Abstract
Background:
It is unclear to which degree Levodopa (L-dopa) remains effective also in the late stage of Parkinson’s disease (PD) and to which degree motor fluctuations and dyskinesias remain a problem.
Objective:
To assess responsiveness of motor symptomatology to L-dopa in a group of patients with late stage PD. Moreover, to investigate the extent to which motor fluctuations and dyskinesias occur.
Methods:
Thirty PD patients in Hoehn and Yahr (HY) stages IV and V in “on” were included. L-dopa responsiveness was assessed with a standardized L-dopa test in the defined “off” and defined “on” states. Motor function was assessed by the Unified PD Rating Scale (UPDRS) III and timed tests. Motor fluctuations and dyskinesias were assessed by the UPDRS IV. The participants were further monitored for 10 days with a mobile movement-analyses-system, the Parkinson’s Kinetigraph (PKG). The median (q1–q3) L-dopa equivalent daily dose (LEDD) was 799 (536–973) mg.
Results:
The UPDRS III score improved with ≥15% in 15 (50%) and with ≥30% in six (20%) participants during the L-dopa test. The median (q1–q3) UPDRS III score in “off” was 46 (37–53) and in “on” 36 (28–46). Twenty-one (70%) of the participants reported either predictable or unpredictable “off” fluctuations (items 36–37). The prevalence of dyskinesias (item 32, duration of dyskinesias ≥1) was 47%. The PKG indicated that dyskinesias primarily were mild and that a majority had a pronounced “off” symptomatology, spending a large proportion of the day either asleep or very inactive.
Conclusions:
Half of a group of patients with late stage PD had an L-dopa response of ≥15% on the UPDRS III. According to the UPDRS IV, a majority of the patients had motor fluctuations and about half had dyskinesias, although the PKG results suggested that these were not very severe.
Keywords
INTRODUCTION
Levodopa (L-dopa) is the most effective symptomatic treatment for the progressive dopamine deficiency in Parkinson’s disease (PD). However, it is well recognized that a majority of PD patients develop L-dopa-induced motor complications (i.e., motor fluctuations and dyskinesias), in many cases already within a few years of treatment with L-dopa [1, 2].
In the late and most severe stages of PD, i.e., Hoehn and Yahr (HY) [3] stages IV and V, patients become increasingly immobile, experience frequent falls and become increasingly dependent in activities of daily living (ADL) [4, 5]. The time from disease onset to these late stages is approximately 14 years [3] and a person lives on average about 5 years in this phase of the disease [4, 6]. In HY stage IV the patient is able to walk and stand independently, though in need of help with ADL-activities, while in stage V the patient is wheelchair-bound or confined to bed unless aided by another person [3]. The prevalence of this late stage group is expected to increase due to increased life expectancy and improved PD specific as well as general health care [6, 7].
The benefit of L-dopa medication may decrease in the late stages of the disease [8]. However, L-dopa responsiveness in patients in late stage PD is not well understood and has not been systematically analyzed [8]. Moreover, it has been suggested that patients in late stage PD are an undertreated group [8–10], where many of the patients no longer receive specialist care and are often excluded in research studies, therefore little is known about their clinical picture [11].
An L-dopa test provides immediate information on L-dopa responsiveness of motor symptoms and also predicts the response to chronic treatment [12]. However, the L-dopa test is carried out at one time point using a single dose and can yield false negative results [13]. A long-term assessment of motor function can be obtained by monitoring with the Parkinson’s Kinetigraph (PKG), which also gives quantitative information about the occurrence of motor fluctuations and dyskinesias.
The aim of this study was to assess responsiveness of motor symptomatology to L-dopa in a group of patients with late stage PD. Moreover, to investigate the extent to which motor fluctuations and dyskinesias occur.
MATERIAL AND METHODS
Participants and recruitment
Participants were recruited from the region of southern Sweden through neurology departments and the community based health care system. Inclusion criteria were: HY stages IV and V in “on” and/or having a substantial need of help with ADL (≤50% on the Schwab and England Scale) [14], as well as having been diagnosed with PD for a minimum of seven years. All participants had been treated by a neurologist or geriatrician (two cases) and all had the diagnosis idiopathic PD. Exclusion criteria were: cognitive symptoms that started before the PD diagnosis, a score of <15 on the Mini Mental State Examination (MMSE, score range 0–30, higher = better) [15]. Because bradykinesia prevented some (seven) participants from completing all items, the MMSE result was alternatively expressed as a percentage of the maximum score of the completed items. In a full MMSE a score of ≥24 (≥80%) is regarded as normal [16].
Thirty-three consecutive patients from the baseline assessment of the Swedish part of the European multicenter study Care for Late Stage Parkinsonism (CLaSP) [17], fulfilled the inclusion criteria and were asked to participate in the L-dopa test and to wear the PKG logger. One patient declined participation and two were excluded because the PKG monitoring was not completed.
The study was approved by the Regional Ethical Review Board in Lund, Sweden (JPND HC-559-002). Written informed consent was obtained by the participants.
Procedure
Two health care professionals (PD nurse, physiotherapist) with long experience in the field carried out the L-dopa test in the homes of the participants. In most cases, a family member of the participant was present. A first baseline assessment in practically defined “on” under the patients’ normal medication was made about a week prior to the L-dopa test.
