration-approved eye-tracking assessment system (EyeKnow; Beijing CAS-Ruiyi Information Technology Co., Ltd). The system comprises a tablet computer and a VR headset featuring an eye-tracking camera, with a sampling rate of 120 Hz. The VR headset serves to guide the subject's eye movements and record oculomotor data. Prior to testing, participants completed a nine-point calibration procedure to achieve a maximum calibration error no greater than 2° radius. Participants followed a predesigned protocol to assess central fixation, SP, prosaccade (PS), and AS. Data were processed in real-time using the system's integrated processing module (the detailed procedures are described in the supplementary materials).
The EyeKnow system has been rigorously validated throughout the registration and approval process to ensure their feasibility, reliability, stability, precision, and safety. The utility of these systems has also been confirmed in previous publications.27,28
Image acquisition and data processing
All participants underwent brain MRI scans using a 3.0 T MRI scanner (Discovery CT750 HD, GE Healthcare). High-resolution T1-weighted images were acquired with the following parameters: number of slices=190; repetition time (TR) = 8.5 ms; echo time (TE) = 3.2 ms; flip angle=12°; field of view=256 × 256 mm; voxel size=1 × 1 × 1 mm; and slice thickness=1.0 mm. Total scan duration was 4 min and 43 s. MRI scans in DICOM format were converted to NIFTI using MRIcro software and processed with the CAT12 toolbox in SPM12 (Wellcome Trust Center for Neuroimaging) via MATLAB R2022a (MathWorks, Natick, MA, USA). Voxel-based morphometry (VBM) analysis was performed, involving image segmentation and spatial normalization using the DARTEL method to generate probability maps for gray matter (GM), white matter (WM), and cerebrospinal fluid (CSF). 29 Image quality was assessed through manual visual inspection and SPM's quality metrics. Gray matter volume (GMV) images were smoothed with an 8 mm Gaussian filter. All these tasks were performed in adherence to the protocols outlined in the Cat12 manual.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics 31.0 (SPSS Inc.), with a significance threshold of p < 0.05 (two-tailed). The Shapiro-Wilk and Levene tests were applied to confirm the normal distribution of data and homogeneity of variances, respectively. Continuous data were presented as mean ± standard deviation (SD) or median (interquartile range), with parametric data analyzed using t-tests. Categorical data were expressed as counts (%) and assessed via chi-squared (χ2) tests. Non-parametric data analyses were carried