Abstract
Planned discontinuation or acute unplanned cessation of oral dopaminergic medications might result in a severe relapse of Parkinson's symptoms or, sporadically, in life-limiting withdrawal syndromes. Unplanned cessation may occur due to dysphagia or decreased alertness, amongst other reasons. Planned acute discontinuation occurs during surgery or a medical necessity for a ‘nil per os’ policy (such as hospitalizations for gastrointestinal diseases). Non-oral alternatives are available, such as transdermal rotigotine, subcutaneous apomorphine, and levodopa delivered subcutaneously or via an enteral tube. Selecting the best treatment can be difficult and should be based upon clinical considerations, patient preference and be tailored to the care setting. These considerations will differ during the course of disease. For example, more invasive treatment options can be considered in hospitalized persons with early to moderate-stage disease, whereas symptomatic palliative treatments are more appropriate towards the end of life. Here, we discuss several practical considerations for three, partially overlapping, but conceptually distinct moments at which acute discontinuation or cessation events occur: during hospitalization (including surgery), late-stage disease and end of life. We stress the need for prevention and early advance care planning and present a stepwise pharmacological approach to address unplanned acute cessation or planned discontinuation.
Plain-language summary
People with Parkinson's disease can encounter situations in which they suddenly have to stop taking their oral antiparkinson medication. These situations can be divided into two broad categories. One is unplanned cessation of medication, such as in swallowing problems or in decreased consciousness. The other is planned acute discontinuation of medication for medical reasons, such as surgery or certain bowel diseases. If not handled correctly, sudden withdrawal of antiparkinson medication can lead to an increase in Parkinson's complaints or even in life-threatening illness. This article describes several ways to handle and anticipate planned and unplanned withdrawal of oral antiparkinson medication, and provides a practical guideline for healthcare professionals.
Introduction
Oral levodopa is the cornerstone of treatment for persons with Parkinson's disease (PwP).1,2 It is the most effective and safest dopaminergic treatment, both in early and late stages of Parkinson's disease (PD).2–4 As the disease progresses, higher and more frequent dosages of levodopa are required to combat response fluctuations.1,2 In advanced stages, persons may require levodopa as frequently as every 2–3 h, and the daily dose can be escalated above 2000 mg. 2 Other oral dopaminergic medications, such as dopamine receptor agonists (DRAs), catechol-O-methyltransferase inhibitors, amantadine and monoamine oxidase B (MAO-B) inhibitors, may be introduced to alleviate response fluctuations.1–3
Acute cessation of levodopa might result in a severe relapse of PD symptoms, especially for persons with higher daily doses.5,6 This includes a worsening of motor symptoms, such as akinesia, rigidity or tremor, and also of non-motor symptoms, such as anxiety and pain. In rare cases, acute cessation causes parkinsonism-hyperpyrexia syndrome.7,8 This potentially life-limiting syndrome is a specific form of neuroleptic malignant syndrome and consists of severe rigidity, autonomic dysfunction (e.g., hyperthermia, tachycardia), rhabdomyolysis and disturbances of consciousness. Therefore, preventing or treating acute levodopa cessation is an important aspect of good quality of care. It should be noted that acute dysfunction of deep brain stimulation can trigger parkinsonism-hyperpyrexia syndrome as well. 9
Acute withdrawal of DRAs carries a risk as well in the form of the dopamine agonist withdrawal syndrome. This entails neuropsychiatric symptoms and autonomic dysregulation and is more likely to occur in PwP who have higher daily DRA doses as well as a history of impulse control disorders. 10
Other dopaminergic medications generally carry a lower risk of complications from acute cessation. A notable exception is amantadine: while it can usually be safely stopped periprocedurally, acute withdrawal carries a risk of dystonia and is also associated with a greater chance of postprocedural dyskinesias and worse recovery. In rare cases, amantadine withdrawal can trigger parkinsonism-hyperpyrexia syndrome. 11 Caution is therefore advised.
In this review, we distinguish unintentional or unplanned cessation of levodopa on the one hand, and intentional acute discontinuation on the other hand. Unintentional gradual but also acute cessation may occur naturally during the disease course, for example as a result of dysphagia, 12 decreased alertness, 13 drug shortages or pharmacy/care provider error. Intentional acute discontinuation can occur during surgery under general anesthesia 14 or during medical necessity for a ‘nil per os’ policy (such as hospitalization for gastrointestinal diseases, see Box 1).15,16
Reasons for discontinuation or acute unplanned cessation of oral dopaminergic medication.
General anesthesia
Nil per os regime
Sedative medication
Medication has run out / medication shortage
Pharmacy or prescriber error
Decreased alertness/ cognitive decline
Delirium
Behavioral problems
Dysphagia
Nausea / vomiting
Severe gastroparesis / constipation
Recent gastrointestinal surgery
Ileus
Other comorbid gastro-intestinal disease
Specific clinical considerations with respect to cessation and discontinuation differ during the course of disease. For example, more invasive treatment options can be considered in hospitalized persons with early or moderate-stage disease, compared to end-of-life disease, where comfort care is more appropriate. 17 For the purpose of this review, we will discuss the typical clinical considerations for three, partially overlapping, but conceptually distinct moments when acute withdrawal events may occur: during hospitalization (including surgery), in late-stage disease and at end of life. For each of these moments, this review will give an up-to-date practice-oriented overview of clinical considerations for persons with acute unplanned cessation or discontinuation of oral dopaminergic medication. In addition, we propose a stepwise pharmacological strategy to address unplanned cessation or discontinuation of oral dopaminergic medications. Finally, we hope that this review helps raising further awareness for the risks associated with medication withdrawal in PwP, so that clinicians can deploy proactive strategies to avoid these from happening in the first place.