Clinical evaluation
The study protocol consisted of two major parts: (i) an L-dopa test for an immediate assessment of motor function and (ii) the PKG registration for continuous long-term assessment of motor function. In addition, motor complications were assessed clinically.
Immediate assessment of motor function (L-dopa test)
L-dopa responsiveness was assessed by an L-dopa test, with a standardized test protocol according to CAPSIT-PD [18, 19]. Participants were first assessed in the morning in the defined “off” condition; at least 12 hours after having taken the previous dose of dopaminergic medication [18, 19] and before their morning L-dopa medication, in a fasting condition. They were then given a standardized dose of L-dopa (200 mg tablet Madopark Quick dissolved in a glass of water) and assessed again in the defined “on” condition [18, 19]. This occurred when the patient and the investigator agreed that the best therapeutic L-dopa effect had been reached, typically after approximately 60 minutes. Each patient was observed clinically for about three hours. Three patients with known severe orthostatic hypotension received domperidone (10 mg three times per day, starting 12 hours before the test) prior to testing.
Motor function was evaluated [18] by the following assessments: The motor section (part III) of the Unified PD Rating Scale (UPDRS; UPDRS III); score range 0–108 points, higher = worse [20]. Bradykinesia was assessed by the sum of UPDRS III items: 23 (finger taps), 24 (hand movements), 25 (rapid alternating movements of hands), 26 (foot agility) and 31 (body bradykinesia and hypokinesia). Axial signs were defined as the sum of UPDRS III items: 18 (speech), 19 (facial expression), 22 (neck rigidity only), 27 (arising from a chair), 28 (posture), 29 (gait) and 30 (postural stability) [21]. Gait test of 7 meters×2, including turning. The patients were instructed to walk as fast as possible. Time, number of steps and number of freezing episodes were noted. Hand-arm movements between two points, 30 cm apart during 20 seconds, were conducted twice with each hand and the mean number of movement cycles for each hand was counted. Moving the hand from one point to the other and back was counted as one movement cycle. In addition, orthostatic blood pressure was measured (supine and one and three minutes after standing up). Some of the patients were unable to perform all aspects of the orthostatic blood pressure measurement, in those cases a modified testing was performed; from supine to sitting (eight cases) or from sitting to standing (four cases).
Twenty-four (80%) of the patients performed gait testing. One patient was unable to walk in “off”, another could perform the full distance in “off” but only half of it in “on”. In other cases (four patients in “off”, five in “on”), due to either space restrictions in the participant’s home or inability to walk the full distance, the distance was modified and gait speed (m/s) was calculated based on the completed distance.
Thresholds for L-dopa effect
L-dopa responsiveness was defined as an improvement in the UPDRS III score of at least 15% [12]. According to CAPSIT-PD [18], the L-dopa test should induce at least a 33% decrease in the UPDRS III score, as a criterion for surgical PD therapy. A clinically meaningful effect, i.e., the clinically important difference (CID) in the UPDRS III score has been estimated as 2.5 points for minimal, 5.2 points for moderate and 10.8 for large meaningful change [22].
For the timed tests of gait and hand-arm movements, there are no defined thresholds in CAPSIT-PD. However, in other clinical investigations a 20% threshold has been suggested for gait testing [23], while a 15% improvement of hand-arm movements has been considered an adequate threshold for detecting dopaminergic responsiveness [24].
Medication
A full medication list was obtained from each participant and the L-dopa equivalent daily dose (LEDD) was calculated according to a standardized formula [25].
Clinical assessments of motor fluctuations and dyskinesias
Motor fluctuations and dyskinesias were assessed with the UPDRS part IV (complications of therapy, score range 0–23, higher = worse) [20]. Fluctuations were assessed by items 36–39, predictable “off” fluctuations were assessed by item 36, unpredictable “off” fluctuations by item 37 and % of the day in “off” by item 39. Dyskinesias were assessed by items 32–34, duration of dyskinesias was assessed by item 32 and disability of dyskinesias by item 33. Dyskinesias in association with the L-dopa test were assessed using the Clinical Dyskinesia Rating Scale (CDRS) [18, 26].
Other clinical assessments
Daytime sleepiness was evaluated by item 3 of the Non-Motor Symptoms Scale (NMSS); score range 0–12, higher = worse [27].
Depressive symptoms were assessed by the Geriatric Depression Scale (GDS-30; score range 0–30, higher = worse) [28]. Depression = scores ≥10, cut-off validated for PD [29].
Continuous assessment of motor function (accelerometry)
Long-term motor function was assessed with a mobile movement recording system, the Parkinson’s Kinetigraph (PKG) [30]. The PKG system consists of a data logger and algorithms that provide continuous registration of movements. This movement recording system has been validated and used in previous studies [30, 31]. Scores for bradykinesia (BKS) and dyskinesias (DKS) are calculated every two minutes from 9:00–18:00 over a certain number of days [30]. Detailed information on immobility (percent time immobile, PTI), inactivity (percent time inactive, PTIn), variation in dyskinesias and bradykinesia (fluctuation dyskinesia score, FDS) and percent time above the threshold for “off” (proportion of time in the period 9:00–18:00 that an individual’s BKS is >26) is also provided.