out using Mann-Whitney U tests. Oculomotor parameters across groups were compared using t-tests or Mann-Whitney U tests. Diagnostic accuracy was evaluated using receiver operating characteristic (ROC) curves, with optimal thresholds determined by the Youden index and area under the curve (AUC) values compared using the DeLong method. Correlations between oculomotor parameters and clinical variables were examined using Spearman tests. Multiple regression, adjusting for age, gender, and total intracranial volume (TIV), was employed to investigate associations between oculomotor parameters and GMV in brain regions showing significant group differences (p < 0.05, family-wise error correction).
Results
Demographic and clinical characteristics
Demographic and clinical data are presented in Table 1. The study comprised 150 subjects: 100 MSA patients and 50 HCs. No significant differences were observed in age, sex, body mass index (BMI), or years of education between MSA patients and HCs. Among MSA patients, 56 (56%) were male, with a median age of 55.00 (10.0) years. For HCs, 23 (46%) were male, with a median age of 59.00 (10.0) years.
Demographic and clinical characteristics.
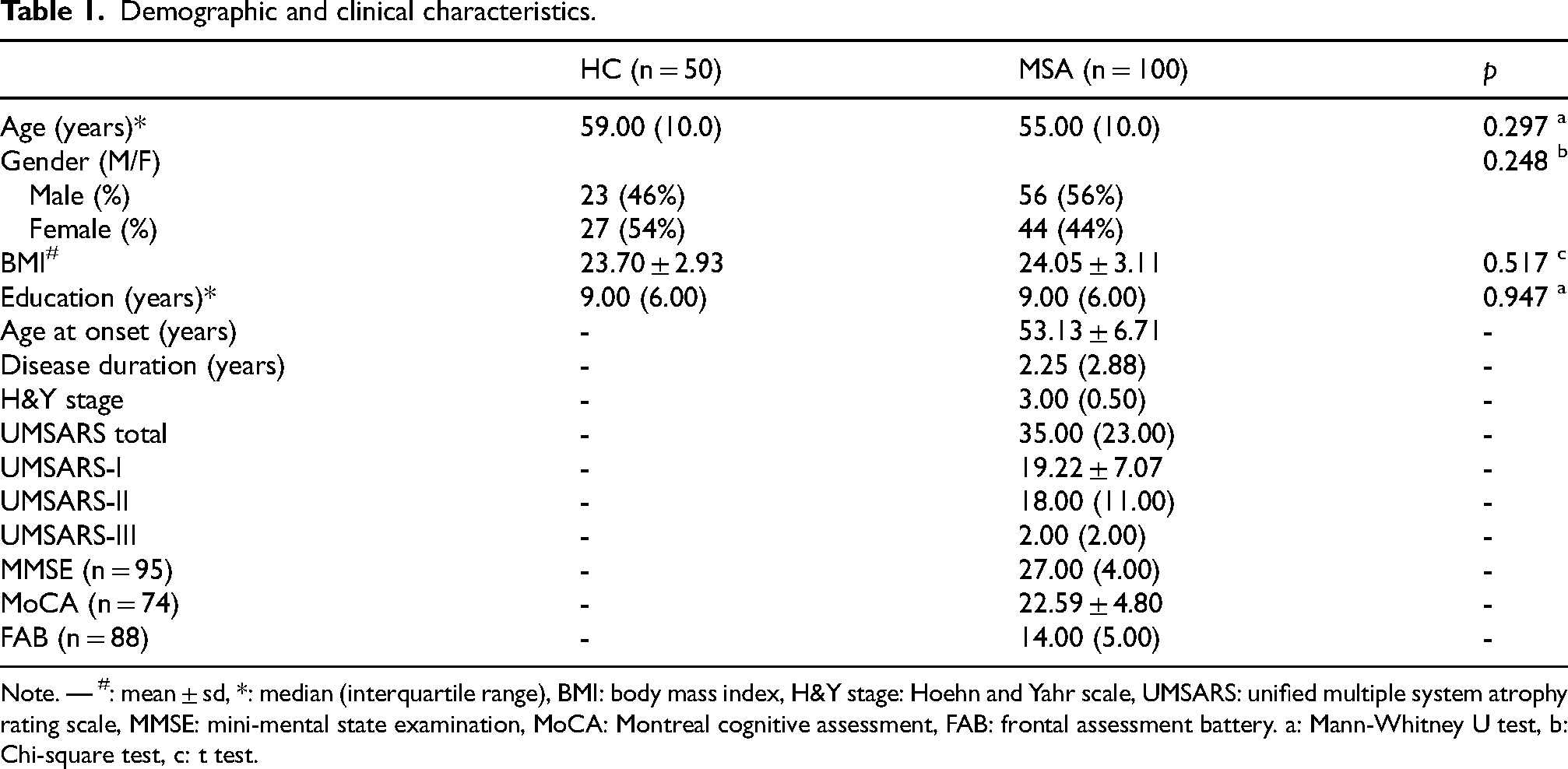
Note. — #: mean ± sd, *: median (interquartile range), BMI: body mass index, H&Y stage: Hoehn and Yahr scale, UMSARS: unified multiple system atrophy rating scale, MMSE: mini-mental state examination, MoCA: Montreal cognitive assessment, FAB: frontal assessment battery. a: Mann-Whitney U test, b: Chi-square test, c: t test.
The group differences of oculomotor parameters
Figure 1 illustrates examples of the four eye movement tasks for a typical MSA and HC. In summary, MSA patients exhibited inferior performance compared to HCs across four aspects of oculomotor function (Table 2). The detailed analysis results of these four aspects are as follows.
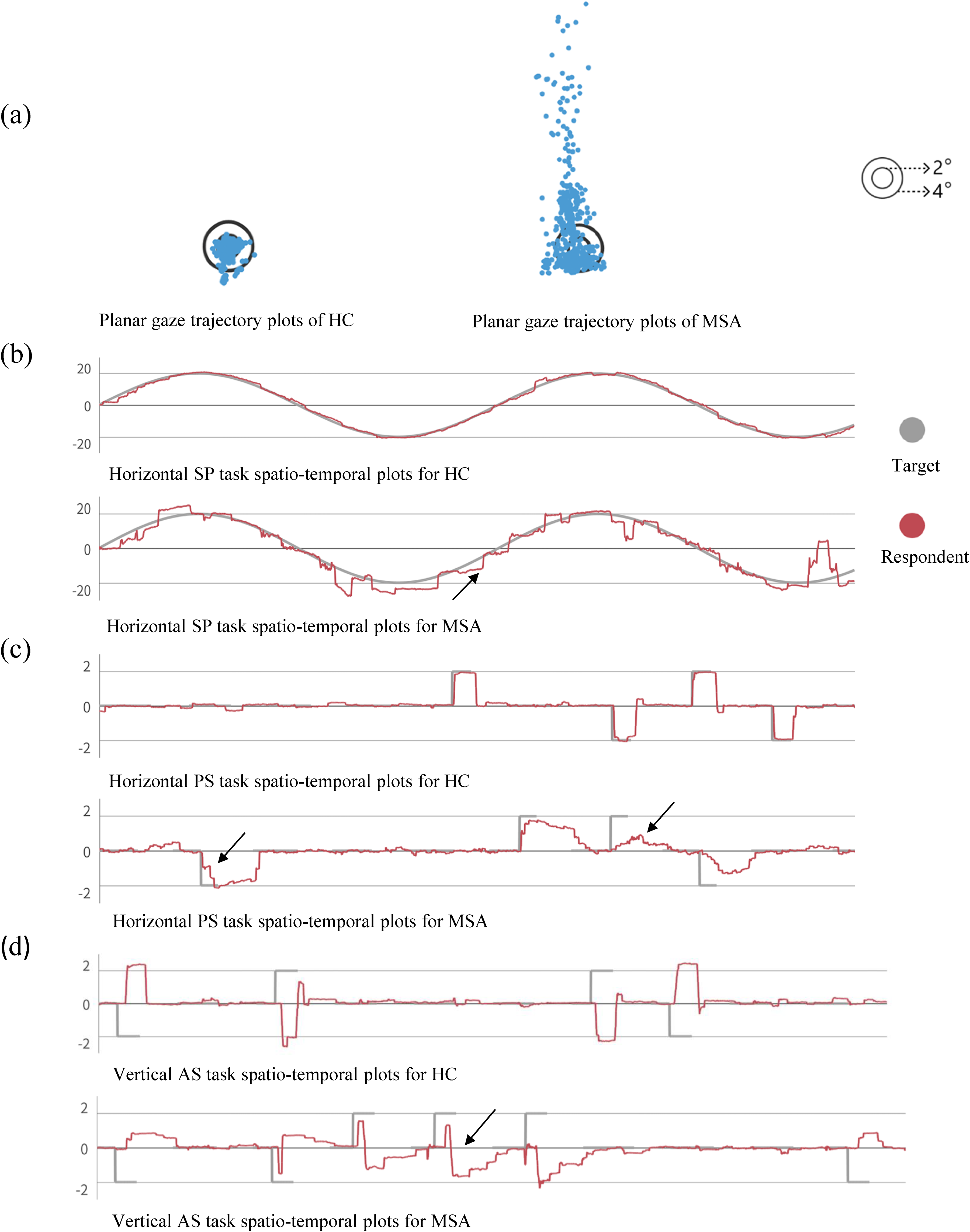
Examples of the four eye movement tasks for a typical MSA and a typical HC. (a) In the fixation task, planar gaze trajectories for the MSA group showed more frequent deviations from the target. (b) The trajectories demonstrate the position changes of the target point (gray line) and participant gaze position (red line). In the smooth pursuit (SP) task, trajectories of the MSA group's paths were disorganized, characterized by frequent catch-up saccades. (c) The horizontal prosaccade (PS) task analysis showed that the MSA group had slower saccades and exhibited saccadic hypometria. (d) In the vertical antisaccade (AS) task, the MSA group demonstrated a higher error rate and extended correction reaction time.