Intentional discontinuation of oral dopaminergic medication during hospitalization
Surgery
For patients undergoing surgery, physicians should ideally prevent discontinuation of dopaminergic medication. To achieve this, surgeons should ask four questions in the preoperative stage:
- Are there alternatives to surgery? - Are there alternatives to general anesthesia? - Is a nil per os regimen absolutely necessary? - In the case of (very) long surgery: can non-enteral dopaminergic medication be used?
Are there alternatives to surgery?
As a general rule of thumb in all patients, it should be considered whether the benefit-to-harm ratio favors surgery over conservative treatment. This ratio can be skewed towards harm in PwP,18–21 and more likely so in frail patients. Compared to matched controls, PwP undergoing general, visceral, thoracic, vascular, or trauma surgery have an increased duration of postoperative hospitalization, increased risk of perioperative morbidity (for example postoperative falls) and are less often discharged home.18,19 Furthermore, hospitalization of PwP can worsen parkinsonian symptoms (related at least in part to the stress that accompanies the admission), cause delirium and reduce the ability for self-care.19–21 If surgery is the best option, recommended preoperative screening for PwP includes consulting a specialized PD nurse or neurologist to advise on dopaminergic treatments. For specific individuals, a speech-language therapist can be asked to assess the risk of dysphagia. For those with suboptimal endurance and strength, rehabilitative therapy prior to surgery can be considered (‘prehabilitative’ therapy). 22 Also, for frail patients, involvement of geriatricians reduces perioperative functional decline and mortality.23,24 Involvement of an anesthesiologist who is aware of the particular needs of PwP is instrumental. For instance, certain drugs which are commonly used periprocedurally are contraindicated in PwP (examples include ketamine, metoclopramide and haloperidol). Other drugs should be used with caution. An example of this latter category is inhalation anesthetics, because of their risk of autonomic dysregulation. The decision to administer, and the selection of, opioids to PwP should be made after considering possible side effects and interactions. Opioids with serotonin reuptake properties (these include dextromethorphan, methadone, pethidine, propoxyphene and tramadol) have the potential to cause serotonin syndrome when combined with other serotonergic medications, which include MAO-B inhibitors used for PD such as selegiline, rasagiline and safinamide. Also, PwP who are administered opioids are at increased risk of postoperative delirium.25,26 In addition, particular opioids (fentanyl and alfentanil) may worsen rigidity.22,27
Are there alternatives to general anesthesia?
The use of local or regional anesthesia might prevent discontinuation of dopaminergic medication, compared to general anesthesia.22,28,29 In addition to the risk for discontinuation of dopaminergic medication, relative contraindications for general anesthesia are frequently present in PwP. For example, PwP have more severe dysautonomia (creating risk of perioperative hypotension), 30 and higher risk of aspiration pneumonia. 31 Airway management can be challenging because of factors such as cervical and laryngeal dystonia. Also, severe adverse events were reported following general anesthesia, such as a prolonged diminished consciousness and acute respiratory problems.29,32–34 Postprocedural confusion and delirium are very common in PwP and they result in a longer recuperation period before discharge is possible. Furthermore, benefits of local and regional anesthesia include the ability to detect post-operative complications earlier, such as nausea or pain.28,29 However, it should be noted that there are also other considerations which may favor general anesthesia. For example, for cataract surgery in PwP, the need for good positioning and the ability to lie still prompts most ophthalmologists to prefer general anesthesia. 35 Tremor could also interfere with monitoring devices during surgery. 29 Finally, many surgical procedures are simply not possible under local or regional anesthesia.
Is a nil per os regime absolutely necessary?
In the hours leading up to surgery, surgeons should consider if a strict nil per os regime is really necessary. In most cases, intake of dopaminergic medications with limited amounts of water (max 15 cc) is deemed safe.21,36 If nil per os is necessary, surgeons can strive to minimize the number of missed medications.21,37 For example, as most medication is administered during daytime, arrangements could be made to plan surgery at the earliest occasion in the morning, limiting the nil per os period mostly to the nighttime. Furthermore, the antiparkinson medication should be resumed as soon as possible after the surgery.
Can non-enteral dopaminergic medication be used?
In case of (very) long surgical procedures or otherwise protracted periods of discontinuation of oral medication, non-enteral dopaminergic medication can be considered. Plannable procedures such as elective surgery or scheduled hospitalization provide the PwP and their physician with the relative luxury of being able to plan ahead with regard to the antiparkinson medication. Non-enteral dopaminergic medication, such as subcutaneous levodopa or apomorphine, or transdermal rotigotine, can then be initiated and stabilized before the commencement of the nil per os period.