The PKG-logger was placed on the patient’s most affected arm after the L-dopa test and was worn for 10 days. Collected data were transferred to a server where the results were analyzed using a proprietary algorithm to calculate and graph the respective variables regarding the patients’ mobility throughout the day. In this way, an objective and quantitative picture of the variations in the patient’s motor state is given [30].
PKG values used in this study
All PKG values below were calculated every two minutes during the period 9:00–18:00.
BKS:
a bradykinesia score (BKS), known as the median BKS, which correlates with the UPDRS III [30, 31].
DKS:
a dyskinesia score (DKS), known as the median DKS, which correlates with the Abnormal Involuntary Movement Score [30, 31]. Scores >7 are regarded as clinically evident dyskinesias.
PTI:
percent time immobile. Immobility means that the logger, while being worn by the participant, was entirely still for a two minute period. This has been shown to correlate with sleep in polysomnographic recordings [32].
PTIn:
percent time inactive. This is an estimate of time spent with very little motor activity that is not due to sleep, nor is it directly due to low BKS. It is frequently but not always correlated with the PTI. To be inactive is to be very inert and to have less movement in a two minute period than what might be associated with reading a book.
FDS:
the fluctuation dyskinesia score provides a measure of variation (or fluctuation) in BKS and DKS [33]. An FDS of <8.0 is low and >12.8 is high. Individuals with an FDS of <8.0 have low DKS and high BKS, with very little response to each dose of medication. Patients who are consistently “off” for most or all of the day have low FDS. When the FDS is >12.8 there is greater variation in both BKS and DKS, particularly in the DKS. Patients with peaks of dyskinesia or bradykinesia may have high FDS.
Percent Time Above Threshold:
the proportion of time that an individual’s BKS is >26. This is the point at which tremor reemerges for many individuals [34] and which an expert panel (unpublished results) considers to be “off”.
Statistical Analyses
Descriptive data are given by median and first and third quartiles (q1–q3) and frequencies and percentages, as appropriate.
L-dopa responsiveness was calculated as the absolute differences (“off”-“on”) in scores. In addition, the UPDRS III was also evaluated as the relative difference (“off”-“on”)/”off”×100. Differences were tested statistically by the Wilcoxon Sign-Rank Test. In addition, PKG data were correlated (Spearman) with clinical variables and compared between sub-groups (Mann-Whitney U-test).
P-values of < 0.05 were considered significant. Analyses were performed using IBM SPSS version 22.0 (IBM 211 Corporation, Armonk, NY, USA).
RESULTS
Demographic data
The study sample consisted of 30 individuals (Table 1). The median age was 83 and the median PD duration was 12 years. Nineteen (63%) of the participants were in HY stage IV and 11 (37%) in stage V. In HY stage IV, 21% lived in a nursing home, while in HY stage V the proportion in nursing homes was 55%. The median (q1–q3) LEDD was 799 (536–973) mg. Further data on PD therapy are presented in Table 2. Twenty-one out of the thirty patients (70%) had orthostatic hypotension, defined as a decrease in blood pressure of≥20 mm Hg systolic or≥10 mm Hg diastolic, after three minutes in standing as compared to lying down [35, 36], during the L-dopa test.
Participants: demographics and clinical characteristics, N = 30
q1–q3, first and third quartiles. PD, Parkinson’s disease; HY, Hoehn and Yahr staging scale (score range I–V, higher = worse); S&E, Schwab and England activities of daily living scale (score range 0–100, higher = better); UPDRS, Unified PD Rating Scale, part I = mentation, behaviour and mood (score range 0–16, higher = worse); part II = activities in daily living (score range 0–52; higher = worse); part III = motor examination (score range 0–108, higher = worse); part IV = complications of therapy (score range 0–23, higher = worse); GDS-30, the Geriatric Depression Scale (score range 0–30, higher = worse), depression = scores ≥10; MMSE, Mini-mental state examination (score range 0–30, higher = better, ≥24 normal/no cognitive impairment). *1 missing (GDS-30); **Baseline assessment in practically defined “on” under the patients’ normal medication. All assessments were carried out during baseline examination.
Participant therapeutic data, N = 30
LEDD, Levodopa (L-dopa) equivalent daily dose; monotherapy, therapy with only L-dopa, Carbidopa and L-dopa/Benserazide; Combination, combination of L-dopa with Dopamine agonists, Amantadine, COMT-inhibitors and/or MAO-B inhibitors; COMT-inhibitors, Catechol-O-methyl transferase Inhibitors; MAO-B inhibitors, Monoamine Oxidase B Inhibitors.
Immediate assessment of motor function (L-dopa test)
The median (q1–q3) UPDRS III score in “off” was 46 (37–53.3) and in “on” 36 (27.5–46), with a significant median improvement of 15.5% (7.6–26.5%), p < 0.001 (Table 3). The median UPDRS III score during normal medication was 42 (28.75–50) (Table 1). The UPDRS III score improved during the L-dopa test with ≥15% [12] in 15 (50%) and with ≥30% in six (20%) participants (Fig. 1). Statistically significant improvements were found in all UPDRS III item scores, except for posture and postural instability (Table 3).