Oculomotor characteristics of HC and MSA.
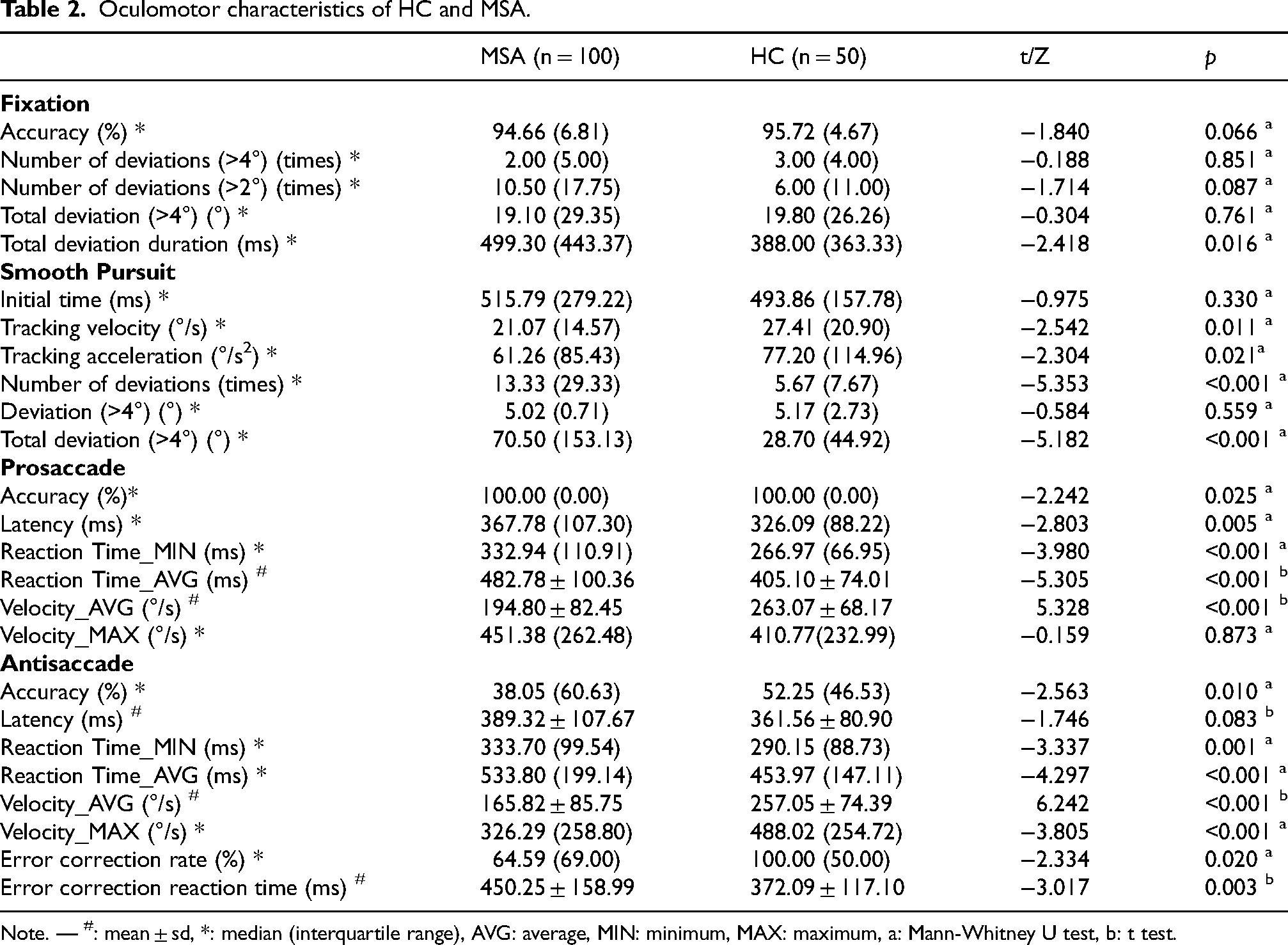
Note. — #: mean ± sd, *: median (interquartile range), AVG: average, MIN: minimum, MAX: maximum, a: Mann-Whitney U test, b: t test.
