Abstract
Background
Parkinsonism following hypoxic ischemic damage of the basal ganglia is an uncommon phenomenon that has been infrequently reported. However, only a few cases have noted improvement of symptoms with dopaminergic therapy. We report the clinical and imaging features of five patients with post-anoxic parkinsonism responsive to dopamine supplementation.
Objective
To describe a retrospective case series of five cases of dopamine-responsive post-anoxic parkinsonism.
Methods
We identified all the cases using the Mayo Clinic Data Management System utilizing advanced data explorer search engine for any patients evaluated for post anoxic parkinsonism and its associated acronyms from 2000–2024. Clinical features, neuroimaging, medication trials, and responses were obtained from chart review of identified patients.
Results
Five patients met the inclusion criteria. All patients underwent anoxic events followed by development of parkinsonism. Patients exhibited parkinsonism described as combinations of bradykinesia, rigidity, tremor, and postural instability. All patients underwent evaluation by a neurologist, MRI imaging, and treatment by dopaminergic agents. Of the five patients, four received carbidopa/levodopa whereas one received a dopamine agonist. All patients were clinically followed for a median of approximately 4 years and showed improvement in parkinsonism.
Conclusions
Parkinsonism following a hypoxic ischemic insult is a rare occurrence but response to dopaminergic therapy in those cases is even more scarcely described. Our cases series provides important implications for treatment options for patients with post anoxic parkinsonism.
Plain language summary
Signs and symptoms of parkinsonism could develop rarely after damage of the basal ganglia region of the brain from lack of blood flow or oxygen. In those cases, an even smaller group of patients have been reported to respond to dopamine therapy. We describe the clinical details, imaging, and medication regimens of five patients with post-anoxic parkinsonism that improved with dopamine medications. Five patients were identified by searching a patient database for terms that described parkinsonism after anoxic events. Those who responded to dopamine medications were identified and their charts were reviewed for presenting signs and symptoms, medical evaluations, imaging results, and response to various therapeutics including medications that affected the dopamine system. Those cases showed various presentations of parkinsonism after anoxic events that responded well to medications that increase dopamine in the brain which could be helpful for clinicians to treat future patients with similar presentations.
Introduction
Anoxic brain injury can result from insults including circulatory, cardiac, and respiratory etiologies ranging from most commonly, cardiac arrest to severe hypotension, respiratory arrest, carbon monoxide inhalation, and drug overdose.1,2 A wide variety of movement disorders due to hypoxic ischemic brain injury has been well documented. Postanoxic akinetic-rigid syndromes resembling parkinsonism have been noted in patients with damage in the lenticular nucleus and striatum.3–5
In the past, limited anecdotal reports of clinical improvement to levodopa treatment in post anoxic parkinsonism have been inconsistent and unpredictable. PET and SPECT studies have shown decreased pre-synaptic uptake of dopamine, predicting a theoretical positive response to dopaminergic therapy.6,7 In contrast, MR imaging is limited to its ability to demonstrate postsynaptic region involvement only, thus, cannot be relied upon to predict levodopa response. In contrast, patients that exhibit parkinsonism with no clear MRI abnormalities may have a higher likelihood of levodopa response due to a suspected presynaptic neuronal insult. This limitation in the ability to predict levodopa response in postanoxic parkinsonism patients remains a clinical obstacle for both the clinician and patients. We report a case series of five patients with postanoxic parkinsonism that improved after dopaminergic therapy.
Methods
Using Mayo Clinic Data Management System, patients were identified from a search of Mayo Clinic Rochester patients evaluated from 2000 to 2024. The search was completed using inclusive search terms of postanoxic parkinsonism, anoxic parkinsonism, and other associated synonyms including post hypoxic parkinsonism, hypoxic parkinsonism, and ischemic parkinsonism. Exclusion criteria included any patients that were: < 18 years of age, did not have response to dopaminergic agents, prior neurodegenerative disease diagnosis, or parkinsonism prior to the ischemic event. Six patients originally met inclusion criteria with one patient excluded as she was treated with levodopa and did not respond.