L-dopa effect on motor function in late stage PD (HY IV-V), N = 30
Values are presented as median (q1–q3, first and third quartiles) if nothing else is stated. PD, Parkinson’s disease; HY, Hoehn and Yahr staging scale (score range I–V, higher = worse); UPDRS III, Unified PD Rating Scale, part III = motor examination (score range 0–108; higher = worse); CDRS, the Clinical Dyskinesia Rating Scale (score range 0–28, higher = worse); *2 missing (due to not able to perform the test); **in “off” 7 missing, in “on” 6 missing (due to not able to walk), modified/shorter length of gait test, participants in “off” 4, in “on” 5; Bradykinesia (score range 0–36), sum of UPDRS III items: 23 (finger taps, score range 0–8), 24 (hand movements, score range 0–8), 25 (rapid alternating movements of hands, score range 0–8), 26 (foot agility, score range 0–8) and 31 (body bradykinesia and hypokinesia, score range 0–4); Axial signs (score range 0–28), sum of UPDRS III items: 18 (speech, score range 0–4), 19 (facial expression, score range 0–4), 22 (neck rigidity, score range 0–4), 27 (arising from a chair, score range 0–4), 28 (posture, score range 0–4), 29 (gait, score range 0–4) and 30 (postural stability, score range 0–4). Bold p-values are significant at p < 0.05 following Bonferroni corrections.
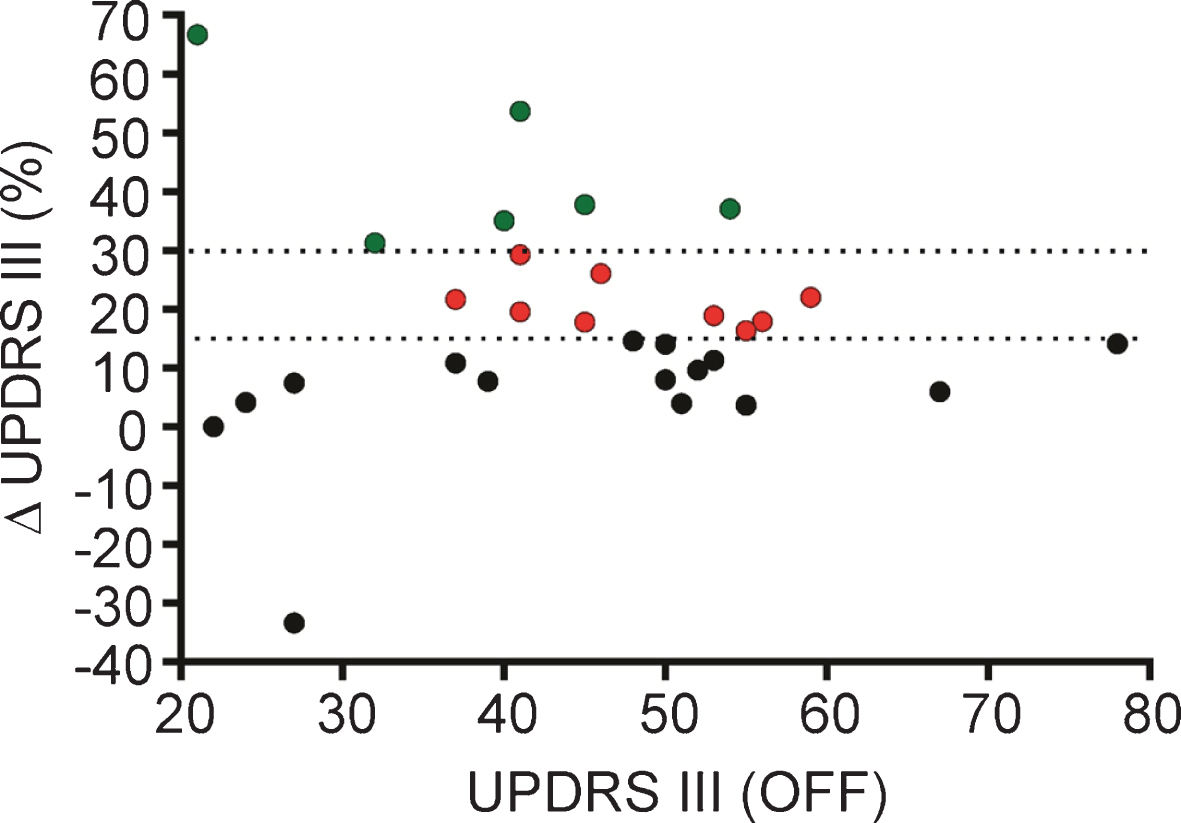
The UPDRS III in “off” plotted against the percentage improvement following the L-dopa. It illustrates the number of participants with an improvement in the UPDRS III score during the L-dopa test of ≥15% (red and green circles n = 15, 50%) and ≥30% (green circles n = 6, 20%). One of the patients who did not have a significant L-dopa effect (i.e., ≥15% improvement in the UPDRS III score during the L-dopa test, black circles) had a worsened UPDRS score in “on” compared to “off” due to medication side effects, in spite of the presence of other signs of having a significant L-dopa effect (difference -33).
A minimal meaningful change in the UPDRS III score (≥2.5 points) was found in 24 (80%) patients; a moderate meaningful change (≥5.2 points) in 19 (63%) and a large meaningful change (≥10.8 points) was observed in 9 (30%) of the participants [22].