Unplanned hospitalizations
Most hospitalizations in PwP are acute and are caused by complications of PD, such as infections, psychiatric problems, falls and motor problems.38,39 Those who are elderly or who have progressed to advanced disease stages have a higher risk of being hospitalized, and also an increased risk of unplanned cessation of dopaminergic treatment. Causes for discontinuation of dopaminergic medications are nausea or a nil per os regime (for example, in persons with intestinal obstructions).15,16
Arrangements to prevent or cope with unplanned cessation of dopaminergic treatment should be in place prior to hospitalization. These include swallowing therapy to decrease dysphagia and early referral to the neurologist or Parkinson's disease nurse. In PwP who (e.g., because of motor fluctuations or therapy-resistant tremor) qualify for one of the various device-aided therapies, e.g., deep brain stimulation, levodopa/carbidopa with or without entacapone intestinal gel therapy, and subcutaneous apomorphine or levodopa pump therapy, should not suffer doctor's delay in their installment.22,40 The risk for unplanned cessation of dopaminergic medications is by itself not enough to initiate such advanced treatments. However, for those in whom these treatments are already in place, the risk of unplanned cessation of dopaminergic medications is obviously diminished as a non-oral treatment route is available. Neurologists or specialized PD nurses should therefore strive to reduce unnecessary delays in the introduction of these therapies. In advanced disease stages, which are often complicated by levodopa-resistant axial symptoms and cognitive impairment, introduction of device-aided therapies may no longer be possible. In addition, even if advanced treatments are in place, unplanned cessation of dopaminergic treatments may occur. For example, major gastrointestinal surgeries could impair intrajejunal levodopa/carbidopa gel therapy, or a delirium could warrant a marked reduction of the DRA dose.
Unintentional cessation of oral dopaminergic medication in late-stage Parkinson's disease
Late-stage disease is characterized by dependency upon others, diminished physical/cognitive ability, and frailty. 41 Several causes of unplanned cessation events are more prevalent in late-stage disease compared to earlier disease stages, such as dysphagia, delirium, hospitalization and comorbid diseases.42,43 In late-stage PD, cessation events might appear to be acute, but frequently they are not. For example, a gradual disease progression with escalating motor problems culminates in what appears to be a sudden dysphagia, 44 or an underrecognized gradual cognitive decline predisposes PwP to the development of an acute delirium. 45 These examples illustrate the need for timely recognition and initiation of treatment for motor and non-motor symptoms.
Any physician treating a person with late-stage PD with unplanned cessation of dopaminergic medication should ask three questions:
- What non-oral treatments are safe and effective? - What non-oral alternatives are feasible in the current care setting? - What are the person's wishes with regard to non-oral alternatives?
What non-oral treatments are safe and effective?
Effective non-oral treatments are available, but persons with late-stage disease likely have more intolerance. Rotigotine is a DRA that is available as a transdermal patch, with a beneficial effect on motor and non-motor symptoms. 46 While it has a good safety profile, it can sometimes induce psychotic symptoms or worsen autonomic symptoms, such as hypotension. 47 It cannot fully replace high doses of oral levodopa. The equivalency factor from levodopa to transdermal rotigotine is 30:1 and the maximum dose of rotigotine is 16 mg/24 h, corresponding to 480 mg levodopa. 48 Caution must be exercised, however, as a DRA-naïve person cannot be suddenly started on the maximum dose of 16 mg/24 h, as this could potentially be accompanied by a high risk of autonomic dysregulation and/or neuropsychiatric adverse effects. Therefore, the dose should be titrated up gradually. Immediate relief of PD symptoms can thus not be achieved. Another aspect that should be considered is that (especially in a palliative setting), the cutaneous absorption of medication can be adversely affected by factors such as sweating, cachexia and reduced dermal blood circulation. 49 This may limit the efficacy of a rotigotine patch.
Apomorphine is a potent DRA that is administered subcutaneously. It is administered either through intermittent (pen) injections or through continuous infusion with a removable infusion pump. 50 Apomorphine has a short half-life of 30 min and the clinical use for intermittent (pen) injections is therefore mostly limited to ‘rescue’ treatment of OFF episodes. For a response with longer duration, continuous subcutaneous infusion is required.
Sublingual and rectal formulations of apomorphine were developed, but are generally not available for clinical use.51,52 There is conflicting information on safety of apomorphine in late-stage disease. Apomorphine is generally safe, even for those with cognitive dysfunction, 53 but analysis of reasons for discontinuation of apomorphine treatment showed hallucinations, confusion and somnolence as most frequent. 54 The presence of neuropsychiatric symptoms predicts discontinuation of apomorphine treatment. 55 However, apomorphine can be continued if a patient is adequately treated for the neuropsychiatric symptoms.56,57 Given its efficacy, apomorphine can be considered as an alternative for patients who acutely need to stop high doses of oral levodopa. To prevent intolerance, pretreatment with an anti-emetic such as domperidone is needed. The levodopa equivalency factor from levodopa to apomorphine is 10:1. 48 However, given the ‘all-or-nothing’ pharmacodynamics of apomorphine, a conversion factor is seldom used in practice to obtain the apomorphine dose. Instead, the dose is titrated up from 1 mg/h until a satisfactory response is achieved. 58
Oral levodopa is the cornerstone of antiparkinsonian treatment, but the most frequent administration route of non-oral levodopa—using an enteral tube—is invasive. 59 Administering continuous levodopa/carbidopa intestinal gel through a percutaneous endoscopic gastrostomy with a jejunal extension is rarely feasible in acute unplanned situations as this requires careful planning and titration. 59 Pulverized and dissolved levodopa tablets can be administered through a nasogastric tube.
The introduction of a nasogastric (NG) tube for levodopa administration should be carefully weighed against the expected duration of the inability to swallow, the state and prognosis of the PwP and whether or not there is also an intention of using the tube for nutrition. In persons with dementia, enteral tube feeding did not reduce morbidity or mortality and did not improve quality of life. 60 Enteral tube placement in this patient category therefore comes with complex ethical considerations regarding the acceptability of life-sustaining treatments. 61 Starting tube feeding can extend life beyond what is potentially welcomed by some PwP.