The demographic, clinical, and imaging variables were extracted from Epic electronic medical record by a neurologist (T.L.). Variables including sex, age, anoxic event, medication trials, medication response, and imaging results were recorded from chart review. All patients completed a brain MRI at Mayo clinic and the available images and radiology reports were reviewed.
Results
Patient cases
Patient 1
A 32-year-old right-handed male suffered an out-of-hospital respiratory arrest secondary to a massive bilateral pulmonary embolism. The patient had extended pulseless activity and asystole with multiple unsuccessful attempts of intubation and resuscitation before arrival at the hospital. Upon transfer to the hospital, the patient was reintubated with eventual return of spontaneous circulation with intravenous vasopressor support. The hospital course was later complicated by gastrointestinal bleed, hypotension, and rhabdomyolysis. MRI brain showed subtle T2 hyperintensity bilaterally in the caudate heads, putamen, globus pallidus, and medial temporal lobes (Figure 1). Approximately 1 month after his anoxic event, the patient was discharged to inpatient rehab at which time physical medicine and rehabilitation (PMR) physicians described parkinsonism with significant bradykinesia. Upon recovery from the initial hypoxic event and inpatient rehab stay, the patient presented to the outpatient neurology clinic for evaluation of spells and abnormal movements. The patient was evaluated by a Mayo movement disorders specialist who noted marked facial masking, anarthria, moderate neck rigidity, bradykinesia, decreased arm swing, and severe freezing and shuffling gait. A trial of levodopa was started and titrated up to a total daily dose of 750 mg in three divided doses in combination with carbidopa taken on an empty stomach. On neurological examination at a 12-month follow-up, the patient was noted to have improved bradykinesia, increased heel strike during gait, and significant resolution of gait freezing.
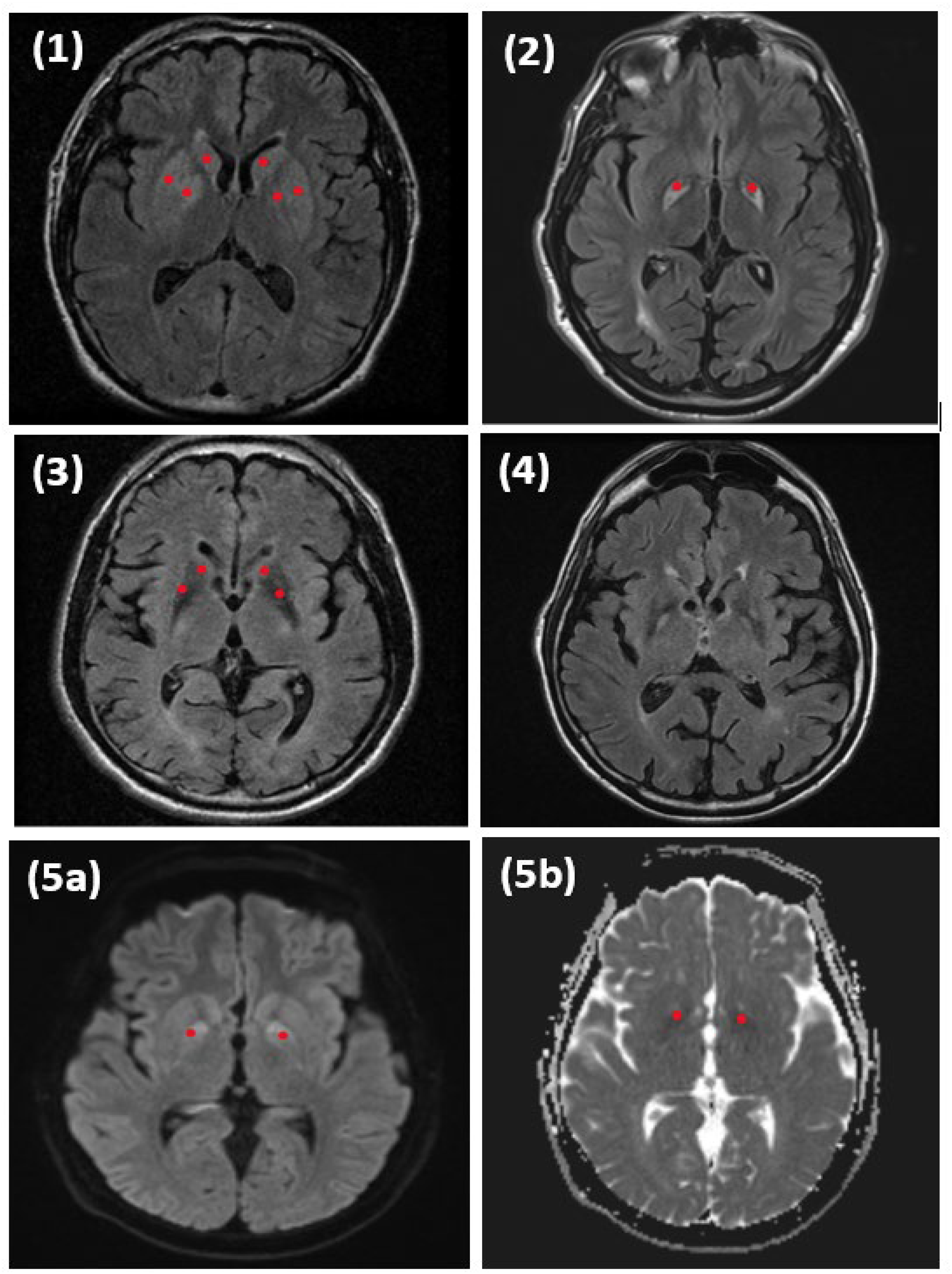
Axial MRI sequences of patients at the level of the basal ganglia.
Patient 2