Fixation stability
Patients with MSA exhibited significantly longer total deviation duration than HCs (p = 0.016). No significant differences were observed in accuracy (p = 0.066), number of deviations (>4°) (p = 0.851), number of deviations (>2°) (p = 0.087) or total deviation (>4°) (p = 0.761).
Smooth pursuit
The MSA group showed significantly SP impairment with decreased tracking velocity (p = 0.011) and acceleration (p = 0.021) versus HCs. Both number of deviations (p < 0.001) and total deviation (p < 0.001) were significantly higher in MSA patients. Initial time (p = 0.330) and deviation (>4°) (p = 0.559) did not differ between groups.
Prosaccade
MSA patients exhibited reduced accuracy (p = 0.025) and longer latency (p = 0.005) during target-directed saccades compared to controls. Additionally, the reaction time of PS for the MSA group was significantly slower than HCs, both average (p < 0.001) and minimum (p < 0.001). Average saccadic velocity was significantly reduced in MSA patients (p < 0.001), but maximum saccadic velocity showed no significant difference (p = 0.873).
Antisaccade
The MSA group had significantly lower AS accuracy (p = 0.010) and slower reaction times, both average (p < 0.001) and minimum (p = 0.001) compared to HCs. Both average (p < 0.001) and maximum (p < 0.001) AS velocities were significantly reduced in MSA patients. Latency showed no significant difference (p = 0.083). Error correction rate (p = 0.020) was significantly lower in MSA than HCs with higher error correction reaction time (ms) (p = 0.003).
No significant differences in oculomotor parameters were observed between MSA-C and MSA-P subtypes (Supplementary Table 1).
Diagnostic values of oculomotor parameters
ROC curves were generated to investigate whether oculomotor parameters could distinguish MSA patients from HCs. The AS average velocity showed an AUC of 0.793 (sensitivity, 0.722 and specificity, 0.787), followed by SP number of deviations with an AUC of 0.772 (sensitivity, 0.794 and specificity, 0.673) and PS average velocity with an AUC of 0.732 (sensitivity, 0.602 and specificity, 0.816). A combined ROC analysis of AS average velocity, SP number of deviations and PS average velocity improved discrimination, achieving an AUC of 0.814 (sensitivity, 0.772 and specificity, 0.800) (Figure 2).
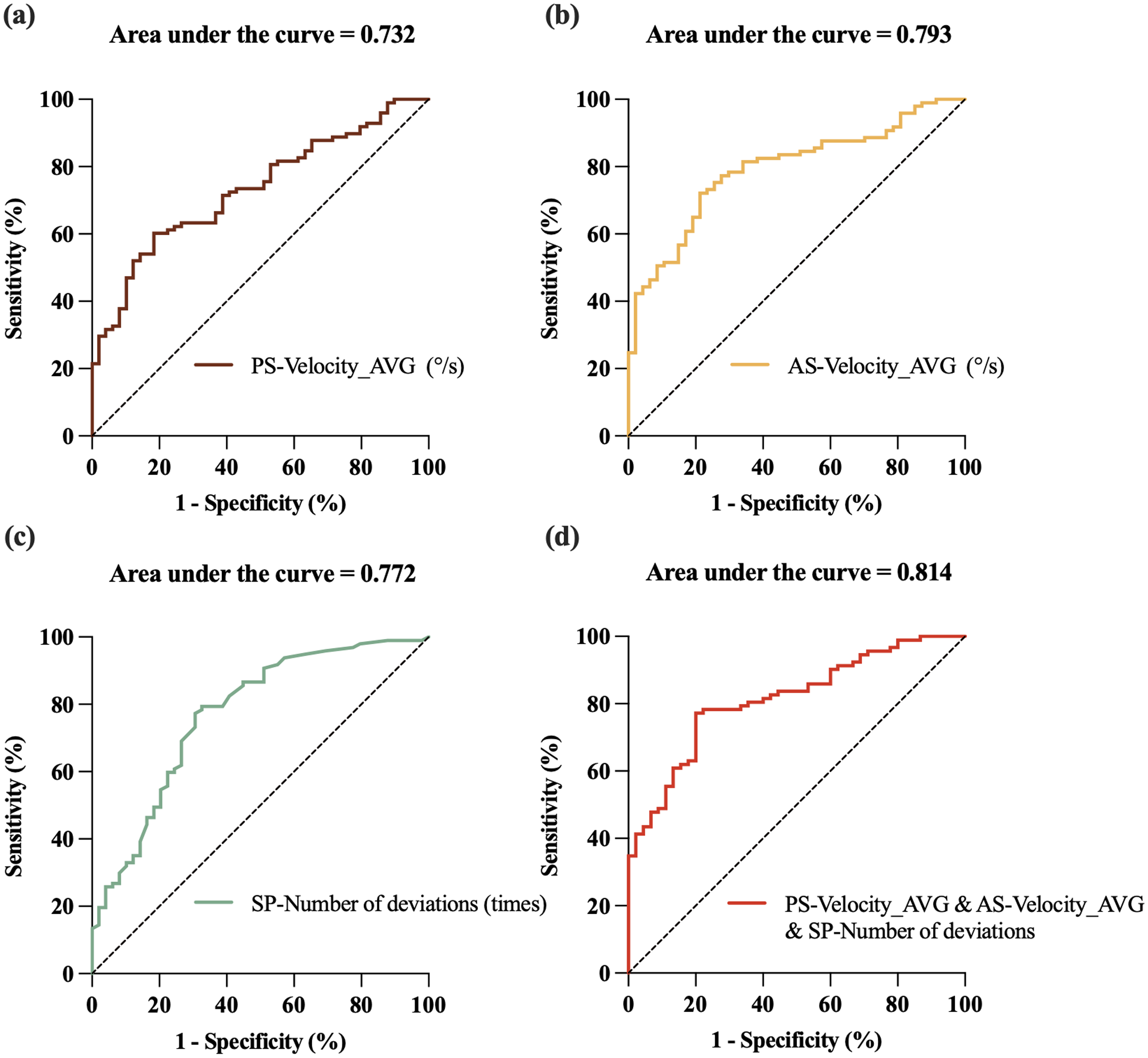
ROC curves to evaluate the use of oculomotor parameters for the discrimination of MSA patients from HCs. ROC, receiver operating characteristic curve, MSA: multiple system atrophy, HC: health control, SP: smooth pursuit, PS: prosaccade, AS: antisaccade, AVG: average.