In palliative-care settings, for example in advanced cancer, patients can experience gastroparesis. 62 Levodopa administration via an NG tube might thus be less effective, and the effect less predictable, in this population. Conversely, in situations in which swallowing is expected to be transiently impaired (such as following an acute stroke), using an NG tube for levodopa administration may very well be a good temporary solution.
If enteral tube placement is not preferred and DRAs are not tolerated, a theoretical alternative would be the use of rectal levodopa formulations. Pharmacokinetic studies into rectal suppository administration of levodopa are scarce and contradictory. Whilst one study reported comparable levodopa blood concentrations following either oral or rectal administration, a later study did not find rectally-administered levodopa/carbidopa to reach the circulation in measurable quantities 63 and an even later case report found a negligible serum concentration of levodopa after rectal levodopa/carbidopa administration. 64 A further case report, following through on an earlier suggestion that the lack of rectal absorption may be linked to the rectum's alkaline milieu, described a positive clinical response to a strongly acidic rectal suspension of levodopa/carbidopa (prepared by adding citric acid to a suspension of pulverized levodopa in water/glycerol). 65 Pharmacokinetic analyses were not performed, however. The other case report, conversely, used the same acidic formulation and found negligible absorption. 64 The uncertainty surrounding rectal levodopa's pharmacological properties and the cumbersomeness of having to repeatedly manually prepare a suspension and administer it by enema makes this an improbable candidate for non-oral levodopa administration.
Recently, inhaled and subcutaneous levodopa have become available for several countries. Outside of specific countries (such as Japan), 66 intravenous administration of levodopa is generally not available for clinical use and remains reserved for scientific research. The marked acidity of levodopa solutions means that they need to be heavily diluted for infusion, which makes its use impractical and carries a risk of hypervolemia as well as phlebitis. 67 In a retrospective observational study of PwP undergoing gastric or colorectal surgery, perioperative administration of intravenous levodopa reduced the length of hospital stay, compared to perioperative withholding of antiparkinson medication, but it did not reduce the rate of complications or mortality rate. 66
What non-oral alternatives are feasible in the current care setting?
Treatment options are frequently limited by the care setting. A majority of persons in late-stage disease are less mobile and frequently unable to attend outpatient clinics for expert treatment. 68 Nearly half of PwP with late-stage disease live in care facilities. 69 The formal and informal carers in nursing home settings are less equipped to utilize more invasive strategies, such as subcutaneous apomorphine and non-oral levodopa treatments.70,71 In our experience, knowledge and experience in handling these therapies is frequently absent. Physicians should take these limitations into consideration while choosing the treatment options and/or consider arranging training of healthcare providers.
Regardless of the care setting, initiation of advanced therapies generally requires planning. The pen or pump for the non-oral medication, as well as the medication cartridges, should be ordered; a specialist nurse should be available to perform the initiation (dose-finding and monitoring for adverse effects); and premedication for several days can be required (such as domperidone in case of initiation of apomorphine). This makes installment of these non-oral alternatives in a setting of acute oral medication cessation often impractical or impossible.
What are the PwP's wishes with regard to non-oral alternatives?
Although treatment optimization might benefit persons with late-stage disease, even with the best treatments, they typically experience a higher symptom burden and a lower quality of life compared to earlier stages.72–74 As a result, the goals of care shift and are less focused on restoring maximum health and more aimed towards optimizing comfort. In this stage, ascertaining the right goals of care also includes a careful assessment of the person's wishes for intervention to cope with the consequences of an unplanned cessation event. Clinicians also have the responsibility to provide information on non-oral alternatives and their implications for daily life, side effects and the possible need for extra help in case of a subcutaneous/intrajejunal therapy. Providing information and organizing care based on a patient's preferences is embedded in advance care planning. 75 The process of advance care planning enables individuals to define goals and preferences for future medical treatment and care, to discuss these goals and preferences with family and healthcare providers, and to record and review these preferences if appropriate. 76 Studies in other fields have shown that advance care planning can improve quality of life of patients, improve patient satisfaction with end-of-life care, and lead to better quality of communication between patient, family caregivers and healthcare professionals.77–81 In the process of advance care planning, specific attention should be paid to unplanned cessation of dopaminergic treatments. Physicians should discuss a person's treatment wishes for more invasive treatment options, such as enteral tube placement. Also, treatment options that require a different setting, such as hospitalization, should be discussed, as many persons wish to abstain from hospital admissions. These advance care planning discussions should preferably be held early in the disease course, as later on in the disease many PwP will experience cognitive decline or difficulty expressing themselves.
Unplanned cessation of oral dopaminergic medication at the end of life
At the end of life, acute cessation of oral medication is common. In this case, many drugs are often ceased permanently, with the remainder preferably being administered via subcutaneous infusion in most settings.
82
This can even be achieved with the help of family carers.
83
When it comes to antiparkinson therapy, however, full substitution of oral dopaminergic medication is not always a self-evident treatment strategy. Several experts argue that diminishing and tapering off the dopaminergic medication is, at least in some PwP, possible with limited negative consequences.64,84 Others suggest to continue the use of dopaminergic treatments for as long as until the moment of death, including more invasive treatments such as apomorphine.
85
Likely, at the end of life, care needs and wishes of PwP are not uniform.
86
In our opinion, at the end of life the treating physician should ask the following three questions:
- Which symptoms determine the level of comfort and quality of dying? - What are the most effective and feasible treatment options in the person's preferred place of death? - What are the person's wishes with regard to life-sustaining treatments and end-of-life care?
Which symptoms determine the level of comfort and quality of dying?