A 36-year-old right-handed male suffered a heroin overdose resulting in a 5-day coma, anoxic brain injury, and prolonged hospitalization. One year after his anoxic event, he presented to Mayo neurology for evaluation for significant mobility issues described as marked slowness in all activities. It was unclear when his motor symptoms started after his anoxic event. MRI brain showed T2 hyperintensity of bilateral substantia nigra and bilateral T2 hyperintensity of globus pallidus with mild hemosiderin deposition (Figure 1). A neurologist evaluated the patient and noted significant physical exam findings of marked parkinsonism described as hypokinetic dysarthria, marked rigidity of all limbs, generalized bradykinesia, and severely slowed gait with freezing. Prior to the first evaluation by neurology, the patient was treated with baclofen and trihexyphenidyl with no improvement in his symptoms. Those medications were later tapered off. The patient was started on levodopa and up titrated to a total daily dose of 900 mg, in 3 divided doses with carbidopa. On follow-up evaluation 1 year later, the patient had moderate improvement in rigidity, bradykinesia of his hands, and mildly improved speech paucity while consistently maintained on dopamine repletion.
Patient 3
A 45-year-old right-handed male suffered a myocardial infarction and subsequent cardiac arrest with a prolonged resuscitation. He received emergent resuscitation and cardiac catheterization followed by prolonged hospitalization in the intensive care unit. His hospital course was complicated by retroperitoneal hematoma, gastrointestinal bleeding, and pancreatitis. After discharge to physical rehabilitation approximately a few weeks later, the patient showed signs of mood lability, cognitive slowing, and motor impairment. MRI brain showed T2 hyperintensities of the bilateral caudate, putamen, and anterior internal capsule (Figure 1). On initial evaluation by local physicians, the patient was noted to exhibit parkinsonism. Initial treatment with ropinirole, 1 mg TID, improved his paucity of movements. Unfortunately, months later the patient's motor impairments worsened despite the increase of ropinirole to 3 mg TID which was later tapered off with significant worsening of symptoms described as akinetic. Mayo Neurology later evaluated patient approximately 14 months after the anoxic event with neurological examination notable for decreased attention, mildly hypokinetic speech, hypomimia, decreased blink rate, absent right arm swing with moderate bradykinesia and en-bloc turning during gait. Pramipexole was initiated then titrated up to 1 mg TID with significant improvement in rigidity and bradykinesia during follow up neurologic examination.