The participants who had ≥30% L-dopa effect on the UPDRS III score had significantly (p = 0.02) higher score on the MMSE, i.e., better cognition than the participants with less improvement in the UPDRS III score during the L-dopa test (Fig. 2A).
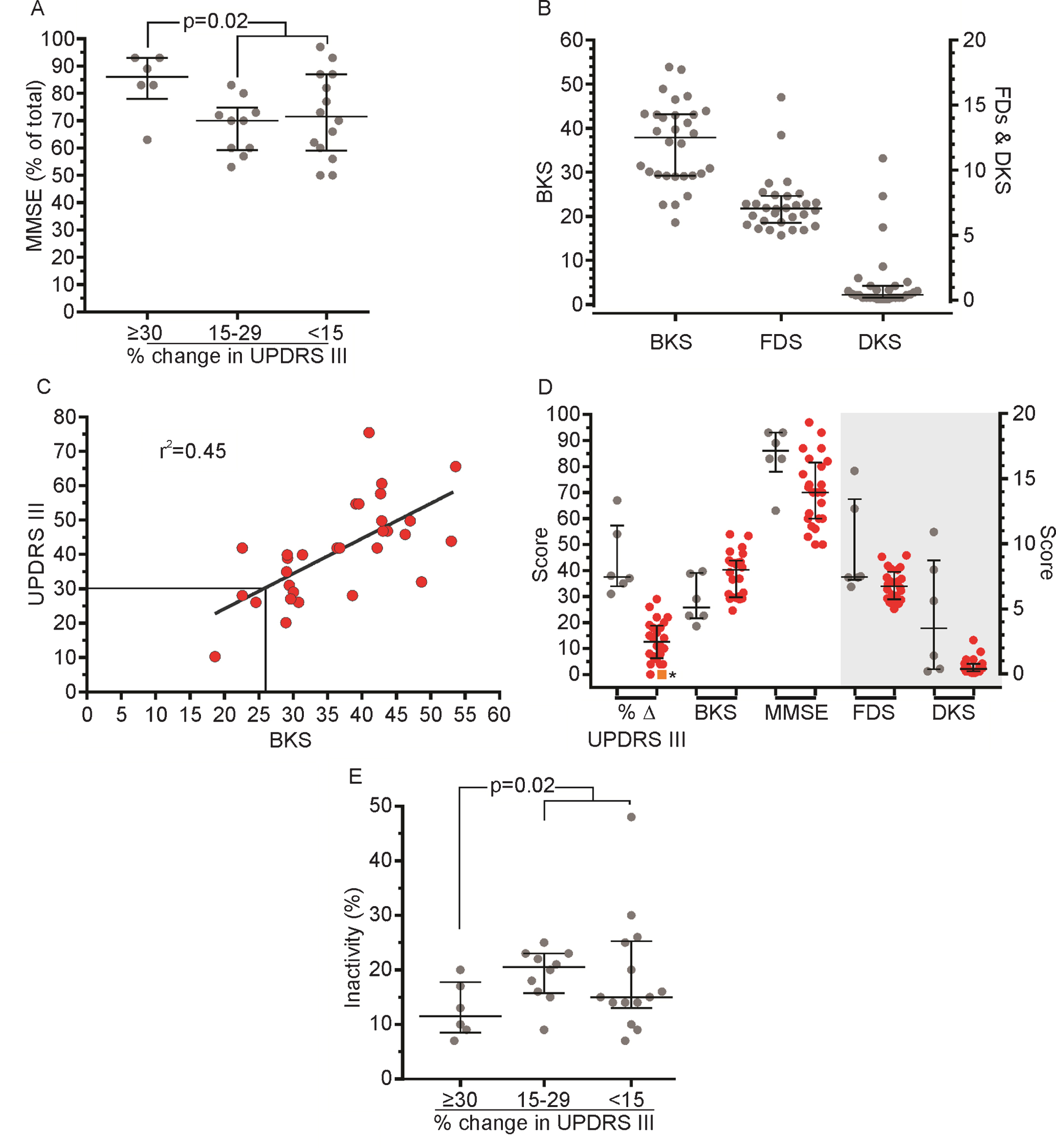
(A) Participants are sorted according to the amplitude of the response to the L-dopa test ≥30%, 15–29% and <15% and plotted against the MMSE expressed as % of the total score. Participants with an L-dopa response (≥30%) had significantly (p = 0.02) higher score on the MMSE than individuals with less improvement in the UPDRS III score during the L-dopa test. (B) The distribution of BKS, FDS and DKS. The participants represented by the three highest FDS (circles) were the same individuals with the three highest DKS. (C) The correlation (coefficient of determination, r2 = 0.45) between the UPDRS III (baseline assessment) score and the BKS. (D) The various scores grouped according to whether the % change in the UPDRS III score was ≥30% (grey circles) or <30% (red circles). The left y-axis represents % change in the UPDRS III scores during the L-dopa test, BKS and MMSE scores (% of total), respectively. The right y-axis represents the FDS and the DKS (grey shaded area). (*orange square: Δ –33, see Fig. 1). (E) Participants are sorted according to the amplitude of the response to the L-dopa test ≥30%, 15–29% and <15% and plotted against the PTIn. Individuals with less improvement in the UPDRS III score during the L-dopa test were significantly (p = 0.02) more inactive than those with an L-dopa response ≥30%. Error bars indicate the median, 25th and 75th percentiles.