Associations between oculomotor parameters and clinical variables
Correlations between clinical characteristics and oculomotor parameters were analyzed for 100 MSA patients (Table 1&2). SP number of deviations and total deviation were positively correlated with disease duration (r = 0.331, p < 0.001 and r = 0.334, p < 0.001, respectively). PS average velocity and SP number of deviations were significantly associated with H&Y stage (r = -0.332, p < 0.001 and r = 0.325, p = 0.001, respectively). PS average velocity was negatively correlated with UMSARS total and UMSARS-II scores (r = -0.354, p < 0.001 and r = -0.379, p < 0.001, respectively). AS average velocity was negatively correlated with UMSARS-II scores (r = -0.286, p = 0.005), while SP number of deviations was positively correlated with UMSARS-IV scores (r = 0.349, p < 0.001). AS error correction rate was negatively correlated with UMSARS total and UMSARS-II scores (r = -0.413, p < 0.001 and r = −0.429, p < 0.001, respectively). Additionally, after adjusting for educational level, oculomotor parameters showed significant correlations with cognitive scores. AS accuracy and error correction rate were positively correlated with MOCA scores (r* = 0.375, p* = 0.001 and r* = 0.409, p* < 0.001, respectively).PS latency was negatively correlated with MMSE scores (r* = -0.403, p* < 0.001). PS average reaction time was correlated with MOCA scores (r* = -0.325, p* = 0.006) and FAB scores (r* = -0.340, p* = 0.002). AS error correction reaction time was associated with FAB scores (r* = -0.270, p* = 0.031) (Figure 3).
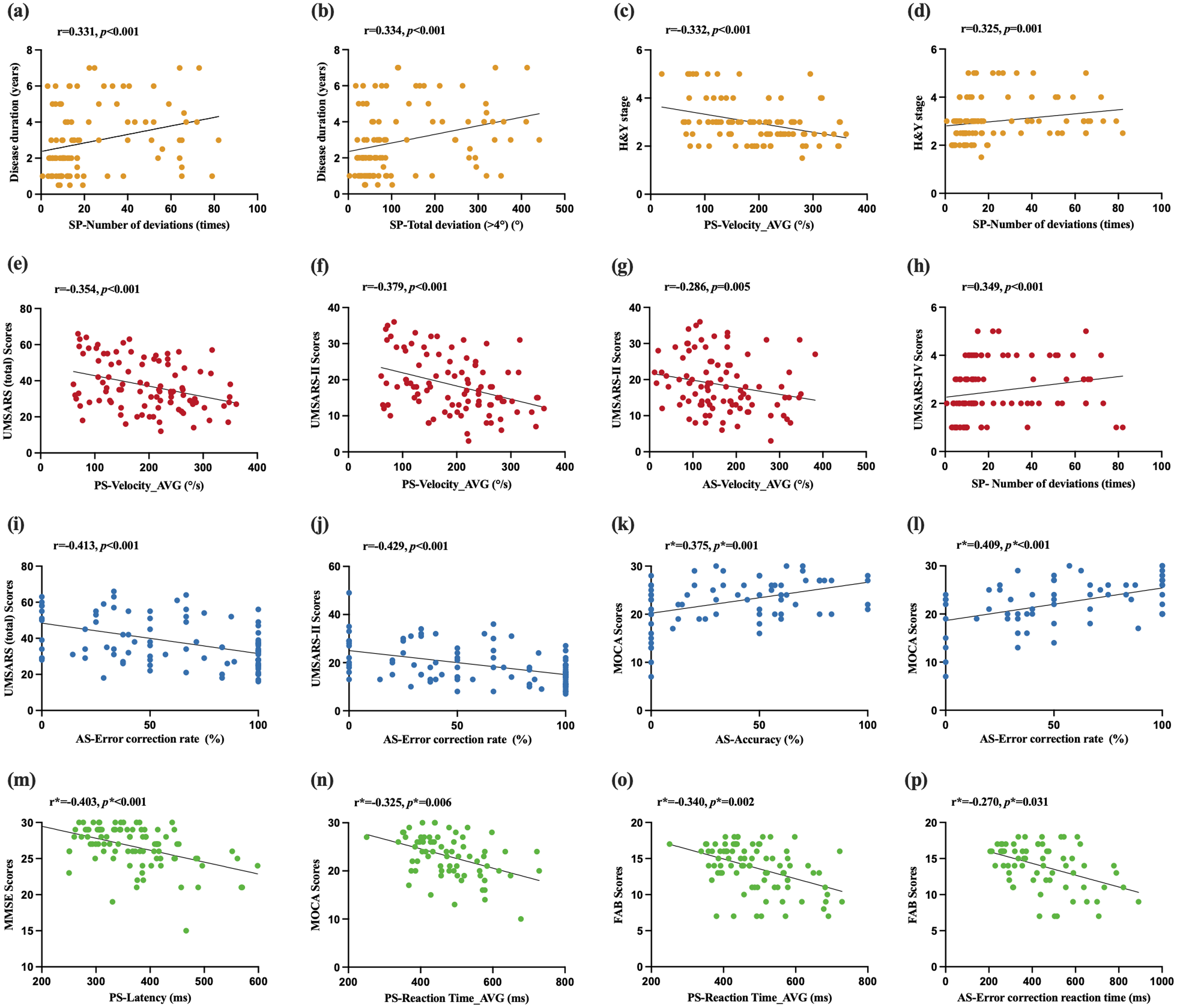
Correlation analysis between oculomotor parameters and clinical variables. r values and p values calculated using Spearman's correlation coefficient analysis are indicated on each dot plot, *: adjusted for education years. PS: prosaccade, AS: antisaccade, SP: smooth pursuit, AVG: average, UMSARS: unified multiple system atrophy rating scale, MMSE: mini-mental state examination, FAB: frontal assessment battery, MoCA: Montreal cognitive assessment.