Patient 4
A 48-year-old right-handed male underwent a Billroth procedure for peptic ulcer disease with subsequent respiratory arrest following the procedure. The patient was hospitalized in the intensive care unit with emergence of cognitive impairment, tremor, and gait disorder at discharge approximately 1 month after the anoxic event. Following speech and physical therapy, the patient continued to show motor impairment described as tremor and gait instability with local neurology evaluation noting findings of parkinsonism. MRI brain showed mild generalized atrophy with no clear abnormalities of the basal ganglia (Figure 1). Pramipexole was started and levodopa was added one year later. Upon evaluation 6 years later by a Mayo movement disorders specialist, the patient noted prior significant improvement of tremor with dopaminergic therapy. He was maintained on pramipexole 1.5/1.5/1 mg daily, in addition to carbidopa/levodopa 25/100 mg, on a 1.5/1.5/1 tablet regimen. Neurological examination was notable for mild hypomimia, increased tone in all limbs, decreased bilateral right greater than left arm swing, moderately decreased stride length, mild bilateral postural tremor, moderate kinetic tremor, and mildly slowed alternating movements. Due to concern for somnolence, pramipexole and levodopa were both tapered off. Approximately 5 years after first evaluation, the patient returned with worsened gait impairment described as shuffling and fenestration of gait, stooped posture, and increased falls requiring a walker. Repeat neurological examination was notable for mild hypomimia, moderate left greater than right arm rigidity, and moderate bilateral bradykinesia with absent left arm swing and shuffling gait. Carbidopa/levodopa 25/100 mg was reinitiated and titrated to one tablet three times daily with significantly improved gait described as upright posture and ability to walk independently without gait aid.
Patient 5
A 66-year-old right-handed male underwent urgent surgical repair of ascending aortic dissection. The surgery was complicated by hypoxia with noted pancreatic ischemia and hypoxic brain damage with noted T2 weighted and FLAIR hyperintensities on MRI images following the event. Approximately 2 weeks post-operatively, the patient experienced significant visual changes, word-finding difficulties, bradykinesia, along with cognitive and gait impairment. After 4 weeks of physical therapy, the patient was able to ambulate but with continued bradykinesia. MRI brain showed restricted diffusion of bilateral globus pallidus on diffusion-weighted images with corresponding ADC signal (Figure 1). Subsequently, 1.5 years later the patient continued to have gait difficulties that acutely worsened in the context of COVID-19 infection which prompted evaluation by neurology. The patient was then evaluated by a Mayo movement disorders specialist who noted dysarthria, shuffling gait, and bradykinesia. A course of carbidopa/levodopa was started and titrated up to a total of 1200 mg of levodopa daily in four doses in combination with 25 mg of carbidopa each dose. PET CT brain was obtained after levodopa therapy was initiated which showed mild patchy FDG hypometabolism in the bilateral caudate and putamen. On neurological examination during follow-up, the patient showed decreased bradykinesia and improved gait. There was a distinct wearing-off effect reported 1 h before the next scheduled levodopa dosage described as overall paucity of movements.
Discussion
The search identified 5 patients who met criteria for inclusion. Table 1 shows the demographic, clinical, imaging features along with medication dosages of our 5 patients. All the patients were male (100%) and age of onset of symptoms ranged from 32 to 66 years, with mean of 45.4 and median of 45. All patients underwent hypoxic ischemic insults: circulatory, respiratory, or cardiac arrest. Three of the five patients were documented to have prolonged periods of anoxia and resuscitation with no clearly specified time of hypoxia. No patients reported or had a documented history of parkinsonism or any neurodegenerative disorder prior to anoxic event. All five patients were evaluated by a Mayo staff neurologist and were clinically followed for a median of 4 years. All patients exhibited parkinsonism including symptoms of hypomimia, bradykinesia, dysarthria, rigidity, and/or gait imbalance. Four patients were treated with carbidopa/levodopa, while one patient was maintained on pramipexole. All patients (or caretakers) reported a beneficial response with dopaminergic therapy, which was sustained. Of those patients who remained on carbidopa/levodopa, the levodopa ranged from 300 mg to 1200 mg daily in divided doses. Notably, patient 4 presented with observed asymmetric parkinsonism with a possible 1-month latency after anoxic event that responded well to low dose levodopa that raises the possibility of a separate development of idiopathic Parkinson's disease.