Twenty-four (80%) out of the 30 patients were able to perform gait testing. Both gait speed and number of steps improved by >20% in nine participants (38%). An improvement of >15% in hand-arm movements was observed in 13/28 (46%) individuals on the right side and 12/28 (43%) on the left side.
Prevalence of motor fluctuations and dyskinesias
Twenty-one (70%) of the participants reported that they experience either predictable or unpredictable “off” fluctuations (UPDRS IV, items 36–37). All participants reported that they spend at least some part of the day (1% or more; ≥1 on UPDRS IV item 39) in “off”.
The prevalence of dyskinesias according to item 32 of the UPDRS IV (duration of dyskinesias; ≥1) was 47% (14/30 individuals). Disabling dyskinesias (disability of dyskinesias; ≥1 on item 33) were present in 3 (10%) participants.
Only three participants had evident dyskinesias according to the PKG registration (thus a DKS above the mean of individuals without PD). Two of these three participants also had elevated FDS.
Additional PKG results on motor function
Long-term (10 days) motor function, as measured by the PKG, is illustrated in Fig. 2B. The median BKS in this study was 37. This result indicates that a majority of the participants had a pronounced “off” symptomatology, as BKS >26 is by an expert panel considered to be “off” (unpublished results) and is the point when tremor reemerges. Further, it correlates (r2 = 0.45) with the UPDRS III (baseline assessment, Fig. 2C).
The participants with a ≥30% L-dopa improvement in the UPDRS III score were characterized by having lower bradykinesia score and higher MMSE, dyskinesia and fluctuation dyskinesia scores than those with less improvement in the UPDRS III during the L-dopa test (Fig. 2D), who were significantly (p = 0.02) more inactive (Fig. 2E).
According to the PKG, many of the participants spent a large proportion of the day either asleep or very inactive. The average participant spent 20% of the day asleep and 20% of the day very inactive; which constitute 40% of the day either asleep or markedly inactive. The most severe cases spent close to 100% of the day in an “off”-like (BKS >26) condition (Fig. 3A). Those who were very inactive also showed a higher degree of bradykinesia, as shown with both the BKS and the UPDRS III score. The participants who spent a larger percent of the time inactive had more severe bradykinesia, higher UPDRS III (baseline assessment) score and a smaller improvement in the UPDRS III score during the L-dopa test (Fig. 3B). There is a correlation between percent time inactive and median BKS (r2 = 0.71) (Fig. 3C).
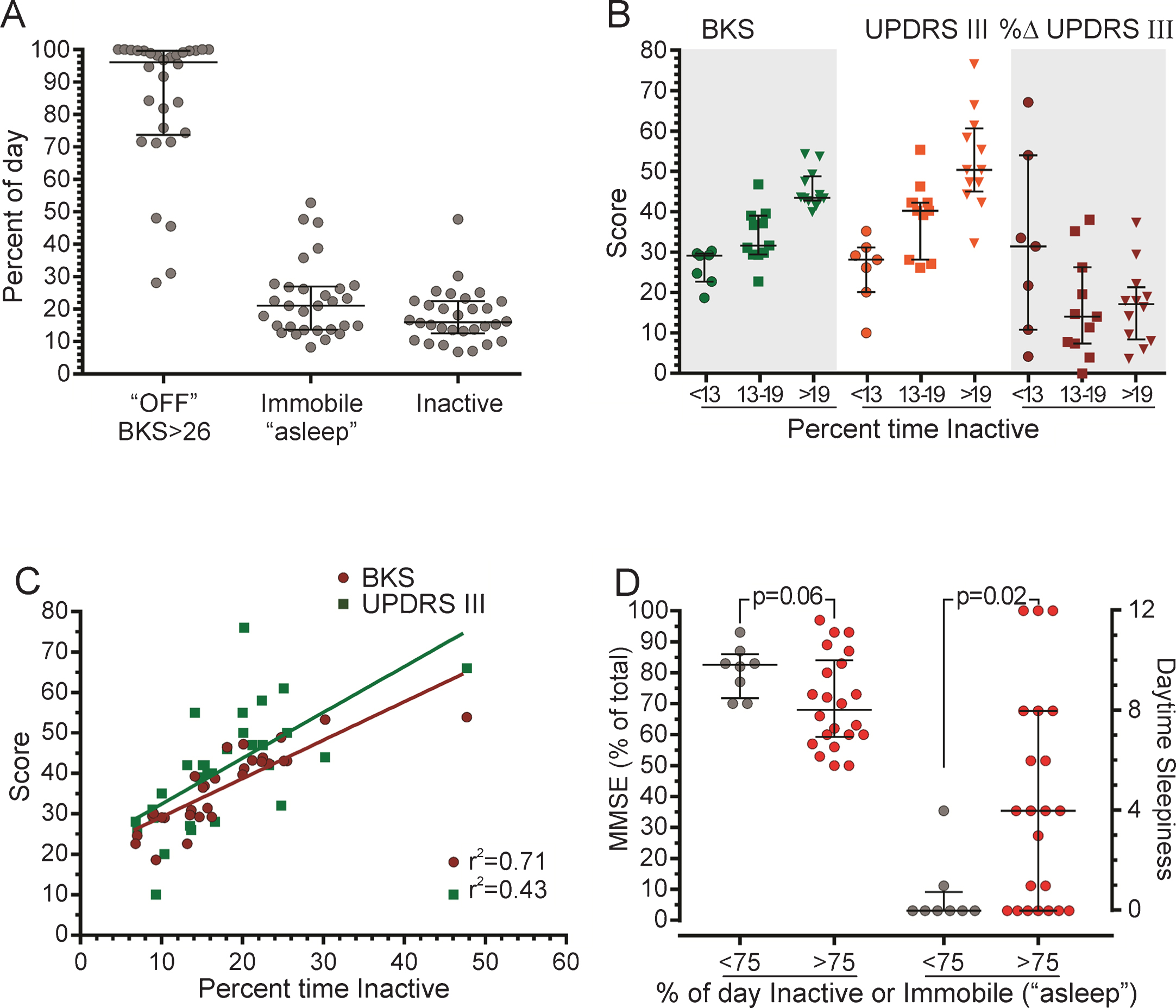
(A) The percent of the period between 09:00–18:00 that the individual was above the threshold for “off” according to the BKS, immobile (PTI, “asleep”) or inactive (PTIn). (B) The BKS (green), the UPDRS III (baseline assessment, orange) and the percent change in the UPDRS III following the L-dopa test (% Δ, red), sorted into cases where the PTIn was <13 (circles), 13–19 (squares) or >19 (triangles). (C) The UPDRS III scores (green squares) and the BKS (red circles) plotted against the PTIn. The linear regression and the coefficient of determination for the UPDRS III (baseline assessment) and BKS are shown. (D) Participants are sorted according to their response to the MMSE expressed as % of the total score (left y-axis) and the NMSS Q3 (daytime sleepiness), right y-axis. The PTIn and PTI are shown combined on the x-axis as <75 (grey circles) and >75 (red circles) % of day inactive or immobile (“asleep”). Error bars indicate the median, 25th and 75th percentiles. L-dopa, levodopa; UPDRS III, Unified PD Rating Scale, part III = motor examination (score range 0–108, higher = worse); MMSE, Mini-Mental State Examination (score range 0–30, higher = better); NMSS, the Non-Motor Symptoms Scale (NMSS), item 3 daytime sleepiness (score range 0–12, higher = worse); PKG, the Parkinson’s KinetiGraph; PKG values: BKS, bradykinesia score; DKS, dyskinesia score; FDS, fluctuation dyskinesia score; PTI, percent time immobile; PTIn, percent time inactive.