Associations between oculomotor parameters and brain atrophy
Correlation analyses were conducted in 100 MSA patients and 50 HCs with high-resolution MRI and concurrent eye-tracking data to examine relationships between regional gray matter atrophy and oculomotor performance. MSA patients exhibited significant cerebellar and frontal lobe atrophy compared to HCs (Supplementary Table 2). In multivariate analysis adjusted for age, gender, and TIV, vermis atrophy was significantly associated with PS average velocity in MSA patients. Bilateral cerebellar hemisphere volumes were positively correlated with AS average velocity. In the SP task, number of deviations was significantly correlated with atrophy in the right cerebellum, right middle frontal gyrus, and left superior frontal gyrus (p < 0.05, family-wise error correction) (Figure 4 and Table 3). No significant correlations were observed in HCs (p > 0.05, family-wise error correction). These findings indicated that oculomotor measures (PS and AS average velocity, SP number of deviations) and GMV in specific brain regions (cerebellum and frontal lobe) not only showed significant alterations in MSA but also demonstrated oculomotor-volumetric couplings compared to HCs.
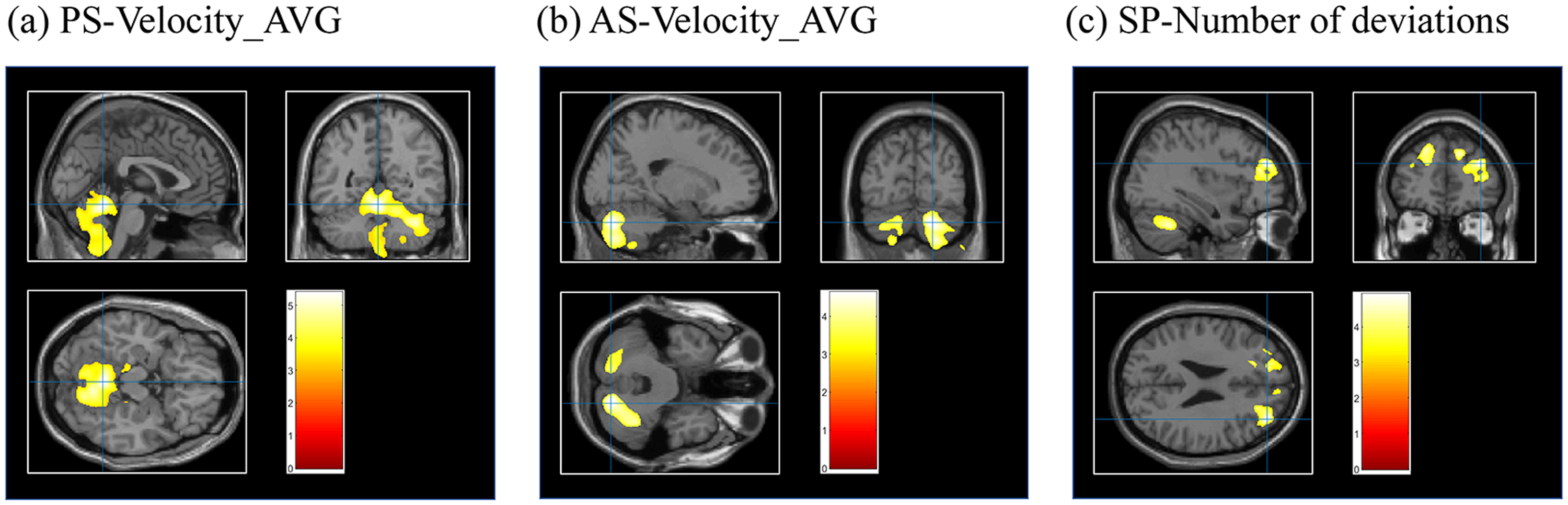
MRI T1W1 whole-brain voxel-based analysis VBM results. Regions of gray matter volumes associated with oculomotor parameters in MSA (p < 0.05, family-wise error correction). a: PS-Velocity_AVG (+); b: AS-Velocity_AVG (+); c: SP-Number of deviations (-). PS: prosaccade, AS: antisaccade, SP: smooth pursuit, AVG: average. The color bar represents t-values.