The major causes of death in PwP are infections (pneumonia, urinary tract infections with tissue invasion), falls or cardiac disease. As a result, symptomatic control includes the treatment of traumatic injuries or any respiratory and cardiac distress. 87 The most frequent end-of-life symptoms in PwP are pain, respiratory secretions and agitation. 88 In the 72 h before death, severe motor symptoms were present in only 6% of persons in one study. 88 Whilst opioids and benzodiazepines are best avoided in earlier disease stages, these treatments are the mainstay of treatment at the end of life.82,89 If a person has sufficient symptom control with palliative treatments, including palliative sedation, one can opt to omit dopaminergic treatment altogether – although sudden cessation carries a risk of parkinsonism-hyperpyrexia syndrome.
What are the most effective and feasible treatment options in the person's preferred place of death?
Few available studies report on the effectiveness and feasibility of treatments at the end of life. Based on these studies, transdermal rotigotine is the most widely used non-oral strategy at the end of life. 90 In a UK-based cohort of 51 PwP at the end-of-life, 55% had dopaminergic medication substituted with transdermal rotigotine at death. 88 However, evidence regarding its effectiveness is scarce, with case studies showing conflicting results of rotigotine, two reporting improvement and two others reporting insufficient effect.91–94 In addition, great caution is advised in prescribing DRAs to patients who are at high risk of hallucinations, confusion or autonomic dysregulation. In a study in which people with advanced PD were started on a rotigotine patch to replace their oral dopaminergic agents, as many as 24% of subjects developed a delirium. 95
End-of-life use of subcutaneous apomorphine and subcutaneous scopolamine (for tremor) were successful treatments in the persons in whom rotigotine was insufficient. However, improvement with apomorphine and scopolamine came with adverse events, such as nausea, vomiting and dry mouth. All these cases were in a hospital or hospice setting, which is not the preferred place of death for most persons, as most wish to die at home. 96 At home, the general practitioner is the first physician to treat troublesome symptoms. 97 In our experience, general practitioners have sufficient experience with palliative symptomatic treatments, but struggle with the non-oral dopaminergic options in the period shortly before the end-of-life phase. Therefore, collaboration between general practitioner, (specialized) pharmacist and movement disorders expert is necessary.
What are the person's wishes with regard to life-sustaining treatments and end-of-life care?
With the current lack of evidence, we think it even more important to orient treatment choice towards the wishes of the PwP. It is an interesting finding that utilization of palliative care services, including a discussion on the goals of care, decreases the number of invasive therapies. 70 At the end of life, persons may prioritize comfort over survival, and the best treatment is the one that is most successful at achieving this goal, with the lowest treatment burden. In settings with limited availability of non-oral dopaminergic treatments, symptomatic palliative treatments—such as analgesics and benzodiazepines—are more feasible. However, for those PwP with a need and wish for more invasive non-oral dopaminergic strategies, options should be in place.
Stepwise pharmacological strategies for unplanned cessation or medically-indicated discontinuation of dopaminergic medication
If a PwP cannot take oral dopaminergic medication, oral levodopa and oral dopamine agonists should be substituted with alternatives. It is generally considered safe to temporarily omit rasagiline, selegiline, entacapone and tolcapone, 27 and amantadine with caution. Many non-oral dopaminergic treatments were developed, but not all are widely available. See Table 1 for considerations on all alternatives on safety, efficacy, complexity, requirements and dosing. Table 2 contains alternatives that are also mentioned in the literature but that are, for various reasons, not readily applicable. For the assignment of the qualification ‘readily applicable’ or ‘not readily applicable’ to the various options, several aspects were considered, including commercial availability, efficacy, safety and available evidence. It should be noted that ‘readily applicable’ does not necessarily imply a lack of preconditions or complexity: e.g., for device-aided therapies, specialist nursing care may be necessary in addition to specific equipment, as well as premedication and (in some jurisdictions) prior approval by healthcare insurance. Table 2 is provided to give an overview of various described treatment suggestions which the authors cannot currently recommend. Rather than an endorsement of these treatment options, this table is intended as a practical reference for the reader to make an informed decision on the use or non-use of the alternatives provided, for cases in which the alternatives in Table 1 are not available, contraindicated or have already been tried unsuccessfully.
Readily applicable methods for treating acute discontinuation or unplanned cessation of oral dopaminergic medication (in alphabetical order).