Demographic, clinical and imaging features of post-anoxic parkinsonism patients.
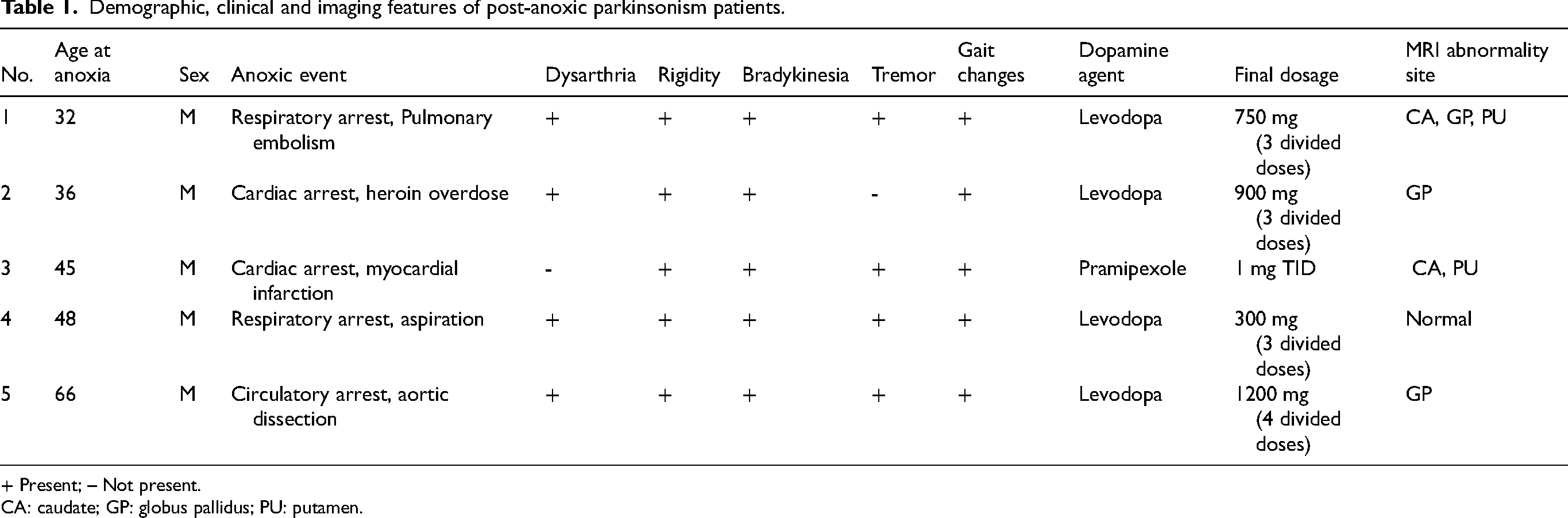
+ Present; – Not present.
CA: caudate; GP: globus pallidus; PU: putamen.
All 5 patients underwent MRI brain imaging. MRI reports and images were reviewed and pallidal or striatal abnormalities were noted in 4 of the 5 patients, implying a postsynaptic involvement. In those patients, T2 hyperintensities were found in various regions of the basal ganglia including the caudate, putamen and globus pallidus. In one patient, MRI diffusion weighted imaging showed clear abnormalities in the globus pallidus. No patients underwent DAT scans.