MRI T1W1 whole-brain voxel-based analysis VBM results: regions of gray matter volumes associated with oculomotor parameters in MSA (p < 0.05, family-wise error correction).

Note. MNI: Montreal Neurological Institute, L: left, R: right.
Discussions
In this study, we investigated oculomotor function in patients with MSA and its relationships with clinical characteristics and regional brain volumes. Using a portable and efficient eye-tracking system integrated with VR, we found that MSA patients exhibited significantly impaired performance compared to HCs across four oculomotor tasks: fixation stability, SP, PS, and AS. Notably, AS average velocity, SP number of deviations, and PS average velocity could help differentiate MSA from HC. Correlation analyses revealed significant associations between oculomotor parameters and clinical measures. MRI-based volumetric analyses further revealed significant correlations between oculomotor parameters and regional brain atrophy in MSA patients. Specifically, PS average velocity correlated with vermis atrophy, while AS average velocity showed associations with bilateral cerebellar hemisphere volumes. Additionally, SP number of deviations was linked to atrophy in both cerebellar and frontal gyrus regions. These findings suggest that quantitative assessments of eye movement may serve as potential biomarkers for MSA diagnosis and progression monitoring.
Eye-tracking technology has emerged as a promising tool for neurological assessments, enabling precise quantification of eye movements.11,30 Our study found that MSA patients had significant impaired SP and gaze-holding function, consistent with prior research. 31 Specifically, MSA patients exhibited decreased SP gain, increased gaze-evoked nystagmus and frequent square-wave jerks during gaze-holding test. 32 These deficits are thought to be cerebellum-related ocular signs, caused by lesions in the cerebellar flocculus and paraflocculus. Additionally, MSA patients displayed prolonged PS latencies compared to HCs. Saccadic movement impairment is common in MSA patients. 12 Prolonged reflexive saccade latency may result from lesions in brainstem burst neurons and their connections to the superior colliculus, delaying saccade initiationn.33,34 Although slowed saccades were once considered exclusionary for MSA, 35 recent studies confirm prolonged saccades and reduced saccadic velocity in MSA compared to HCs. 36 The cerebellum also plays a role in the function of the cortical-superior colliculus circuit by connecting with brainstem saccade generators to modulate the amplitude and peak velocity of saccades. 37
The AS task is a well-established paradigm for assessing frontostriatal function by measuring the ability to suppress reflexive saccades toward a sudden peripheral target and generate voluntary saccade in the opposite direction. 38 This task evaluates both inhibitory control and voluntary saccade generation. 39 While AS impairments, such as increased error rates and prolonged latencies, are well-documented in Parkinson's disease (PD) and Alzheimer's disease (AD),20,40,41 AS performance in MSA remains understudied. Our findings revealed that MSA patients exhibited decreased AS accuracy and error correction rate, longer reaction times and error correction times than HCs, indicating impairment of the frontalstriatal network, a feature shared across these neurodegenerative disorders. Compared to PD, MSA patients may display more severe AS dysfunction, likely due to combined degeneration of fronto-basal ganglia and cerebellar-brainstem pathways, resulting in pronounced deficits in inhibitory control and voluntary saccade generation. 38
Given the pronounced oculomotor deficits in MSA patients, we further evaluated their potential as diagnostic biomarkers. Reliable biomarkers are important for MSA diagnosis, as it presents complex and variable symptoms. 42 Compared to clinical scales, quantitative oculomotor parameters provide sensitive and reliable measures.43,44 Our study revealed that AS and PS average velocity, SP number of deviations effectively differentiated MSA patients from HCs. Oculomotor abnormalities have been reported in idiopathic rapid eye movement sleep behavior disorder (iRBD),45,46 a prodromal stage of α-synucleinopathies, suggesting their potential as early neurodegenerative markers. However, oculomotor parameters showed limited ability to differentiate MSA-P from MSA-C subtypes, aligning with previous studies that reported no significant differences across multiple oculomotor parameters.47–49 This may reflect a shared underlying pathophysiology, as postmortem studies have demonstrated that olivopontocerebellar neurons are affected in both subtypes even at early stages, establishing cerebellar dysfunction as a common pathological substrate.50,51 Although one study reported differences, 12 we speculate that this discrepancy may stem from variations in patient cohorts or experimental conditions.
The relationship between eye movements and clinical scales in MSA has been scarcely reported. Our study found that SP, PS and AS ocular parameters were strongly correlated with clinical scores. These findings suggest that progressive alterations in oculomotor neurocircuitries are linked to MSA pathogenesis and neurodegeneration. 52 Beyond motor burden, 53 eye movement alterations also reflect cognitive impairments. Stuart et al. demonstrated that saccadic characteristics help identify patients who might be at risk for rapid cognitive decline. 54 Our study identified associations between PS and AS performance and cognitive assessments, indicating that saccade performance profiles may reflect cognitive function in MSA patients. As potential treatments to slow MSA progression are developed, 55 our findings suggest that portable VR-based quantitative ocular measures could serve as valuable tools to assess disease progression and treatment effects in future trials.