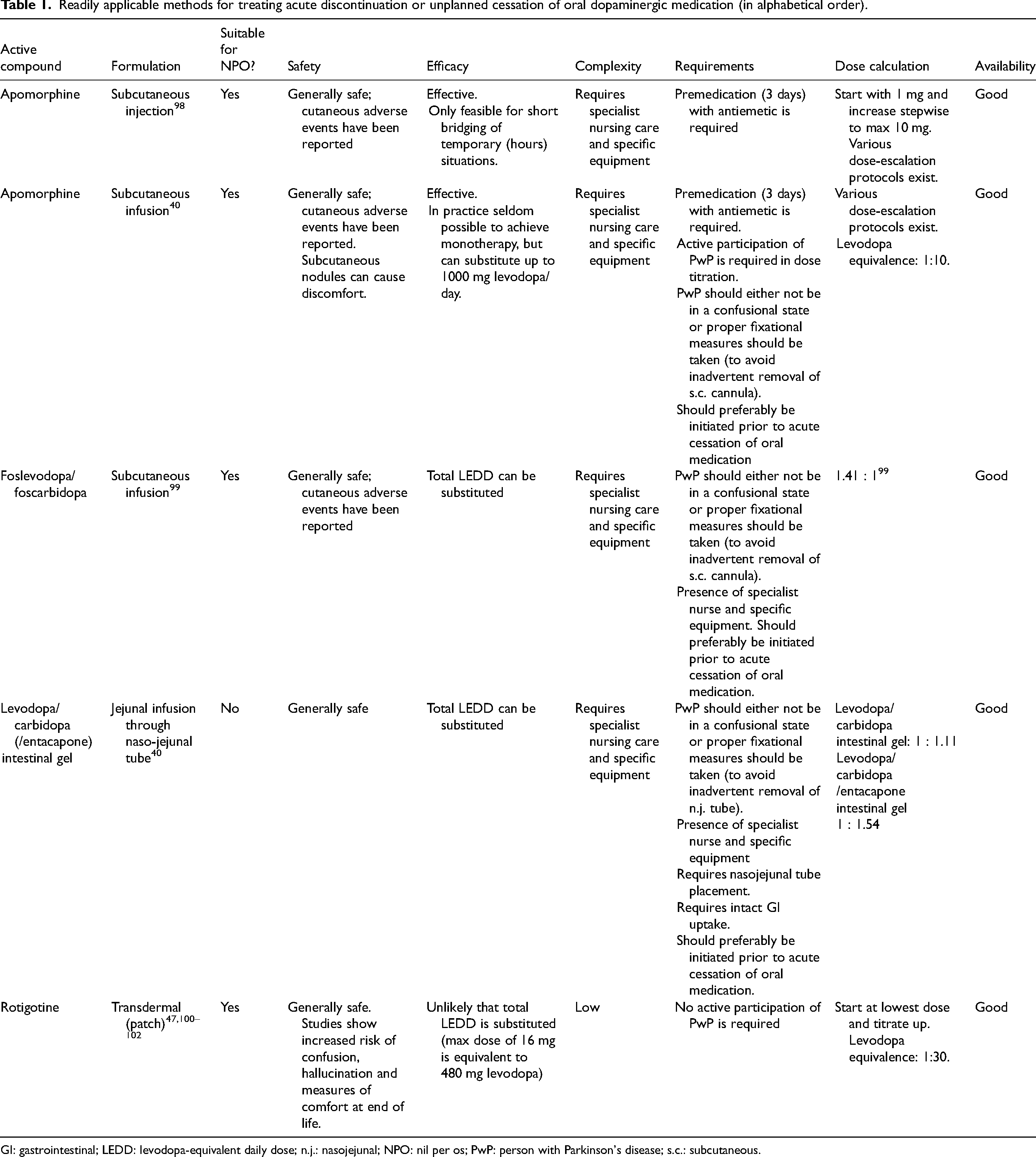
GI: gastrointestinal; LEDD: levodopa-equivalent daily dose; n.j.: nasojejunal; NPO: nil per os; PwP: person with Parkinson's disease; s.c.: subcutaneous.
Other reported, but not readily applicable, methods (in alphabetical order).
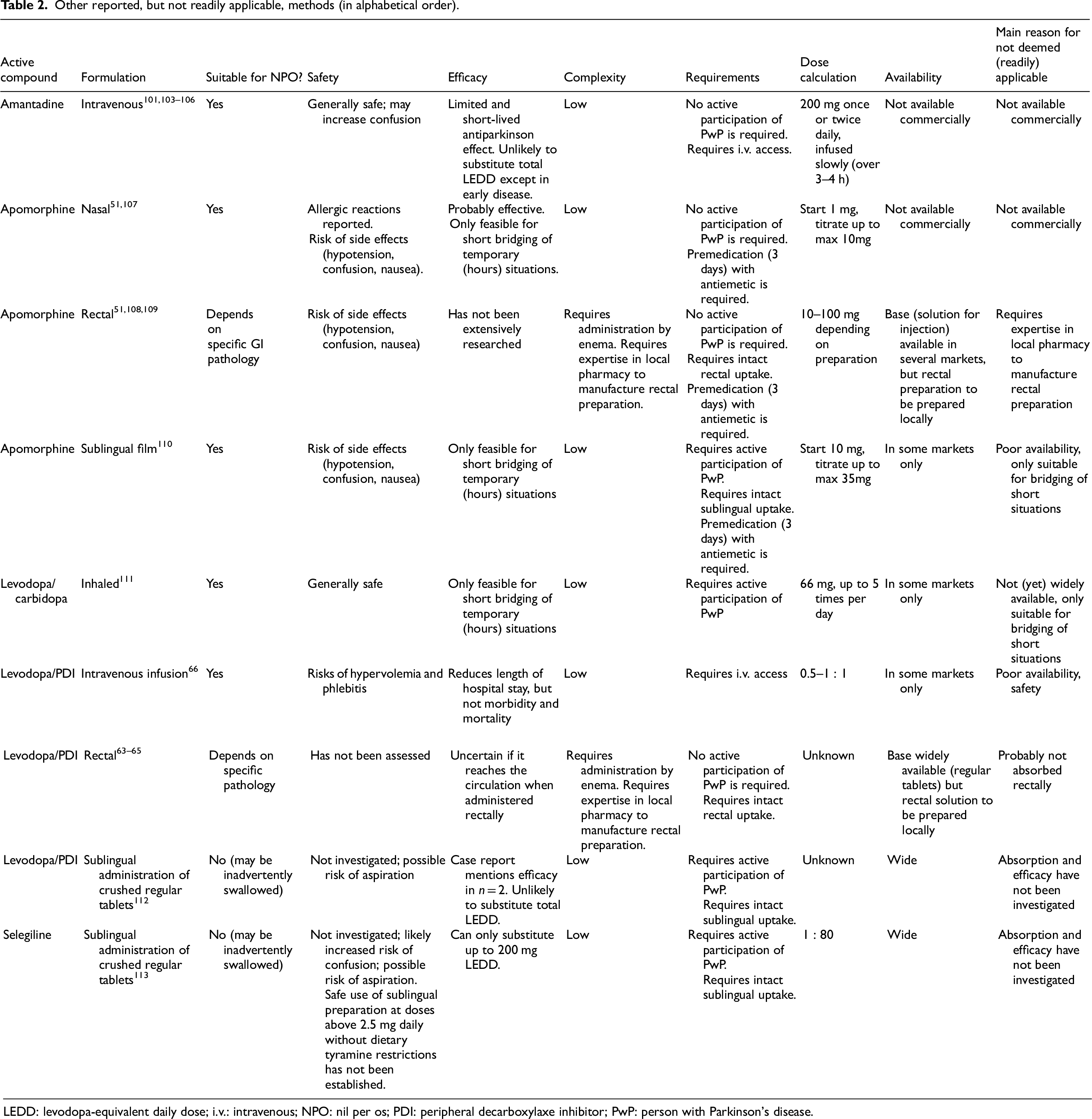
LEDD: levodopa-equivalent daily dose; i.v.: intravenous; NPO: nil per os; PDI: peripheral decarboxylaxe inhibitor; PwP: person with Parkinson's disease.