Cognition tended to be lower and daytime sleepiness was significantly higher in individuals with more immobility and inactivity. There was a tendency that the more active participants had higher cognition, as measured by the MMSE, than those who spent >75% of the day asleep or very inactive. The participants who spent >75% of the day asleep or very inactive also reported significantly more daytime sleepiness, as assessed by item 3 of the NMSS (p = 0.02), than the more active participants (Fig. 3D).
DISCUSSION
L-dopa effect and motor complications
The degree of L-dopa response in late stage PD has been unclear. This study suggests that half of the late stage PD patients had a significant improvement of ≥15% in motor function according to the UPDRS III and around 40–50% showed improvements (above the 15% and 20% thresholds) in specific tests of gait and upper limb mobility following a standardized L-dopa test.
As a substantial amount of late stage PD patients still demonstrate significant L-dopa responsiveness, the value of continuously optimizing treatment with dopaminergic therapy should be emphasized. In late stage PD, as for PD patients in general, the treating physician should not rely solely on the results of an L-dopa test, but titrate up dopaminergic medication until there is no more symptomatic gain of raising the dosage or when side effects outweigh gains. This could also include continuing treatment with continuous dopaminergic treatment with apomorphine or L-dopa-carbidopa intestinal gel (LCIG) pump therapy. When informing about dopaminergic therapy, the message should be that there will most likely always be benefits of dopaminergic therapy, even if there is a development of symptoms which respond less well to L-dopa, such as freezing of gait (FOG) and cognitive impairment.
Although not including a control group of patients with less severe PD, our results are in agreement with previous observations [8] that there is a tendency towards a somewhat decreased L-dopa response in late stage PD patients as compared to in earlier disease stages [12]. The majority of the patients had motor fluctuations and about half of them had dyskinesias as estimated by the UPDRS IV, although the extent and severity of motor complications were not very pronounced according to the PKG registrations. These results provide new knowledge to this field, as L-dopa effect and the occurrence of motor complications in late stage PD have previously been scarcely studied.
PKG results
The PKG results suggest that patients with late stage PD is a very sedentary group, spending a large proportion of the day either immobile or very inactive. The vast majority of the participants spent close to 100% of the day in an “off”-like state, as measured by the PKG’s score for bradykinesia. These observations are similar to the results of a previous study, where a large proportion of late stage PD patients were found to be “off” during most of the day [9]. However, there was a tendency that those with good L-dopa effect had less bradykinesia, better cognition and somewhat more fluctuations and dyskinesias than those with a poorer L-dopa effect. Furthermore, we observed a relation between the time spent inactive, cognition and daytime sleepiness, where participants who were more inactive had poorer cognition and more daytime sleepiness, than more active individuals. The PKG provides a possibility to get an insight on motor performance during the day, as compared to how the patients present during a clinical assessment. There are likely further probabilistic relations between bradykinesia, apathy, cognition and daytime sleepiness that are not well differentiated, in distinguishing between what a patient can or cannot do due to e g bradykinesia as compared to what he or she actually performs in a real life condition.