Movement disorders due to post anoxic damage have been reported as presentations of myoclonus, chorea, tics, dystonia, parkinsonism and including a progressive supranuclear palsy syndrome.1,3,7–10 Post-anoxic parkinsonism/akinetic rigid syndromes have been described but clear improvement with dopamine therapy was only reported in a small minority of cases.5,8 Note that the basal ganglia is particularly vulnerable to ischemic cell death due to a higher concentration of excitatory glutamate receptors and increased metabolic demand. 2 Prior literature of post anoxic movement disorders found younger individuals presented with more dystonic syndromes and older individuals presenting as more akinetic rigid syndromes with a proposed aging of nigrostriatal system. 3 There was no clear documentation of the timeline of appearance of parkinsonism after the inciting event; however, most of our cohort exhibited symptoms and signs soon after hospital discharge.
Among prior reports of post-anoxic parkinsonism, the location of MRI lesions has primarily been in the globus pallidus. This was consistent with 3 of our 5 patients. One of our patients had abnormalities localized to the putamen and caudate. Another patient had no lesions on MRI imaging. Interestingly in patient 5, FDG PET scan showed mild patchy hypometabolism in bilateral caudate and putamen after initiation of levodopa therapy. Studies in the past have shown baseline FDG PET hypermetabolism of the pallidothalamic circuitry in idiopathic Parkinson's disease with similar hypometabolism in the putamen in levodopa responsive idiopathic Parkinson's disease after levodopa therapy.11–13 This finding further suggests the possibility of presynaptic dopaminergic transmission disruption in patient 5. Obviously, lack of pathology on imaging as in one of our patients does not exclude possibility of unseen disruption of the dopaminergic system- and highlights the possibility of other imaging modalities for evaluation of levodopa response.
Of note, four of our five patients had lesions on the postsynaptic side of the dopaminergic synapse (i.e., globus pallidus or striatum). Intuitively, carbidopa levodopa should not be very effective if the pathology is exclusively postsynaptic. A previously theorized mechanism of parkinsonism after anoxic damage proposed incomplete postsynaptic striatal D2-receptor loss with an expected theoretical response to dopamine as shown in our case series. 7 Additionally, it seems likely that these postsynaptic findings were also associated with presynaptic dopaminergic insults, given the fact that patients responded, albeit incompletely. Presynaptic insults would obviously appear on DAT imaging but that was not done in our cohort. Additionally, this highlights the limitations of MRI imaging modalities to evaluate only specific parts of the dopaminergic system.
There exist limitations to this study due to its single center design and retrospective nature. Different neurologists examined each patient with purely descriptive clinical examinations with no validated scale to evaluate parkinsonism or clinical improvement which could introduce inter-rater variability and observer bias. Our study did not include one patient with postanoxic parkinsonism that did not respond to levodopa for comparison which limits its significance owing partly to the small number of patients available that met the exclusion and inclusion criteria in this single center study. Other limitations included the lack of certain data such as time latency from anoxic event to development of parkinsonism symptoms that were not available partly due to the retrospective nature of the study. Additionally, DaT scan information was not available for the patients to evaluate for pre-synaptic dopaminergic transmission to confirm the presence of presynaptic insults. Taken together, these limitations could limit the generalizability of the study findings.
Although our patient numbers are limited, this is the among the largest case series of patients with dopamine responsive post-anoxic parkinsonism. This suggests that parkinsonism time-locked to a post anoxic insult should be treated with dopaminergic therapy and ideally, levodopa. This is advisable even in the setting of brain MRI findings of postsynaptic pathology.
Footnotes
Acknowledgments
We thank the patients that provided permission for their information to be used in this research.
Ethical considerations
This research was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans.
Consent to participate
All patients included in this study provided informed consent for their information to be used.
Consent for publication
Not applicable
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: James Bower receives research support from Novartis and Amylyx Pharmaceuticals. Tina Liu, J. Eric Ahlskog, and Orhun Kantarci have no relevant conflicts of interest to report. Rodolfo Savica is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
Data availability
The data supporting the findings of this study are available upon request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