Below, we will discuss a stepwise protocol consisting of the widely available treatment options (Figure 1).
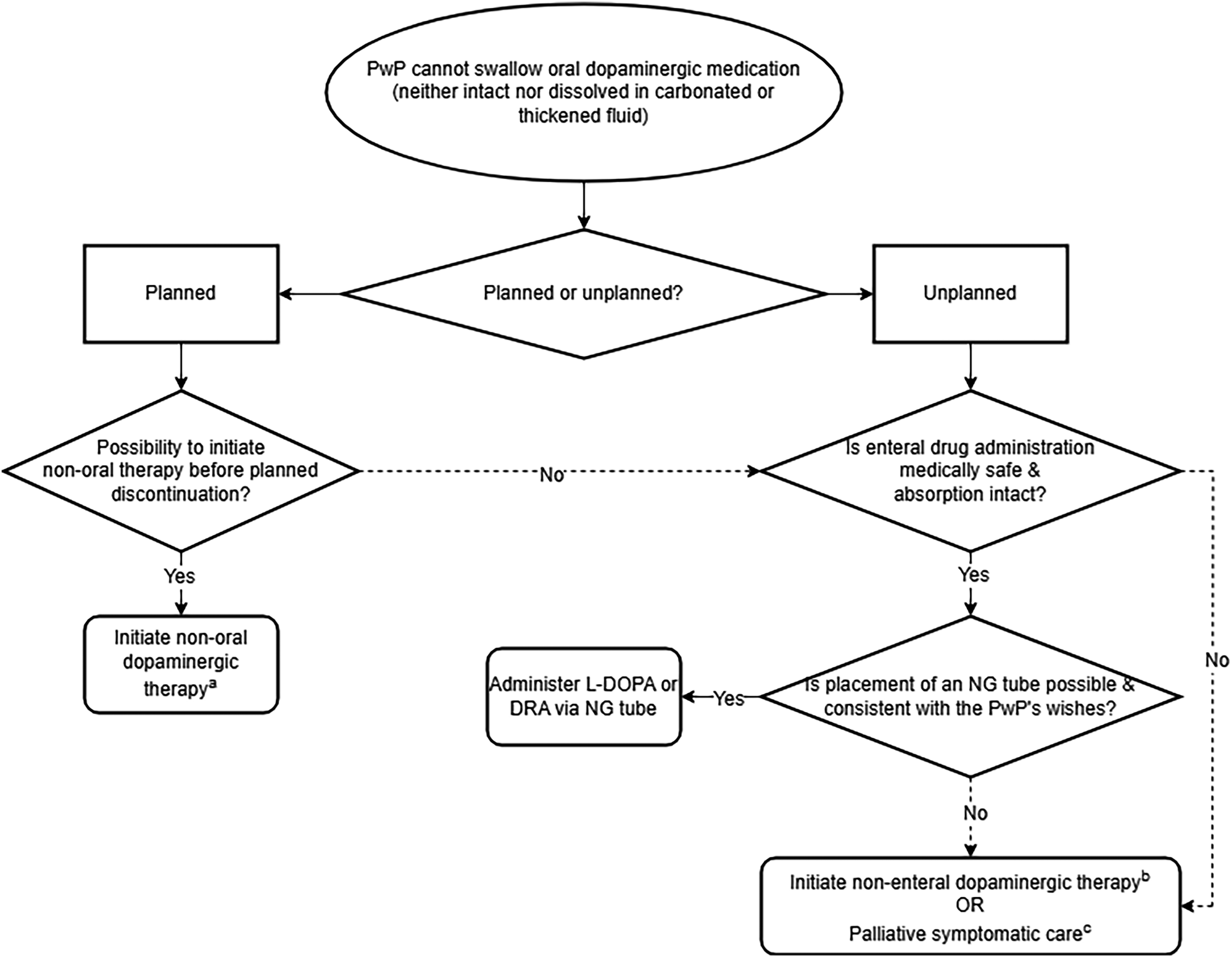
Stepwise pharmacological strategy for discontinuation/cessation of dopaminergic medication.