Comparison between clinical assessments and PKG results
Half of our sample had a good L-dopa effect, while they according to the PKG registrations spend a considerable amount of the day in an “off”-like state, immobile or very inactive. This suggests the possibility of factors other than pharmacological response influencing the immobility of this group; e g cognition, apathy and daytime sleepiness. This adds up to the already reduced mobility that comes with the disease progression, as defined by the HY stages [3]. Another possible reason for this discrepancy about what patients perform during an L-dopa test as compared to in their everyday state, could be suboptimal dopaminergic treatment as well as an undertreatment of typical non-motor symptoms (NMS), such as cognition, apathy and depressive symptoms.
The severity of motor fluctuations and dyskinesias according to the PKG registrations, as well as when observing the participants clinically during the L-dopa test and under regular treatment, does not appear very pronounced, in spite of being present in many of the participants according to the UPDRS IV. The PKG registrations suggested that a number of the participants with a non-significant (<15%) [12] L-dopa effect during testing still exhibited signs of motor fluctuations.
Dopaminergic undertreatment
It has been suggested that late stage PD patients are undertreated [9, 10]. One reason for this may be increased complications such as orthostatic hypotension [37] and hallucinations [4]. In addition, treating physicians may not be aware that motor function of these patients may still be improved with optimized dopaminergic therapy.
However, the LEDD estimates of the current sample do not suggest undertreatment. The median LEDD in this study was about 800 mg, which appears fairly representative of late stage PD [8]. This does not exclude that some patients may have been undertreated. The PKG registrations provide a valuable complement to the L-dopa testing, with additional information on long-term motor function of this patient group.
Strengths, limitations and future perspectives
As the majority of the participants were identified through hospital outpatient neurology clinics, there is a risk that the sample cannot be considered completely representative, as those with the most severe disease may not be able to attend the hospital clinics any longer. However, attempts were made to reach also those who do not have contact with the hospital clinics, although this lead to recruitment of participants in relatively few cases.
The risk of getting false negative L-dopa test results [13] should be considered. There is a risk that one underestimates the results and that, in spite of little or no detectable L-dopa response in the test situation, the patient can still benefit during long-term treatment.
All participants had the diagnosis idiopathic PD, given by specialists (neurologist, in two cases geriatrician). However, one cannot completely rule out that single participants may have had atypical Parkinsonism, which could explain limited L-dopa response in these cases. However, this reflects clinical reality and does therefore not necessarily challenge the relevance or validity of our observations.
The PKG is a validated movement recording system [30, 31]. However, a special difficulty in this study was that when patients use a mobility device such as a four wheeled walker with the wrists held still on the handles, there could be a risk that the PKG registrations would interpret the patients as less mobile than they are.
This study was carried out in the homes of the participants due to their severe disability. A limitation could be that the test procedures were less standardized than in a hospital setting. On the other hand, an advantage of home testing is that it in contrast to data collection in a clinical setting reflects the patients’ function under real life conditions. Home testing was well accepted by the participants and in spite of probably being more resource consuming than in a clinical setting, it is likely a necessary approach for many of these severely affected patients to be able to participate.
A strength in this study was the excellent inclusion rate, with only one patient declining participation and another two being excluded due to non-compliance. The participants were from a geographically widespread area in the southern region of Sweden and were consecutively admitted to the study from the baseline assessment of the CLaSP study [17]. There was a slight majority of men, which reflects the PD population at large [38]. Two thirds of the participants were in HY stage IV and one third in stage V, which also appears representative for late stage PD at large.
A major strength of this study is that we were able to recruit and conduct L-dopa tests in the late and most severe disease stages of PD, which hardly has been documented before. However, there is a need for additional studies in larger samples to better understand and allow for firmer conclusions regarding the dopaminergic responsiveness of different types of therapies in this population. In addition to motor, also non-motor effects should be investigated. The effect of L-dopa on NMS and the occurrence of NMS fluctuations are subject to a separate study (manuscript in preparation).
Conclusions
In conclusion, about half of patients with late stage PD appear to exhibit a significant L-dopa response (≥15% on the UPDRS III). A majority experience motor fluctuations and about half have dyskinesias, although the PKG registrations suggest that these are not very severe in this population. It is thus important to strive towards optimized dopaminergic therapy also in the late and most severe stages of PD.
CONFLICT OF INTEREST
Professor Malcolm Horne has developed the Parkinson’s Kinetigraph (PKG; Global Kinetics Corporation) and has a financial interest in this instrument. However, the PKG is only one out of several components in this study.
Footnotes
ACKNOWLEDGMENTS
The CLaSP consortium.
The main funding of this project: a JPND grant provided by the Swedish Research Council (VR), CLaSP/JPND HC-559-002; MultiPark, the strategic research area for neuroscience at Lund University; the Swedish Parkinson Foundation; the Swedish Parkinson Academy, the Faculty of Medicine at Lund University.