Neuroimaging analysis revealed significant GM loss in the cerebellum and frontal lobe in MSA patients compared to HCs, consistent with prior studies.56,57 However, previous neuroimaging studies only reported correlations between SP and cerebellar/ pontine volumes.21,22 Here, we expanded these findings by revealing associations between multiple oculomotor parameters and regional atrophy. Specifically, SP performance correlated with atrophy in both cerebellum and frontal cortex, indicating that SP impairment in MSA involves both motor control and higher-order cognitive regulation. 58 Reduced PS average velocity was linked to atrophy in the cerebellar vermis, a region that receives input from the nucleus reticularis tegmenti pontis and modulates brainstem burst neurons via the fastigial oculomotor region, thereby disrupting saccadic regulation. 59 This is distinct from PD, where oculomotor deficits primarily reflect dysfunction within the basal ganglia-thalamocortical circuits. 20 Similarly, bilateral cerebellar hemisphere volumes positively correlated with AS velocity, aligning with evidence that cerebellar hemispheres support higher-order oculomotor control.60–62 Overall, these findings highlight the importance of the “cerebello-prefrontal circuit” in MSA-related oculomotor deficits and reflect underlying pathological changes. Thus, oculomotor parameters may serve as potential biomarkers for detecting circuit dysfunction and monitoring the efficacy of interventions in future clinical trials.
The VR-based eye-tracking assessment offers several translational and clinical values. First, it enabled a novel finding: markedly reduced PS and AS average velocities, combined with increased SP number of deviations in MSA. These parameters demonstrated diagnostic value and were significantly associated with clinical severity scores. Unlike traditional eye-tracking systems, the VR headset allows for the presentation of visual motion stimuli in a head-unrestrained, immersive environment, which may reveal oculomotor impairments through more naturalistic saccades and enhance the ecological validity of these parameters as potential biomarkers.18,63 Second, this approach provides an objective, portable, and user-friendly alternative, enabling efficient testing in clinical or outpatient settings with minimal equipment. This enhances accessibility for patients with mobility issues or in resource-limited environments and holds promise for future remote telemedicine applications, as studies have shown that eye-tracking VR interfaces can emulate physician tasks for remote diagnosis of neurodegenerative diseases. 64 Finally, our findings reveal specific brain-behavior associations, demonstrating that oculomotor parameters are closely linked to atrophy in distinct brain regions, particularly the cerebellum. To our knowledge, this is the first study to use VR eye-tracking to explore MSA oculomotor dysfunction and its relationships with regional brain volumes, positioning these parameters as novel, non-invasive proxies for assessing the functional integrity of specific neural circuits and facilitating rapid evaluation for early diagnosis and progression monitoring in MSA.
The current study has several limitations. First, the cross-sectional design limits conclusions about the progression of oculomotor dysfunction and gray matter atrophy in MSA over time. Future longitudinal studies are needed to track oculomotor and imaging changes in MSA cohorts. Second, our control group did not include patients with other neurodegenerative disorders such as PD and progressive supranuclear palsy (PSP), which limits the validation of the diagnostic specificity of the oculomotor parameters in differentiating MSA from other neurodegenerative conditions. Future studies involving additional disease control groups are needed to further explore the differential diagnostic value of these oculomotor parameters.
In conclusion, our study provides comprehensive insights into the VR-based quantitative oculomotor analysis and its association with regional brain atrophy in MSA. Notably, parameters such as PS and AS average velocities, and SP number of deviations, hold promise as potential biomarkers for MSA diagnosis and disease severity assessment.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251412237 - Supplemental material for VR-based quantitative oculomotor analysis and association with regional brain atrophy in MSA
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251412237 for VR-based quantitative oculomotor analysis and association with regional brain atrophy in MSA by You Fu, Linlin Wan, Zhao Chen, Chunrong Wang, Daji Chen, Riwei Ouyang, Xiaokang Wu, Xiafei Long, Kefang Du, Xinying Xiao, Ruqing He, Linliu Peng, Rong Qiu, Beisha Tang and Hong Jiang in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We thank all the participants for their involvement in this study.
Authors’ roles
YF and HJ: Design; YF, LLW, ZC, CRW, DJC, RWO, XKW, XFL, KFD, XYX and RQH: Execution and Analysis; YF: Writing of the first draft; LLW, LLP, RQ, BST and HJ: Editing of final version of the manuscript.
Financial disclosure/funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key R&D Program of China (No. 2021YFA0805200 to H Jiang), the National Natural Science Foundation of China (U25A2058, 82571433, 82171254 to H Jiang; 82571632, 82371272 to Z Chen; 82301628 to L Wan; 82401496 to L Peng), the Natural Science Foundation of Hunan Province (No. 2024JJ3050 to H Jiang; 2025JJ20078 to Z Chen; 2024JJ6638 to L Wan), the Major Scientific Research Project for High-level Health Talent in Hunan Province (No. R2023047 to H Jiang), Furong Lab Research Project (No. 2023SK2084 to H Jiang), MIIT Project for Open Competition and Leadership in AI Medical Device Innovation (No. 2025-AISJK-01009 to H Jiang), the Central South University Research Programme of Advanced Interdisciplinary Study (No. 2023QYJC010 to H Jiang), Postdoctoral Fellowship Program of CPSF (GZB20230870 to L Peng) and China Postdoctoral Science Foundation (2024M753690 to L Peng).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