In PwP who have mild dysphagia (i.e., swallowing is still safe and possible, but the person experiences difficulty swallowing certain types of food or tablets/capsules), it should be ascertained whether, as step 2, the PwP can swallow the medication when taken with carbonated water, as this has been shown to be easier to swallow in people with swallowing dysfunction due to diffuse Lewy-body disease. 114 If not, it should be assessed whether they can swallow thickened fluids. 25 Levodopa and immediate-release dopamine agonists can be dispersed or pulverized and dissolved in (carbonated or thickened) fluids or in soft foods such as applesauce or pudding. To enhance the stability of the suspension 115 and to improve gastrointestinal absorption of levodopa,116,117 the solution can be made slightly acidic, for example by adding ascorbid acid or lemon juice. This might not be necessary in case of carbonated water, which is acidic in itself. Controlled-release dopamine agonists can be switched to immediate-release preparations (taking into account the different bioavailabilities) although this may come with an increase in adverse events. It is worthwhile to proactively consult a PD nurse, speech-language therapist or pharmacist for individualized advice.
If medication truly cannot be taken by mouth, the following steps can be taken:
As step 1, it should be ascertained whether the medication withdrawal is planned or unplanned. In planned situations, it should be evaluated beforehand whether initiation of non-oral dopaminergic therapy (subcutaneous/intrajejunal levodopa, subcutaneous apomorphine, transdermal rotigotine, deep brain stimulation) before the planned event is possible.
As step 2, the physician should evaluate whether enteral drug administration is safe and there is no suspicion of major gastrointestinal malabsorption. If unsafe or marred by malabsorption, the enteral route should be bypassed (step 3). If the PwP cannot swallow a carbonated or thickened levodopa suspension, the physician should ascertain whether placement of a nasogastric tube is possible and consistent with the PwP's wishes.22,25 If so, similar to the dissolution of dispersed or pulverized medication in thickened fluid, levodopa and immediate-release dopamine agonists can be dissolved in water for administration through the tube. As many PwP have gastroparesis, the tip of the tube should ideally be placed in the jejunum. 40 However, a gastroenterologist or radiologist is needed for such tube placement. In most cases, regular tube placing can be tried first and jejunal placing considered if the effect is dissatisfactory.
As step 3, if no gastrointestinal route is available, several other treatments are possible.22,25 First, a rotigotine transdermal patch can (partially) substitute dopaminergic treatment.14,47,100 This has several caveats, as described above. The equivalency factor from levodopa is 30:1 48 and rotigotine substitutes dopaminergic medications up to a maximum daily dose of 480 mg, although gradual dose titration is necessary. Often, this is not high enough, but it does allow for the prevention of a total cessation of dopaminergic medication.
Alternatively, subcutaneous apomorphine can be considered.40,118 Apomorphine allows for a levodopa-equivalent daily dose higher than 480 mg. It can be used either with short-acting single injections or via continuous pump therapy, depending on the duration of the cessation of oral therapy. However, for the best result, prior information on the effective and safe individual dose of apomorphine is needed, as the response to apomorphine can best be described as ‘all or nothing’ and does not simply correspond to response to the converted levodopa dose. This is only available for those that underwent a prior apomorphine challenge. Furthermore, the PwP should be pretreated with an anti-emetic such as domperidone for several days before apomorphine can be initiated, as apomorphine is a powerful emetic. This limits the use of apomorphine in the acute setting without pretreatment. Also, apomorphine is not available in all countries.
A third option is the use of subcutaneous levodopa.99,118,119 The choice between these three strategies depend on the person's wishes, individual daily dose needed, expected tolerance to side effects and availability of products.
If no dopaminergic alternative is feasible or wanted, palliative symptomatic treatment is the final step. Palliative symptomatic treatments aim to improve quality of life by treating the symptoms irrespective of the underlying causes of the symptoms. 120 For persons experiencing an unplanned cessation of oral dopaminergic medication, symptoms that may occur are severe rigidity, tremor, pain, anxiety, confusion and, in rare cases, parkinsonism-hyperpyrexia syndrome. The palliative symptomatic treatment of severe rigidity, tremor, and pain in PwP does not differ from symptomatic treatment in other persons. Non-oral analgesics include paracetamol suppositories and opioids administered transdermally, subcutaneously or intravenously. For anxiety and confusion, lorazepam or midazolam can be administered subcutaneously. If symptoms are refractory, palliative sedation can be employed. Depending on the symptom severity and the wishes of PwP and their near ones, this can be administered either intermittently or continuously, and with the intent of either superficial or deep sedation.
Conclusions
This review summarized the challenges and considerations with respect to non-oral treatment following (acute) planned discontinuation and unplanned cessation of oral dopaminergic treatment. We hope that the practical sequential steps outlined in the flowchart can assist clinicians in making an optimal, person-centered decision, tailored to the specific medical context at hand.
Footnotes
Acknowledgements
The authors would like to thank the peer reviewers for their thorough review of the paper and well-grounded comments, which have strongly contributed to the improvement of this paper.
The Radboudumc Centre of Expertise for Parkinson & Movement Disorders was supported by a center of excellence grant by the Parkinson's Foundation. MB and BB are supported by research grants from ParkinsonNL (project P2021-18), the Dutch Digestive Foundation (project WOO 21-05), Stichting Woelse Waard and Stichting Alkemade-Keuls. MB is a recipient of the Edmond J. Safra Fellowship in Movement Disorders.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Bas Bloem is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
